Calcium: The Missing Link in Auxin Action
Abstract
:1. Auxin and Ca2+: Essential Elements of Plant Development
2. The Source of Auxin-Induced Ca2+ Signals
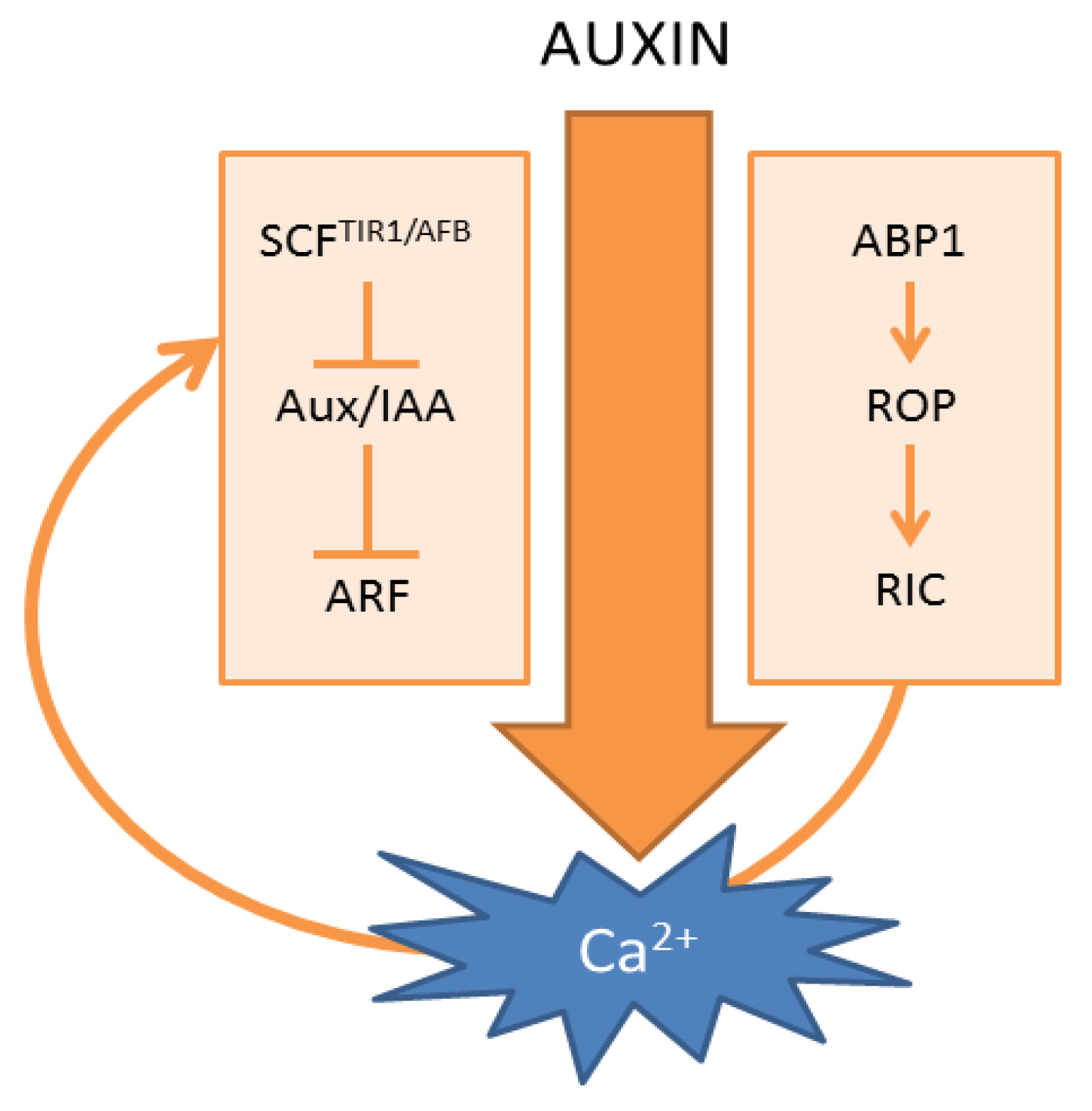
3. Auxin Receptors for Auxin-Induced Ca2+ Signals
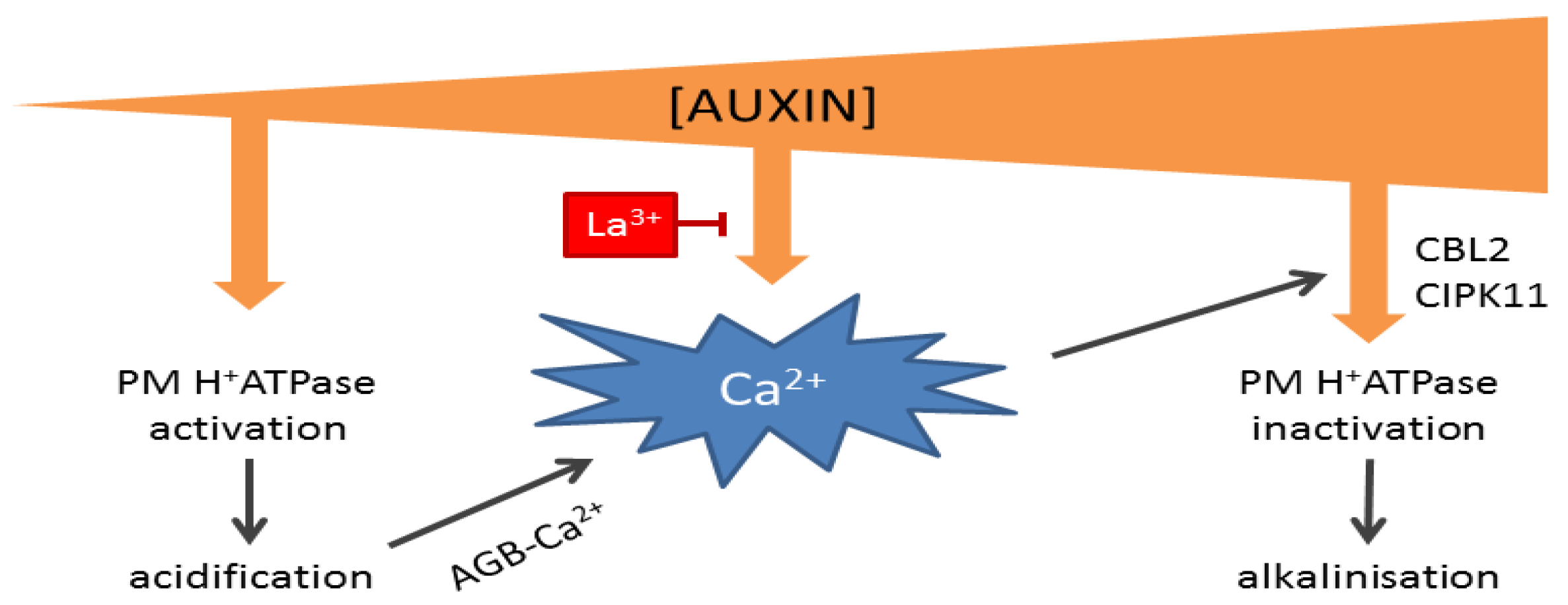
4. Calcium Modulates pH and Growth
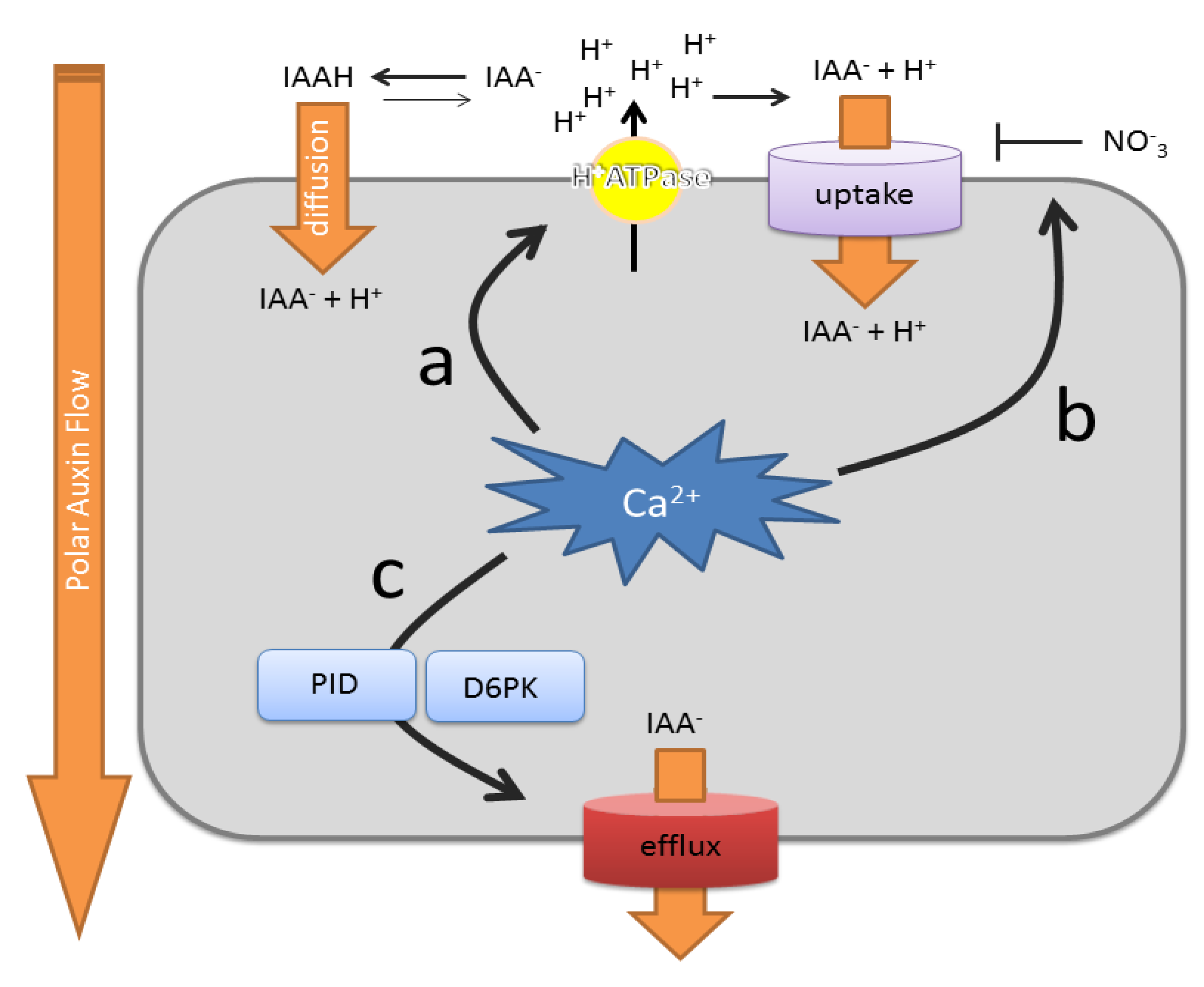
5. Ca2+ Controls the Rate of Auxin Transport
6. Ca2+ in the Balance of Exocytosis and Endocytosis
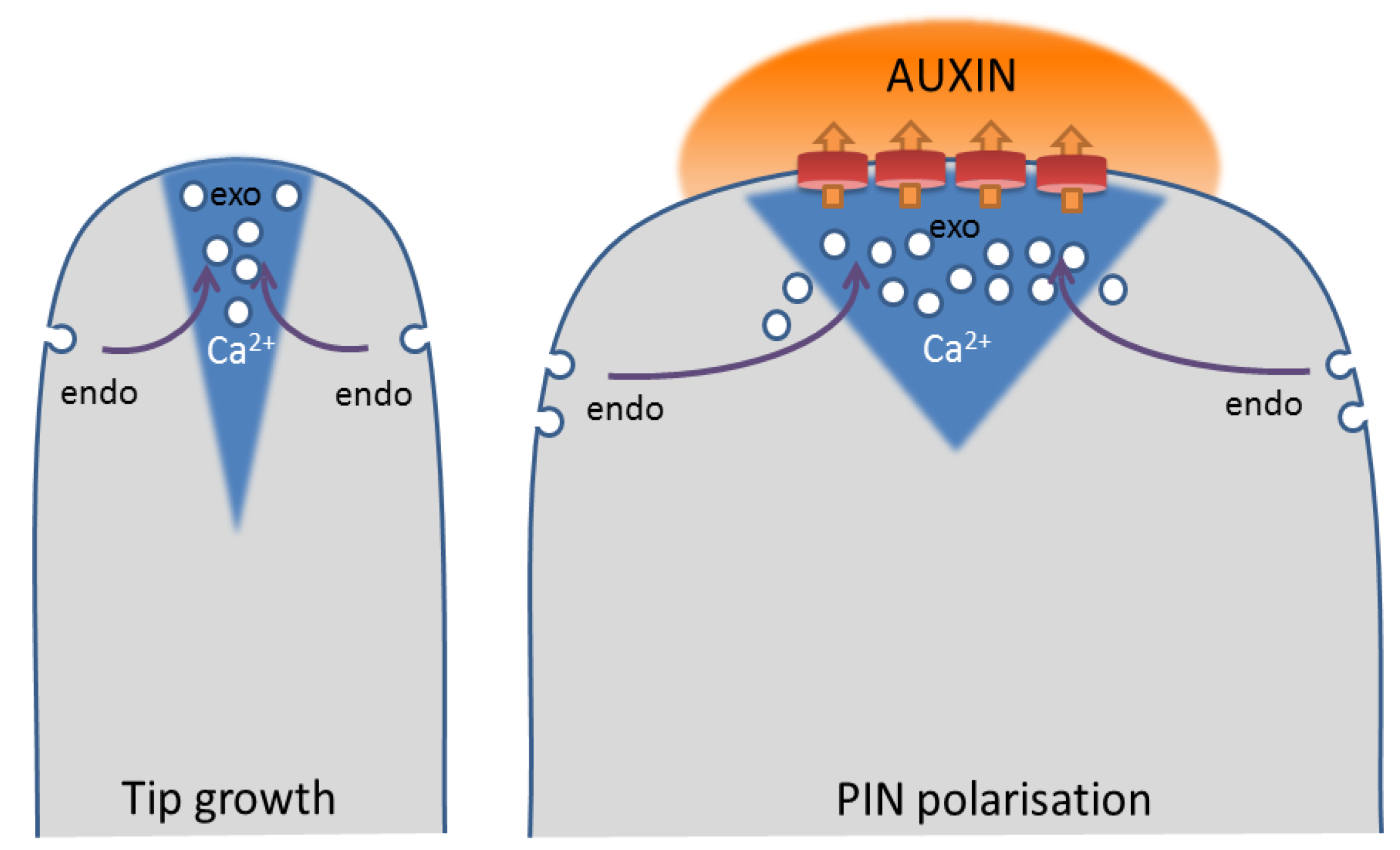
7. Ca2+ As a Means to Change Auxin Flow Direction
8. The Ca2+-Auxin Interplay during Gravitropism
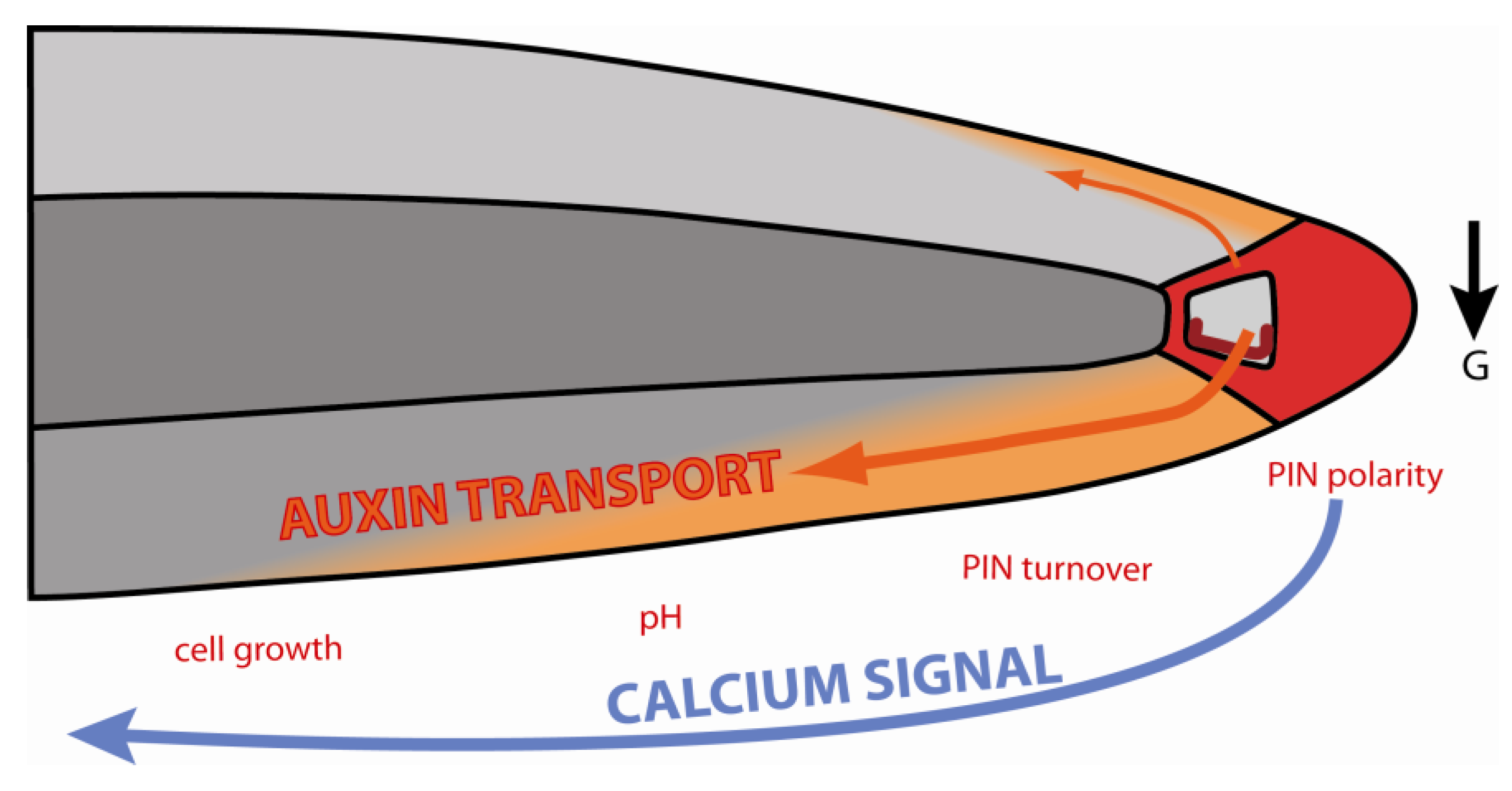
9. Conclusions and Future Perspectives
Acknowledgements
Conflicts of Interest
References
- Vanneste, S.; Friml, J. Auxin: A trigger for change in plant development. Cell 2009, 136, 1005–1016. [Google Scholar] [CrossRef]
- Tanaka, H.; Dhonukshe, P.; Brewer, P.B.; Friml, J. Spatiotemporal asymmetric auxin distribution: A means to coordinate plant development. Cell Mol. Life Sci. 2006, 63, 2738–2754. [Google Scholar]
- Napier, R.M.; Venis, M.A. Tansley review No-79-auxin action and auxin-binding proteins. New Phytol. 1995, 129, 167–201. [Google Scholar]
- Bennett, T.; Scheres, B. Root development-two meristems for the price of one? Curr. Top. Dev. Biol. 2010, 91, 67–102. [Google Scholar] [CrossRef]
- Lavenus, J.; Goh, T.; Roberts, I.; Guyomarc’h, S.; Lucas, M.; de Smet, I.; Fukaki, H.; Beeckman, T.; Bennett, M.; Laplaze, L. Lateral root development in Arabidopsis: Fifty shades of auxin. Trends Plant Sci. 2013, 18, 450–458. [Google Scholar]
- Reinhardt, D.; Pesce, E.R.; Stieger, P.; Mandel, T.; Baltensperger, K.; Bennett, M.; Traas, J.; Friml, J.; Kuhlemeier, C. Regulation of phyllotaxis by polar auxin transport. Nature 2003, 426, 255–260. [Google Scholar]
- Benkova, E.; Michniewicz, M.; Sauer, M.; Teichmann, T.; Seifertova, D.; Jurgens, G.; Friml, J. Local, efflux-dependent auxin gradients as a common module for plant organ formation. Cell 2003, 115, 591–602. [Google Scholar] [CrossRef]
- Barkoulas, M.; Hay, A.; Kougioumoutzi, E.; Tsiantis, M. A developmental framework for dissected leaf formation in the Arabidopsis relative cardamine hirsuta. Nat. Genet. 2008, 40, 1136–1141. [Google Scholar] [CrossRef]
- Scarpella, E.; Marcos, D.; Friml, J.; Berleth, T. Control of leaf vascular patterning by polar auxin transport. Genes Dev. 2006, 20, 1015–1027. [Google Scholar] [CrossRef]
- Pagnussat, G.C.; Alandete-Saez, M.; Bowman, J.L.; Sundaresan, V. Auxin-dependent patterning and gamete specification in the Arabidopsis female gametophyte. Science 2009, 324, 1684–1689. [Google Scholar]
- Zadnikova, P.; Petrasek, J.; Marhavy, P.; Raz, V.; Vandenbussche, F.; Ding, Z.; Schwarzerova, K.; Morita, M.T.; Tasaka, M.; Hejatko, J.; et al. Role of pin-mediated auxin efflux in apical hook development of Arabidopsis thaliana. Development 2010, 137, 607–617. [Google Scholar]
- Vandenbussche, F.; Petrasek, J.; Zadnikova, P.; Hoyerova, K.; Pesek, B.; Raz, V.; Swarup, R.; Bennett, M.; Zazimalova, E.; Benkova, E.; et al. The auxin influx carriers AUX1 and LAX3 are involved in auxin-ethylene interactions during apical hook development in Arabidopsis thaliana Seedlings. Development 2010, 137, 597–606. [Google Scholar] [CrossRef]
- Toyota, M.; Gilroy, S. Gravitropism and mechanical signaling in plants. Am. J. Bot. 2013, 100, 111–125. [Google Scholar] [CrossRef]
- Takahashi, H.; Miyazawa, Y.; Fujii, N. Hormonal interactions during root tropic growth: Hydrotropism versus gravitropism. Plant Mol. Biol. 2009, 69, 489–502. [Google Scholar] [CrossRef]
- Friml, J.; Wisniewska, J.; Benkova, E.; Mendgen, K.; Palme, K. Lateral relocation of auxin efflux regulator PIN3 mediates tropism in Arabidopsis. Nature 2002, 415, 806–809. [Google Scholar]
- Ding, Z.; Galvan-Ampudia, C.S.; Demarsy, E.; Langowski, L.; Kleine-Vehn, J.; Fan, Y.; Morita, M.T.; Tasaka, M.; Fankhauser, C.; Offringa, R.; et al. Light-mediated polarization of the PIN3 auxin transporter for the phototropic response in Arabidopsis. Nat. Cell Biol. 2011, 13, 447–452. [Google Scholar]
- Christie, J.M.; Yang, H.; Richter, G.L.; Sullivan, S.; Thomson, C.E.; Lin, J.; Titapiwatanakun, B.; Ennis, M.; Kaiserli, E.; Lee, O.R.; et al. phot1 inhibition of ABCB19 primes lateral auxin fluxes in the shoot apex required for phototropism. PLoS Biol. 2011, 9, e1001076. [Google Scholar] [CrossRef]
- Tao, Y.; Ferrer, J.L.; Ljung, K.; Pojer, F.; Hong, F.; Long, J.A.; Li, L.; Moreno, J.E.; Bowman, M.E.; Ivans, L.J.; et al. Rapid synthesis of auxin via a new tryptophan-dependent pathway is required for shade avoidance in plants. Cell 2008, 133, 164–176. [Google Scholar]
- Jones, A.R.; Kramer, E.M.; Knox, K.; Swarup, R.; Bennett, M.J.; Lazarus, C.M.; Leyser, H.M.; Grierson, C.S. Auxin transport through non-hair cells sustains root-hair development. Nat. Cell Biol. 2009, 11, 78–84. [Google Scholar] [CrossRef]
- Ikeda, Y.; Men, S.; Fischer, U.; Stepanova, A.N.; Alonso, J.M.; Ljung, K.; Grebe, M. Local auxin biosynthesis modulates gradient-directed planar polarity in Arabidopsis. Nat. Cell Biol. 2009, 11, 731–738. [Google Scholar] [CrossRef]
- Irving, H.R.; Gehring, C.A.; Parish, R.W. Changes in cytosolic pH and calcium of guard cells precede stomatal movements. Proc. Natl. Acad. Sci. USA 1992, 89, 1790–1794. [Google Scholar] [CrossRef]
- Ding, Z.; Wang, B.; Moreno, I.; Duplakova, N.; Simon, S.; Carraro, N.; Reemmer, J.; Pencik, A.; Chen, X.; Tejos, R.; et al. ER-localized auxin transporter PIN8 regulates auxin homeostasis and male gametophyte development in Arabidopsis. Nat. Commun. 2012, 3. [Google Scholar] [CrossRef]
- Kim, J.I.; Murphy, A.S.; Baek, D.; Lee, S.W.; Yun, D.J.; Bressan, R.A.; Narasimhan, M.L. Yucca6 over-expression demonstrates auxin function in delaying leaf senescence in Arabidopsis thaliana. J. Exp. Bot. 2011, 62, 3981–3992. [Google Scholar]
- Lim, P.O.; Lee, I.C.; Kim, J.; Kim, H.J.; Ryu, J.S.; Woo, H.R.; Nam, H.G. Auxin response Factor 2 (Arf2) plays a major role in regulating auxin-mediated leaf longevity. J. Exp. Bot. 2010, 61, 1419–1430. [Google Scholar]
- Sorefan, K.; Girin, T.; Liljegren, S.J.; Ljung, K.; Robles, P.; Galvan-Ampudia, C.S.; Offringa, R.; Friml, J.; Yanofsky, M.F.; Ostergaard, L. A regulated auxin minimum is required for seed dispersal in Arabidopsis. Nature 2009, 459, 583–586. [Google Scholar]
- Goetz, M.; Vivian-Smith, A.; Johnson, S.D.; Koltunow, A.M. Auxin response factor 8 is a negative regulator of fruit initiation in Arabidopsis. Plant Cell 2006, 18, 1873–1886. [Google Scholar] [CrossRef]
- Ellis, C.M.; Nagpal, P.; Young, J.C.; Hagen, G.; Guilfoyle, T.J.; Reed, J.W. Auxin response factor1 and auxin response factor2 regulate senescence and floral organ abscission in Arabidopsis thaliana. Development 2005, 132, 4563–4574. [Google Scholar] [CrossRef]
- Grunewald, W.; Cannoot, B.; Friml, J.; Gheysen, G. parasitic nematodes modulate PIN-mediated auxin transport to facilitate infection. PLoS Pathog. 2009, 5, e1000266. [Google Scholar] [CrossRef] [Green Version]
- Navarro, L.; Dunoyer, P.; Jay, F.; Arnold, B.; Dharmasiri, N.; Estelle, M.; Voinnet, O.; Jones, J.D. A plant miRNA contributes to antibacterial resistance by repressing auxin signaling. Science 2006, 312, 436–439. [Google Scholar]
- Iglesias, M.J.; Terrile, M.C.; Bartoli, C.G.; D’ippolito, S.; Casalongue, C.A. Auxin signaling participates in the adaptative response against oxidative stress and salinity by interacting with redox metabolism in Arabidopsis. Plant Mol. Biol. 2010, 74, 215–222. [Google Scholar] [CrossRef]
- Ding, Z.; Friml, J. Auxin regulates distal stem cell differentiation in Arabidopsis Roots. Proc. Natl. Acad. Sci. USA 2010, 107, 12046–12051. [Google Scholar] [CrossRef]
- Himanen, K.; Boucheron, E.; Vanneste, S.; de Almeida Engler, J.; Inze, D.; Beeckman, T. Auxin-mediated cell cycle activation during early lateral root initiation. Plant Cell 2002, 14, 2339–2351. [Google Scholar] [CrossRef] [Green Version]
- Vanneste, S.; de Rybel, B.; Beemster, G.T.; Ljung, K.; de Smet, I.; van Isterdael, G.; Naudts, M.; Iida, R.; Gruissem, W.; Tasaka, M.; et al. Cell cycle progression in the pericycle is not sufficient for solitary root/IAA14-mediated lateral root initiation in Arabidopsis thaliana. Plant Cell 2005, 17, 3035–3050. [Google Scholar]
- Tromas, A.; Braun, N.; Muller, P.; Khodus, T.; Paponov, I.A.; Palme, K.; Ljung, K.; Lee, J.Y.; Benfey, P.; Murray, J.A.; et al. The auxin binding protein 1 is required for differential auxin responses mediating root growth. PLoS One 2009, 4, e6648. [Google Scholar] [CrossRef]
- Xu, T.; Nagawa, S.; Yang, Z. Uniform auxin triggers the rho gtpase-dependent formation of interdigitation patterns in pavement cells. Small Gtpases 2011, 2, 227–232. [Google Scholar]
- Abas, L.; Benjamins, R.; Malenica, N.; Paciorek, T.; Wisniewska, J.; Moulinier-Anzola, J.C.; Sieberer, T.; Friml, J.; Luschnig, C. Intracellular Trafficking and proteolysis of the Arabidopsis auxin-efflux facilitator PIN2 are involved in root gravitropism. Nat. Cell Biol. 2006, 8, 249–256. [Google Scholar] [CrossRef]
- Baster, P.; Robert, S.; Kleine-Vehn, J.; Vanneste, S.; Kania, U.; Grunewald, W.; de Rybel, B.; Beeckman, T.; Friml, J. SCF(TIR1/AFB)-auxin signalling regulates PIN vacuolar trafficking and auxin fluxes during root gravitropism. EMBO J. 2013, 32, 260–274. [Google Scholar]
- Paciorek, T.; Zazimalova, E.; Ruthardt, N.; Petrasek, J.; Stierhof, Y.D.; Kleine-Vehn, J.; Morris, D.A.; Emans, N.; Jurgens, G.; Geldner, N.; et al. Auxin Inhibits endocytosis and promotes its own efflux from cells. Nature 2005, 435, 1251–1256. [Google Scholar] [CrossRef]
- Robert, S.; Kleine-Vehn, J.; Barbez, E.; Sauer, M.; Paciorek, T.; Baster, P.; Vanneste, S.; Zhang, J.; Simon, S.; Covanova, M.; Hayashi, K.; et al. ABP1 mediates auxin inhibition of clathrin-dependent endocytosis in Arabidopsis. Cell 2010, 143, 111–121. [Google Scholar] [CrossRef]
- Sauer, M.; Robert, S.; Kleine-Vehn, J. Auxin: Simply complicated. J. Exp. Bot. 2013, 64, 2565–2577. [Google Scholar] [CrossRef]
- Ljung, K. Auxin metabolism and homeostasis during plant development. Development 2013, 140, 943–950. [Google Scholar] [CrossRef]
- Lokerse, A.S.; Weijers, D. Auxin enters the matrix—Assembly of response machineries for specific outputs. Curr. Opin. Plant Biol. 2009, 12, 520–526. [Google Scholar] [CrossRef]
- Chapman, E.J.; Estelle, M. Mechanism of auxin-regulated gene expression in plants. Annu. Rev. Genet. 2009, 43, 265–285. [Google Scholar] [CrossRef]
- Vanstraelen, M.; Benkova, E. Hormonal interactions in the regulation of plant development. Annu. Rev. Cell. Dev. Biol. 2012, 28, 463–487. [Google Scholar] [CrossRef]
- Bothwell, J.H.; Ng, C.K. The evolution of Ca2+ signalling in photosynthetic eukaryotes. New Phytol. 2005, 166, 21–38. [Google Scholar] [CrossRef]
- Verret, F.; Wheeler, G.; Taylor, A.R.; Farnham, G.; Brownlee, C. Calcium channels in photosynthetic eukaryotes: Implications for evolution of calcium-based signalling. New Phytol. 2010, 187, 23–43. [Google Scholar] [CrossRef]
- Hepler, P.K. Calcium: A central regulator of plant growth and development. Plant Cell 2005, 17, 2142–2155. [Google Scholar] [CrossRef]
- Kudla, J.; Batistic, O.; Hashimoto, K. Calcium signals: The lead currency of plant information processing. Plant Cell 2010, 22, 541–563. [Google Scholar] [CrossRef]
- Dodd, A.N.; Kudla, J.; Sanders, D. The language of calcium signaling. Annu. Rev. Plant Biol. 2010, 61, 593–620. [Google Scholar] [CrossRef]
- Shishova, E.; Lindberg, S. Auxin-induced cytosol acidification in wheat leaf protoplasts depends on external concentration of Ca2+. J. Plant Physiol. 1999, 155, 190–196. [Google Scholar]
- Shishova, M.; Lindberg, S. Auxin induces an increase of Ca2+ concentration in the cytosol of wheat leaf protoplasts. J. Plant Physiol. 2004, 161, 937–945. [Google Scholar] [CrossRef]
- Felle, H. Auxin causes oscillations of cytosolic free calcium and pH in Zea-Mays coleoptiles. Planta 1988, 174, 495–499. [Google Scholar] [CrossRef]
- Gehring, C.A.; Irving, H.R.; Parish, R.W. Effects of auxin and abscisic-acid on cytosolic calcium and ph in plant-cells. Proc. Natl. Acad. Sci. USA 1990, 87, 9645–9649. [Google Scholar]
- Shishova, M.; Yemelyanov, V.; Rudashevskaya, E.; Lindberg, S. A shift in sensitivity to auxin within development of maize seedlings. J. Plant. Physiol. 2007, 164, 1323–1330. [Google Scholar] [CrossRef]
- Monshausen, G.B.; Miller, N.D.; Murphy, A.S.; Gilroy, S. Dynamics of auxin-dependent Ca2+ and pH signaling in root growth revealed by integrating high-resolution imaging with automated computer vision-based analysis. Plant J. 2011, 65, 309–318. [Google Scholar] [CrossRef]
- Wheeler, G.L.; Brownlee, C. Ca2+ signalling in plants and green algae—Changing channels. Trends Plant Sci. 2008, 13, 506–514. [Google Scholar] [CrossRef]
- Cousson, A.; Vavasseur, A. Putative involvement of cytosolic Ca2+ and gtp-binding proteins in cyclic-GMP-mediated induction of stomatal opening by auxin in Commelina communis L. Planta 1998, 206, 308–314. [Google Scholar] [CrossRef]
- Ayling, S.M.; Clarkson, D.T. The cytoplasmic streaming response of tomato root hairs to auxin; the role of calcium. Aust. J. Plant Physiol. 1996, 23, 699–708. [Google Scholar] [CrossRef]
- Shishova, M.F.; Lindberg, S.; Polevoi, V.V. Auxin activation of Ca2+ transport across the plasmalemma of plant cells. Russ. J. Plant Physiol. 1999, 46, 626–633. [Google Scholar]
- Shishova, M.F.; Inge-Vechtomova, N.I.; Vykhvalov, K.A.; Rudashevskaya, E.L.; Polevoi, V.V. Auxin-dependent transport of K+ and Ca2+ across the membrane of plasmalemma vesicles from coleoptile cells. Russ. J. Plant. Physiol. 1998, 45, 67–73. [Google Scholar]
- Thomine, S.; Zimmermann, S.; Vanduijn, B.; Barbier-Brygoo, H.; Guern, J. Calcium-channel antagonists induce direct inhibition of the outward rectifying potassium channel in tobacco protoplasts. FEBS Lett. 1994, 340, 45–50. [Google Scholar] [CrossRef]
- Ordenes, V.R.; Moreno, I.; Maturana, D.; Norambuena, L.; Trewavas, A.J.; Orellana, A. In vivo analysis of the calcium signature in the plant Golgi apparatus reveals unique dynamics. Cell Calcium 2012, 52, 397–404. [Google Scholar] [CrossRef]
- Krebs, M.; Held, K.; Binder, A.; Hashimoto, K.; den Herder, G.; Parniske, M.; Kudla, J.; Schumacher, K. FRET-based genetically encoded sensors allow high-resolution live cell imaging of Ca2+ Dynamics. Plant J. 2012, 69, 181–192. [Google Scholar] [CrossRef]
- Mehlmer, N.; Parvin, N.; Hurst, C.H.; Knight, M.R.; Teige, M.; Vothknecht, U.C. A toolset of aequorin expression vectors for in planta studies of subcellular calcium concentrations in Arabidopsis thaliana. J. Exp. Bot. 2012, 63, 1751–1761. [Google Scholar] [CrossRef]
- Costa, A.; Drago, I.; Behera, S.; Zottini, M.; Pizzo, P.; Schroeder, J.I.; Pozzan, T.; lo Schiavo, F. H2O2 in plant peroxisomes: An in vivo analysis uncovers a Ca2+-dependent scavenging system. Plant J. 2010, 62, 760–772. [Google Scholar] [CrossRef]
- Logan, D.C.; Knight, M.R. Mitochondrial and cytosolic calcium dynamics are differentially regulated in plants. Plant Physiol. 2003, 133, 21–24. [Google Scholar] [CrossRef]
- Felle, H.; Brummer, B.; Bertl, A.; Parish, R.W. Indole-3-acetic-acid and fusicoccin cause cytosolic acidification of corn coleoptile cells. Proc. Natl. Acad. Sci. USA 1986, 83, 8992–8995. [Google Scholar] [CrossRef]
- Brummer, B.; Bertl, A.; Potrykus, I.; Felle, H.; Parish, R.W. Evidence That fusicoccin and indole-3-acetic-acid induce cytosolic acidification of Zea-Mays cells. FEBS Lett. 1985, 189, 109–114. [Google Scholar] [CrossRef]
- Jurado, S.; Abraham, Z.; Manzano, C.; Lopez-Torrejon, G.; Pacios, L.F.; Del Pozo, J.C. The Arabidopsis cell cycle f-box protein Skp2a binds to auxin. Plant Cell 2010, 22, 3891–3904. [Google Scholar] [CrossRef]
- Sauer, M.; Kleine-Vehn, J. Auxin binding protein 1: The outsider. Plant Cell 2011, 23, 2033–2043. [Google Scholar] [CrossRef]
- Venis, M.A.; Napier, R.M.; Oliver, S. Molecular analysis of auxin-specific signal transduction. Plant Growth Regul. 1996, 18, 1–6. [Google Scholar] [CrossRef]
- Barbier-Brygoo, H.; Ephritikhine, G.; Klambt, D.; Ghislain, M.; Guern, J. Functional evidence for an auxin receptor at the plasmalemma of tobacco mesophyll protoplasts. Proc. Natl. Acad. Sci. USA 1989, 86, 891–895. [Google Scholar] [CrossRef]
- Tian, H.; Klambt, D.; Jones, A.M. Auxin-binding protein 1 does not bind auxin within the endoplasmic reticulum despite this being the predominant subcellular location for this hormone receptor. J. Biol. Chem. 1995, 270, 26962–26969. [Google Scholar] [CrossRef]
- Felle, H.; Peters, W.; Palme, K. The electrical response of maize to auxins. Biochim. Biophys. Acta 1991, 1064, 199–204. [Google Scholar]
- Barbier-Brygoo, H.; Ephritikhine, G.; Klambt, D.; Maurel, C.; Palme, K.; Schell, J.; Guern, J. Perception of the auxin signal at the plasma-membrane of tobacco mesophyll protoplasts. Plant J. 1991, 1, 83–93. [Google Scholar] [CrossRef]
- Thiel, G.; Blatt, M.R.; Fricker, M.D.; White, I.R.; Millner, P. Modulation of K+ channels in vicia stomatal guard cells by peptide homologs to the auxin-binding protein C-terminus. Proc. Natl. Acad. Sci. USA 1993, 90, 11493–11497. [Google Scholar] [CrossRef]
- Zimmermann, S.; Thomine, S.; Guern, J.; Barbier-Brygoo, H. An anion current at the plasma-membrane of tobacco protoplasts shows ATP-dependent voltage regulation and is modulated by auxin. Plant J. 1994, 6, 707–716. [Google Scholar]
- Xu, T.; Wen, M.; Nagawa, S.; Fu, Y.; Chen, J.G.; Wu, M.J.; Perrot-Rechenmann, C.; Friml, J.; Jones, A.M.; Yang, Z. Cell surface- and rho GTPase-based auxin signaling controls cellular interdigitation in Arabidopsis. Cell 2010, 143, 99–110. [Google Scholar] [CrossRef]
- Nagawa, S.; Xu, T.; Lin, D.; Dhonukshe, P.; Zhang, X.; Friml, J.; Scheres, B.; Fu, Y.; Yang, Z. Rop GTPase-dependent actin microfilaments promote pin1 polarization by localized inhibition of clathrin-dependent endocytosis. PLoS Biol. 2012, 10, e1001299. [Google Scholar] [CrossRef] [Green Version]
- Chen, X.; Naramoto, S.; Robert, S.; Tejos, R.; Lofke, C.; Lin, D.; Yang, Z.; Friml, J. ABP1 and ROP6 GTPase signaling regulate clathrin-mediated endocytosis in Arabidopsis roots. Curr. Biol. 2012, 22, 1326–1332. [Google Scholar] [CrossRef]
- Marten, I.; Lohse, G.; Hedrich, R. Plant-growth hormones control voltage-dependent activity of anion channels in plasma-membrane of guard-cells. Nature 1991, 353, 758–762. [Google Scholar]
- Gehring, C.A.; Mcconchie, R.M.; Venis, M.A.; Parish, R.W. Auxin-binding-protein antibodies and peptides influence stomatal opening and alter cytoplasmic pH. Planta 1998, 205, 581–586. [Google Scholar] [CrossRef]
- Cho, D.; Villiers, F.; Kroniewicz, L.; Lee, S.; Seo, Y.J.; Hirschi, K.D.; Leonhardt, N.; Kwak, J.M. Vacuolar CAX1 and CAX3 influence auxin transport in guard cells via regulation of apoplastic pH. Plant Physiol. 2012, 160, 1293–1302. [Google Scholar] [CrossRef]
- Yamagami, M.; Haga, K.; Napier, R.M.; Iino, M. Two distinct signaling pathways participate in auxin-induced swelling of pea epidermal protoplasts. Plant Physiol. 2004, 134, 735–747. [Google Scholar] [CrossRef]
- Tromas, A.; Paque, S.; Stierle, V.; Quettier, A.L.; Muller, P.; Lechner, E.; Genschik, P.; Perrot-Rechenmann, C. Auxin-binding protein 1 is a negative regulator of the SCF(TIR1/AFB) pathway. Nat. Commun. 2013, 4. [Google Scholar] [CrossRef]
- Singla, B.; Chugh, A.; Khurana, J.P.; Khurana, P. An early auxin-responsive Aux/IAA gene from wheat (Triticum aestivum) is induced by epibrassinolide and differentially regulated by light and calcium. J. Exp. Bot. 2006, 57, 4059–4070. [Google Scholar] [CrossRef]
- Thimann, K.V. Hormones and the analysis of growth. Plant Physiol. 1938, 13, 437–449. [Google Scholar] [CrossRef]
- Takahashi, K.; Hayashi, K.; Kinoshita, T. Auxin activates the plasma membrane H+-atpase by phosphorylation during hypocotyl elongation in Arabidopsis. Plant Physiol. 2012, 159, 632–641. [Google Scholar]
- Heyn, A.N.J. Molecular-Basis of auxin-regulated extension growth and role of dextranase. Proc. Natl. Acad. Sci. USA 1981, 78, 6608–6612. [Google Scholar] [CrossRef]
- Heyn, A.N.J. Dextranase activity and auxin-induced cell elongation in coleoptiles of Avena. Biochem. Biophys. Res. Commun. 1970, 38, 831–837. [Google Scholar] [CrossRef]
- Claussen, M.; Luthen, H.; Blatt, M.; Bottger, M. Auxin-induced growth and its linkage to potassium channels. Planta 1997, 201, 227–234. [Google Scholar]
- Philippar, K.; Buchsenschutz, K.; Edwards, D.; Loffler, J.; Luthen, H.; Kranz, E.; Edwards, K.J.; Hedrich, R. The Auxin-induced K+ channel gene Zmk1 in maize functions in coleoptile growth and is required for embryo development. Plant Mol. Biol. 2006, 61, 757–768. [Google Scholar]
- Thiel, G.; Brüdern, A.; Gradmann, D. Small inward rectifying K+ channels in coleoptiles: Inhibition by external Ca2+ and function in cell elongation. J. Membr. Biol. 1996, 149, 9–20. [Google Scholar]
- Rayle, D.L.; Cleland, R.E. Evidence that auxin-induced growth of soybean hypocotyls involves proton excretion. Plant Physiol. 1980, 66, 433–437. [Google Scholar]
- Hager, A. Role of the plasma membrane H+-atpase in auxin-induced elongation growth: Historical and new aspects. J. Plant Res. 2003, 116, 483–505. [Google Scholar] [CrossRef]
- Luthen, H.; Bigdon, M.; Bottger, M. Reexamination of the acid growth theory of auxin action. Plant Physiol. 1990, 93, 931–939. [Google Scholar] [CrossRef]
- Hager, A.; Menzel, H.; Krauss, A. Experiments and hypothesis concerning primary action of auxin in elongation growth. Planta 1971, 100, 47–75. [Google Scholar]
- Moloney, M.M.; Elliott, M.C.; Cleland, R.E. Acid growth effects in maize roots—Evidence for a link between auxin-economy and proton extrusion in the control of root-growth. Planta 1981, 152, 285–291. [Google Scholar] [CrossRef]
- Tode, K.; Luthen, H. Fusicoccin- and IAA-induced elongation growth share the same pattern of K+ dependence. J. Exp. Bot. 2001, 52, 251–255. [Google Scholar] [CrossRef]
- Sondergaard, T.E.; Schulz, A.; Palmgren, M.G. Energization of transport processes in plants. roles of the plasma membrane H+-atpase. Plant Physiol. 2004, 136, 2475–2482. [Google Scholar] [CrossRef]
- Duby, G.; Poreba, W.; Piotrowiak, D.; Bobik, K.; Derua, R.; Waelkens, E.; Boutry, M. Activation of plant plasma membrane H+-ATPase by 14-3-3 proteins is negatively controlled by two phosphorylation sites within the H+-ATPase C-terminal region. J. Biol. Chem. 2009, 284, 4213–4221. [Google Scholar]
- Fuglsang, A.T.; Guo, Y.; Cuin, T.A.; Qiu, Q.; Song, C.; Kristiansen, K.A.; Bych, K.; Schulz, A.; Shabala, S.; Schumaker, K.S.; et al. Arabidopsis Protein Kinase PKS5 inhibits the plasma membrane H+-atpase by preventing interaction with 14-3-3 Protein. Plant Cell 2007, 19, 1617–1634. [Google Scholar]
- Staal, M.; de Cnodder, T.; Simon, D.; Vandenbussche, F.; van der Straeten, D.; Verbelen, J.P.; Elzenga, T.; Vissenberg, K. Apoplastic alkalinization is instrumental for the inhibition of cell elongation in the arabidopsis root by the ethylene precursor 1-aminocyclopropane-1-carboxylic acid. Plant Physiol. 2011, 155, 2049–2055. [Google Scholar] [CrossRef]
- Hasenstein, K.H.; Evans, M.L. Calcium dependence of rapid auxin action in maize roots. Plant Physiol. 1986, 81, 439–443. [Google Scholar]
- Lamport, D.T.; Varnai, P. Periplasmic arabinogalactan glycoproteins act as a calcium capacitor that regulates plant growth and development. New Phytol. 2013, 197, 58–64. [Google Scholar] [CrossRef]
- Schenck, D.; Christian, M.; Jones, A.; Luthen, H. Rapid auxin-induced cell expansion and gene expression: A four-decade-old question revisited. Plant Physiol. 2010, 152, 1183–1185. [Google Scholar] [CrossRef]
- Rück, A.; Palme, K.; Venis, M.A.; Napier, R.M.; Felle, R.H. Patch-clamp analysis establishes a role for an auxin-binding protein in the auxin stimulation of plasma-membrane current in Zea-mays protoplasts. Plant J. 1993, 4, 41–46. [Google Scholar]
- Ivashikina, N.; Deeken, R.; Fischer, S.; Ache, P.; Hedrich, R. AKT2/3 subunits render guard cell K+ Channels Ca2+ sensitive. J. Gen. Physiol. 2005, 125, 483–492. [Google Scholar] [CrossRef]
- Cheong, Y.H.; Pandey, G.K.; Grant, J.J.; Batistic, O.; Li, L.; Kim, B.G.; Lee, S.C.; Kudla, J.; Luan, S. Two calcineurin b-like calcium sensors, interacting with protein kinase CIPK23, regulate leaf transpiration and root potassium uptake in Arabidopsis. Plant J. 2007, 52, 223–239. [Google Scholar] [CrossRef]
- Lan, W.Z.; Lee, S.C.; Che, Y.F.; Jiang, Y.Q.; Luan, S. Mechanistic analysis of AKT1 regulation by the CBL-CIPK-PP2CA interactions. Mol. Plant 2011, 4, 527–536. [Google Scholar] [CrossRef]
- Ren, X.L.; Qi, G.N.; Feng, H.Q.; Zhao, S.; Zhao, S.S.; Wang, Y.; Wu, W.H. Calcineurin B-like protein CBL10 directly interacts with AKT1 and modulates K+ homeostasis in Arabidopsis. Plant J. 2013, 74, 258–266. [Google Scholar] [CrossRef]
- Li, L.; Kim, B.G.; Cheong, Y.H.; Pandey, G.K.; Luan, S. A Ca2+ signaling pathway regulates a K+ channel for low-K response in Arabidopsis. Proc. Natl. Acad. Sci. USA 2006, 103, 12625–12630. [Google Scholar]
- Li, J.; Lee, Y.R.; Assmann, S.M. Guard cells possess a calcium-dependent protein kinase that phosphorylates the KAT1 potassium channel. Plant Physiol. 1998, 116, 785–795. [Google Scholar] [CrossRef]
- Vicente-Agullo, F.; Rigas, S.; Desbrosses, G.; Dolan, L.; Hatzopoulos, P.; Grabov, A. Potassium carrier TRH1 is required for auxin transport in Arabidopsis roots. Plant J. 2004, 40, 523–535. [Google Scholar] [CrossRef]
- Rigas, S.; Ditengou, F.A.; Ljung, K.; Daras, G.; Tietz, O.; Palme, K.; Hatzopoulos, P. Root gravitropism and root hair development constitute coupled developmental responses regulated by auxin homeostasis in the arabidopsis root apex. New Phytol. 2013, 197, 1130–1141. [Google Scholar] [CrossRef]
- Remy, E.; Cabrito, T.R.; Baster, P.; Batista, R.A.; Teixeira, M.C.; Friml, J.; Sa-Correia, I.; Duque, P. A major facilitator superfamily transporter plays a dual role in polar auxin transport and drought stress tolerance in Arabidopsis. Plant Cell 2013, 25, 901–926. [Google Scholar] [CrossRef] [Green Version]
- Tripathi, V.; Parasuraman, B.; Laxmi, A.; Chattopadhyay, D. CIPK6, A CBL-interacting protein kinase is required for development and salt tolerance in plants. Plant J. 2009, 58, 778–790. [Google Scholar] [CrossRef]
- Held, K.; Pascaud, F.; Eckert, C.; Gajdanowicz, P.; Hashimoto, K.; Corratge-Faillie, C.; Offenborn, J.N.; Lacombe, B.; Dreyer, I.; Thibaud, J.B.; et al. Calcium-dependent modulation and plasma membrane targeting of the AKT2 potassium channel by the CBL4/CIPK6 calcium sensor/protein kinase complex. Cell Res. 2011, 21, 1116–1130. [Google Scholar] [CrossRef]
- Darwin, C.; Darwin, F. The Power of Movement in Plants; Appleton and Co.: New York, NY, USA, 1881. [Google Scholar]
- Marchant, A.; Bhalerao, R.; Casimiro, I.; Eklof, J.; Casero, P.J.; Bennett, M.; Sandberg, G. AUX1 promotes lateral root formation by facilitating indole-3-acetic acid distribution between sink and source tissues in the Arabidopsis Seedling. Plant Cell 2002, 14, 589–597. [Google Scholar] [CrossRef]
- Tang, P.M.; Dela Fuente, R.K. The Transport of indole-3-acetic acid in boron- and calcium-deficient sunflower hypocotyl segments. Plant Physiol. 1986, 81, 646–650. [Google Scholar]
- Tang, P.M.; Dela Fuente, R.K. Boron and calcium sites involved in indole-3-acetic acid transport in sunflower hypocotyl segments. Plant Physiol. 1986, 81, 651–655. [Google Scholar] [CrossRef]
- Dela Fuente, R.K.; Leopold, A.C. Role for calcium in auxin transport. Plant Physiol. 1973, 51, 845–847. [Google Scholar] [CrossRef]
- Evans, M.L.; Young, L.M.; Hasenstein, K.H. The role of calcium in the regulation of hormone transport in gravistimulated Roots. Adv. Space Res. 1992, 12, 211–218. [Google Scholar]
- Migliaccio, F.; Galston, A.W. On the role of calcium in indole-3-acetic acid movement and graviresponse in etiolated pea epicotyls. Plant Growth Regul. 1989, 8, 335–347. [Google Scholar] [CrossRef]
- Raven, J.A. Transport of indole-acetic-acid in plant cells in relation to pH and electrical potential gradients, and its significance for polar IAA Transport. New Phytol. 1975, 74, 163–172. [Google Scholar] [CrossRef]
- Rubery, P.H.; Sheldrake, A.R. Carrier-mediated auxin transport. Planta 1974, 118, 101–121. [Google Scholar] [CrossRef]
- rubery, P.H.; sheldrake, A.R. Effect of pH and surface charge on cell uptake of auxin. Nature 1973, 244, 285–288. [Google Scholar] [CrossRef]
- Goldsmith, M.H.; Goldsmith, T.H.; Martin, M.H. Mathematical analysis of the chemosmotic polar diffusion of auxin through plant tissues. Proc. Natl. Acad. Sci. USA 1981, 78, 976–980. [Google Scholar] [CrossRef]
- Peret, B.; Swarup, K.; Ferguson, A.; Seth, M.; Yang, Y.; Dhondt, S.; James, N.; Casimiro, I.; Perry, P.; Syed, A.; et al. AUX/LAX genes encode a family of auxin influx transporters that perform distinct functions during arabidopsis development. Plant Cell 2012, 24, 2874–2885. [Google Scholar] [CrossRef] [Green Version]
- Bennett, M.J.; Marchant, A.; Green, H.G.; May, S.T.; Ward, S.P.; Millner, P.A.; Walker, A.R.; Schulz, B.; Feldmann, K.A. Arabidopsis AUX1 gene: A permease-like regulator of root gravitropism. Science 1996, 273, 948–950. [Google Scholar]
- Kubes, M.; Yang, H.; Richter, G.L.; Cheng, Y.; Mlodzinska, E.; Wang, X.; Blakeslee, J.J.; Carraro, N.; Petrasek, J.; Zazimalova, E.; et al. The Arabidopsis concentration-dependent influx/efflux transporter abcb4 regulates cellular auxin levels in the root epidermis. Plant J. 2012, 69, 640–654. [Google Scholar] [CrossRef]
- Krouk, G.; Lacombe, B.; Bielach, A.; Perrine-Walker, F.; Malinska, K.; Mounier, E.; Hoyerova, K.; Tillard, P.; Leon, S.; Ljung, K.; et al. Nitrate-regulated auxin transport by Nrt1.1 defines a mechanism for nutrient sensing in plants. Dev. Cell 2010, 18, 927–937. [Google Scholar] [CrossRef]
- Yang, Y.; Hammes, U.Z.; Taylor, C.G.; Schachtman, D.P.; Nielsen, E. High-affinity auxin transport by the AUX1 influx carrier protein. Curr. Biol. 2006, 16, 1123–1127. [Google Scholar] [CrossRef]
- Swarup, R.; Kramer, E.M.; Perry, P.; Knox, K.; Leyser, H.M.; Haseloff, J.; Beemster, G.T.; Bhalerao, R.; Bennett, M.J. Root gravitropism requires lateral root cap and epidermal cells for transport and response to a mobile auxin signal. Nat. Cell Biol. 2005, 7, 1057–1065. [Google Scholar]
- Swarup, K.; Benkova, E.; Swarup, R.; Casimiro, I.; Peret, B.; Yang, Y.; Parry, G.; Nielsen, E.; de Smet, I.; Vanneste, S.; et al. The auxin influx carrier Lax3 promotes lateral root emergence. Nat. Cell Biol. 2008, 10, 946–954. [Google Scholar] [CrossRef] [Green Version]
- Bainbridge, K.; Guyomarc’h, S.; Bayer, E.; Swarup, R.; Bennett, M.; Mandel, T.; Kuhlemeier, C. Auxin influx carriers stabilize phyllotactic patterning. Genes Dev. 2008, 22, 810–823. [Google Scholar] [CrossRef]
- Cheng, N.H.; Pittman, J.K.; Barkla, B.J.; Shigaki, T.; Hirschi, K.D. The Arabidopsis Cax1 mutant exhibits impaired ion homeostasis, development, and hormonal responses and reveals interplay among vacuolar transporters. Plant Cell 2003, 15, 347–364. [Google Scholar] [CrossRef]
- Pickett, F.B.; Wilson, A.K.; Estelle, M. The Aux1 mutation of Arabidopsis confers both auxin and ethylene resistance. Plant Physiol. 1990, 94, 1462–1466. [Google Scholar] [CrossRef]
- Kamimoto, Y.; Terasaka, K.; Hamamoto, M.; Takanashi, K.; Fukuda, S.; Shitan, N.; Sugiyama, A.; Suzuki, H.; Shibata, D.; Wang, B.; et al. Arabidopsis ABCB21 is a facultative auxin importer/exporter regulated by cytoplasmic auxin concentration. Plant Cell Physiol. 2012, 53, 2090–2100. [Google Scholar] [CrossRef]
- Ho, C.H.; Lin, S.H.; Hu, H.C.; Tsay, Y.F. CHL1 functions as a nitrate sensor in plants. Cell 2009, 138, 1184–1194. [Google Scholar] [CrossRef]
- Petrasek, J.; Mravec, J.; Bouchard, R.; Blakeslee, J.J.; Abas, M.; Seifertova, D.; Wisniewska, J.; Tadele, Z.; Kubes, M.; Covanova, M.; et al. PIN proteins perform a rate-limiting function in cellular auxin efflux. Science 2006, 312, 914–918. [Google Scholar] [CrossRef]
- Yang, H.; Murphy, A.S. Functional expression and characterization of Arabidopsis ABCB, AUX 1 and PIN auxin transporters in Schizosaccharomyces pombe. Plant J. 2009, 59, 179–191. [Google Scholar]
- Blakeslee, J.J.; Bandyopadhyay, A.; Lee, O.R.; Mravec, J.; Titapiwatanakun, B.; Sauer, M.; Makam, S.N.; Cheng, Y.; Bouchard, R.; Adamec, J.; et al. Interactions among PIN-formed and P-glycoprotein auxin transporters in Arabidopsis. Plant Cell 2007, 19, 131–147. [Google Scholar] [CrossRef]
- Mravec, J.; Kubes, M.; Bielach, A.; Gaykova, V.; Petrasek, J.; Skupa, P.; Chand, S.; Benkova, E.; Zazimalova, E.; Friml, J. Interaction of PIN and PGP transport mechanisms in auxin distribution-dependent development. Development 2008, 135, 3345–3354. [Google Scholar] [CrossRef]
- Henrichs, S.; Wang, B.; Fukao, Y.; Zhu, J.; Charrier, L.; Bailly, A.; Oehring, S.C.; Linnert, M.; Weiwad, M.; Endler, A.; et al. Regulation of ABCB1/PGP1-catalysed auxin transport by linker phosphorylation. EMBO J. 2012, 31, 2965–2980. [Google Scholar]
- Titapiwatanakun, B.; Murphy, A.S. Post-transcriptional regulation of auxin transport proteins: Cellular trafficking, protein phosphorylation, protein maturation, ubiquitination, and membrane composition. J. Exp. Bot. 2009, 60, 1093–1107. [Google Scholar] [CrossRef]
- Cho, M.; Lee, S.H.; Cho, H.T. P-glycoprotein4 displays auxin efflux transporter-like action in Arabidopsis root hair cells and tobacco cells. Plant Cell 2007, 19, 3930–3943. [Google Scholar] [CrossRef]
- Geisler, M.; Blakeslee, J.J.; Bouchard, R.; Lee, O.R.; Vincenzetti, V.; Bandyopadhyay, A.; Titapiwatanakun, B.; Peer, W.A.; Bailly, A.; Richards, E.L.; et al. Cellular efflux of auxin catalyzed by the arabidopsis MDR/PGP transporter AtPGP1. Plant J. 2005, 44, 179–194. [Google Scholar] [CrossRef]
- Bouchard, R.; Bailly, A.; Blakeslee, J.J.; Oehring, S.C.; Vincenzetti, V.; Lee, O.R.; Paponov, I.; Palme, K.; Mancuso, S.; Murphy, A.S.; et al. Immunophilin-like twisted DWARF1 modulates auxin efflux activities of Arabidopsis P-glycoproteins. J. Biol. Chem. 2006, 281, 30603–30612. [Google Scholar] [CrossRef]
- Wang, B.; Bailly, A.; Zwiewka, M.; Henrichs, S.; Azzarello, E.; Mancuso, S.; Maeshima, M.; Friml, J.; Schulz, A.; Geisler, M. Arabidopsis TWISTED DWARF1 functionally interacts with auxin exporter ABCB1 on the root plasma membrane. Plant Cell 2013, 25, 202–214. [Google Scholar] [CrossRef] [Green Version]
- Willige, B.C.; Ahlers, S.; Zourelidou, M.; Barbosa, I.C.; Demarsy, E.; Trevisan, M.; Davis, P.A.; Roelfsema, M.R.; Hangarter, R.; Fankhauser, C.; et al. D6PK AGCVIII kinases are required for auxin transport and phototropic hypocotyl bending in Arabidopsis. Plant Cell 2013, 25, 1674–1688. [Google Scholar] [CrossRef]
- Benjamins, R.; Ampudia, C.S.; Hooykaas, P.J.; Offringa, R. Pinoid-mediated signaling involves calcium-binding proteins. Plant Physiol. 2003, 132, 1623–1630. [Google Scholar] [CrossRef]
- Yang, T.; Poovaiah, B.W. Molecular and biochemical evidence for the involvement of calcium/calmodulin in auxin action. J. Biol. Chem. 2000, 275, 3137–3143. [Google Scholar] [CrossRef]
- Spartz, A.K.; Lee, S.H.; Wenger, J.P.; Gonzalez, N.; Itoh, H.; Inze, D.; Peer, W.A.; Murphy, A.S.; Overvoorde, P.J.; Gray, W.M. The SAUR19 subfamily of small auxin up RNA genes promote cell expansion. Plant J. 2012, 70, 978–990. [Google Scholar] [CrossRef]
- Dela Fuente, R.K. Role Of calcium in the polar secretion of indoleacetic acid. Plant Physiol. 1984, 76, 342–346. [Google Scholar] [CrossRef]
- Grunewald, W.; Friml, J. The march of the pins: Developmental plasticity by dynamic polar targeting in plant cells. EMBO J. 2010, 29, 2700–2714. [Google Scholar] [CrossRef]
- Cousin, M.A. Synaptic vesicle endocytosis: Calcium works overtime in the nerve terminal. Mol. Neurobiol. 2000, 22, 115–128. [Google Scholar] [CrossRef]
- Dolensek, J.; Skelin, M.; Rupnik, M.S. Calcium dependencies of regulated exocytosis in different endocrine cells. Physiol. Res. 2011, 60, S29–S38. [Google Scholar]
- Chrispeels, M.J.; Varner, J.E. Gibberellic acid-enhanced synthesis and release of alpha-amylase and ribonuclease by isolated barley and aleurone layers. Plant Physiol. 1967, 42, 398–406. [Google Scholar] [CrossRef]
- Bush, D.S.; Cornejo, M.J.; Huang, C.N.; Jones, R.L. Ca-stimulated secretion of alpha-amylase during development in barley aleurone protoplasts. Plant Physiol. 1986, 82, 566–574. [Google Scholar] [CrossRef]
- Sticher, L.; Penel, C.; Greppin, H. Calcium requirement for the secretion of peroxidases by plant cell suspensions. J. Cell Sci. 1981, 48, 345–353. [Google Scholar]
- Castillo, F.J.; Penel, C.; Greppin, H. Peroxidase release induced by ozone in sedum album leaves: Involvement of Ca. Plant Physiol. 1984, 74, 846–851. [Google Scholar] [CrossRef]
- Campanoni, P.; Blatt, M.R. Membrane trafficking and polar growth in root hairs and pollen tubes. J. Exp. Bot. 2007, 58, 65–74. [Google Scholar] [CrossRef]
- Coelho, S.M.B.; Brownlee, C.; Bothwell, J.H.F. A Tip-high, Ca2+-interdependent, reactive oxygen species gradient is associated with polarized growth in fucus serratus zygotes. Planta 2008, 227, 1037–1046. [Google Scholar] [CrossRef]
- Kroeger, J.; Geitmann, A. The pollen tube paradigm revisited. Curr. Opin. Plant Biol. 2012, 15, 618–624. [Google Scholar] [CrossRef]
- Samaj, J.; Muller, J.; Beck, M.; Bohm, N.; Menzel, D. Vesicular trafficking, cytoskeleton and signalling in root hairs and pollen tubes. Trends Plant Sci. 2006, 11, 594–600. [Google Scholar] [CrossRef]
- Homann, U.; Tester, M. Ca2+-independent and Ca2+/GTP-binding protein-controlled exocytosis in a plant cell. Proc. Natl. Acad. Sci. USA 1997, 94, 6565–6570. [Google Scholar]
- Tester, M.; Zorec, R. Cytoplasmic calcium stimulates exocytosis in a plant secretory cell. Biophys. J. 1992, 63, 864–867. [Google Scholar] [CrossRef]
- Sutter, J.U.; Homann, U.; Thiel, G. Ca2+-stimulated exocytosis in maize coleoptile cells. Plant Cell 2000, 12, 1127–1136. [Google Scholar]
- Thiel, G.; Sutter, J.U.; Homann, U. Ca2+-sensitive and Ca2+-Insensitive exocytosis in maize coleoptile protoplasts. Pflugers Arch. 2000, 439, R152–R153. [Google Scholar] [CrossRef]
- Carroll, A.D.; Moyen, C.; van Kesteren, P.; Tooke, F.; Battey, N.H.; Brownlee, C. Ca2+, annexins, and gtp modulate exocytosis from maize root cap protoplast. Plant Cell 1998, 10, 1267–1276. [Google Scholar]
- Sutter, J.U.; Denecke, J.; Thiel, G. Synthesis of vesicle cargo determines amplitude of Ca2+-sensitive exocytosis. Cell Calcium 2012, 52, 283–288. [Google Scholar] [CrossRef]
- Jones, R.L.; Carbonell, J. Regulation of the synthesis of barley aleurone alpha-amylase by gibberellic acid and calcium Ions. Plant Physiol. 1984, 76, 213–218. [Google Scholar] [CrossRef]
- Kleine-Vehn, J.; Wabnik, K.; Martiniere, A.; Langowski, L.; Willig, K.; Naramoto, S.; Leitner, J.; Tanaka, H.; Jakobs, S.; Robert, S.; et al. Recycling, clustering, and endocytosis jointly maintain PIN auxin carrier polarity at the plasma membrane. Mol. Syst. Biol. 2011, 7. [Google Scholar] [CrossRef]
- Camacho, L.; Malho, R. Endo/exocytosis in the pollen tube apex is differentially regulated by Ca2+ and GTPases. J. Exp. Bot. 2003, 54, 83–92. [Google Scholar] [CrossRef]
- Cole, R.A.; Fowler, J.E. Polarized growth: Maintaining focus on the tip. Curr. Opin. Plant Biol. 2006, 9, 579–588. [Google Scholar] [CrossRef]
- Lee, Y.J.; Szumlanski, A.; Nielsen, E.; Yang, Z. Rho-GTPase-dependent filamentous actin dynamics coordinate vesicle targeting and exocytosis during tip growth. J. Cell Biol. 2008, 181, 1155–1168. [Google Scholar] [CrossRef]
- Xiang, Y.; Huang, X.; Wang, T.; Zhang, Y.; Liu, Q.; Hussey, P.J.; Ren, H. Actin binding protein 29 from lilium pollen plays an important role in dynamic actin remodeling. Plant Cell 2007, 19, 1930–1946. [Google Scholar] [CrossRef]
- Ketelaar, T.; Galway, M.E.; Mulder, B.M.; Emons, A.M. Rates of exocytosis and endocytosis in arabidopsis root hairs and pollen tubes. J. Microsc. 2008, 231, 265–273. [Google Scholar] [CrossRef]
- Derksen, J.; Rutten, T.; Lichtscheidl, I.K.; Dewin, A.H.N.; Pierson, E.S.; Rongen, G. Quantitative-analysis of the distribution of organelles in tobacco pollen tubes—Implications for exocytosis and endocytosis. Protoplasma 1995, 188, 267–276. [Google Scholar] [CrossRef]
- Lisboa, S.; Scherer, G.E.; Quader, H. Localized endocytosis in tobacco pollen tubes: Visualisation and dynamics of membrane retrieval by a fluorescent phospholipid. Plant Cell Rep. 2008, 27, 21–28. [Google Scholar] [CrossRef]
- Moscatelli, A.; Ciampolini, F.; Rodighiero, S.; Onelli, E.; Cresti, M.; Santo, N.; Idilli, A. Distinct endocytic pathways identified in tobacco pollen tubes using charged nanogold. J. Cell Sci. 2007, 120, 3804–3819. [Google Scholar] [CrossRef]
- Moscatelli, A.; Idilli, A.I.; Rodighiero, S.; Caccianiga, M. Inhibition of actin polymerisation by low concentration latrunculin B affects endocytosis and alters exocytosis in shank and tip of tobacco pollen tubes. Plant Biol. 2012, 14, 770–782. [Google Scholar] [CrossRef]
- Zonia, L.; Munnik, T. Vesicle trafficking dynamics and visualization of zones of exocytosis and endocytosis in tobacco pollen tubes. J. Exp. Bot. 2008, 59, 861–873. [Google Scholar] [CrossRef]
- Wabnik, K.; Govaerts, W.; Friml, J.; Kleine-Vehn, J. Feedback models for polarized auxin transport: An emerging trend. Mol. Biosyst. 2011, 7, 2352–2359. [Google Scholar] [CrossRef]
- Wu, Y.; Zhao, S.; Tian, H.; He, Y.; Xiong, W.; Guo, L. CPK3-phosphorylated RhoGDI1 is essential in the development of Arabidopsis seedlings and leaf epidermal cells. J. Exp. Bot. 2013, 64, 3327–3338. [Google Scholar] [CrossRef]
- Lee, Y.J.; Yang, Z. Tip growth: Signaling in the apical dome. Curr. Opin. Plant Biol. 2008, 11, 662–671. [Google Scholar] [CrossRef]
- Marhavy, P.; Bielach, A.; Abas, L.; Abuzeineh, A.; Duclercq, J.; Tanaka, H.; Parezova, M.; Petrasek, J.; Friml, J.; Kleine-Vehn, J.; et al. Cytokinin modulates endocytic trafficking of PIN1 auxin efflux carrier to control plant organogenesis. Dev. Cell 2011, 21, 796–804. [Google Scholar] [CrossRef]
- Shinohara, N.; Taylor, C.; Leyser, O. Strigolactone can promote or inhibit shoot branching by triggering rapid depletion of the auxin efflux protein PIN1 from the plasma membrane. PLoS Biol. 2013, 11, e1001474. [Google Scholar] [CrossRef]
- Löfke, C.; Zwiewka, M.; Heilmann, I.; van Montagu, M.C.; Teichmann, T.; Friml, J. Asymmetric gibberellin signaling regulates vacuolar trafficking of PIN auxin transporters during root gravitropism. Proc. Natl. Acad. Sci. USA 2013, 110, 3627–3632. [Google Scholar]
- Willige, B.C.; Isono, E.; Richter, R.; Zourelidou, M.; Schwechheimer, C. Gibberellin regulates PIN-formed abundance and is required for auxin transport-dependent growth and development in Arabidopsis thaliana. Plant Cell 2011, 23, 2184–2195. [Google Scholar] [CrossRef]
- Du, Y.; Tejos, R.; Beck, M.; Himschoot, E.; Li, H.; Robatzek, S.; Vanneste, S.; Friml, J. Salicylic acid interferes with clathrin-mediated endocytic protein trafficking. Proc. Natl. Acad. Sci. USA 2013, 110, 7946–7951. [Google Scholar] [CrossRef]
- Feraru, E.; Friml, J. PIN polar targeting. Plant Physiol. 2008, 147, 1553–1559. [Google Scholar] [CrossRef] [Green Version]
- Wisniewska, J.; Xu, J.; Seifertova, D.; Brewer, P.B.; Ruzicka, K.; Blilou, I.; Rouquie, D.; Benkova, E.; Scheres, B.; Friml, J. Polar PIN localization directs auxin flow in plants. Science 2006, 312. [Google Scholar] [CrossRef]
- Friml, J.; Vieten, A.; Sauer, M.; Weijers, D.; Schwarz, H.; Hamann, T.; Offringa, R.; Jurgens, G. Efflux-dependent auxin gradients establish the apical-basal axis of Arabidopsis. Nature 2003, 426, 147–153. [Google Scholar] [CrossRef]
- Rakusova, H.; Gallego-Bartolome, J.; Vanstraelen, M.; Robert, H.S.; Alabadi, D.; Blazquez, M.A.; Benkova, E.; Friml, J. Polarization of PIN3-dependent auxin transport for hypocotyl gravitropic response in Arabidopsis thaliana. Plant J. 2011, 67, 817–826. [Google Scholar] [CrossRef]
- Sauer, M.; Balla, J.; Luschnig, C.; Wisniewska, J.; Reinohl, V.; Friml, J.; Benkova, E. Canalization of auxin flow by AUX/IAA-ARF-dependent feedback regulation of PIN polarity. Genes Dev. 2006, 20, 2902–2911. [Google Scholar] [CrossRef]
- Huang, F.; Zago, M.K.; Abas, L.; van Marion, A.; Galvan-Ampudia, C.S.; Offringa, R. Phosphorylation of conserved PIN motifs directs arabidopsis pin1 polarity and auxin transport. Plant Cell 2010, 22, 1129–1142. [Google Scholar] [CrossRef]
- Zhang, J.; Nodzynski, T.; Pencik, A.; Rolcik, J.; Friml, J. PIN Phosphorylation is sufficient to mediate pin polarity and direct auxin transport. Proc. Natl. Acad. Sci. USA 2010, 107, 918–922. [Google Scholar] [CrossRef]
- Friml, J.; Yang, X.; Michniewicz, M.; Weijers, D.; Quint, A.; Tietz, O.; Benjamins, R.; Ouwerkerk, P.B.; Ljung, K.; Sandberg, G.; et al. A pinoid-dependent binary switch in apical-basal PIN polar targeting directs auxin efflux. Science 2004, 306, 862–865. [Google Scholar] [CrossRef]
- Michniewicz, M.; Zago, M.K.; Abas, L.; Weijers, D.; Schweighofer, A.; Meskiene, I.; Heisler, M.G.; Ohno, C.; Zhang, J.; Huang, F.; et al. Antagonistic regulation of pin phosphorylation by PP2A and PINOID directs auxin flux. Cell 2007, 130, 1044–1056. [Google Scholar] [CrossRef]
- Kleine-Vehn, J.; Huang, F.; Naramoto, S.; Zhang, J.; Michniewicz, M.; Offringa, R.; Friml, J. PIN auxin efflux carrier polarity is regulated by pinoid kinase-mediated recruitment into GNOM-independent trafficking in Arabidopsis. Plant Cell 2009, 21, 3839–3849. [Google Scholar] [CrossRef] [Green Version]
- Babourina, O.; Newman, I.; Shabala, S. Blue light-induced kinetics of H+ and Ca2+ fluxes in etiolated wild-type and phototropin-mutant arabidopsis seedlings. Proc. Natl. Acad. Sci. USA 2002, 99, 2433–2438. [Google Scholar] [CrossRef]
- Chen, X.; Lin, W.H.; Wang, Y.; Luan, S.; Xue, H.W. An inositol polyphosphate 5-phosphatase functions in phototropin1 signaling in Arabidopis by altering cytosolic Ca2+. Plant Cell 2008, 20, 353–366. [Google Scholar] [CrossRef]
- Perera, I.Y.; Hung, C.Y.; Brady, S.; Muday, G.K.; Boss, W.F. A universal role for inositol 1,4,5-trisphosphate-mediated signaling in plant gravitropism. Plant Physiol. 2006, 140, 746–760. [Google Scholar] [CrossRef]
- Zhang, J.; Vanneste, S.; Brewer, P.B.; Michniewicz, M.; Grones, P.; Kleine-Vehn, J.; Lofke, C.; Teichmann, T.; Bielach, A.; Cannoot, B.; et al. Inositol trisphosphate-induced Ca2+ signaling modulates auxin transport and PIN polarity. Dev. Cell 2011, 20, 855–866. [Google Scholar] [CrossRef]
- Gehring, C.A.; Williams, D.A.; Cody, S.H.; Parish, R.W. Phototropism and geotropism in maize coleoptiles are spatially correlated with increases in cytosolic free calcium. Nature 1990, 345, 528–530. [Google Scholar] [CrossRef]
- Plieth, C.; Trewavas, A.J. Reorientation of seedlings in the earth’s gravitational field induces cytosolic calcium transients. Plant Physiol. 2002, 129, 786–796. [Google Scholar] [CrossRef]
- Toyota, M.; Furuichi, T.; Tatsumi, H.; Sokabe, M. Cytoplasmic calcium increases in response to changes in the gravity vector in hypocotyls and petioles of Arabidopsis seedlings. Plant Physiol. 2008, 146, 505–514. [Google Scholar]
- Band, L.R.; Wells, D.M.; Larrieu, A.; Sun, J.; Middleton, A.M.; French, A.P.; Brunoud, G.; Sato, E.M.; Wilson, M.H.; Peret, B.; et al. Root gravitropism is regulated by a transient lateral auxin gradient controlled by a tipping-point mechanism. Proc. Natl. Acad. Sci. USA 2012, 109, 4668–4673. [Google Scholar]
- Lee, J.S.; Mulkey, T.J.; Evans, M.L. Inhibition of polar calcium movement and gravitropism in roots treated with auxin-transport inhibitors. Planta 1984, 160, 536–543. [Google Scholar] [CrossRef]
- Lee, J.S.; Mulkey, T.J.; Evans, M.L. Gravity-induced polar transport of calcium across root tips of maize. Plant Physiol. 1983, 73, 874–876. [Google Scholar] [CrossRef]
- Bjorkman, T.; Leopold, A.C. Effect of inhibitors of auxin transport and of calmodulin on a gravisensing-dependent current in maize roots. Plant Physiol. 1987, 84, 847–850. [Google Scholar] [CrossRef]
- Lee, J.S.; Mulkey, T.J.; Evans, M.L. Reversible loss of gravitropic sensitivity in maize roots after tip application of calcium chelators. Science 1983, 220, 1375–1376. [Google Scholar]
- Friedman, H.; Meir, S.; Rosenberger, I.; Halevy, A.H.; Kaufman, P.B.; Philosoph-Hadas, S. Inhibition of the gravitropic response of snapdragon spikes by the calcium-channel blocker lanthanum chloride. Plant Physiol. 1998, 118, 483–492. [Google Scholar] [CrossRef]
- Daye, S.; Biro, R.L.; Roux, S.J. Inhibition of gravitropism in oat coleoptiles by the calcium chelator, ethyleneglycol-bis-(beta-aminoethyl ether)-N,N'-tetraacetic acid. Plant Physiol. 1984, 61, 449–454. [Google Scholar] [CrossRef]
- Biro, R.L.; Hale, C.C.; Wiegand, O.F.; Roux, S.J. Effects of chlorpromazine on gravitropism in avena coleoptiles. Ann. Bot. 1982, 50, 737–745. [Google Scholar]
- Stinemetz, C.L.; Kuzmanoff, K.M.; Evans, M.L.; Jarrett, H.W. Correlation between calmodulin activity and gravitropic sensitivity in primary roots of maize. Plant Physiol. 1987, 84, 1337–1342. [Google Scholar] [CrossRef]
- Baldwin, K.L.; Strohm, A.K.; Masson, P.H. Gravity Sensing and signal transduction in vascular plant primary roots. Am. J. Bot. 2013, 100, 126–142. [Google Scholar] [CrossRef]
- Kleine-Vehn, J.; Ding, Z.; Jones, A.R.; Tasaka, M.; Morita, M.T.; Friml, J. Gravity-induced PIN transcytosis for polarization of auxin fluxes in gravity-sensing root cells. Proc. Natl. Acad. Sci. USA 2010, 107, 22344–22349. [Google Scholar]
- Ottenschlager, I.; Wolff, P.; Wolverton, C.; Bhalerao, R.P.; Sandberg, G.; Ishikawa, H.; Evans, M.; Palme, K. Gravity-regulated differential auxin transport from columella to lateral root cap cells. Proc. Natl. Acad. Sci. USA 2003, 100, 2987–2991. [Google Scholar]
- Luschnig, C.; Gaxiola, R.A.; Grisafi, P.; Fink, G.R. Eir1, a root-specific protein involved in auxin transport, is required for gravitropism in Arabidopsis thaliana. Genes Dev. 1998, 12, 2175–2187. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Vanneste, S.; Friml, J. Calcium: The Missing Link in Auxin Action. Plants 2013, 2, 650-675. https://doi.org/10.3390/plants2040650
Vanneste S, Friml J. Calcium: The Missing Link in Auxin Action. Plants. 2013; 2(4):650-675. https://doi.org/10.3390/plants2040650
Chicago/Turabian StyleVanneste, Steffen, and Jiří Friml. 2013. "Calcium: The Missing Link in Auxin Action" Plants 2, no. 4: 650-675. https://doi.org/10.3390/plants2040650



