Verbesina alternifolia Tolerance to the Holoparasite Cuscuta gronovii and the Impact of Drought
Abstract
:1. Introduction
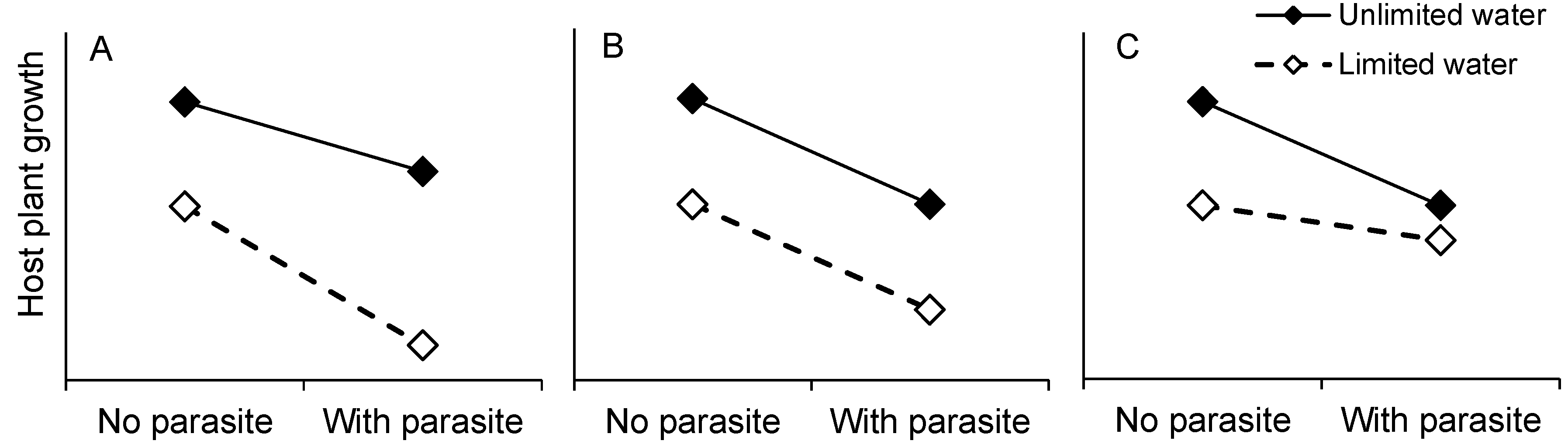
2. Results and Discussion
2.1. Host Growth
| A | Pillai’s trace | Standardized canonical coefficient | ||||||
| df | F | P | Shoot | Root | Shoot 2 | Root 2 | ||
| Leaf length | 2,161 | 18.78 | <0.0001 | 1.844 | 0.956 | - | - | |
| Water | 4,324 | 42.13 | <0.0001 | 2.935 | −0.463 | −1.520 | 2.836 | |
| Parasite | 2,161 | 352.01 | <0.0001 | 1.494 | 1.281 | - | - | |
| Parasite*Water | 4,324 | 19.84 | <0.0001 | 1.891 | 0.910 | −2.711 | 2.725 | |
| B | V. alternifolia shoot mass contrast | df | F | P | ||||
| Control vs. Pulsed | 1,162 | 52.29 | <0.0001 | |||||
| Control vs. Continuous | 1,162 | 69.78 | <0.0001 | |||||
| Pulsed & Continuous | 1,162 | 1.53 | 0.2172 | |||||
| V. alternifolia root mass contrast | df | F | P | |||||
| Control vs. Pulsed | 1,162 | 23.89 | <0.0001 | |||||
| Control vs. Continuous | 1,162 | 61.66 | <0.0001 | |||||
| Pulsed & Continuous | 1,162 | 9.39 | 0.0026 | |||||
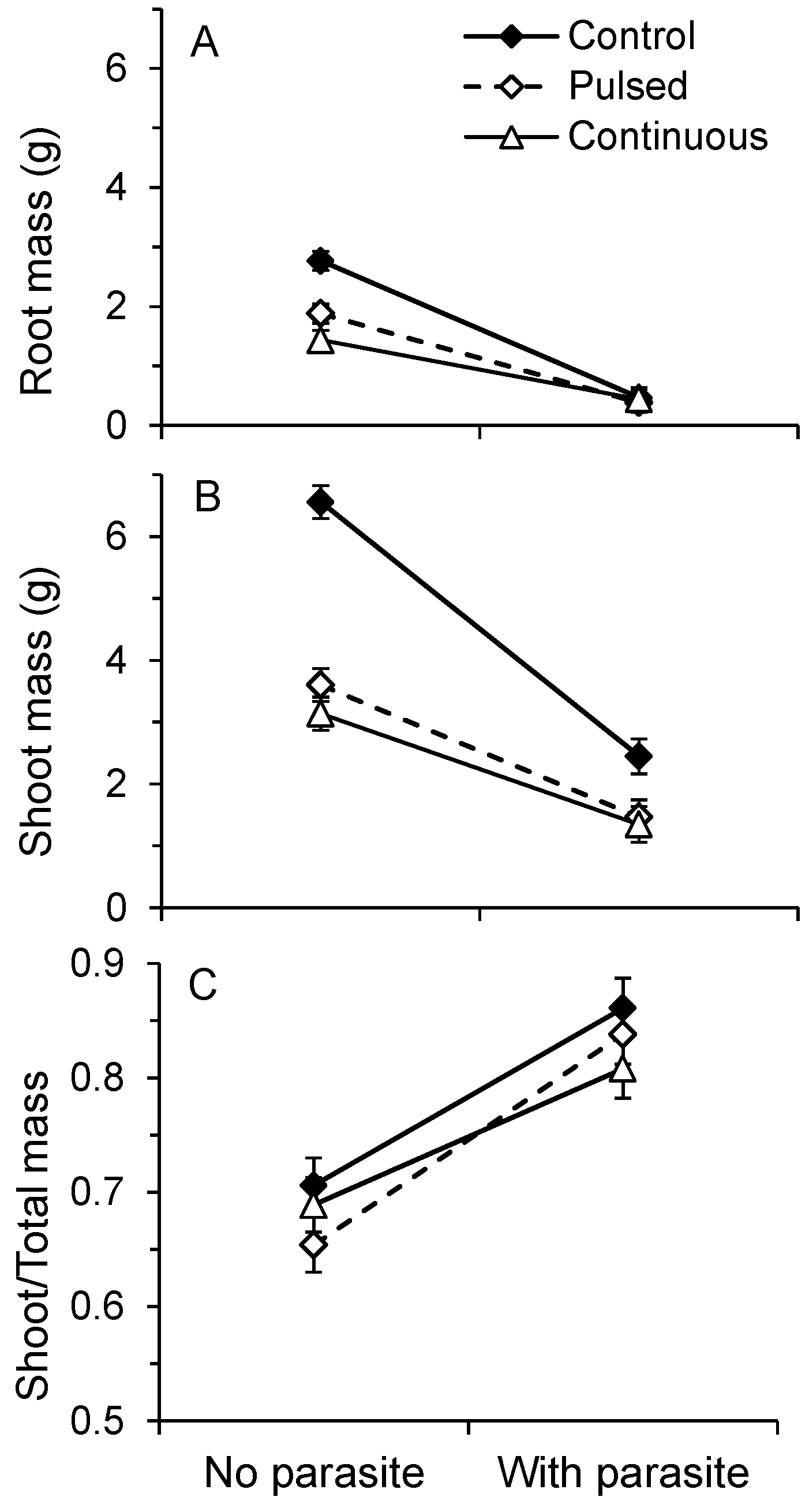
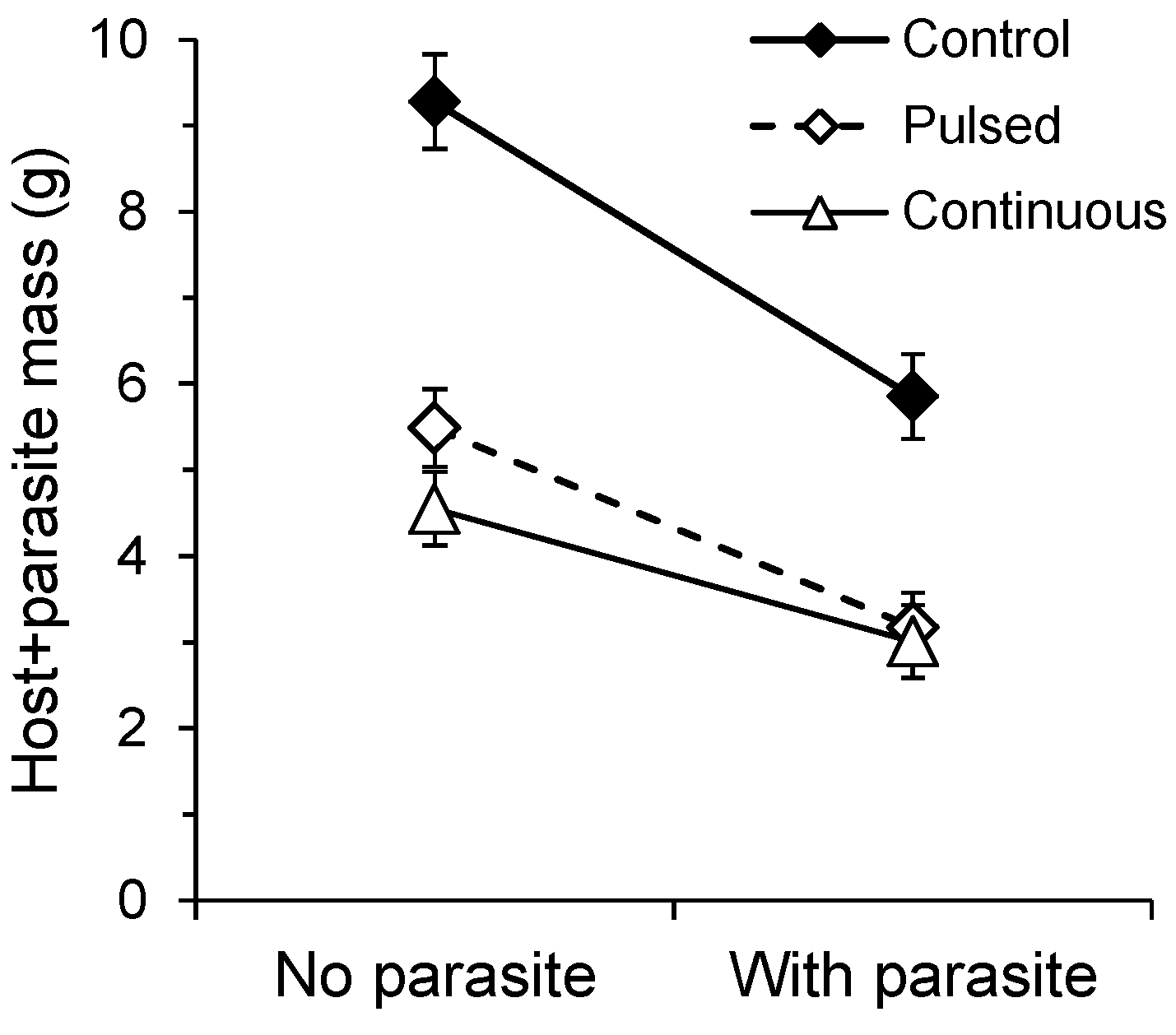
2.2. Pot Productivity
2.3. Requirements and Utility of the LRM for Holoparasite-Host Interactions
3. Experimental
3.1. Study Organisms
3.2. Experimental Procedure
3.2.1. Parasite Treatment
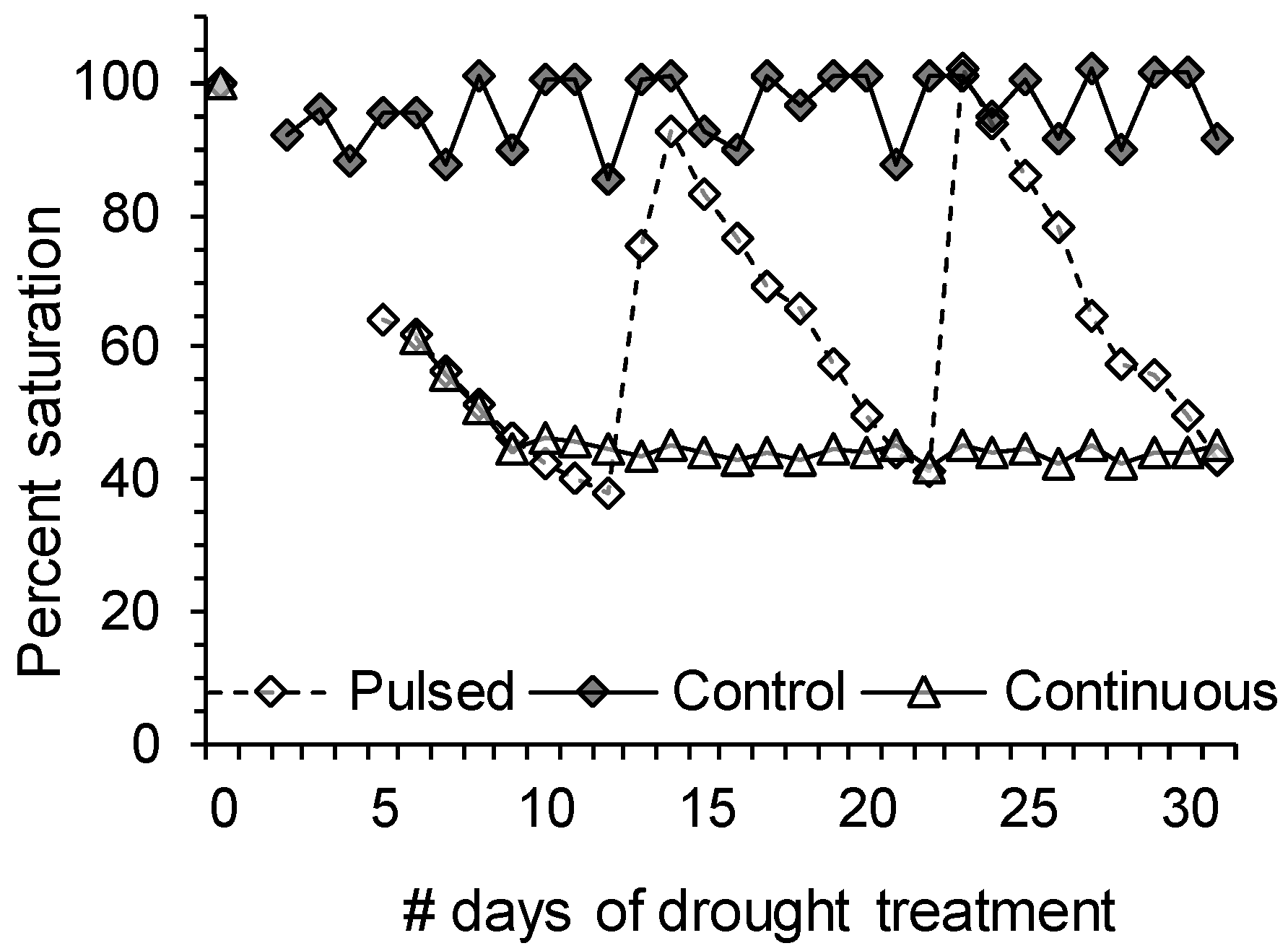
3.2.2. Drought Stress Treatment
3.2.3. Harvest
3.3. Statistical Analysis
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Watling, J.R.; Press, M.C. Impacts of infection by parasitic angiosperms on host photosynthesis. Plant Biol. 2001, 3, 244–250. [Google Scholar] [CrossRef]
- Bollard, E.G. Transport in the xylem. Annu. Rev. Plant Physiol. 1960, 11, 141–166. [Google Scholar] [CrossRef]
- Mooney, H.A. The carbon balance of plants. Annu. Rev. Ecol. Syst. 1972, 3, 315–346. [Google Scholar]
- Thompson, M.V. Phloem: The long and the short of it. Trends Plant Sci. 2006, 11, 26–32. [Google Scholar] [CrossRef]
- Wardlaw, I.F. The control and pattern of movement of carbohydrates in plants. Bot. Rev. 1968, 34, 79–105. [Google Scholar] [CrossRef]
- Hsiao, T.C. Plant responses to water stress. Annu. Rev. Plant Physiol. 1973, 24, 519–570. [Google Scholar] [CrossRef]
- Gutbrodt, B.; Mody, K.; Dorn, S. Drought changes plant chemistry and causes contrasting responses in lepidopteran herbivores. Oikos 2011, 120, 1732–1740. [Google Scholar] [CrossRef]
- Hawkes, C.V.; Sullivan, J.J. The impact of herbivory on plants in different resource conditions: A metaanalysis. Ecology 2001, 82, 2045–2058. [Google Scholar] [CrossRef]
- Joern, A.; Mole, S. The plant stress hypothesis and variable responses by blue grama grass (Bouteloua gracilis) to water, mineral nitrogen, and insect herbivory. J. Chem. Ecol. 2005, 31, 2069–2090. [Google Scholar] [CrossRef]
- Robinson, E.A.; Ryan, G.D.; Newman, J.A. A meta-analytical review of the effects of elevated CO2 on plant-arthropod interactions highlights the importance of interacting environmental and biological variables. New Phytol. 2012, 194, 321–336. [Google Scholar] [CrossRef]
- Sadras, V.O.; Wilson, L.J.; Lally, D.A. Water deficit enhanced cotton resistance to spider mite herbivory. Ann. Bot. 1998, 81, 273–286. [Google Scholar] [CrossRef]
- Sun, Y.; Ding, J.; Frye, M.J. Effects of resource availability on tolerance of herbivory in the invasive Alternanthera philoxeroides and the native Alternanthera sessilis. Weed Res. 2009, 50, 527–536. [Google Scholar]
- Marquardt, E.S.; Pennings, S.C. Constraints on host use by a parasitic plant. Oecologia 2010, 164, 177–184. [Google Scholar] [CrossRef]
- Pennings, S.C.; Callaway, R.M. Parasitic plants: Parallels and contrasts with herbivores. Oecologia 2002, 131, 479–489. [Google Scholar] [CrossRef]
- Núñez-Farfán, J.; Fornoni, J.; Valverde, P.L. The evolution of resistance and tolerance to herbivores. Annu. Rev. Evol. Syst. 2007, 38, 541–566. [Google Scholar] [CrossRef]
- Rosenthal, J.P.; Kotanen, P.M. Terrestrial plant tolerance to herbivory. Trends Ecol. Evol. 1994, 9, 145–148. [Google Scholar] [CrossRef]
- Strauss, S.Y.; Agrawal, A.A. The ecology and evolution of plant tolerance to herbivory. Trends Ecol. Evol. 1999, 14, 179–185. [Google Scholar] [CrossRef]
- Belsky, A.J. Does herbivory benefit plants? A review of the evidence. Am. Nat. 1986, 127, 870–892. [Google Scholar]
- Ferraro, D.O.; Oesterheld, M. Effect of defoliation on grass growth. A quantitative review. Oikos 2002, 98, 125–133. [Google Scholar] [CrossRef]
- Meyer, G.A.; Root, R.B. Effects of herbivorous insects and soil fertility on reproduction of goldenrod. Ecology 1993, 74, 1117–1128. [Google Scholar] [CrossRef]
- Rand, T.A. Competition, facilitation, and compensation for insect herbivory in an annual salt marsh forb. Ecology 2004, 85, 2046–2052. [Google Scholar] [CrossRef]
- Wise, M.J.; Abrahamson, W.G. Beyond the compensatory continuum: Environmental resource levels and plant tolerance of herbivory. Oikos 2005, 109, 417–428. [Google Scholar] [CrossRef]
- Hibberd, J.M.; Quick, W.P.; Press, M.C.; Scholes, J.D.; Jeschke, W.D. Solute fluxes from tobacco to the parasitic angiosperm Orobanche cernua and the influence of infection on host carbon and nitrogen relations. Plant Cell Environ. 1999, 22, 937–947. [Google Scholar] [CrossRef]
- Jeschke, W.D.; Baig, A.; Hilpert, A. Sink-stimulated photosynthesis, increased transpiration and increased demand dependent stimulation of nitrate uptake: Nitrogen and carbon relations in the parasitic association Cuscuta reflexa–Coleus blumei. J. Exp. Bot. 1997, 48, 915–925. [Google Scholar] [CrossRef]
- Allen, C.D.; Macalady, A.K.; Chenchouni, H.; Bachelet, D.; McDowell, N.; Vennetier, M.; Kitzberger, T.; Rigling, A.; Breshears, D.D.; Hogg, E.H.; et al. A global overview of drought and heat-induced tree mortality reveals emerging climate change risks for forests. For. Ecol. Manag. 2010, 259, 660–684. [Google Scholar] [CrossRef]
- Novoplansky, A.; Goldberg, D.E. Effects of water pulsing on individual performance and competitive hierarchies in plants. J. Veg. Sci. 2001, 12, 199–208. [Google Scholar] [CrossRef]
- Huberty, A.F.; Denno, R.F. Plant water stress and its consequences for herbivorous insects: A new synthesis. Ecology 2004, 85, 1383–1398. [Google Scholar] [CrossRef]
- Mody, K.; Eichenberger, D.; Dorn, S. Stress magnitude matters: Different intensities of pulsed water stress produce non-monotonic resistance responses of host plants to insect herbivores. Ecol. Entomol. 2009, 34, 133–143. [Google Scholar] [CrossRef]
- Xu, Z.; Zhou, G.; Shimizu, H. Plant responses to drought and rewatering. Plant Signal. Behav. 2010, 5, 649–654. [Google Scholar] [CrossRef]
- Wise, M.J.; Abrahamson, W.G. Effects of resource availability on tolerance of herbivory: A review and assessment of three opposing models. Am. Nat. 2007, 169, 443–454. [Google Scholar] [CrossRef]
- Kerkhoff, A.J.; Enquist, B.J. Multiplicative by nature: Why logarithmic transformation is necessary in allometry. J. Theor. Biol. 2009, 257, 519–521. [Google Scholar] [CrossRef]
- Mascaro, J.; Litton, C.M.; Hughes, R.F.; Uowolo, A.; Schnitzer, S.A. Minimizing bias in biomass allometry: Model selection and log-transformation of data. Biotropica 2011, 43, 649–653. [Google Scholar] [CrossRef]
- McConnaughay, K.D.M.; Coleman, J.S. Biomass allocation in plants: Ontogeny or optimality? A test along three resource gradients. Ecology 1999, 80, 2581–2593. [Google Scholar] [CrossRef]
- Shen, H.; Hong, L.; Ye, W.; Cao, H.; Wang, Z. The influence of the Holoparasitic plant Cuscuta campestris on the growth and photosynthesis of its host Mikania micrantha. J. Exp. Bot. 2007, 58, 2929–2937. [Google Scholar] [CrossRef]
- Jeschke, W.D.; Rath, N.; Baumel, P.; Czygan, F.C.; Proksch, P. Modeling the flow and partitioning of carbon and nitrogen in the holoparasite Cuscuta reflexa Roxb and its host Lupinus albus L. 1. Methods for estimating net flows. J. Exp. Bot. 1994, 45, 791–800. [Google Scholar] [CrossRef]
- Evans, B.A. Pattern of Drought and Host Plant-Holoparasite Relations: An Examination of Resistance and Tolerance. Master’s Thesis, Illinois State University, Normal, IL, USA, 2012. [Google Scholar]
- Chaves, M.M.; Maroco, J.P.; Pereira, J.S. Understanding plant responses to drought—From genes to the whole plant. Funct. Plant Biol. 2003, 30, 239–264. [Google Scholar] [CrossRef]
- Ghannoum, O. C4 photosynthesis and water stress. Ann. Bot. 2009, 103, 635–644. [Google Scholar] [CrossRef]
- Heschel, M.S.; Riginos, C. Mechanisms of selection for drought stress tolerance and avoidance in Impatiens capensis. Am. J. Bot. 2005, 92, 37–44. [Google Scholar] [CrossRef]
- Sletvold, N.; Agren, J. Variation in tolerance to drought among Sandinavian populations of Arabidopsis lyrata. Evol. Ecol. 2012, 26, 559–557. [Google Scholar] [CrossRef]
- Gleason, H.A.; Cronquist, A. Manual of Vascular Plants of Northeastern United States and Adjacent Canada, 2nd ed.; New York Botanical Garden: New York, NY, USA, 1991. [Google Scholar]
- Costea, M.; Tardif, F.J. The biology of Canadian weeds. 133. Cuscuta campestris Yuncker, C. gronovii Willd. Ex Schult., C. umbrosa Beyr. Ex Hook., C. epithymum (L.) L. and C. epilinum Weihe. Can. J. Plant Sci. 2006, 86, 293–316. [Google Scholar] [CrossRef]
- Sandler, H.A. Dodder—Cuscuta gronovii Willd; Cranberry Experiment Station, University of Massachusetts: Wareham, MA, USA, 2001. [Google Scholar]
- Parker, C.; Riches, C.R. Parasitic Weeds of the World. Biology and Control; CAB International: Wallingford, UK, 1993. [Google Scholar]
- Gaertner, E.E. Studies of seed germination, seed identification, and host relationships in dodders, Cuscuta spp. Cornell Exp. Stn. Mem. 1950, 294, 1–56. [Google Scholar]
- Devlin, R.M.; Deubert, K.H. Control of swamp dodder (Cuscuta gronovii) on cranberry bogs with butralin. Proc. Annu. Meet. Northeast. Weed Sci. Soc. 1980, 11, 112–113. [Google Scholar]
- Kelly, C.K. Resource choice in Cuscuta europaea. Proc. Natl. Acad. Sci. USA 1992, 89, 12194–12197. [Google Scholar] [CrossRef]
- Birschwilks, M.; Haupt, S.; Hofius, D.; Neumann, S. Transfer of phloem-mobile substances from the host plants to the holoparasite Cuscuta sp. J. Exp. Bot. 2006, 57, 911–921. [Google Scholar] [CrossRef]
- Vaughn, K.C. Conversion of the searching hyphae of dodder into xylic and phloic hyphae: A cytochemical and immunocytochemical investigation. Int. J. Plant Sci. 2006, 167, 1099–1114. [Google Scholar] [CrossRef]
- Hibberd, J.M.; Jeschke, D. Solute flux into parasitic plants. J. Exp. Bot. 2001, 52, 2043–2049. [Google Scholar] [CrossRef]
- Niinemets, Ü.; Portsmuth, A.; Tena, D.; Tobias, M.; Matesanz, S.; Valladares, F. Do we underestimate the importance of leaf size in plant economics? Disproportional scaling of support costs within the spectrum of leaf physiognomy. Ann. Bot. 2007, 100, 283–303. [Google Scholar] [CrossRef]
- Dawson, J.H.; Musselman, L.J.; Wolswinkel, P.; Dörr, L. Biology and control of Cuscuta. Rev. Weed Sci. 1994, 6, 265–317. [Google Scholar]
- Lanini, T.; Kogan, M. Biology and management of Cuscuta in crops. Cien. Inv. Agr. 2005, 32, 165–179. [Google Scholar]
- Grewell, B. Parasite facilitates plant species coexistence in a coastal wetland. Ecology 2008, 89, 1481–1488. [Google Scholar] [CrossRef]
- Press, M.C.; Phoenix, G.K. Impacts of parasitic plants on natural communities. New Phytol. 2005, 166, 737–751. [Google Scholar] [CrossRef]
- Yu, H.; Yu, F.-H.; Miao, S.-L.; Dong, M. Holoparasitic Cuscuta campestris suppresses invasive Mikania micrantha and contributes to native community recovery. Biol. Conserv. 2008, 141, 2653–2661. [Google Scholar] [CrossRef]
- Parry, M.L.; Canziani, O.F.; Palutikof, J.P.; van der Linden, P.J.; Hanson, C.E. IPCC Fourth Assesment Report (AR4). Climate Change 2007: Impacts, Adaptation and Vulnerability. Contribution of Working Group II to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change; Cambridge University Press: Cambridge, UK, 2007. [Google Scholar]
- Milnes, K.J.; Davies, W.J.; Rodwell, J.S.; Francis, B.J. The responses of Briza media and Koeleria macrantha to drought and re-watering. Funct. Ecol. 1998, 12, 665–672. [Google Scholar] [CrossRef]
- Rennenberg, H.; Dannenmann, M.; Gessler, A.; Kreuzwieser, J.; Simon, J.; Papen, H. Nitrogen balance in forest soils: Nutritional limitation of plants under climate change stresses. Plant Biol. 2009, 11, 4–23. [Google Scholar] [CrossRef]
- Phoenix, G.K.; Press, M.C. Effects of climate change on parasitic plants: The root hemiparasitic Orobanchaceae. Folia Geobot. 2005, 40, 205–216. [Google Scholar] [CrossRef]
- Parmesan, C. Ecological and evolutionary responses to recent climate change. Annu. Rev. Ecol. Evol. Syst. 2006, 37, 637–669. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Evans, B.; Borowicz, V. Verbesina alternifolia Tolerance to the Holoparasite Cuscuta gronovii and the Impact of Drought. Plants 2013, 2, 635-649. https://doi.org/10.3390/plants2040635
Evans B, Borowicz V. Verbesina alternifolia Tolerance to the Holoparasite Cuscuta gronovii and the Impact of Drought. Plants. 2013; 2(4):635-649. https://doi.org/10.3390/plants2040635
Chicago/Turabian StyleEvans, Bethany, and Victoria Borowicz. 2013. "Verbesina alternifolia Tolerance to the Holoparasite Cuscuta gronovii and the Impact of Drought" Plants 2, no. 4: 635-649. https://doi.org/10.3390/plants2040635




