Calcium Signals from the Vacuole
Abstract
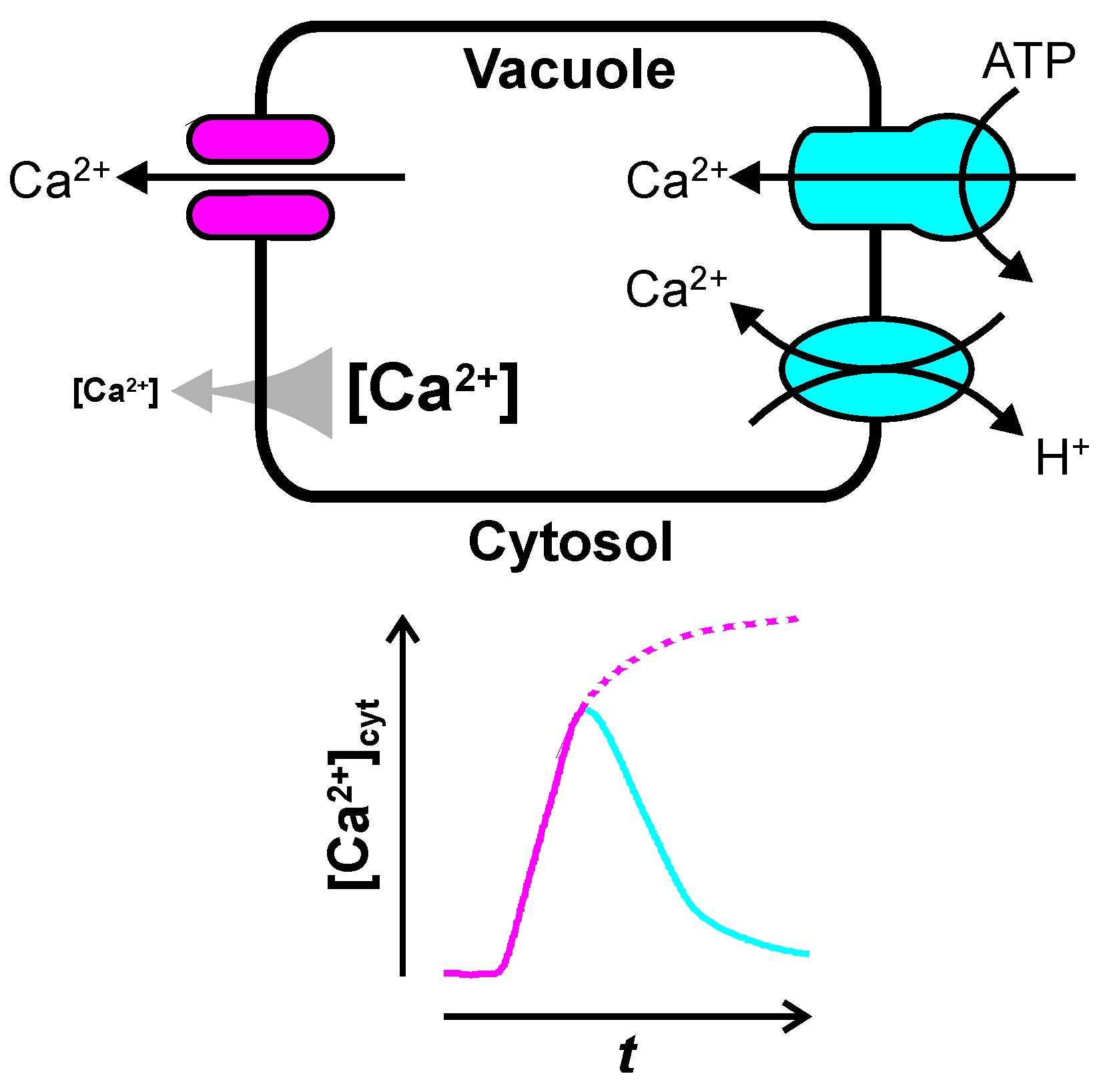
:1. The Vacuole—A Huge Intracellular Ca2+ Store
2. Ca2+ Release from the Vacuole
2.1. Inositol Trisphosphate-Dependent Vacuolar Ca2+ Release

2.2. Ca2+ Mobilization by Inositol Hexakisphosphate
2.3. Cyclic Adenosine Diphosphoribose (cADPR)-Induced Vacuolar Ca2+ Release
2.4. The Slow Vacuolar / Two Pore Channel (SV/TPC1)
2.4.1. Is There Ca2+-Induced Ca2+ Release by the SV Channel?
2.4.2. Is TPC1 Involved in Ca2+ Signaling?
2.5. Cyclic Nucleotide-Gated Channels
2.6. Transient Receptor Potential Ca2+ Channels
3. Ca2+ Uptake into the Vacuole
3.1. P-Type Ca2+-ATPases
3.2. Ca2+:H+ Exchanger (CAX)
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Felle, H. Cytoplasmic free calcium in Riccia fluitans L. and Zea mays L.: Interaction of Ca2+ and pH? Planta 1988, 176, 248–255. [Google Scholar] [CrossRef]
- Bethmann, B.; Thaler, M.; Simonis, W.; Schönknecht, G. Electrochemical potential gradients of H+, K+, Ca2+, and Cl− across the tonoplast of the green alga Eremosphaera viridis. Plant Physiol. 1995, 109, 1317–1326. [Google Scholar]
- Felle, H.H.; Hepler, P.K. The cytosolic Ca2+ concentration gradient of Sinapis alba root hairs as revealed by Ca2+-selective microelectrode tests and fura-dextran ratio imaging. Plant Physiol. 1997, 114, 39–45. [Google Scholar]
- Plieth, C. Plant calcium signaling and monitoring: Pros and cons and recent experimental approaches. Protoplasma 2001, 218, 1–23. [Google Scholar] [CrossRef]
- Perez, V.; Wherrett, T.; Shabala, S.; Muniz, J.; Dobrovinskaya, O.; Pottosin, I. Homeostatic control of slow vacuolar channels by luminal cations and evaluation of the channel-mediated tonoplast Ca2+ fluxes in situ. J. Exp. Bot. 2008, 59, 3845–3855. [Google Scholar] [CrossRef]
- Conn, S.; Gilliham, M. Comparative physiology of elemental distributions in plants. Ann. Bot. 2010, 105, 1081–1102. [Google Scholar] [CrossRef]
- Pottosin, I.I.; Martínez-Estévez, M.; Dobrovinskaya, O.R.; Muñiz, J.; Schönknecht, G. Mechanism of luminal Ca2+ and Mg2+ action on the vacuolar slowly activating channels. Planta 2004, 219, 1057–1070. [Google Scholar] [CrossRef]
- Dadacz-Narloch, B.; Beyhl, D.; Larisch, C.; López-Sanjurjo, E.J.; Reski, R.; Kuchitsu, K.; Müller, T.D.; Becker, D.; Schönknecht, G.; Hedrich, R. A novel calcium binding site in the slow vacuolar cation channel TPC1 senses luminal calcium levels. Plant Cell 2011, 23, 2696–2707. [Google Scholar] [CrossRef]
- Walker, D.J.; Leigh, R.A.; Miller, A.J. Potassium homeostasis in vacuolate plant cells. Proc. Natl. Acad. Sci. USA 1996, 93, 10510–10514. [Google Scholar] [CrossRef]
- Bertl, A.; Blumwald, E.; Coronado, R.; Eisenberg, R.; Findlay, G.P.; Gradmann, D.; Hille, B.; Köhler, K.; Kolb, H.A.; MacRobbie, E.A.C.; et al. Electrical measurements on endomembranes. Science 1992, 258, 873–874. [Google Scholar]
- Taiz, L. The plant vacuole. J. Exp. Biol. 1992, 172, 113–122. [Google Scholar]
- Martinoia, E.; Meyer, S.; de Angeli, A.; Nagy, R. Vacuolar transporters in their physiological context. Annu. Rev. Plant Biol. 2012, 63, 183–213. [Google Scholar] [CrossRef]
- Hedrich, R. Ion channels in plants. Physiol. Rev. 2012, 92, 1777–1811. [Google Scholar] [CrossRef]
- Isayenkov, S.; Isner, J.C.; Maathuis, F.J.M. Vacuolar ion channels: Roles in plant nutrition and signalling. FEBS Lett. 2010, 584, 1982–1988. [Google Scholar] [CrossRef]
- Spalding, E.P.; Harper, J.F. The ins and outs of cellular Ca2+ transport. Curr. Opin. Plant Biol. 2011, 14, 715–720. [Google Scholar] [CrossRef]
- Jammes, F.; Hu, H.-C.; Villiers, F.; Bouten, R.; Kwak, J.M. Calcium-permeable channels in plant cells. FEBS J. 2011, 278, 4262–4276. [Google Scholar] [CrossRef]
- Bose, J.; Pottosin, I.; Shabala, S.S.; Palmgren, M.G.; Shabala, S. Calcium efflux systems in stress signalling and adaptation in plants. Front. Plant Sci. 2011. [Google Scholar] [CrossRef]
- Swarbreck, S.; Colaco, R.; Davies, J. Plant calcium-permeable channels. Plant Physiol. 2013. [Google Scholar] [CrossRef]
- Dodd, A.N.; Kudla, J.; Sanders, D. The language of calcium signaling. Annu. Rev. Plant Biol. 2010, 61, 593–620. [Google Scholar] [CrossRef]
- Stael, S.; Wurzinger, B.; Mair, A.; Mehlmer, N.; Vothknecht, U.C.; Teige, M. Plant organellar calcium signalling: An emerging field. J. Exp. Bot. 2011, 433, 1–9. [Google Scholar]
- Peiter, E. The plant vacuole: Emitter and receiver of calcium signals. Cell Calcium 2011, 50, 120–128. [Google Scholar] [CrossRef]
- Pittman, J.K. Vacuolar Ca2+ uptake. Cell Calcium 2011, 50, 139–146. [Google Scholar] [CrossRef]
- Webb, A.A.R.; Larman, M.G.; Montgomery, L.T.; Taylor, J.E.; Hetherington, A.M. The role of calcium in ABA-induced gene expression and stomatal movements. Plant J. 2001, 26, 351–362. [Google Scholar] [CrossRef]
- Gilroy, S.; Fricker, M.D.; Read, N.D.; Trewavas, A.J. Role of calcium in signal transduction of Commelina guard cells. Plant Cell 1991, 3, 333–344. [Google Scholar]
- McAinsh, M.R.; Brownlee, C.; Hetherington, A.M. Visualizing changes in cytosolic-free calcium during the response of stomatal guard cells to abscisic acid. Plant Cell 1992, 4, 1113–1122. [Google Scholar]
- McAinsh, M.R.; Webb, A.A.R.; Taylor, J.E.; Hetherington, A.M. Stimulus-induced oscillations in guard cell cytosolic free calcium. Plant Cell 1995, 7, 1207–1219. [Google Scholar]
- Grabov, A.; Blatt, M.R. Membrane voltage initiates Ca2+ waves and potentiates Ca2+ increases with abscisic acid in stomatal guard cells. Proc. Natl. Acad. Sci. USA 1998, 95, 4778–4783. [Google Scholar] [CrossRef]
- Knight, H.; Trewavas, A.J.; Knight, M.R. Cold calcium signaling in Arabidopsis involves two cellular pools and a change in calcium signature after acclimation. Plant Cell 1996, 8, 489–503. [Google Scholar]
- Knight, H.; Trewavas, A.J.; Knight, M.R. Calcium signalling in Arabidopsis thaliana responding to drought and salinity. Plant J. 1997, 12, 1067–1078. [Google Scholar]
- Foskett, J.K.; White, C.; Cheung, K.-H.; Mak, D.-O.D. Inositol trisphosphate receptor Ca2+ release channels. Physiol. Rev. 2007, 87, 593–658. [Google Scholar] [CrossRef]
- Berridge, M.J. Inositol trisphosphate and calcium signalling. Nature 1993, 361, 315–325. [Google Scholar] [CrossRef]
- Krinke, O.; Novotna, Z.; Valentova, O.; Martinec, J. Inositol trisphosphate receptor in higher plants: is it real? J. Exp. Bot. 2007, 58, 361–376. [Google Scholar]
- Coté, G.G.; Crain, R.C. Why do plants have phosphoinositides? BioEssays 1994, 16, 39–46. [Google Scholar] [CrossRef]
- Gilroy, S.; Read, N.D.; Trewavas, A.J. Elevation of cytoplasmic calcium by caged calcium or caged inositol trisphosphate initiates stomatal closure. Nature 1990, 346, 769–771. [Google Scholar] [CrossRef]
- Monteiro, D.; Liu, Q.; Lisboa, S.; Scherer, G.E.F.; Quader, H.; Malhó, R. Phosphoinositides and phosphatidic acid regulate pollen tube growth and reorientation through modulation of [Ca2+]c and membrane secretion. J. Exp. Bot. 2005, 56, 1665–1674. [Google Scholar] [CrossRef]
- Tucker, E.B.; Boss, W.F. Mastoparan-induced intracellular Ca2+ fluxes may regulate cell-to-cell communication in plants. Plant Physiol. 1996, 111, 459–467. [Google Scholar]
- Maathuis, F.J. Ligand-gated ion channels. In Membrane Transport in Plants; Blatt, M.R., Ed.; Blackwell Publishing: Oxford, UK, 2004; pp. 193–220. [Google Scholar]
- Bothwell, J.H.F.; Ng, C.K.-Y. The evolution of Ca2+ signalling in photosynthetic eukaryotes. New Phytol. 2005, 166, 21–38. [Google Scholar] [CrossRef]
- Burki, F.; Okamoto, N.; Pombert, J.-F.; Keeling, P.J. The evolutionary history of haptophytes andcryptophytes: Phylogenomic evidence for separate origins. Proc. Biol. Sci. 2012, 279, 2246–2254. [Google Scholar] [CrossRef]
- Saier, M.H., Jr.; Yen, M.R.; Noto, K.; Tamang, D.G.; Elkan, C. The Transporter Classification Database: recent advances. Nucleic Acids Res. 2009, 37, D274–D278. [Google Scholar] [CrossRef]
- Saier, M.H., Jr.; Tran, C.V.; Barabote, R.D. TCDB: the Transporter Classification Database for membrane transport protein analyses and information. Nucleic Acids Res. 2006, 34, D181–D186. [Google Scholar] [CrossRef]
- Pazour, G.J.; Agrin, N.; Leszyk, J.; Witman, G.B. Proteomic analysis of a eukaryotic cilium. J. Cell Biol. 2005, 170, 103–113. [Google Scholar] [CrossRef]
- Merchant, S.S.; Prochnik, S.E.; Vallon, O.; Harris, E.H.; Karpowicz, S.J.; Witman, G.B.; Terry, A.; Salamov, A.; Fritz-Laylin, L.K.; Marechal-Drouard, L.; et al. The Chlamydomonas genome reveals the evolution of key animal and plant functions. Science 2007, 318, 245–250. [Google Scholar] [CrossRef]
- Prochnik, S.E.; Umen, J.; Nedelcu, A.M.; Hallmann, A.; Miller, S.M.; Nishii, I.; Ferris, P.; Kuo, A.; Mitros, T.; Fritz-Laylin, L.K.; et al. Genomic analysis of organismal complexity in the multicellular green alga Volvox carteri. Science 2010, 329, 223–226. [Google Scholar] [CrossRef]
- Kuin, H.; Koerten, H.; Ghijsen, W.E.J.M.; Munnik, T.; van den Ende, H.; Musgrave, A. Chlamydomonas contains calcium stores that are mobilized when phospholipase C is activated. Planta 2000, 210, 286–294. [Google Scholar] [CrossRef]
- Wheeler, G.L.; Brownlee, C. Ca2+ signalling in plants and green algae—Changing channels. Trends Plant Sci. 2008, 13, 506–514. [Google Scholar] [CrossRef]
- Förster, B. Injected inositol 1,4,5-trisphosphate activates Ca2+-sensitive K+ channels in the plasmalemma of Eremosphaera viridis. FEBS Lett. 1990, 269, 197–201. [Google Scholar] [CrossRef]
- Price, D.C.; Chan, C.X.; Yoon, H.S.; Yang, E.C.; Qiu, H.; Weber, A.P.M.; Schwacke, R.; Gross, J.; Blouin, N.A.; Lane, C.; et al. Cyanophora paradoxa genome elucidates origin of photosynthesis in algae and plants. Science 2012, 335, 843–847. [Google Scholar] [CrossRef]
- Keeling, P.J.; Palmer, J.D. Horizontal gene transfer in eukaryotic evolution. Nat. Rev. Genet. 2008, 9, 605–618. [Google Scholar] [CrossRef]
- Coelho, S.M.; Taylor, A.R.; Ryan, K.P.; Sousa-Pinto, I.; Brown, M.T.; Brownlee, C. Spatiotemporal patterning of reactive oxygen production and Ca2+ wave propagation in Fucus rhizoid cells. Plant Cell 2002, 14, 2369–2381. [Google Scholar] [CrossRef]
- Cock, J.M.; Sterck, L.; Rouze, P.; Scornet, D.; Allen, A.E.; Amoutzias, G.; Anthouard, V.; Artiguenave, F.; Aury, J.M.; Badger, J.H.; et al. The Ectocarpus genome and the independent evolution of multicellularity in brown algae. Nature 2010, 465, 617–621. [Google Scholar] [CrossRef]
- Schumaker, K.S.; Sze, H. Inositol 1,4,5-trisphosphate releases Ca2+ from vacuolar membrane vesicles of oat roots. J. Biol. Chem. 1987, 262, 3944–3946. [Google Scholar]
- Allen, G.J.; Muir, S.R.; Sanders, D. Release of Ca2+ from individual plant vacuoles by both InsP3 and cyclic ADP-ribose. Science 1995, 268, 735–737. [Google Scholar] [CrossRef]
- Ranjeva, R.; Carrasco, A.; Boudet, A.M. Inositol trisphosphate stimulates the release of calcium from intact vacuoles isolated from Acer cells. FEBS Lett. 1988, 230, 137–141. [Google Scholar] [CrossRef]
- Lommel, C.; Felle, H.H. Transport of Ca2+ across the tonoplast of intact vacuoles from Chenopodium album L suspension cells: ATP-dependent import and inositol-1,4,5-trisphosphate-induced release. Planta 1997, 201, 477–486. [Google Scholar] [CrossRef]
- Martinec, J.; Feltl, T.; Scanlon, C.H.; Lumsden, P.J.; Machackova, I. Subcellular localization of a high affinity binding site for D-myo-inositol 1,4,5-trisphosphate from Chenopodium rubrum. Plant Physiol. 2000, 124, 475–483. [Google Scholar] [CrossRef]
- Muir, S.R.; Sanders, D. Inositol 1,4,5-trisphosphate-sensitive Ca2+ release across nonvacuolar membranes in cauliflower. Plant Physiol. 1997, 114, 1511–1521. [Google Scholar]
- Alexandre, J.; Lassalles, J.P.; Kado, R.T. Opening of Ca2+ channels in isolated red beet vacuole membranes by inositol 1,4,5-trisphosphate. Nature 1990, 343, 567–570. [Google Scholar] [CrossRef]
- Alexandre, J.; Lassalles, J.P. Intracellular Ca2+ release by InsP3 in plants and effect of buffers on Ca2+ diffusion. Phil. Trans. R. Soc. Lond. B 1992, 338, 53–61. [Google Scholar] [CrossRef]
- Allen, G.J.; Sanders, D. Osmotic stress enhances the competence of Beta vulgaris vacuoles to respond to inositol 1,4,5-trisphosphate. Plant J. 1994, 6, 687–695. [Google Scholar]
- Chasan, R.; Schroeder, J.I. Excitation in plant membrane biology. Plant Cell 1992, 4, 1180–1188. [Google Scholar]
- Ping, Z.; Yabe, I.; Muto, S. Identification of K+, Cl−, and Ca2+ channels in the vacuolar membrane of tobacco cell suspension cultures. Protoplasma 1992, 171, 7–18. [Google Scholar] [CrossRef]
- Gelli, A.; Blumwald, E. Calcium retrieval from vacuolar pools. Characterization of a vacuolar calcium channel. Plant Physiol. 1993, 102, 1139–1146. [Google Scholar]
- Pottosin, I.; Wherrett, T.; Shabala, S. SV channels dominate the vacuolar Ca2+ release during intracellular signaling. FEBS Lett. 2009, 583, 921–926. [Google Scholar] [CrossRef]
- Lemtiri-Chlieh, F.; MacRobbie, E.A.; Brearley, C.A. Inositol hexakisphosphate is a physiological signal regulating the K+-inward rectifying conductance in guard cells. Proc. Natl. Acad. Sci. USA 2000, 97, 8687–8692. [Google Scholar] [CrossRef]
- Lemtiri-Chlieh, F.; MacRobbie, E.A.C.; Webb, A.A.R.; Manison, N.F.; Brownlee, C.; Skepper, J.N.; Chen, J.; Prestwich, G.D.; Brearley, C.A. Inositol hexakisphosphate mobilizes an endomembrane store of calcium in guard cells. Proc. Natl. Acad. Sci. USA 2003, 100, 10091–10095. [Google Scholar] [CrossRef]
- Guse, A.H. Biochemistry, biology, and pharmacology of cyclic adenosine diphosphoribose (cADPR). Curr. Med. Chem. 2004, 11, 847–855. [Google Scholar] [CrossRef]
- Clapper, D.L.; Walseth, T.F.; Dargie, P.J.; Lee, H.C. Pyridine nucleotide metabolites stimulate calcium release from sea urchin egg microsomes desensitized to inositol trisphosphate. J. Biol. Chem. 1987, 262, 9561–9568. [Google Scholar]
- Galione, A.; Lee, H.C.; Busa, W.B. Ca2+-induced Ca2+ release in sea urchin egg homogenates:modulation by cyclic ADP-ribose. Science 1991, 253, 1143–1146. [Google Scholar] [CrossRef]
- Galione, A.; Chuang, K.-T. Pyridine nucleotide metabolites and calcium release from intracellular stores. In Calcium Signaling; Islam, M.S., Ed.; Springer: Dordrecht, The Netherlands, 2012; Volume 740, pp. 305–323. [Google Scholar]
- Venturi, E.; Pitt, S.; Galfré, E.; Sitsapesan, R. From eggs to hearts: What is the link between cyclic ADP-ribose and ryanodine receptors? Cardiovasc. Ther. 2012, 30, 109–116. [Google Scholar] [CrossRef]
- Copello, J.A.; Qi, Y.; Jeyakumar, L.H.; Ogunbunmi, E.; Fleischer, S. Lack of effect of cADP-ribose and NAADP on the activity of skeletal muscle and heart ryanodine receptors. Cell Calcium 2001, 30, 269–284. [Google Scholar] [CrossRef]
- Fruen, B.R.; Mickelson, J.R.; Shomer, N.H.; Velez, P.; Louis, C.F. Cyclic ADP-ribose does not affect cardiac or skeletal muscle ryanodine receptors. FEBS Lett. 1994, 352, 123–126. [Google Scholar] [CrossRef]
- Lukyanenko, V.; Györke, I.; Wiesner, T.F.; Györke, S. Potentiation of Ca2+ release by cADP-ribose in the heart is mediated by enhanced SR Ca2+ uptake into the sarcoplasmic reticulum. Circ. Res. 2001, 89, 614–622. [Google Scholar] [CrossRef]
- Zheng, J.; Wenzhi, B.; Miao, L.; Hao, Y.; Zhang, X.; Yin, W.; Pan, J.; Yuan, Z.; Song, B.; Ji, G. Ca2+ release induced by cADP-ribose is mediated by FKBP12.6 proteins in mouse bladder smooth muscle. Cell Calcium 2010, 47, 449–457. [Google Scholar] [CrossRef]
- Muir, S.R.; Sanders, D. Pharmacology of Ca2+ release from red beet microsomes suggests the presence of ryanodine receptor homologs in higher plants. FEBS Lett. 1996, 395, 39–42. [Google Scholar] [CrossRef]
- Moyen, C.; Hammond-Kosack, K.E.; Jones, J.; Knight, M.R.; Johannes, E. Systemin triggers an increase of cytoplasmic calcium in tomato mesophyll cells: Ca2+ mobilization from intra- and extracellular compartments. Plant Cell Environ. 1998, 21, 1101–1111. [Google Scholar] [CrossRef]
- Dodd, A.N.; Gardner, M.J.; Hotta, C.T.; Hubbard, K.E.; Dalchau, N.; Love, J.; Assie, J.M.; Robertson, F.C.; Jakobsen, M.K.; Goncalves, J.; et al. The Arabidopsis circadian clock incorporates a cADPR-based feedback loop. Science 2007, 318, 1789–1792. [Google Scholar] [CrossRef]
- Dodd, A.N.; Gardner, M.J.; Hotta, C.T.; Hubbard, K.E.; Dalchau, N.; Robertson, F.C.; Love, J.; Sanders, D.; Webb, A.A.R. Response to comment on “The Arabidopsis circadian clock incorporates a cADPR-based feedback loop”. Science 2009, 326, 230. [Google Scholar]
- Xu, X.; Graeff, R.; Xie, Q.; Gamble, K.L.; Mori, T.; Johnson, C.H. Comment on “The Arabidopsis circadian clock incorporates a cADPR-based feedback loop”. Science 2009, 326, 230. [Google Scholar]
- Tang, R.H.; Han, S.; Zheng, H.; Cook, C.W.; Choi, C.S.; Woerner, T.E.; Jackson, R.B.; Pei, Z.M. Coupling diurnal cytosolic Ca2+ oscillations to the CAS-IP3 pathway in Arabidopsis. Science 2007, 315, 1423–1426. [Google Scholar] [CrossRef]
- Wu, Y.; Kuzma, J.; Marechal, E.; Graeff, R.; Lee, H.C.; Foster, R.; Chua, N.H. Abscisic acid signaling through cyclic ADP-ribose in plants. Science 1997, 278, 2126–2130. [Google Scholar] [CrossRef]
- Leckie, C.P.; McAinsh, M.R.; Allen, G.J.; Sanders, D.; Hetherington, A.M. Abscisic acid-inducedstomatal closure mediated by cyclic ADP-ribose. Proc. Natl. Acad. Sci. USA 1998, 95, 15837–15842. [Google Scholar]
- Sanchez, J.P.; Duque, P.; Chua, N.H. ABA activates ADPR cyclase and cADPR induces a subset of ABA-responsive genes in Arabidopsis. Plant J. 2004, 38, 381–395. [Google Scholar] [CrossRef]
- MacRobbie, E.A.C. ABA activates multiple Ca2+ fluxes in stomatal guard cells, triggering vacuolar K+(Rb+) release. Proc. Natl. Acad. Sci. USA 2000, 97, 12361–12368. [Google Scholar] [CrossRef]
- Bauer, C.S.; Plieth, C.; Bethmann, B.; Popescu, O.; Hansen, U.-P.; Simonis, W.; Schönknecht, G. Strontium-induced repetitive calcium spikes in a unicellular green alga. Plant Physiol. 1998, 117, 545–557. [Google Scholar] [CrossRef]
- Bauer, C.S.; Simonis, W.; Schönknecht, G. Different xanthines cause membrane potential oscillations in a unicellular green alga pointing to a ryanodine/cADPR receptor Ca2+ channel. Plant Cell Physiol. 1999, 40, 453–456. [Google Scholar] [CrossRef]
- Mackrill, J.J. Ryanodine receptor calcium release channels: An evolutionary perspective. In Calcium Signaling; Islam, M.S., Ed.; Springer: Dordrecht, The Netherlands, 2012; Volume 740, pp. 159–182. [Google Scholar]
- Navazio, L.; Mariani, P.; Sanders, D. Mobilization of Ca2+ by cyclic ADP-ribose from the endoplasmic reticulum of cauliflower florets. Plant Physiol. 2001, 125, 2129–2138. [Google Scholar] [CrossRef]
- Hedrich, R.; Flügge, U.-I.; Fernandez, J.M. Patch-clamp studies of ion transport in isolated plant vacuoles. FEBS Lett. 1986, 204, 228–232. [Google Scholar] [CrossRef]
- Hedrich, R.; Neher, E. Cytoplasmic calcium regulates voltage-dependent ion channels in plant vacuoles. Nature 1987, 329, 833–835. [Google Scholar] [CrossRef]
- Colombo, R.; Cerana, R.; Lado, P.; Peres, A. Voltage-dependent channels permeable to K+ and Na+ in the membrane of Acer pseudoplantanus vacuoles. J. Membr. Biol. 1988, 103, 227–236. [Google Scholar] [CrossRef]
- Hedrich, R.; Barbier-Brygoo, H.; Felle, H.; Flügge, U.-I.; Lüttge, U.; Maathuis, F.J.M.; Marx, S.; Prins, H.B.A.; Raschke, K.; Schnabl, H.; et al. General mechanisms for solute transport across the tonoplast of plant vacuoles: A patch-clamp survey of ion channels and proton pumps. Bot. Acta 1988, 101, 7–13. [Google Scholar]
- Peiter, E.; Maathuis, F.J.M.; Mills, L.N.; Knight, H.; Pelloux, J.; Hetherington, A.M.; Sanders, D. The vacuolar Ca2+-activated channel TPC1 regulates germination and stomatal movement. Nature 2005, 434, 404–408. [Google Scholar] [CrossRef]
- Hedrich, R.; Marten, I. TPC1—SV channels gain shape. Mol. Plant 2011, 4, 428–441. [Google Scholar] [CrossRef]
- Ward, J.M.; Schroeder, J.I. Calcium-activated K+ channels and calcium-induced calcium release by slow vacuolar ion channels in guard cell vacuoles implicated in the control of stomatal closure. Plant Cell 1994, 6, 669–683. [Google Scholar]
- Pantoja, O.; Gelli, A.; Blumwald, E. Voltage-dependent calcium channels in plant vacuoles. Science 1992, 255, 1567–1570. [Google Scholar]
- Pottosin, I.I.; Schönknecht, G. Vacuolar calcium channels. J. Exp. Bot. 2007, 58, 1559–1569. [Google Scholar] [CrossRef]
- Johannes, E.; Sanders, D. The voltage-gated Ca2+ release channel in the vacuolar membrane of sugar beet resides in two activity states. FEBS Lett. 1995, 365, 1–6. [Google Scholar] [CrossRef]
- Johannes, E.; Sanders, D. Lumenal calcium modulates unitary conductance and gating of a plant vacuolar calcium release channel. J. Membr. Biol. 1995, 146, 211–224. [Google Scholar]
- Allen, G.J.; Sanders, D. Calcineurin, a type 2B protein phosphatase, modulates the Ca2+-permeable slow vacuolar ion channel of stomatal guard cells. Plant Cell 1995, 7, 1473–1483. [Google Scholar]
- Pottosin, I.I.; Dobrovinskaya, O.R.; Muniz, J. Conduction of monovalent and divalent cations in the slow vacuolar channel. J. Membr. Biol. 2001, 181, 55–65. [Google Scholar]
- Pottosin, I.I.; Tikhonova, L.I.; Hedrich, R.; Schönknecht, G. Slowly activating vacuolar channels can not mediate Ca2+-induced Ca2+ release. Plant J. 1997, 12, 1387–1398. [Google Scholar]
- Bewell, M.A.; Maathuis, F.J.M.; Allen, G.J.; Sanders, D. Calcium-induced calcium release mediated by a voltage-activated cation channel in vacuolar vesicles from red beet. FEBS Lett. 1999, 458, 41–44. [Google Scholar] [CrossRef]
- Pei, Z.M.; Ward, J.M.; Schroeder, J.I. Magnesium sensitizes slow vacuolar channels to physiological cytosolic calcium and inhibits fast vacuolar channels in fava bean guard cell vacuoles. Plant Physiol. 1999, 121, 977–986. [Google Scholar] [CrossRef]
- Carpaneto, A.; Cantu, A.M.; Gambale, F. Effects of cytoplasmic Mg2+ on slowly activating channels in isolated vacuoles of Beta vulgaris. Planta 2001, 213, 457–468. [Google Scholar] [CrossRef]
- Ivashikina, N.; Hedrich, R. K+ currents through SV-type vacuolar channels are sensitive to elevated luminal sodium levels. Plant J. 2005, 41, 606–614. [Google Scholar] [CrossRef]
- Carpaneto, A.; Cantu, A.M.; Gambale, F. Redox agents regulate ion channel activity in vacuoles from higher plant cells. FEBS Lett. 1999, 442, 129–132. [Google Scholar] [CrossRef]
- Gradogna, A.; Scholz-Starke, J.; Gutla, P.V.K.; Carpaneto, A. Fluorescence combined with excised patch: Measuring calcium currents in plant cation channels. Plant J. 2009, 58, 175–182. [Google Scholar] [CrossRef]
- Gambale, F.; Bregante, M.; Stragapede, F.; Cantu, A.M. Ionic channels of the sugar beet tonoplast are regulated by a multi-ion single-file permeation mechanism. J. Membr. Biol. 1996, 154, 69–79. [Google Scholar] [CrossRef]
- Banks, J.A.; Nishiyama, T.; Hasebe, M.; Bowman, J.L.; Gribskov, M.; dePamphilis, C.; Albert, V.A.; Aono, N.; Aoyama, T.; Ambrose, B.A.; et al. The Selaginella genome identifies genetic changes associated with the evolution of vascular plants. Science 2011, 332, 960–963. [Google Scholar] [CrossRef]
- Linz, K.W.; Köhler, K. Vacuolar ion currents in the primitive green alga Eremosphaera viridis: The electrical properties are suggestive of both the Characeae and higher plants. Protoplasma 1994, 179, 34–45. [Google Scholar] [CrossRef]
- Blanc, G.; Agarkova, I.; Grimwood, J.; Kuo, A.; Brueggeman, A.; Dunigan, D.; Gurnon, J.; Ladunga, I.; Lindquist, E.; Lucas, S.; et al. The genome of the polar eukaryotic microalga Coccomyxa subellipsoidea reveals traits of cold adaptation. Genome Biol. 2012, 13, R39. [Google Scholar] [CrossRef]
- Blanc, G.; Duncan, G.; Agarkova, I.; Borodovsky, M.; Gurnon, J.; Kuo, A.; Lindquist, E.; Lucas, S.; Pangilinan, J.; Polle, J.; et al. The Chlorella variabilis NC64A genome reveals adaptation to photosymbiosis, coevolution with viruses, and cryptic sex. Plant Cell 2010, 22, 2943–2955. [Google Scholar] [CrossRef]
- Collén, J.; Porcel, B.; Carré, W.; Ball, S.G.; Chaparro, C.; Tonon, T.; Barbeyron, T.; Michel, G.; Noel, B.; Valentin, K.; et al. Genome structure and metabolic features in the red seaweed Chondrus crispus shed light on evolution of the Archaeplastida. Proc. Natl. Acad. Sci. USA 2013, 110, 5247–5252. [Google Scholar] [CrossRef] [Green Version]
- Nakamura, Y.; Sasaki, N.; Kobayashi, M.; Ojima, N.; Yasuike, M.; Shigenobu, Y.; Satomi, M.; Fukuma, Y.; Shiwaku, K.; Tsujimoto, A.; et al. The first symbiont-free genome sequence of marine red alga, Susabi-nori Pyropia yezoensis. PLoS One 2013, 8, e57122. [Google Scholar] [CrossRef]
- Ishibashi, K.; Suzuki, M.; Imai, M. Molecular cloning of a novel form (two-repeat) protein related to voltage-gated sodium and calcium channels. Biochem. Biophys. Res. Commun. 2000, 270, 370–376. [Google Scholar] [CrossRef]
- Calcraft, P.J.; Ruas, M.; Pan, Z.; Cheng, X.; Arredouani, A.; Hao, X.; Tang, J.; Rietdorf, K.; Teboul, L.; Chuang, K.T.; et al. NAADP mobilizes calcium from acidic organelles through two-pore channels. Nature 2009, 459, 596–600. [Google Scholar] [CrossRef]
- Rybalchenko, V.; Ahuja, M.; Coblentz, J.; Churamani, D.; Patel, S.; Kiselyov, K.; Muallem, S. Membrane potential regulates nicotinic acid adenine dinucleotide phosphate (NAADP) dependence of the pH- and Ca2+-sensitive organellar two-pore channel TPC1. J. Biol. Chem. 2012, 287, 20407–20416. [Google Scholar]
- Zhu, M.X.; Ma, J.; Parrington, J.; Galione, A.; Mark Evans, A. TPCs: Endolysosomal channels for Ca2+ mobilization from acidic organelles triggered by NAADP. FEBS Lett. 2010, 584, 1966–1974. [Google Scholar] [CrossRef]
- Lee, H.C. A unified mechanism of enzymatic synthesis of two calcium messengers: Cyclic ADP-ribose and NAADP. Biol. Chem. 1999, 380, 785–793. [Google Scholar]
- Lee, H.C.; Aarhus, R. A derivative of NADP mobilizes calcium stores insensitive to inositol trisphosphate and cyclic ADP-ribose. J. Biol. Chem. 1995, 270, 2152–2157. [Google Scholar] [CrossRef]
- Genazzani, A.A.; Galione, A. Nicotinic acid-adenine dinucleotide phosphate mobilizes Ca2+ from a thapsigargin-insensitive pool. Biochem. J. 1996, 315, 721–725. [Google Scholar]
- Chini, E.N.; Dousa, T.P. Nicotinate-adenine dinucleotide phosphate-induced Ca2+-release does not behave as a Ca2+-induced Ca2+-release system. Biochem. J. 1996, 316, 709–711. [Google Scholar]
- Cancela, J.M.; Churchill, G.C.; Galione, A. Coordination of agonist-induced Ca2+-signalling patterns by NAADP in pancreatic acinar cells. Nature 1999, 398, 74–76. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, X.; Dong, X.-P.; Samie, M.; Li, X.; Cheng, X.; Goschka, A.; Shen, D.; Zhou, Y.; Harlow, J.; et al. TPC proteins are phosphoinositide-activated sodium-selective ion channels in endosomes and lysosomes. Cell 2012, 151, 372–383. [Google Scholar] [CrossRef]
- Lin-Moshier, Y.; Walseth, T.F.; Churamani, D.; Davidson, S.M.; Slama, J.T.; Hooper, R.; Brailoiu, E.; Patel, S.; Marchant, J.S. Photoaffinity labeling of nicotinic acid adenine dinucleotide phosphate (NAADP) targets in mammalian cells. J. Biol. Chem. 2012, 287, 2296–2307. [Google Scholar] [CrossRef]
- Navazio, L.; Bewell, M.A.; Siddiqua, A.; Dickinson, G.D.; Galione, A.; Sanders, D. Calcium release from the endoplasmic reticulum of higher plants elicited by the NADP metabolite nicotinic acid adenine dinucleotide phosphate. Proc. Natl. Acad. Sci. USA 2000, 97, 8693–8698. [Google Scholar]
- Islam, M.M.; Munemasa, S.; Hossain, M.A.; Nakamura, Y.; Mori, I.C.; Murata, Y. Roles of AtTPC1, vacuolar two pore channel 1, in Arabidopsis stomatal closure. Plant Cell Physiol. 2010, 51, 302–311. [Google Scholar] [CrossRef]
- Ranf, S.; Wunnenberg, P.; Lee, J.; Becker, D.; Dunkel, M.; Hedrich, R.; Scheel, D.; Dietrich, P. Loss of the vacuolar cation channel, AtTPC1, does not impair Ca2+ signals induced by abiotic and biotic stresses. Plant J. 2008, 53, 287–299. [Google Scholar]
- Bonaventure, G.; Gfeller, A.; Proebsting, W.M.; Hortensteiner, S.; Chetelat, A.; Martinoia, E.; Farmer, E.E. A gain-of-function allele of TPC1 activates oxylipin biogenesis after leaf wounding in Arabidopsis. Plant J. 2007, 49, 889–898. [Google Scholar] [CrossRef]
- Beyhl, D.; Hörtensteiner, S.; Martinoia, E.; Farmer, E.E.; Fromm, J.; Marten, I.; Hedrich, R. The fou2 mutation in the major vacuolar cation channel TPC1 confers tolerance to inhibitory luminal calcium. Plant J. 2009, 58, 715–723. [Google Scholar] [CrossRef]
- Bonaventure, G.; Gfeller, A.; Rodriguez, V.M.; Armand, F.; Farmer, E.E. The fou2 gain-of-function allele and the wild-type allele of Two Pore Channel 1 contribute to different extents or by different mechanisms to defense gene expression in Arabidopsis. Plant Cell Physiol. 2007, 48, 1775–1789. [Google Scholar] [CrossRef]
- Armengaud, P.; Breitling, R.; Amtmann, A. The potassium-dependent transcriptome of Arabidopsis reveals a prominent role of jasmonic acid in nutrient signaling. Plant Physiol. 2004, 136, 2556–2576. [Google Scholar] [CrossRef]
- Gilliham, M.; Athman, A.; Tyerman, S.D.; Conn, S.J. Cell-specific compartmentation of mineral nutrients is an essential mechanism for optimal plant productivity—Another role for TPC1? Plant Signal. Behav. 2011, 6, 1656–1661. [Google Scholar] [CrossRef]
- Conn, S.J.; Berninger, P.; Broadley, M.R.; Gilliham, M. Exploiting natural variation to uncover candidate genes that control element accumulation in Arabidopsis thaliana. New Phytol. 2012, 193, 859–866. [Google Scholar] [CrossRef]
- Furuichi, T.; Mori, I.C.; Takahashi, K.; Muto, S. Sugar-induced increase in cytosolic Ca2+ in Arabidopsis thaliana whole plants. Plant Cell Physiol. 2001, 42, 1149–1155. [Google Scholar] [CrossRef]
- Furuichi, T.; Cunningham, K.W.; Muto, S. A putative two pore channel AtTPC1 mediates Ca2+ flux in Arabidopsis leaf cells. Plant Cell Physiol. 2001, 42, 900–905. [Google Scholar] [CrossRef]
- Kadota, Y.; Furuichi, T.; Ogasawara, Y.; Goh, T.; Higashi, K.; Muto, S.; Kuchitsu, K. Identification of putative voltage-dependent Ca2+-permeable channels involved in cryptogein-induced Ca2+ transients and defense responses in tobacco BY-2 cells. Biochem. Biophys. Res. Commun. 2004, 317, 823–830. [Google Scholar] [CrossRef]
- Hamada, H.; Kurusu, T.; Okuma, E.; Nokajima, H.; Kiyoduka, M.; Koyano, T.; Sugiyama, Y.; Okada, K.; Koga, J.; Saji, H.; et al. Regulation of a proteinaceous elicitor-induced Ca2+ influx and production of phytoalexins by a putative voltage-gated cation channel, OsTPC1, in cultured rice cells. J. Biol. Chem. 2012, 287, 9931–9939. [Google Scholar] [CrossRef]
- Kurusu, T.; Yagala, T.; Miyao, A.; Hirochika, H.; Kuchitsu, K. Identification of a putative voltage-gated Ca2+ channel as a key regulator of elicitor-induced hypersensitive cell death and mitogen-activated protein kinase activation in rice. Plant J. 2005, 42, 798–809. [Google Scholar] [CrossRef]
- Larisch, N.; Schulze, C.; Galione, A.; Dietrich, P. An N-terminal dileucine motif directs two-pore channels to the tonoplast of plant cells. Traffic 2012, 13, 1012–1022. [Google Scholar] [CrossRef]
- Amodeo, G.; Escobar, A.; Zeiger, E. A cationic channel in the guard cell tonoplast of Allium cepa. Plant Physiol. 1994, 105, 999–1006. [Google Scholar]
- Carpaneto, A.; Cantu, A.M.; Busch, H.; Gambale, F. Ion channels in the vacuoles of the seagrass Posidonia oceanica. FEBS Lett. 1997, 412, 236–240. [Google Scholar] [CrossRef]
- Wherrett, T.; Shabala, S.; Pottosin, I. Different properties of SV channels in root vacuoles from near isogenic Al-tolerant and Al-sensitive wheat cultivars. FEBS Lett. 2005, 579, 6890–6894. [Google Scholar] [CrossRef]
- Hashimoto, K.; Saito, M.; Hidetoshi, I.; Matsuoka, H. Evidence for the plasma membrane localization of a putative voltage-dependent Ca2+ channel, OsTPC1, in rice. Plant Biotechnol. 2005, 22, 235–239. [Google Scholar] [CrossRef]
- Dadacz-Narloch, B.; Kimura, S.; Kurusu, T.; Farmer, E.E.; Becker, D.; Kuchitsu, K.; Hedrich, R. On the cellular site of two-pore channel TPC1 action in the Poaceae. New Phytol. 2013. [Google Scholar] [CrossRef]
- Chen, Z.-H.; Hills, A.; Bätz, U.; Amtmann, A.; Lew, V.L.; Blatt, M.R. Systems dynamic modeling of the stomatal guard cell predicts emergent behaviors in transport, signaling, and volume control. Plant Physiol. 2012, 159, 1235–1251. [Google Scholar] [CrossRef]
- Hills, A.; Chen, Z.-H.; Amtmann, A.; Blatt, M.R.; Lew, V.L. OnGuard, a computational platform for quantitative kinetic modeling of guard cell physiology. Plant Physiol. 2012, 159, 1026–1042. [Google Scholar] [CrossRef]
- Yuen, C.C.Y.; Christopher, D.A. The group IV-A cyclic nucleotide-gated channels, CNGC19 and CNGC20, localize to the vacuole membrane in Arabidopsis thaliana. AoB Plants 2013, 5, plt012. [Google Scholar] [CrossRef]
- Fischer, C.; Kugler, A.; Hoth, S.; Dietrich, P. An IQ domain mediates the interaction with calmodulin in a plant cyclic nucleotide-gated channel. Plant Cell Physiol. 2013, 54, 573–584. [Google Scholar] [CrossRef]
- Dietrich, P.; Anschütz, U.; Kugler, A.; Becker, D. Physiology and biophysics of plant ligand-gated ion channels. Plant Biol. 2010, 12, 80–93. [Google Scholar] [CrossRef]
- Chin, K.; Moeder, W.; Yoshioka, K. Biological roles of cyclic-nucleotide-gated ion channels in plants: What we know and dont know about this 20 member ion channel family. Botany 2009, 87, 668–677. [Google Scholar] [CrossRef]
- Zelman, A.K.; Dawe, A.; Berkowitz, G.A.; Gehring, C. Evolutionary and structural perspectives of plant cyclic nucleotide gated cation channels. Front. Plant Sci. 2012. [Google Scholar] [CrossRef]
- Kugler, A.; Kohler, B.; Palme, K.; Wolff, P.; Dietrich, P. Salt-dependent regulation of a CNG channel subfamily in Arabidopsis. BMC Plant Biol. 2009, 9, 140. [Google Scholar]
- Quiles-Pando, C.; Rexach, J.; Navarro-Gochicoa, M.T.; Camacho-Cristóbal, J.J.; Herrera-Rodríguez, M.B.; González-Fontes, A. Boron deficiency increases the levels of cytosolic Ca2+ and expression of Ca2+-related genes in Arabidopsis thaliana roots. Plant Physiol. Biochem. 2013, 65, 55–60. [Google Scholar] [CrossRef]
- Palmer, C.P.; Zhou, X.L.; Lin, J.; Loukin, S.H.; Kung, C.; Saimi, Y. A TRP homolog in Saccharomyces cerevisiae forms an intracellular Ca2+-permeable channel in the yeast vacuolar membrane. Proc. Natl. Acad. Sci. USA 2001, 98, 7801–7805. [Google Scholar]
- Cunningham, K.W. Acidic calcium stores of Saccharomyces cerevisiae. Cell Calcium 2011, 50, 129–138. [Google Scholar] [CrossRef]
- Scrase-Field, S.A.; Knight, M.R. Calcium: Just a chemical switch? Curr. Opin. Plant Biol. 2003, 6, 500–506. [Google Scholar] [CrossRef]
- McAinsh, M.R.; Pittman, J.K. Shaping the calcium signature. New Phytol. 2009, 181, 275–294. [Google Scholar] [CrossRef]
- Zhu, X.; Feng, Y.; Liang, G.; Liu, N.; Zhu, J.-K. Aequorin-based luminescence imaging reveals stimulus- and tissue-specific Ca2+ dynamics in Arabidopsis plants. Mol. Plant 2013, 6, 444–455. [Google Scholar] [CrossRef]
- Tracy, F.E.; Gilliham, M.; Dodd, A.N.; Webb, A.A.R.; Tester, M. NaCl-induced changes in cytosolic free Ca2+ in Arabidopsis thaliana are heterogeneous and modified by external ionic composition. Plant Cell Environ. 2008, 31, 1063–1073. [Google Scholar] [CrossRef]
- Allen, G.J.; Chu, S.P.; Harrington, C.L.; Schumacher, K.; Hoffmann, T.; Tang, Y.Y.; Grill, E.; Schroeder, J.I. A defined range of guard cell calcium oscillation parameters encodes stomatal movements. Nature 2001, 411, 1053–1057. [Google Scholar] [CrossRef]
- Bonza, M.C.; de Michelis, M.I. The plant Ca2+-ATPase repertoire: Biochemical features and physiological functions. Plant Biol. 2011, 13, 421–430. [Google Scholar] [CrossRef]
- Huda, K.M.K.; Banu, M.S.A.; Tuteja, R.; Tuteja, N. Global calcium transducer P-type Ca2+-ATPases open new avenues for agriculture by regulating stress signalling. J. Exp. Bot. 2013, 64, 3099–3109. [Google Scholar] [CrossRef]
- Pedersen, C.N.S.; Axelsen, K.B.; Harper, J.F.; Palmgren, M.G. Evolution of plant P-type ATPases. Front. Plant Sci. 2012, 3, 31. [Google Scholar]
- Geisler, M.; Frangne, N.; Gomes, E.; Martinoia, E.; Palmgren, M.G. The ACA4 gene of Arabidopsis encodes a vacuolar membrane calcium pump that improves salt tolerance in yeast. Plant Physiol. 2000, 124, 1814–1827. [Google Scholar] [CrossRef]
- Lee, S.M.; Kim, H.S.; Han, H.J.; Moon, B.C.; Kim, C.Y.; Harper, J.F.; Chung, W.S. Identification of a calmodulin-regulated autoinhibited Ca2+-ATPase (ACA11) that is localized to vacuole membranes in Arabidopsis. FEBS Lett. 2007, 581, 3943–3949. [Google Scholar] [CrossRef]
- Boursiac, Y.; Lee, S.M.; Romanowsky, S.; Blank, R.; Sladek, C.; Chung, W.S.; Harper, J.F. Disruption of the vacuolar calcium-ATPases in Arabidopsis results in the activation of a salicylic acid-dependent programmed cell death pathway. Plant Physiol. 2010, 154, 1158–1171. [Google Scholar] [CrossRef]
- Qudeimat, E.; Faltusz, A.M.C.; Wheeler, G.; Lang, D.; Brownlee, C.; Reski, R.; Frank, W. A PIIB-type Ca2+-ATPase is essential for stress adaptation in Physcomitrella patens. Proc. Natl. Acad. Sci. USA 2008, 105, 19555–19560. [Google Scholar]
- Zhu, X.; Caplan, J.; Mamillapalli, P.; Czymmek, K.; Dinesh-Kumar, S.P. Function of endoplasmic reticulum calcium ATPase in innate immunity-mediated programmed cell death. EMBO J. 2010, 29, 1007–1018. [Google Scholar] [CrossRef]
- Pittman, J. Multiple transport pathways for mediating intracellular pH homeostasis: The contribution of H+/ion exchangers. Front. Plant Sci. 2012, 3, 11. [Google Scholar] [CrossRef]
- Emery, L.; Whelan, S.; Hirschi, K.D.; Pittman, J.K. Protein phylogenetic analysis of Ca2+/cation antiporters and insights into their evolution in plants. Front. Plant Sci. 2012, 3, 1–19. [Google Scholar]
- Manohar, M.; Shigaki, T.; Hirschi, K.D. Plant cation/H+ exchangers (CAXs): Biological functions and genetic manipulations. Plant Biol. 2011, 13, 561–569. [Google Scholar] [CrossRef]
- Cheng, N.-H.; Pittman, J.K.; Shigaki, T.; Lachmansingh, J.; LeClere, S.; Lahner, B.; Salt, D.E.; Hirschi, K.D. Functional association of Arabidopsis CAX1 and CAX3 is required for normal growth and ion homeostasis. Plant Physiol. 2005, 138, 2048–2060. [Google Scholar] [CrossRef]
- Connorton, J.M.; Webster, R.E.; Cheng, N.; Pittman, J.K. Knockout of multiple Arabidopsis cation/H+ exchangers suggests isoform-specific roles in metal stress response, germination and seed mineral nutrition. PLoS One 2012, 7, e47455. [Google Scholar]
- Conn, S.J.; Gilliham, M.; Athman, A.; Schreiber, A.W.; Baumann, U.; Moller, I.; Cheng, N.-H.; Stancombe, M.A.; Hirschi, K.D.; Webb, A.A.R.; et al. Cell-specific vacuolar calcium storage mediated by CAX1 regulates apoplastic calcium concentration, gas exchange, and plant productivity in Arabidopsis. Plant Cell 2011, 23, 240–257. [Google Scholar] [CrossRef]
- Cho, D.; Villiers, F.; Kroniewicz, L.; Lee, S.; Seo, Y.J.; Hirschi, K.D.; Leonhardt, N.; Kwak, J.M. Vacuolar CAX1 and CAX3 influence auxin transport in guard cells via regulation of apoplastic pH. Plant Physiol. 2012, 160, 1293–1302. [Google Scholar] [CrossRef]
- Xu, L.; Zahid, K.R.; He, L.; Zhang, W.; He, X.; Zhang, X.; Yang, X.; Zhu, L. GhCAX3 Gene, a novel Ca2+/H+ exchanger from cotton, confers regulation of cold response and ABA induced signal transduction. PLoS One 2013, 8, e66303. [Google Scholar]
- Zhao, J.; Barkla, B.; Marshall, J.; Pittman, J.; Hirschi, K. The Arabidopsis cax3 mutants display altered salt tolerance, pH sensitivity and reduced plasma membrane H+-ATPase activity. Planta 2008, 227, 659–669. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Schönknecht, G. Calcium Signals from the Vacuole. Plants 2013, 2, 589-614. https://doi.org/10.3390/plants2040589
Schönknecht G. Calcium Signals from the Vacuole. Plants. 2013; 2(4):589-614. https://doi.org/10.3390/plants2040589
Chicago/Turabian StyleSchönknecht, Gerald. 2013. "Calcium Signals from the Vacuole" Plants 2, no. 4: 589-614. https://doi.org/10.3390/plants2040589
APA StyleSchönknecht, G. (2013). Calcium Signals from the Vacuole. Plants, 2(4), 589-614. https://doi.org/10.3390/plants2040589



