1. Introduction
The mature heart of vertebrates is a hollow muscle—the myocardium—whereby the inner and outer surfaces are covered by skins called the endocardium and epicardium, respectively. Up to the end of the 20th century, research on the developing heart mainly focused on the origin and differentiation of the myocardium and endocardium. The epicardium did not receive much recognition. It was generally thought that the definitive myocardium and epicardium both derived from the outer epithelial wall of the early embryonic heart, which was consequently named the myoepicardium. We now know that the so-called myoepicardium consists exclusively of primary myocardial cells and that the epicardial mesothelium, the majority of the subepicardial and intramyocardial fibroblasts, and the coronary blood vessels derive from a primarily extracardiac source of progenitor cells called the proepicardium (PE). We also know that the epicardium and other PE-derived structures play important roles in normal development of the myocardial wall and in the repair of damaged myocardium (see other articles in this issue).
Studies on chick embryos have significantly contributed to our current knowledge about the developmental biology of the PE/epicardium. The chick embryo was the first model organism enabling the German anatomist Remak to describe the accumulation of villous protrusions of the pericardial mesothelium, now known as the PE [
1]. The chick embryo was also the first model organism through which the Polish anatomist Kurkiewicz in 1909 was able to correctly identify these pericardial villi as the source of the embryonic epicardium [
2]. Finally, avian embryos (chicks, quails) were the first model organisms from which microsurgical experiments led to the identification of further derivatives of the PE.
In this article, I describe three “classical” microsurgical experiments that have been developed and used for studying the developmental significance of the PE/epicardium in avian embryos. These are, firstly, the mechanical PE-blocking technique; secondly, the PE-photoablation technique; and thirdly, the orthotopic PE-grafting technique. These microsurgical experiments have significantly contributed to our current knowledge about the roles of the PE/epicardium in development of the vertebrate heart.
2. Mechanical PE-Blocking
The PE-blocking technique is used in experiments that are conducted to see how hearts will develop in the absence or reduced amounts of PE-derived tissues (loss-of-PE-function experiments). It was developed in my lab in the early 1990s and first published in an original research paper in 1993 [
3]. The goal of the PE-blocking technique is to prevent the normal cell transfer from the PE to the heart. In mammalian species (e.g., mice, humans), as well as in some fish species (e.g., zebrafish), PE-derived cells reach the surface of the embryonic heart in the form of free-floating vesicles (for review see [
4]). These vesicles are released into the fluid of the free pericardial cavity where they float passively to the surface of the heart. Such a PE-cell transfer cannot be blocked effectively by a simple microsurgical procedure. In avian, reptilian, and amphibian embryos, however, the PE-cell transfer is normally accomplished via a secondary tissue bridge, which is formed by the firm attachment of PE villi to the opposite surface of the developing ventricles (
Figure 1). This PE-derived tissue bridge is called the sinu
-ventricular ligament since it runs from the ventral wall of the sinus venosus to the dorsal wall of the ventricles [
5]. The goal of the PE-blocking technique, thus, is to prevent the formation of PE-derived tissue bridges to the heart. The reason for choosing a mechanical PE-blocking technique instead of a mechanical PE-excision technique is due to the avian PE occupying the thin and fragile wall of the right horn of the sinus venosus, so that any mechanical damage to the PE may cause lethal bleeding of the embryo. The prevention of damage to the sinus venosus is not the only advantage of this technique; it is also a technically simple and quick microsurgical procedure. A disadvantage of this technique, however, is the fact that it does not lead to a removal of the PE, so that the colonization of the embryonic heart with PE-derived cells is not completely prevented but only severely delayed [
3,
6].
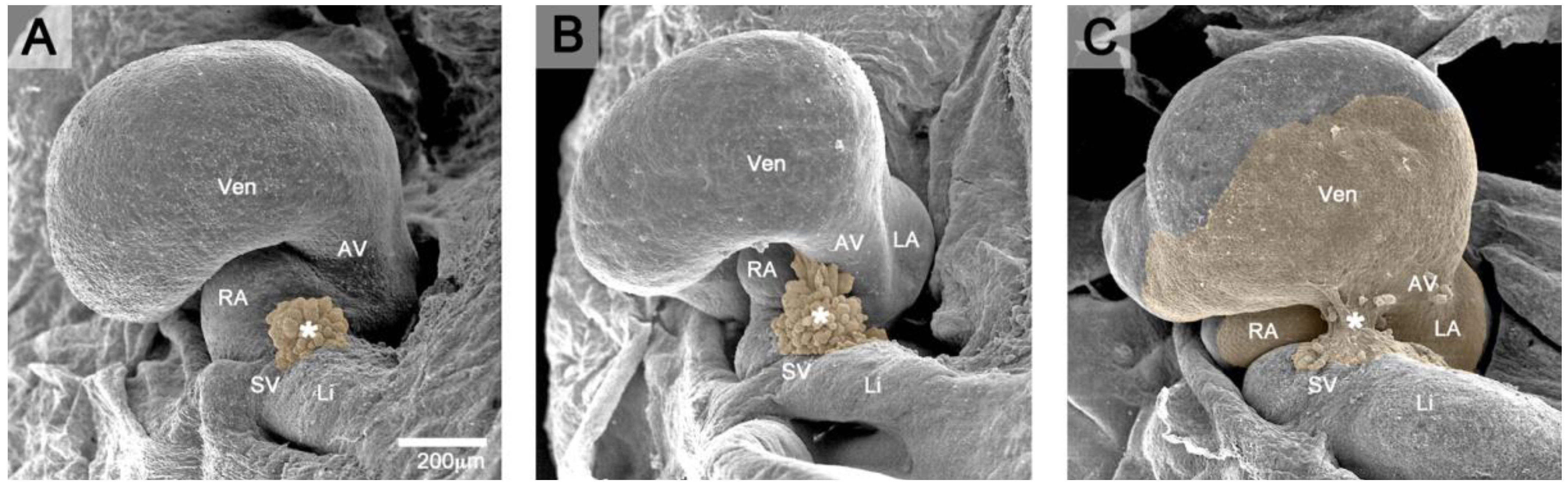
Figure 1.
These scanning electron micrographs, viewed from caudal, show the formation of the normal PE-derived tissue bridge to the heart in three-day-old chick embryos (Hamburger and Hamilton (HH)-stages 16–18). Brown coloration marks the PE and the PE-derived primitive epicardium. (A) At HH-stage 16, the PE appears as a cauliflower-shaped accumulation of mesothelial villous protrusions (*) covering the surface of the right horn of the sinus venosus (SV). (B) At HH-stage 17, the tips of some PE villi (*) adhere to the dorsal surface of the developing ventricles (Vent). (C) At HH-stage 18, a secondary, PE-derived tissue bridge (*) is formed, which bridges the free pericardial cavity between the ventral wall of the sinus venosus and the dorsal wall of the developing ventricles (sinu-ventricular ligament). From the point of its attachment to the heart, the primitive PE-derived epicardium spreads over the naked myocardial surface of the heart. AV: atrio-ventricular canal; LA: future left atrial appendage; Li: liver; RA: future right atrial appendage.
Figure 1.
These scanning electron micrographs, viewed from caudal, show the formation of the normal PE-derived tissue bridge to the heart in three-day-old chick embryos (Hamburger and Hamilton (HH)-stages 16–18). Brown coloration marks the PE and the PE-derived primitive epicardium. (A) At HH-stage 16, the PE appears as a cauliflower-shaped accumulation of mesothelial villous protrusions (*) covering the surface of the right horn of the sinus venosus (SV). (B) At HH-stage 17, the tips of some PE villi (*) adhere to the dorsal surface of the developing ventricles (Vent). (C) At HH-stage 18, a secondary, PE-derived tissue bridge (*) is formed, which bridges the free pericardial cavity between the ventral wall of the sinus venosus and the dorsal wall of the developing ventricles (sinu-ventricular ligament). From the point of its attachment to the heart, the primitive PE-derived epicardium spreads over the naked myocardial surface of the heart. AV: atrio-ventricular canal; LA: future left atrial appendage; Li: liver; RA: future right atrial appendage.
![Jdb 01 00047 g001]()
The embryo is treated
in ovo before the villous protrusions of the PE have established firm contacts to the dorsal wall of the embryonic ventricle (3rd incubation day, at stages 15–17, according to Hamburger and Hamilton [
7]). The egg is windowed over the embryo according to Hara [
8]. The primitive pericardial cavity then is opened by cutting the vitelline and chorionic membranes. This is possible since, in avian species, the primitive pericardial cavity is not separated from the extra-embryonic coelomic cavity during the early phase of embryonic heart development [
9].
In order to prevent the formation of PE-derived tissue bridges to the heart, a small rectangular piece of eggshell membrane is introduced into the primitive pericardial cavity, where it is placed between the PE and the dorsal wall of the embryonic ventricle. Since the cardiac pumping activity can lead to an unwanted displacement of the membrane from this “blocking position,” the membrane must be mechanically anchored at two sides: (1) ventrally at the coelomic wall of the anterior intestinal portal; and (2) dorsally in the furrow between the primitive atrium and sinus venosus (
Figure 2). Further contacts between the “blocking-membrane” and other embryonic or extra-embryonic sides should be avoided since such contacts will lead to the formation of unwanted adhesions between the heart, the implanted membrane, and embryonic or extra-embryonic tissues with the final consequences of development of severe malformations of the heart and body wall. After correct implantation of the blocking-membrane, the egg is closed again with an adhesive tape and returned to the incubator.
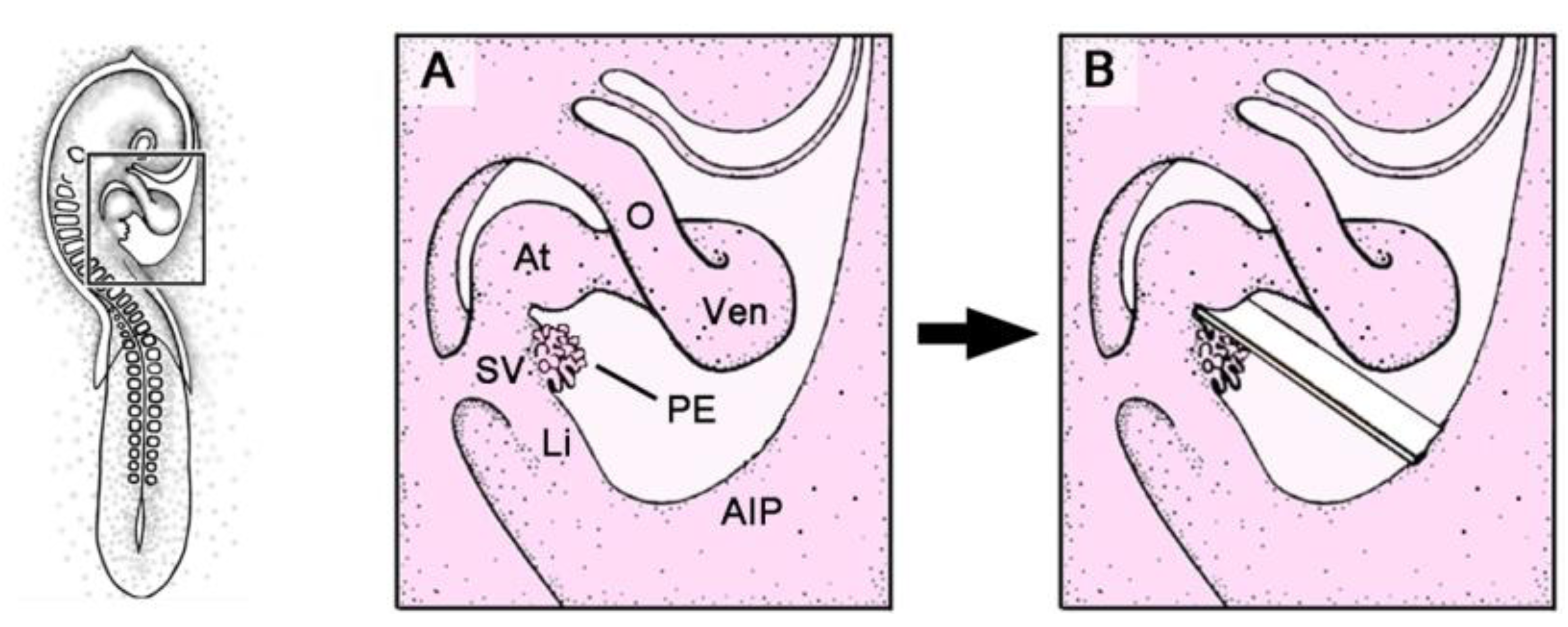
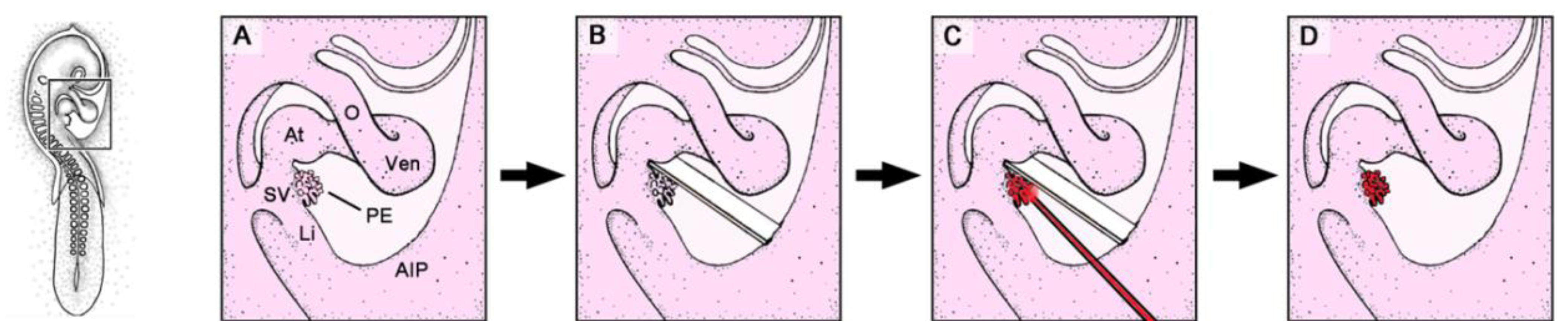
Figure 2.
These schematic drawings depict the mechanical PE-blocking procedure used in avian embryos (chick or quail).
A–
B show details of the boxed area marked in the HH-stage 16 embryo on the left. AIP: coelomic wall of the anterior intestinal portal; O: outflow tract of the embryonic heart loop; other abbreviations as used in
Figure 1.
Figure 2.
These schematic drawings depict the mechanical PE-blocking procedure used in avian embryos (chick or quail).
A–
B show details of the boxed area marked in the HH-stage 16 embryo on the left. AIP: coelomic wall of the anterior intestinal portal; O: outflow tract of the embryonic heart loop; other abbreviations as used in
Figure 1.
For the correct interpretation of the results of PE-blocking experiments, it should be remembered that the goal of the procedure is to prevent the formation of PE-derived tissue bridges to the heart, and thereby to block the direct cell transfer from the PE to the developing heart. Therefore, the criterion for assigning an experimental embryo to the group of successful PE-blocking experiments is the complete lack of PE-derived tissue bridges to the heart, whereas the presence of such tissue bridges must be regarded as the criterion for non-successful operations. According to these criteria, it has been found that the correct implantation of the blocking-membrane principally can lead to the development of two different kinds of hearts, that have since been classified as group-A and group-B hearts [
3]. Group-A hearts represent the group of successful PE-blocking experiments, as they are characterized by the complete lack of any PE-derived tissue bridges to the heart. They only show a PE-derived tissue bridge, which connects the ventral wall of the sinus venosus with the blocking-membrane. This tissue bridge has been named the
sinus cord (
Figure 3). It serves as a pathway over which PE-derived cells colonize the blocking-membrane. Due to its adherence to the anterior intestinal portal, the blocking-membrane does not remain in its initial position behind the heart, but becomes displaced caudally concomitant with the descent of the anterior intestinal portal. The sinus cord subsequently becomes continuously elongated and finally traverses the pericardial and peritoneal cavities. Corresponding to group-A hearts, group-B hearts lack the sinu-ventricular ligament and posses a sinus cord. They show, however, dystopic PE-derived tissue bridges, which connect the sinus cord or the blocking membrane with the developing ventricles. The points of attachment of such dystopic cardiac ligaments to the ventricles usually lie caudal from the normal attachment point of the sinu-ventricular ligament. In some cases, for example, dystopic cardiac ligaments are attached to the apex of the heart (
Figure 3C). Thus, it is likely that, in group-B hearts, the implantation of the blocking-membrane initially has successfully prevented the formation of the sinu-ventricular ligament (indicated by the lack of this ligament and the presence of a sinus cord). With prolonged re-incubation time, however, the PE-derived mesothelium covering the sinus cord or the blocking-membrane must have formed new villous protrusions, which became firmly attached to the dorsal wall of the developing ventricles. Due to the caudal displacement of the blocking-membrane, the points of attachment of these cardiac ligaments lie caudal to the normal attachment point of the sinu-ventricular ligament.
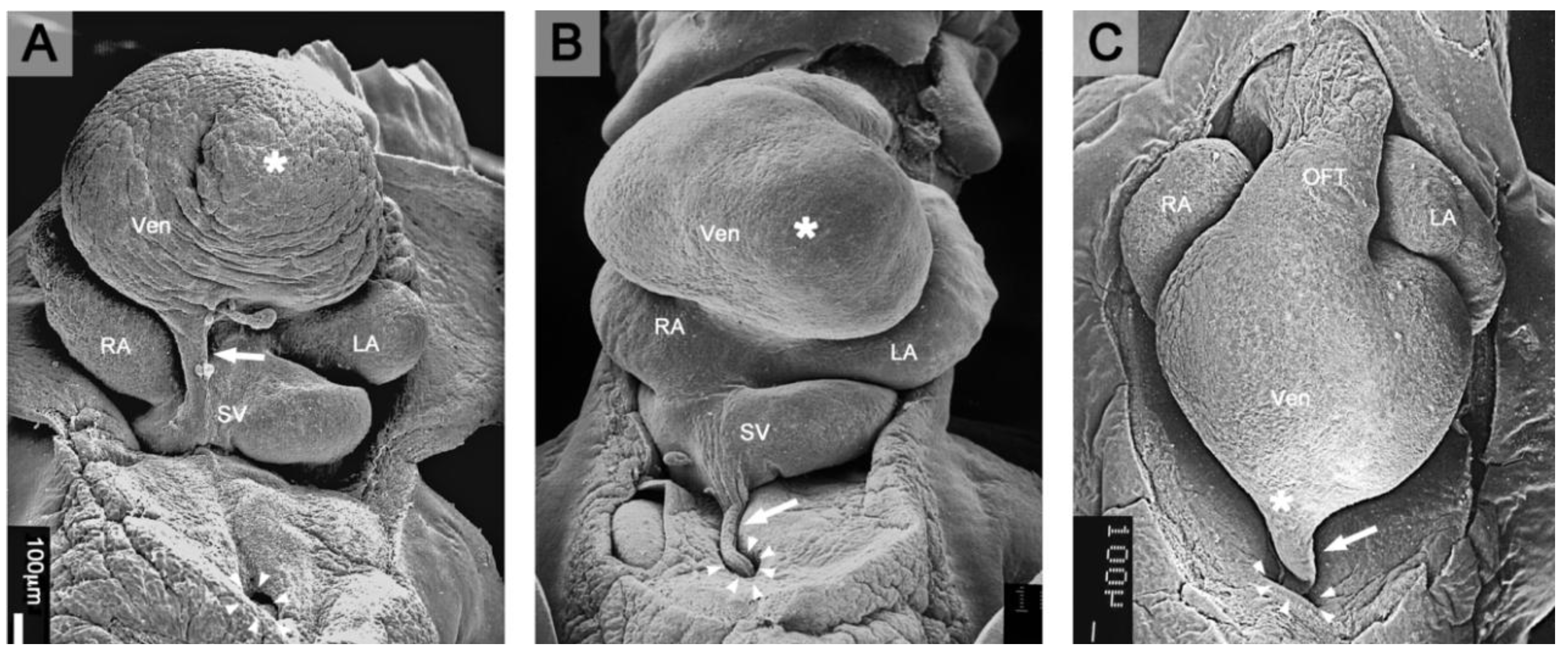
Figure 3.
(
A and
B) These scanning electron micrographs, viewed from ventral caudal, show the ventral wall of the sinus venosus and the dorsal wall of the developing ventricles of a normal chick embryo (
A) and of a group-A experimental chick embryo (
B) from the 6th incubation day (HH-stage 27). For better views, the apices of the developing ventricles (*) were shifted craniad prior to fixation. (
A) In the normal heart, a PE-derived tissue bridge runs from the ventral wall of the sinus venosus to the dorsal wall of the developing ventricles (sinu-ventricular ligament; marked by arrow). (
B) Group-A hearts lack the sinu-ventricular ligament. They show a PE-derived tissue bridge, which connects the ventral wall of the sinus venosus with the blocking-membrane. This tissue bridge is named the
sinus cord (marked by arrow). Due to its adherence to the anterior intestinal portal, the blocking-membrane has been displaced from the pericardial cavity into the peritoneal cavity. Arrowheads mark the border of the initially large ventral opening between the pericardial and peritoneal cavities. (
C) This scanning electron micrograph, viewed from ventral, shows an example of group-B hearts. Such hearts lack the sinu-ventricular ligament but show dystopic PE-derived tissue bridges connecting the sinus cord or the blocking membrane with the developing ventricles. In this group-B heart (HH-stage 29), the dystopic ligament is attached to the apex (*) of the developing ventricles. Abbreviations as used in
Figure 1.
Figure 3.
(
A and
B) These scanning electron micrographs, viewed from ventral caudal, show the ventral wall of the sinus venosus and the dorsal wall of the developing ventricles of a normal chick embryo (
A) and of a group-A experimental chick embryo (
B) from the 6th incubation day (HH-stage 27). For better views, the apices of the developing ventricles (*) were shifted craniad prior to fixation. (
A) In the normal heart, a PE-derived tissue bridge runs from the ventral wall of the sinus venosus to the dorsal wall of the developing ventricles (sinu-ventricular ligament; marked by arrow). (
B) Group-A hearts lack the sinu-ventricular ligament. They show a PE-derived tissue bridge, which connects the ventral wall of the sinus venosus with the blocking-membrane. This tissue bridge is named the
sinus cord (marked by arrow). Due to its adherence to the anterior intestinal portal, the blocking-membrane has been displaced from the pericardial cavity into the peritoneal cavity. Arrowheads mark the border of the initially large ventral opening between the pericardial and peritoneal cavities. (
C) This scanning electron micrograph, viewed from ventral, shows an example of group-B hearts. Such hearts lack the sinu-ventricular ligament but show dystopic PE-derived tissue bridges connecting the sinus cord or the blocking membrane with the developing ventricles. In this group-B heart (HH-stage 29), the dystopic ligament is attached to the apex (*) of the developing ventricles. Abbreviations as used in
Figure 1.
![Jdb 01 00047 g003]()
Since its first publication in 1993, the PE-blocking technique was used in at least 12 further studies, which were conducted to clarify the roles of the epicardium and other PE-derived tissues in normal development of the avian embryonic heart. The observation that group-A hearts usually show defective formation of the epicardium and subepicardial mesenchyme (
Figure 4), while group-B hearts do not develop substantial defects of their epicardial and subepicardial coverings, was the first experimental evidence supporting Kurkiewizc’s view [
2] that the epicardial mesothelium derives from a cauliflower-shaped accumulation of pericardial mesothelial villi, currently called the PE [
3]. Analyses of the development and phenotype of group-A hearts have disclosed that such hearts not only develop deficiencies of the epicardium and subepicardial mesenchyme, but additionally develop (1) defects in coronary vasculature [
6,
10,
11,
12,
13]; (2) a growth defect of the outer compact layer of the ventricular myocardium [
6,
10,
11,
13,
14,
15]; (3) impaired maturation of the working myocardium [
16]; (4) disturbed differentiation of the Purkinje-fiber system [
17]; (5) impaired remodeling of the outflow tract myocardium [
6,
18]; (6) anomalies of the atrio-ventricular endocardial cushions [
6,
11]; and (7) defective formation of the fibrous heart skeleton causing persistent atrio-ventricular myocardial connection with electrical conduction anomalies [
19,
20]. It was also found that the lack of PE-derived epicardium is partially compensated by non-removed PE and pericardial mesothelium, which colonize the heart along its connections with the pericardial wall at the arterial and venous poles of the heart [
3,
6]. The formation of this compensatory epicardium led to a complete mesothelial covering of the heart in every group-A embryo surviving up to the ninth incubation day, which is a three-day delay compared with normal hearts.
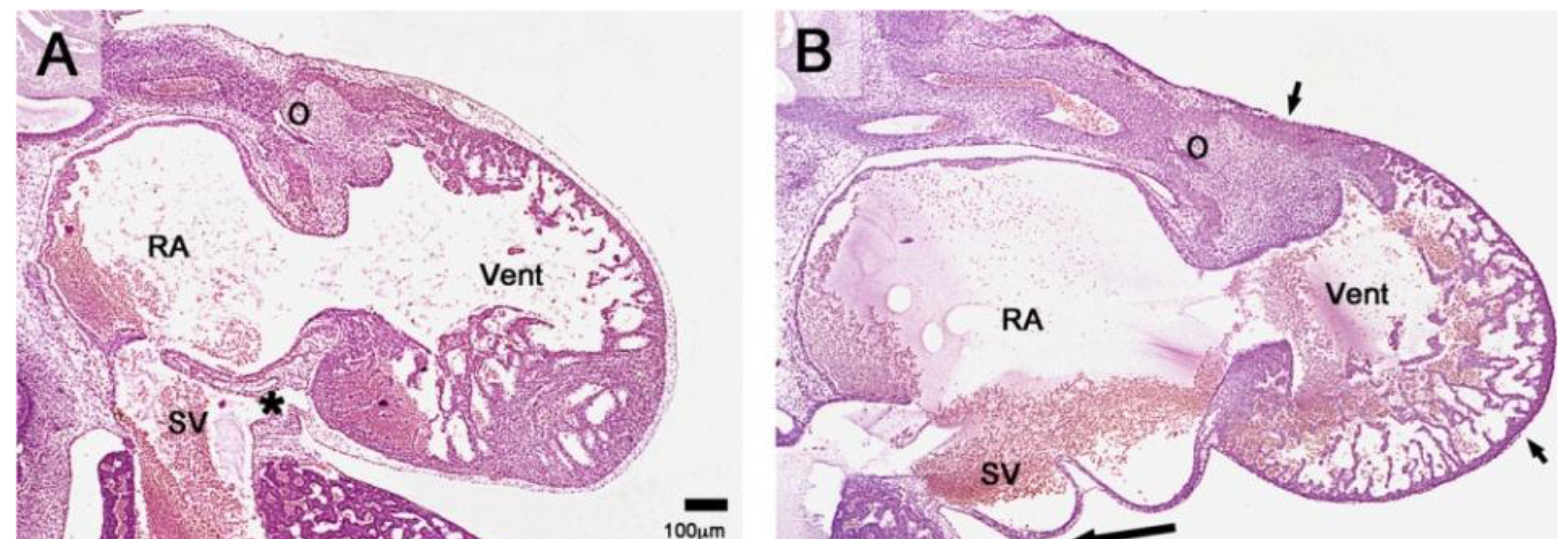
Figure 4.
These sagittal histological sections (Hematoxylin and Eosin (HE) staining) show differences between the phenotypes of hearts from a normal chick embryo and from a group-A experimental chick embryo at HH-stage 29. (
A) The normal heart is completely covered with primitive epicardium and subepicardial mesenchyme. The sinu-ventricular ligament (marked by asterisk) harbors the stem of the developing cardiac veins. (
B) The group-A heart is incompletely covered with primitive epicardium (atria, dorsal wall of the ventricles, distal part of the outflow tract). The ventral wall of the ventricles and the proximal outflow tract still show naked myocardium (short arrows mark the advancing edges of the primitive epicardium). Note the absence of subepicardial mesenchyme and the reduced thickness of the compact layer of the ventricular myocardium (thin myocardium syndrome). Abbreviations as used in
Figure 1; long arrow points to the sinus cord.
Figure 4.
These sagittal histological sections (Hematoxylin and Eosin (HE) staining) show differences between the phenotypes of hearts from a normal chick embryo and from a group-A experimental chick embryo at HH-stage 29. (
A) The normal heart is completely covered with primitive epicardium and subepicardial mesenchyme. The sinu-ventricular ligament (marked by asterisk) harbors the stem of the developing cardiac veins. (
B) The group-A heart is incompletely covered with primitive epicardium (atria, dorsal wall of the ventricles, distal part of the outflow tract). The ventral wall of the ventricles and the proximal outflow tract still show naked myocardium (short arrows mark the advancing edges of the primitive epicardium). Note the absence of subepicardial mesenchyme and the reduced thickness of the compact layer of the ventricular myocardium (thin myocardium syndrome). Abbreviations as used in
Figure 1; long arrow points to the sinus cord.
3. PE-Photoablation
The PE-photoablation technique was developed in my lab in 2003 and first published in 2005 [
21]. The goal of PE-photoablation is the complete elimination of the PE. In contrast to the PE-blocking technique, which is a technically simple and quick microsurgical procedure (see above), PE-photoablation is a technically demanding and time-consuming microsurgical technique. Compared to PE-blocking or mechanical PE-excision techniques, however, it has the great advantage that it facilitates a complete and long-lasting elimination of the PE without significant damage to non-PE tissues. This reduces the amount of compensatory epicardium colonizing the heart via its venous pole. It, thus, seems to be the ideal technique for loss-of-PE-function experiments.
Photoablation techniques use the combined application of two individually non-toxic agents to induce oxygen-dependent tissue damage: (1) a photoactivatable substance, the photosensitizer; and (2) light of a specific wavelength exciting the photosensitizer. In the presence of oxygen, the excitation of the photosensitizer produces reactive oxygen species, which damage the tissues in contact with the photosensitizer. For PE-photoablation, we use the reddish colored dye Rose Bengal (4,5,6,7-tetra-chloro-2',4',5',7'-tetraiodofluorescein sodium salt) as photosensitizer. Use of a colored photosensitizer facilitates selective and complete application of the photosensitizer to the PE under visual control.
The embryo is treated
in ovo at stage 16 according to Hamburger and Hamilton [
7]. At this stage, the PE normally has not made firm contact to the heart and its size is large enough to facilitate selective staining of all of its villi without unwanted staining of surrounding structures. The egg is windowed and the primitive pericardial cavity is opened as described above. After the opening of the primitive pericardial cavity, a small rectangular piece of eggshell membrane is placed in the “blocking-position” between the PE and the dorsal wall of the embryonic ventricle (
Figure 5). This membrane protects the heart from staining with Rose Bengal. The PE then is externally stained with a 1% solution of Rose Bengal (dissolved in physiological saline) by means of a micropipette. Small boluses of the dye solution are repeatedly injected into the pericardial fluid surrounding the PE, where most of the dye is instantly bound to the PE. To prevent staining of non-PE tissues, any unbound dye solution is immediately sucked away by means of a second micropipette. After complete and intense staining of the PE, the protecting membrane is removed from the primitive pericardial cavity and the egg is closed again with an adhesive tape and returned to the incubator. Since Rose Bengal is photoactivated by visible light concomitantly with its application to the PE, there is no need for a specific light exposure phase subsequent to staining of the PE.
The reason for developing the PE-photoablation technique was the fact that, due to the formation of a compensatory epicardium, firstly, from remnants of non-removed PE at the venous heart pole and, secondly, from pericardial mesothelium at the arterial heart pole [
3,
6], the PE-blocking technique was regarded as not suitable for studying long-term effects of epicardium deficiency. We have found that PE-photoablation is a powerful technique for complete and long-lasting removal of the PE. The loss-of-PE-function phenotype corresponds to that observed subsequent to PE-blocking, but the degree of the defects is more severe than in PE-blocking experiments. Although it is not possible to prevent the formation of a compensatory epicardium, its formation is markedly delayed in comparison with PE-blocking experiments. The growth defect of the compact myocardium is also more severe than the thin myocardium phenotype observed in PE-blocking experiments (
Figure 6), and the thinned ventricular wall develops an anomaly not observed in PE-blocking experiments, namely an aneurysma-like outward bulging of those areas devoid of epicardium. The latter finding suggests that the embryonic epicardium may make important contributions to the passive mechanics of the ventricular wall [
21]. The use of PE-photoablation in future studies on avian embryos may help to clarify the hitherto unknown role of epicardial mechanics in cardiac development.
Figure 5.
These schematic drawings depict the PE-photoablation procedure used in avian embryos (chick or quail). A-D show the boxed area marked in the HH-stage 16 embryo on the left.
(A) Opening and inspection of the pericardial cavity.
(B) Implantation of the protecting membrane.
(C) Rose Bengal staining of the PE via a micropipette.
(D) Removal of the protecting membrane. Abbreviations as used in
Figure 2.
Figure 5.
These schematic drawings depict the PE-photoablation procedure used in avian embryos (chick or quail). A-D show the boxed area marked in the HH-stage 16 embryo on the left.
(A) Opening and inspection of the pericardial cavity.
(B) Implantation of the protecting membrane.
(C) Rose Bengal staining of the PE via a micropipette.
(D) Removal of the protecting membrane. Abbreviations as used in
Figure 2.
Figure 6.
Transverse histological sections (HE staining) through the wall of the right ventricle of a normal (A) and epicardium-deficient chick embryo heart (B) from the ninth incubation day (HH-stage 34). Epicardium-deficient hearts develop a severe reduction in thickness of the compact myocardium (thin myocardium syndrome). Comp: compact myocardium; Epi: Epicardium; Subepi: subepicardial mesenchyme; Trab: trabecular myocardium.
Figure 6.
Transverse histological sections (HE staining) through the wall of the right ventricle of a normal (A) and epicardium-deficient chick embryo heart (B) from the ninth incubation day (HH-stage 34). Epicardium-deficient hearts develop a severe reduction in thickness of the compact myocardium (thin myocardium syndrome). Comp: compact myocardium; Epi: Epicardium; Subepi: subepicardial mesenchyme; Trab: trabecular myocardium.
4. Orthotopic PE-Grafting
PE-grafting experiments have been primarily used for PE-fate mapping in avian embryos. The production of chimeras by interspecies grafting of tissues from quail donor embryos into chick host embryos is a well-established experimental approach for clarifying the fate of embryonic structures in avian embryos since the 1970s (for reviews see [
22,
23]). It is based on the existence of structural and immunological differences between the cells of quail and chick embryos, which can be used for the detection of grafted quail cells in chick hosts. Avian embryos, therefore, were the first choice when embryologists and developmental biologists became interested in clarifying the fate of the PE in the 1990s. The microsurgical techniques used for the first PE-grafting experiments were relatively simple. A piece of quail PE-villi, or a piece of quail liver carrying the PE, was loosely placed into the primitive pericardial cavity of a chick host, without blocking or elimination of the host PE [
24,
25,
26,
27]. Although the hearts produced in such experiments receive only a limited contribution of grafted PE-cells, such grafting experiments have significantly contributed to our current knowledge about the PE, since they have shown that the PE not only provides epicardial mesothelial cells but, additionally, provides mesenchymal cells which invade the subepicardium and myocardium, where they differentiate into fibroblasts and cells of the coronary vasculature. A shortcoming of the early PE-grafting procedures, however, is the fact that the donor PE usually contributes only a minority of PE-cells to the host heart, while the host PE still contributes the majority of PE-cells to the host heart. Using such grafting procedures, it is, therefore, not possible to clarify the whole extent of PE-cell contributions to the developing heart. In view of this shortcoming, I developed a new PE-grafting technique called the “orthotopic PE-grafting” procedure, which is outlined in the following paragraphs [
28]. This grafting procedure facilitates an orthotopic attachment of the donor PE to the host heart combined with blocking of the attachment of the host PE to the host heart. The results of such experiments are chimeric hearts in which the tissues derived from the PE are almost completely of donor origin (
Figure 7).
Figure 7.
The orthotopic PE-grafting technique facilitates the production of chimeric hearts in which the derivatives of the PE are almost completely of donor origin. This chimeric heart is from the sixth incubation day (HH-stage 27), which is the time point when the developing chick heart normally is completely covered with epicardium and subepicardial mesenchyme. Quail donor cells (brown color) are immunostained with the anti-quail antibody QCPN (cell nucleus staining). Note that the heart is almost completely covered with donor-derived epicardium and subepicardial mesenchyme. Only the distal portion of the outflow tract is covered with host-derived epicardium (short arrow marks the border between donor and host-derived epicardium). Donor PE-derived subepicardial mesenchymal cells have started to invade the myocardium. The long arrow points to the tissue bridge between the grafted PE and the host heart. Abbreviations as used previously.
Figure 7.
The orthotopic PE-grafting technique facilitates the production of chimeric hearts in which the derivatives of the PE are almost completely of donor origin. This chimeric heart is from the sixth incubation day (HH-stage 27), which is the time point when the developing chick heart normally is completely covered with epicardium and subepicardial mesenchyme. Quail donor cells (brown color) are immunostained with the anti-quail antibody QCPN (cell nucleus staining). Note that the heart is almost completely covered with donor-derived epicardium and subepicardial mesenchyme. Only the distal portion of the outflow tract is covered with host-derived epicardium (short arrow marks the border between donor and host-derived epicardium). Donor PE-derived subepicardial mesenchymal cells have started to invade the myocardium. The long arrow points to the tissue bridge between the grafted PE and the host heart. Abbreviations as used previously.
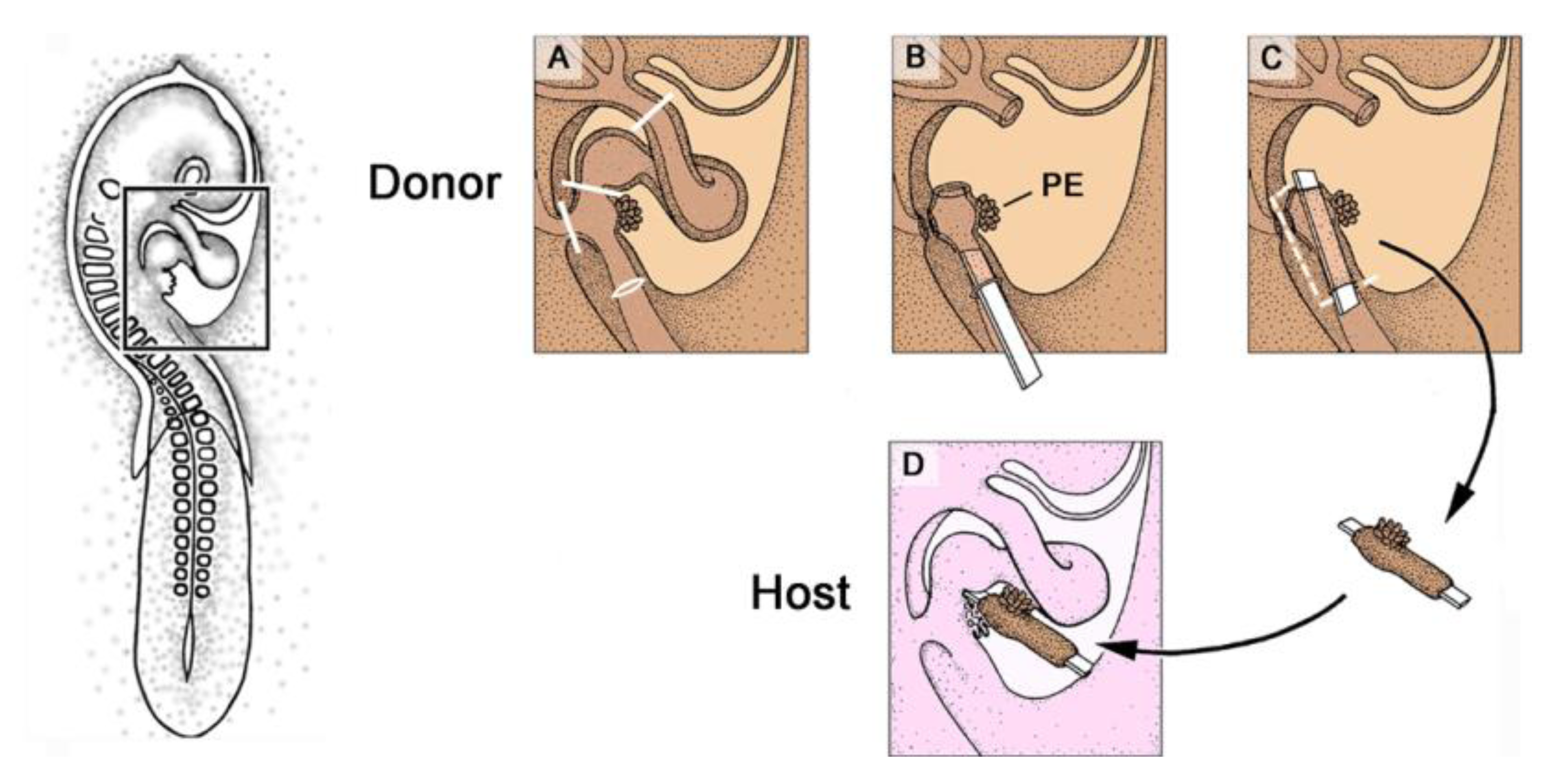
The orthotopic PE-grafting procedure starts with the preparation of the host embryo, which is treated
in ovo before PE-villi have established firm contacts to the heart (stages 16/17, according to Hamburger and Hamilton [
7]). The egg is windowed and the primitive pericardial cavity is opened as described above. After the opening of the primitive pericardial cavity, a small rectangular piece of eggshell membrane is prepared for accurate placement in “blocking” position between the PE and the dorsal wall of the embryonic ventricle (see above). This membrane will serve two functions: (1) as a carrier for the PE-graft, keeping the graft in a position within the pericardial cavity of the host embryo, which facilitates an orthotopic attachment of donor PE-villi to the host heart; (2) as a blocking-membrane preventing the transfer of host PE-cells to the host heart. This “carrier-membrane” is then transferred to the donor embryo, where it will be mounted with a cuff of tissue from the venous heart pole carrying the PE.
Preparation of the donor embryo (stages 16/17, according to Hamburger and Hamilton [
7]) is carried out in a dish with sterile Locke’s solution outside of the egg (
Figure 8A–C). The pericardial cavity is opened (see above) and the heart is removed by two cuts with fine scissors; the first along the sinu-atrial border, and the second through the outflow tract. The rest of the venous heart pole is then disconnected from the body veins, by cuts through the lateral mesocardia, and the right omphalomesenteric vein is opened by an incision into its ventral wall. This opening is used to introduce the “carrier-membrane” (see above) into the lumen of the omphalomesenteric vein and the rest of the venous heart pole. This tissue block is then excised together with the carrier-membrane. The carrier-membrane now is mounted with a cuff of donor tissue carrying the ventral wall of the sinus venosus with the PE. The graft is transferred to the pericardial cavity of the host embryo, where the carrier-membrane is placed in the PE “blocking-position” (
Figure 8D). After correct implantation of the graft, the host egg is closed again with an adhesive tape and returned to the incubator.
Figure 8.
These schematic drawings depict the orthotopic PE-grafting technique used in avian embryos.
A–
D show details of the boxed area marked in the HH-stage 16 embryo on the left.
A–
C: Mounting of the PE-graft on a carrier membrane.
D: After implantation into the pericardial cavity of the host embryo, the membrane keeps the PE-graft in a position facilitating orthotopic attachment of the grafted PE to the heart of the host. The membrane, additionally, blocks the attachment of the host PE to the host heart. Abbreviations as used in
Figure 2.
Figure 8.
These schematic drawings depict the orthotopic PE-grafting technique used in avian embryos.
A–
D show details of the boxed area marked in the HH-stage 16 embryo on the left.
A–
C: Mounting of the PE-graft on a carrier membrane.
D: After implantation into the pericardial cavity of the host embryo, the membrane keeps the PE-graft in a position facilitating orthotopic attachment of the grafted PE to the heart of the host. The membrane, additionally, blocks the attachment of the host PE to the host heart. Abbreviations as used in
Figure 2.
Since its first publication in 1999, the orthotopic PE-grafting technique or modifications of this procedure have been used in several studies on avian embryos [
6,
10,
11,
12,
13,
29,
30,
31,
32,
33,
34]. The results of these experiments have confirmed and complemented the results of the earlier fate-mapping studies using “loose PE-grafting” techniques. The picture about the fate of the avian PE arising from these studies presents as follows: (1) the PE provides the epicardium and subepicardial mesenchyme of the heart, while the epicardium and subepicardial mesenchyme of the great arterial trunks derive from the pericardium at the arterial heart pole (
Figure 7,
Figure 9A) [
28,
32]. (2) PE- and epicardium-derived mesenchymal cells invade the subepicardial [
26], myocardial, and endocardial layers of the heart. They also colonize the atrio-ventricular endocardial cushions but do not colonize the endocardial outflow ridges [
11,
25,
28,
33]. (3) PE- and epicardium-derived mesenchymal cells provide the endothelium, smooth muscle cells, and fibroblast of the coronary blood vessels (
Figure 9) [
11,
12,
25,
26,
28,
29,
31]. (4) PE- and epicardium-derived mesenchymal cells do not substantially contribute to the formation of the cardiac lymphatic system [
34]. (5) PE- and epicardium-derived mesenchymal cells provide the majority of the subepicardial and intramyocardial fibroblasts, and contribute to the formation of the fibrous heart skeleton [
11,
12,
25,
26,
28,
29,
31]. (6) PE- and epicardium-derived mesenchymal cells do not contribute a substantial number of myocardioblasts to the developing avian heart (
Figure 9B) [
28].
Figure 9.
Results from PE-fate-mapping studies using the orthotopic PE-grafting technique for production of quail–chick chimeras. Quail donor cells (brown color) are immunostained with the anti-quail antibodies QCPN or QH1. (A,B) PE-derived cells start to form the main stems of the coronary arteries at HH-stage 34, when PE-derived vascular endothelial sprouts grow into the aortic valve sinus (A; QCPN staining). At HH-stage 36, PE-derived smooth muscle cells start to form the media (B; QCPN staining). Note also the border between PE-derived epicardium, which covers the heart, and non-PE-derived epicardium, which covers the great arterial trunks (arrow). (C,D) The grafted PE provides the coronary endothelium (C; QH1 endothelial staining), the smooth muscle cells of the coronary arteries and the majority of the intramyocardial mesenchyme (D; QCPN cell nucleus staining). Note that there is no immunostaining of myocardial cells, suggesting that the PE does not contribute a significant number of myocardioblast to the avian embryonic heart. Asterisk marks the orifice of the coronary artery. Ao: aorta; Ca: coronary artery.
Figure 9.
Results from PE-fate-mapping studies using the orthotopic PE-grafting technique for production of quail–chick chimeras. Quail donor cells (brown color) are immunostained with the anti-quail antibodies QCPN or QH1. (A,B) PE-derived cells start to form the main stems of the coronary arteries at HH-stage 34, when PE-derived vascular endothelial sprouts grow into the aortic valve sinus (A; QCPN staining). At HH-stage 36, PE-derived smooth muscle cells start to form the media (B; QCPN staining). Note also the border between PE-derived epicardium, which covers the heart, and non-PE-derived epicardium, which covers the great arterial trunks (arrow). (C,D) The grafted PE provides the coronary endothelium (C; QH1 endothelial staining), the smooth muscle cells of the coronary arteries and the majority of the intramyocardial mesenchyme (D; QCPN cell nucleus staining). Note that there is no immunostaining of myocardial cells, suggesting that the PE does not contribute a significant number of myocardioblast to the avian embryonic heart. Asterisk marks the orifice of the coronary artery. Ao: aorta; Ca: coronary artery.
![Jdb 01 00047 g009]()
Due to the high reliability of interspecies quail–chick grafting experiments, the results of such fate-mapping studies may be used as “reference standards” for the evaluation of the outcome of fate-mapping studies made in other vertebrate model systems, such as the mouse or the zebrafish. With respect to the PE, it was found that the results of genetic PE-fate mapping largely corresponded to those made in the aforementioned avian PE-grafting experiments. There seemed to exist, however, two exceptions. The first exception was a possible contribution of PE/epicardium-derived cells to the myocardium. While avian PE-grafting experiments did not provide any evidence for the contribution of a substantial number of PE-derived myocardioblast to the developing heart [
28], genetic PE-fate mapping of mouse PE/epicardium seemed to have identified PE/epicardium-derived cells as hitherto unrecognized myocardial progenitor cells in mouse embryos [
35,
36]. The divergent results have led to a discussion about the reliability of genetic PE-fate mapping [
37,
38,
39]. It was shown, for example, that the “epicardium-specific” Cre-line (TBX18) used in one of two aforementioned mouse studies [
35] has a limited expression in the myocardium of the “naked” embryonic heart [
37]. It is, therefore, questionable whether mouse PE really contributes a substantial number of myocardioblasts to the developing heart. The data from classical fate-mapping studies on avian embryos, as well as recent data from genetic PE-fate mapping in the zebrafish [
40], indicate that, in these species, the natural fates of PE-/epicardium-derived cells are limited to non-myocardial cell lineages. The second exception was the contribution of the PE to the endothelium of the coronary blood vessels. Most of the fate-mapping studies conducted on avian embryos provided evidence for a PE/epicardial origin of the coronary endothelium [
28,
31,
41,
42]. Genetic PE-fate mapping of mouse PE/epicardium using TBX18 and WT-1 Cre lines, on the other hand, did not provide any evidence for a significant contribution of PE-/epicardium-derived cells to the coronary endothelium [
35,
36]. Therefore, the origin of the coronary endothelium of the developing mouse heart has remained obscure until recently when marker analyses have shown that the mouse PE is a source of coronary endothelial cells [
43,
44].
5. Future Perspectives
During the initial phase of the research on the PE, the interest of researchers was mainly focused on classical themes of developmental biology, such as the fate or the functional significance of PE-derived cells. The easy accessibility and the possibility to carry out microsurgical experiments, such as mechanical PE-blocking or PE-grafting, made avian embryos attractive for use in studies addressing classical themes of developmental biology. During the past two decades, however, the focus of PE research has shifted from classical to molecular themes. This was accompanied by a shift in usage of model organisms from birds to mice and zebrafish. Although transgenic technologies are more advanced in the latter two systems at the present time, remarkable progress has been made in the development of methods for manipulating genetic and molecular functions in avian embryos during the past few years. Thus, chick and quail embryos are still regarded as attractive model systems for developmental biology, as well as regenerative medicine [
45,
46]. It is especially the possibility of combining modern molecular techniques, such as gene delivery via
in vivo electroporation, siRNA technology or transgenic quails [
47], with classical microsurgical techniques that make quail and chick embryos attractive for future research. With respect to the research on the PE and epicardium, it is, therefore, likely that the three aforementioned techniques for microsurgical manipulation of the avian PE may be used in future studies in combination with molecular techniques. The orthotopic PE-grafting technique, for example, can be used for the transplantation of PE from transgenic into wild type embryos or
vice versa. Such
in vivo experiments cannot be carried out in zebrafish or mice and may be very helpful for elucidating the function of genes involved in the dialog between the developing epicardium and myocardium, which controls the growth of the compact layer of ventricular myocardium (for reviews see [
48,
49]). Experiments in which the PE from green fluorescent protein-expressing transgenic chicken [
50] will be grafted into a wild-type host may facilitate the
in vivo visualization of the initial phase of epicardial development.