1. Introduction
Rheumatoid arthritis (RA) is a chronic autoimmune disease characterized by autoantibodies, systemic inflammation, persistent synovitis, and irreversible joint cartilage and bone destruction [
1]. Some extra-articular symptoms, such as pleuritis and rheumatoid vasculitis, can cause complications and poor prognosis of RA [
1]. To date, RA affects approximately 1% of the global population [
2]. However, the certain aetiology and the exact pathogenesis of RA remains obscure. Recently, clinical trials and epidemiological data have indicated that the interaction of environmental factors, genetic factors, autoimmunity, endocrine system, and infection contributes to the initiation and progression of RA [
1,
3,
4]. Among them, the dysregulation of innate and adaptive immune response, including aberrant production of immune cells and imbalance of cytokines, were widely demonstrated to participate in RA [
5,
6]. Abundant genetic variants were broadly confirmed to play major roles in RA [
1]. Recently, emerging evidence has revealed that a subset of noncoding RNAs (ncRNAs) also play pivotal roles in RA [
7].
It has been shown that merely 1–2% of human genome encode amino acids and 80% of phenotype-related loci map to noncoding regions across the human genome [
8,
9]. NcRNAs take up a significant portion of non-coding regions [
10]. Currently, the majority of research attention in thousands of ncRNAs coded by mammalian genome has shown that ncRNAs may play pivotal roles in diverse biological functions of RA [
7,
11]. For instance, miRNAs may promote numerous immune processes, and their dysregulation and implications in RA are well accepted [
7], such as miR-155 targeting TAB2 to activation of inflammatory responses [
12], miR-16 targeting 3′-untranslated region (UTR) of TNF-α to be a signature of disease activity [
13], miR-124a acting as a disease biomarker and a potential target for treatment [
14], etc. Importantly, research on another type of ncRNAs named lncRNAs in RA is on the rise. LncRNAs, which were initially considered to be a kind of nonfunctional RNAs, were actually capable of manipulating gene expression, such as gene transcription, RNA splicing, chromosome remodeling and protein transport [
11,
15]. Besides, lncRNAs have the ability to associate with RNA molecules to regulate their translation such as mRNA, miRNA, and interact with proteins to modulate their functions [
16]. For instance, linc-p21 was activated by P53 for cell apoptosis by binding to ribonucleoprotein-K [
17]. miR-21 and GAS5 may inhibit each other’s expression due to negative correlation between the two lncRNAs [
18]. Moreover, lncRNAs as important ceRNAs to impact the distribution of miRNAs on their targets also have received extensive attention [
19].
Increasing evidence has supported significant regulatory roles of lncRNAs in inflammatory responses, such as regulating the differentiation and activation of immune cells [
15]. In this term, several lncRNAs exhibited aberrant expression profiles in inflamed joint tissues of RA. Hotair is one of the earliest identified lncRNAs with abnormal expression in RA patients, it may interact with polycomb repressive complex 2 to regulate chromatin status [
20]. Compared with normal tissues, Hotair overexpressed in RA cartilages [
21]. Stuhlmüller et al. found that H19 expression was up-regulated in RA synovial tissue and might be induced by cytokines in synovial fibroblasts [
22]. Wu et al. showed that linc0597 (BZRAP1-AS1) decreased in peripheral blood mononuclear cells (PBMCs) of patients with RA [
23]. GAS5 originally discovered as a highly expressed ncRNA in growth-retarded cells was another aberrantly expressed lncRNA in RA [
24]. Mayama et al. demonstrated that GAS5 expression decreased in B cells and CD4 + T-cells of RA patients [
25]. Conversely, Moharamoghli et al. reported that GAS5 expression was up-regulated in T-cells of RA patients [
26]. Thus, compelling evidence showing the relevance of lncRNAs with RA is emerging, and in-depth research on the role of lncRNAs in RA is an urgent issue.
To further explore the implications of lncRNAs in RA, this study based on the available evidence hypothesized that H19, linc0597 and GAS5 may be involved in the pathogenesis of RA. A two-stage study of lncRNAs in RA was established. We aimed to investigate the three lncRNAs expression levels in PBMCs of RA patients, as well as explore the associations of lncRNAs expression and their gene single nucleotide polymorphisms (SNPs) with RA susceptibility.
4. Discussion
RA is a complex autoimmune disease resulting from multiple factors, as well as a clinical syndrome spanning several disease subsets [
1]. To the best of our knowledge, almost all of RA disease subsets present persistent synovial inflammation, associated bone, and articular cartilage damage [
1]. These disorders complicated with extra-articular diseases can damage any part of the body [
3]. Recent research has advanced the understanding of RA pathogenesis to the point where lncRNAs biological functions for RA pathogenetic process are underway, and the excavation of novel functions of thousands of lncRNAs promoted us to explore their affects in RA.
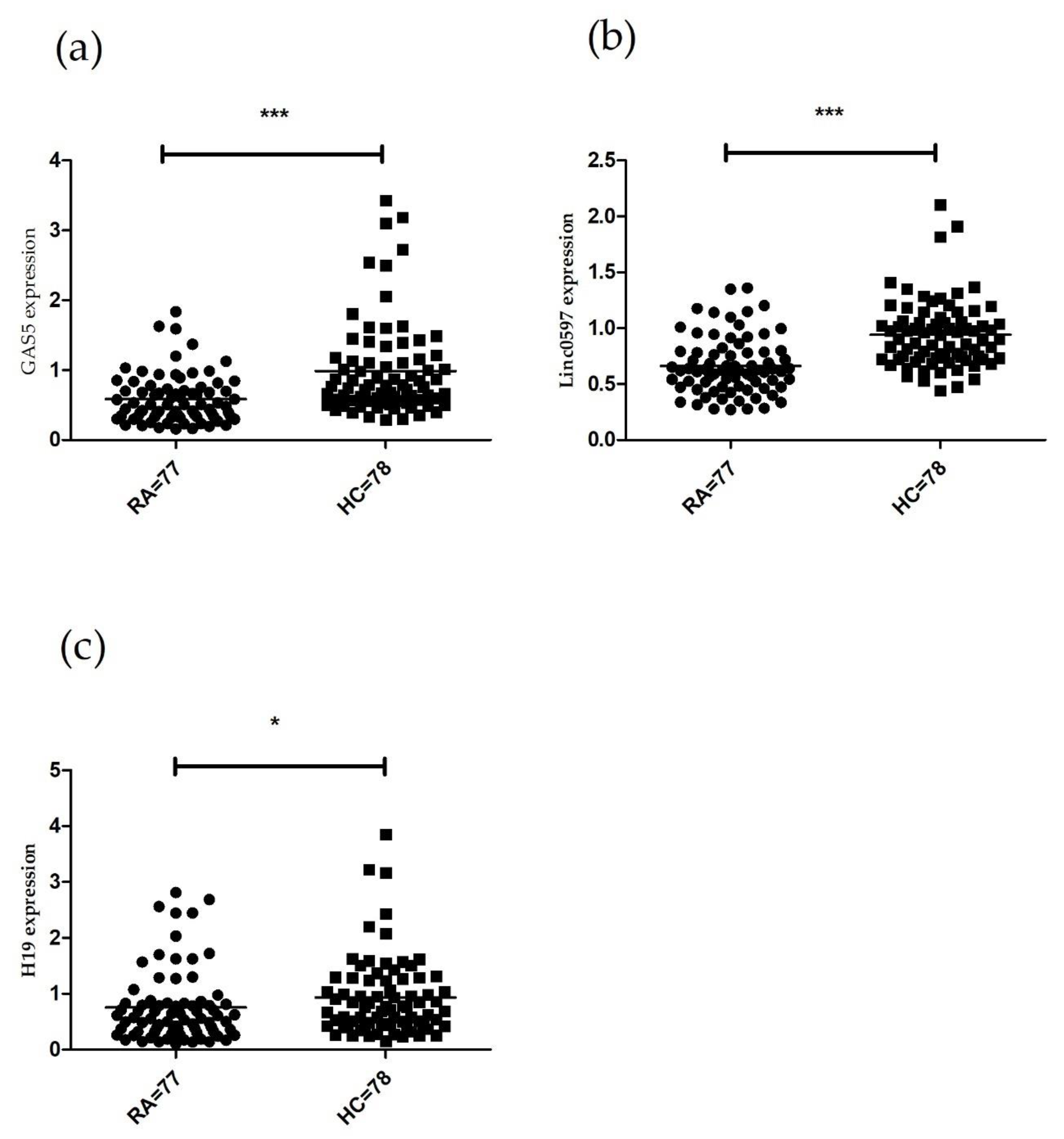
In the present study, we investigated three lncRNAs (H19, GAS5 and linc0597) expression profiles in PBMCs of patients with RA by qT-PCR in the first stage, and further analyzed the correlations between their expression and laboratory parameters of RA patients. The results demonstrated that the expression levels of H19, GAS5 and linc0597 were lower in RA patients. H19 could exert carcinogenesis as precursor of miRNA or ceRNA, such as precursor for miR-675 and modulating let-7 family of miRNAs [
29]. Metabolic stress-induced H19 and its encoded miR-675 could regulate inflammatory and hypoxic conditions by directly affecting COL2A1 [
30]. The expression of H19 in RA synovial fibroblasts was not only induced by cytokines/serum starvation but also correlated with mRNA expression for TIMP-2 [
22]. Taken together, it is fully suggested that H19 may play a pathogenic role in RA. However, Stuhlmüller et al. reported that H19 was overexpressed in RA synovial tissues [
22]. Song et al. indicated that H19 antisense expression was higher in PBMCs of patients with RA [
31]. However, our study showed that the expression level of H19 decreased in PBMCs of patients with RA. The possible explanation is that different locations in the genome of this lncRNA or lncRNAs expression possess tissue specificity. In addition, we also studied GAS5 in patients with RA. Indeed, related research on several other autoimmune diseases has shown its vital roles in a wide range of biological processes. GAS5 was reported to bind glucocorticoid receptor (GR) DNA-binding domain (DBD) and suppress GR-induced transcriptional activity, thereby inducing apoptosis [
32]. Most importantly, GAS5 was demonstrated to promote apoptosis and trigger growth arrest in human T-cells [
33]. All of the above are implicated in the pathogenesis of RA [
34]. Moreover, negative correlations between GAS5 expression and CRP level and hypocomplementemia in patients with RA were also found in our study. CRP is a product of inflammatory response, playing pro-inflammatory roles in RA by activating complements and inducing osteoclast differentiation, and acts as an indicator of treatment efficacy [
35]. Besides, complement system plays central roles in numerous protective immune processes [
36]. Complement C3 and C4 are also one of the most important indicators of RA disease activity [
36]. Thus, we speculate that GAS5 may play a pivotal role in the pathogenesis of RA, and its underlying mechanism is worth further exploration. Furthermore, it is generally acknowledged that TNF-α and IL-6 are documented in RA pathogenesis [
37,
38]. Linc0597 expression level was lower in THP1 macrophages and might regulate the induction of pro-inflammatory TNF-α and IL-6 [
39]. However, linc0597, which was previously found to be highly expressed in PBMCs of patients with RA [
23], showed low expression level in our study. One explanation is that a different internal control was used between two studies. The other explanation is that linc0597 expression may be affected by clinical treatment.
Then, gene polymorphisms of differentially expressed lncRNAs (rs2067051 and rs2075745 for H19; rs6790, rs16847206, and rs6692753 for GAS5; rs2070107, rs2877877, rs2632516, rs2285991, rs8071916, rs12601867, rs13414, rs2680700, and rs4372750 for linc0597) with RA susceptibility were detected in the second stage. Among these fourteen SNPs, rs12601867, rs13414, rs2680700, rs4372750, rs16847206, rs8071916, and rs6692753 were first reported in this study, and rs6790 genotype (GA) and rs2680700 genotype (GT) were found to be associated with RA risk. Previous studies prompted that certain functional SNPs within the promoter region of GAS5 could regulation its expression, and were related to risk of cancers, among which rs6790 were suggested as a biomarker for chemoradiotherapy induced toxic reactions in nasopharyngeal cancer patients [
40,
41,
42]. CHIP-seq data showed that rs6790 was labeled as an active promoter region or enhancer region position, and silico analysis suggested that this SNP also possessed a significant feature of e-QTL, both of them verified its contribution in the genetic variance of GAS5 expression [
41]. Similarly, rs6790 was reported to play inhibitory functions of GAS5 on the transcriptional activity of GR in autoimmune disease [
43], GAS5 knock-down could attenuate the progression of experimental autoimmune encephalomyelitis (EAE) and promote remyelination [
44]. Since no studies reported on linc0597 rs2680700, we tried to explore its potential functions through bioinformatics databases. rs2680700 is also labeled as an enhancer region according to ChIP-seq data, and it has a strong feature of e-QTL in several tissues (
Table S4), suggesting that rs2680700 might exert their roles through affecting the expression of target genes. Taken together, these data provide evidence for functional contribution of rs2680700 in changing linc0597 expression or stability. However, the exact roles of these two SNPs in RA still need further exploration [
45,
46]. Additionally, we attempted to discover the correlations of H19 expression with their gene polymorphisms in RA patients, but still no statistically significant results were found. Similarly, Huang et al. and Zhou et al. showed that H19 (rs2839698, rs3741219 and rs217727) gene polymorphisms were not related to RA risk in a Chinese population [
47,
48], indicating that there might be other mechanisms for H19 in RA pathogenesis.
Presently, more and more studies reveal the impact of lncRNAs on the pathogenesis of RA. There are evidence that lncRNAs regulate sphingomyelin phosphodiesterase 1 (SMPD1), and SMPD1 can regulate differentiation and apoptosis in T-cells [
49]. Additionally, Hotair contributes to RA pathogenesis through activation of matrix metalloproteinases 2 (MMP-2) and MMP-13 in synoviocytes and osteoclasts [
31], and MMPs play vital roles in recruitment of inflammatory cells and degradation of cartilage and bone of RA [
50]. Linc-p21 was confirmed to be negative regulators of NF-κB activity, and NF-κB is a pro-inflammatory transcription factor of RA patients [
51]. At present, emerging evidence proved that lncRNAs might regulate transcript levels of genes in the same genomic region, for instance, lncRNA C5T1 located in TRAF1-C5 influenced C5 mRNA level in RA risk [
52]. Last but not least, the regulation of lncRNAs might be specific in cells of innate immune system [
53]. For instance, Müller et al. reported that 7419 lncRNAs were detected in CD14+ monocytes isolated before and after IL-6 or TNF-α inhibition in RA, among them, only 85 lncRNAs examined exhibited up-regulation and down-regulation by anti-cytokine treatment initiated, and none of lncRNAs identified showed a similar pattern in response to IL-6 versus TNF-α inhibition [
53]. Overall, lncRNAs are emerging as key regulators of activation, differentiation, and expression of immune cells, which may direct or indirect link to cellular and tissue homeostasis of autoimmunity [
54,
55].
Several limitations should be acknowledged in our study. Due to the lack of data on non-steroidal anti-inflammatory drug (NSAIDs), the effects of NSAIDs on the expression of lncRNAs in RA patients might not be known. In addition, this is a hospital-based case-control study, so that a causal relationship was difficultly determined between RA risk and lncRNAs expression.