Prevalence and Determinants of Antibiotic Self-Medication among Adult Patients with Respiratory Tract Infections in the Mboppi Baptist Hospital, Douala, Cameroon: A Cross-Sectional Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design and Setting
2.2. Study Participants and Sampling
2.3. Sample Size
2.4. Study Procedure
Administrative and Ethical Considerations
2.5. Data Collection
2.6. Data Management and Analysis
3. Results
3.1. Characteristics of the Study Population
3.2. Prevalence of Antibiotic Self-Medication
3.3. Factors Associated with Antibiotic Self-Medication
4. Discussion
4.1. Study Limitations
4.2. Policy Implications
5. Conclusions
Ethical Approval and Consent to Participate
Consent for Publication
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Goossens, H.; Ferech, M.; Vander Stichele, R.; Elseviers, M.; ESAC Project Group. Outpatient antibiotic use in Europe and association with resistance: A cross-national database study. Lancet 2005, 365, 579–587. [Google Scholar] [CrossRef]
- Van Boeckel, T.P.; Gandra, S.; Ashok, A.; Caudron, Q.; Grenfell, B.T.; Levin, S.A.; Laxminarayan, R. Global antibiotic consumption 2000 to 2010: An analysis of national pharmaceutical sales data. Lancet Infect. Dis. 2014, 14, 742–750. [Google Scholar] [CrossRef]
- Guidelines for the Regulatory Assessment of Medicinal Products for Use in Self-Medication. Available online: http://apps.who.int/medicinedocs/en/d/Js2218e/ (accessed on 19 March 2018).
- Grigoryan, L.; Haaijer-Ruskamp, F.M.; Burgerhof, J.G.M.; Mechtler, R.; Deschepper, R.; Tambic-Andrasevic, A.; Andrajati, R.; Monnet, D.L.; Cunney, R.; Di Matteo, A.; et al. Self-medication with antimicrobial drugs in Europe. Emerging Infect. Dis. 2006, 12, 452–459. [Google Scholar] [CrossRef] [PubMed]
- Blenkinsopp, A.; Bradley, C. Over the Counter Drugs: Patients, society, and the increase in self medication. BMJ 1996, 312, 629–632. [Google Scholar] [CrossRef] [PubMed]
- Nsimba, S.E.D.; Rimoy, G.H. Self-medication with chloroquine in a rural district of Tanzania: A therapeutic challenge for any future malaria treatment policy change in the country. J. Clin. Pharm. Ther. 2005, 30, 515–519. [Google Scholar] [CrossRef] [PubMed]
- Chipwaza, B.; Mugasa, J.P.; Mayumana, I.; Amuri, M.; Makungu, C.; Gwakisa, P.S. Self-medication with anti-malarials is a common practice in rural communities of Kilosa district in Tanzania despite the reported decline of malaria. Malar. J. 2014, 13, 252. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- World Health Organization. The World Drug situation. Available online: http://who.int/iris/handle/10665/37781 (1998).
- Vanden Eng, J.; Marcus, R.; Hadler, J.L.; Imhoff, B.; Vugia, D.J.; Cieslak, P.R.; Zell, E.; Deneen, V.; McCombs, K.G.; Zansky, S.M.; et al. Consumer attitudes and use of antibiotics. Emerging Infect. Dis. 2003, 9, 1128–1135. [Google Scholar] [CrossRef] [PubMed]
- Muras, M.; Krajewski, J.; Nocun, M.; Godycki-Cwirko, M. A survey of patient behaviours and beliefs regarding antibiotic self-medication for respiratory tract infections in Poland. Arch. Med. Sci. 2013, 9, 854–857. [Google Scholar] [CrossRef] [PubMed]
- Ivanovska, V.; Zdravkovska, M.; Bosevska, G.; Angelovska, B. Antibiotics for upper respiratory infections: Public knowledge, beliefs and self-medication in the Republic of Macedonia. Pril 2013, 34, 59–70. [Google Scholar]
- Grigoryan, L.; Burgerhof, J.G.M.; Degener, J.E.; Deschepper, R.; Lundborg, C.S.; Monnet, D.L.; Scicluna, E.A.; Birkin, J.; Haaijer-Ruskamp, F.M. Self-Medication with Antibiotics and Resistance (SAR) Consortium. Determinants of self-medication with antibiotics in Europe: The impact of beliefs, country wealth and the healthcare system. J. Antimicrob. Chemother. 2008, 61, 1172–1179. J. Antimicrob. Chemother. 2008, 61, 1172–1179. [Google Scholar] [CrossRef] [PubMed]
- Hughes, C.M.; McElnay, J.C.; Fleming, G.F. Benefits and risks of self medication. Drug Saf. 2001, 24, 1027–1037. [Google Scholar] [CrossRef] [PubMed]
- Ruiz, M.E. Risks of self-medication practices. Curr. Drug Saf. 2010, 5, 315–323. [Google Scholar] [CrossRef] [PubMed]
- Lansang, M.A.; Lucas-Aquino, R.; Tupasi, T.E.; Mina, V.S.; Salazar, L.S.; Juban, N.; Limjoco, T.T.; Nisperos, L.E.; Kunin, C.M. Purchase of antibiotics without prescription in Manila, the Philippines. Inappropriate choices and doses. J. Clin. Epidemiol. 1990, 43, 61–67. [Google Scholar] [CrossRef]
- Saradamma, R.D.; Higginbotham, N.; Nichter, M. Social factors influencing the acquisition of antibiotics without prescription in Kerala State, south India. Soc. Sci. Med. 2000, 50, 891–903. [Google Scholar] [CrossRef]
- Radyowijati, A.; Haak, H. Improving antibiotic use in low-income countries: An overview of evidence on determinants. Soc. Sci. Med. 2003, 57, 733–744. [Google Scholar] [CrossRef]
- Promoting Appropriate Drug Use in Missionary Health Facilities in Cameroon—EDM Research Series No. 028: 3. Literature review. Available online: http://apps.who.int/medicinedocs/en/d/Js2238e/6.html (accessed on 20 May 2016).
- Agbor, M.A.; Azodo, C.C. Self medication for oral health problems in Cameroon. Int. Dent. J. 2011, 61, 204–209. [Google Scholar] [CrossRef] [PubMed]
- Charan, J.; Biswas, T. How to Calculate Sample Size for Different Study Designs in Medical Research? Indian J. Psychol. Med. 2013, 35, 121–126. [Google Scholar] [CrossRef] [PubMed]
- 6A.10 - Adjustment Factors for Sample Size Calculations | STAT 509. Available online: https://onlinecourses.science.psu.edu/stat509/node/47 (accessed on 19 March 2018).
- Upper Respiratory Tract Infection Symptoms, Treatment & Contagious. MedicineNet. Available online: https://www.medicinenet.com/upper_respiratory_infection/article.htm (accessed on 16 March 2018).
- Sawair, F.; Baqain, Z.; Abukaraky, A.; Abu Eid, R. Assessment of Self-Medication of Antibiotics in a Jordanian Population. Med. Princ. Pract. 2009, 18, 21–25. [Google Scholar] [CrossRef] [PubMed]
- Godycki-Cwirko, M.; Hood, K.; Nocun, M.; Muras, M.; Goossens, H.; Butler, C.C. Presentation, antibiotic management and associated outcome in Polish adults presenting with acute cough/LRTI. Fam. Prac. 2011, 28, 608–614. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Belkina, T.; Al Warafi, A.; Eltom, E.; Tadjieva, N.; Aleš Antonín, K.; Vlcek, J. Antibiotic use and knowledge in the community of Yemen, Saudi Arabia, and Uzbekistan. J. Infect. Dev. Ctry. 2014, 8, 424–429. [Google Scholar] [CrossRef] [PubMed]
- Al Rasheed, A.; Yagoub, U.; Alkhashan, H.; Abdelhay, O.; Alawwad, A.; Al Aboud, A.; Al Battal, S. Prevalence and Predictors of Self-Medication with Antibiotics in Al Wazarat Health Center, Riyadh City, KSA. BioMed Research International. 2016. Available online: https://www.hindawi.com/journals/bmri/2016/3916874/ (accessed on 19 February 2018).
- Pereko, D.D.; Lubbe, M.S.; Essack, S.Y. Public knowledge, attitudes and behaviour towards antibiotic usage in Windhoek, Namibia. South. Afr. J. Infect. Dis. 2015, 30, 134–137. [Google Scholar] [CrossRef]
- Widayati, A.; Suryawati, S.; de Crespigny, C.; Hiller, J.E. Self medication with antibiotics in Yogyakarta City Indonesia: A cross sectional population-based survey. BMC Res. Notes 2011, 4, 491. [Google Scholar] [CrossRef] [PubMed]
- Vialle-Valentin, C.E.; LeCates, R.F.; Zhang, F.; Desta, A.T.; Ross-Degnan, D. Predictors of antibiotic use in African communities: Evidence from medicines household surveys in five countries. Trop. Med. Int. Health 2012, 17, 211–222. [Google Scholar] [CrossRef] [PubMed]
- Alfalogy, E.H.; Nafadi, H.B.; al Rehaili, S.H.; L-Harbi, B.A.A. Prevalence and Predictors of Self-Medication with Antibiotics for Children in Makkah, Saudi Arabia. Eur. J. Prev. Med. 2017, 5, 60–64. [Google Scholar] [CrossRef]
- Kibuule, D.; Kagoya, H.R.; Godman, B. Antibiotic use in acute respiratory infections in under-fives in Uganda: Findings and implications. Expert Rev. Anti-Infect. Ther. 2016, 14, 863–872. [Google Scholar] [CrossRef] [PubMed] [Green Version]
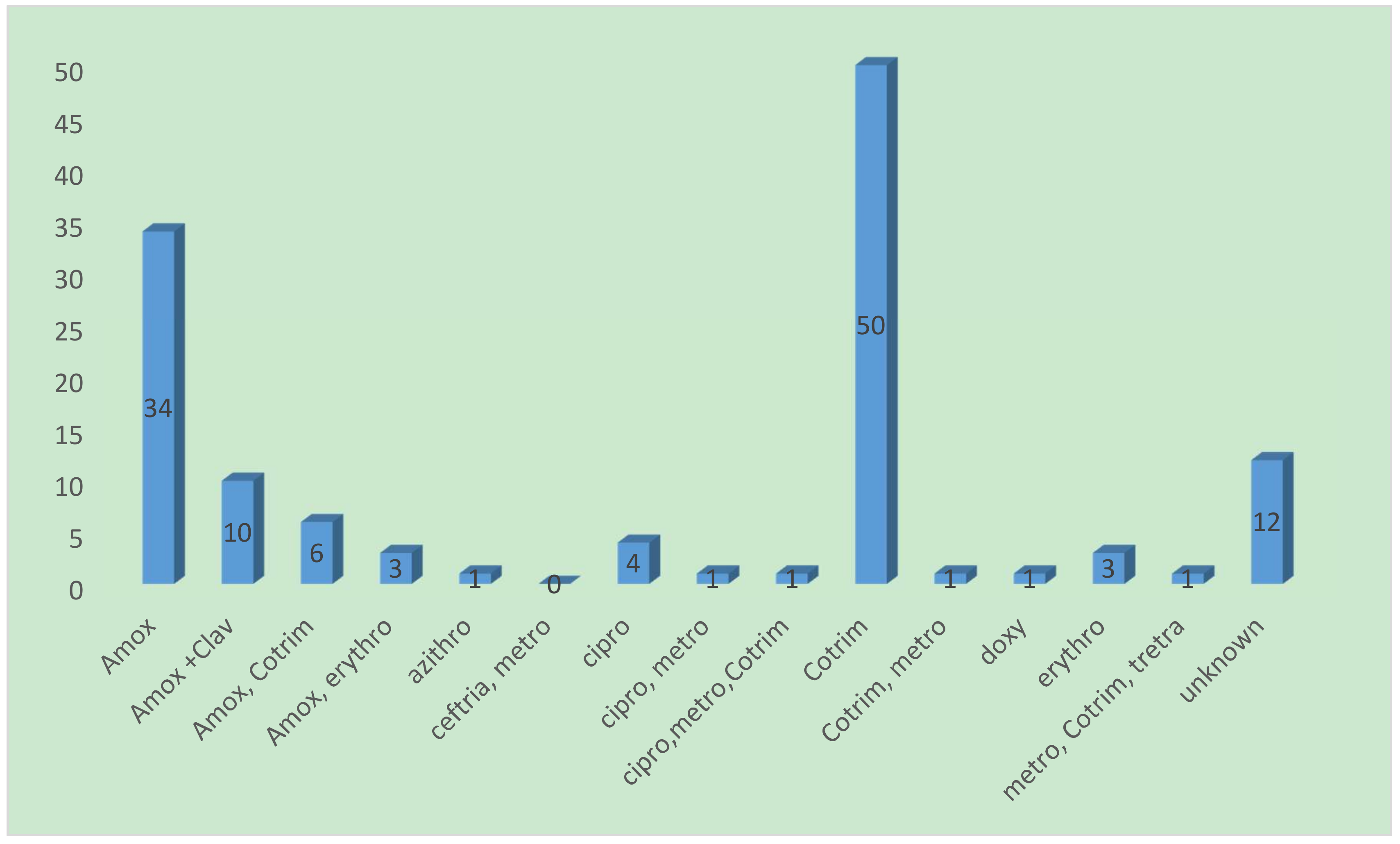
| Variable | Frequency (%) |
|---|---|
| Age (years) | |
| Median (IQR) ** | 35 (27–49) |
| Sex | |
| Male | 138 (44.8%) |
| Female | 170 (55.2%) |
| Occupation | |
| Employed | 152 (49.4%) |
| Unemployed | 156 (50.6%) |
| Marital Status | |
| Married | 140 (45.6%) |
| Single | 133 (43.3%) |
| Widowed | 25 (8.1%) |
| Divorced | 9 (2.9%) |
| Level of Education | |
| None | 29 (9.4%) |
| Primary | 102 (33.1%) |
| Secondary/High school | 124 (40.3%) |
| Tertiary/University | 53 (17.2%) |
| Residence | |
| Urban | 247 (80.2%) |
| Rural | 61 (19.8%) |
| Alcohol(yes) | 150 (48.7%) |
| Smoking(yes) | 32 (10.4%) |
| History of pulmonary tuberculosis (yes) | 26 (8.4%) |
| Variables | Frequency(%) |
|---|---|
| Source of self-medication antibiotic | |
| Home | 10 (7.7%) |
| Friends/relatives | 14 (10.9%) |
| Pharmacy | 80 (62.0%) |
| Chemist | 25 (19.4%) |
| Variable | Antibiotic Self-Medication Frequency (%) | |||
|---|---|---|---|---|
| Yes | No | Total | p-value | |
| Age (years) | 0.289 | |||
| ≤35 | 62 (39%) | 97 (61%) | 159 (51.6%) | |
| >35 | 67 (45%) | 82 (55%) | 149 (48.4%) | |
| Gender | 0.266 | |||
| Male | 53 (38.4%) | 85 (61.6%) | 138 (44.8%) | |
| Female | 76 (44.7%) | 94 (55.3%) | 170 (55.2%) | |
| Residence | 0.318 | |||
| Urban | 100 (40.5%) | 147 (59.5%) | 247 (80.2%) | |
| Rural | 29 (47.5%) | 32 (52.5%) | 61 (19.8%) | |
| Level of Education | 0.464 | |||
| Primary and above | 115(41.2%) | 164(58.8%) | 279(90.6%) | |
| None | 14(48.3%) | 15(51.7%) | 29(9.4%) | |
| Occupation | 0.539 | |||
| Employed | 61 (40.1%) | 91 (59.9%) | 152 (49.4%) | |
| Unemployed | 68 (43.6%) | 88 (56.4%) | 156 (50.6%) | |
| Marital status | 0.462 | |||
| Married/cohabiting | 62 (44.3%) | 78 (55.7%) | 140 (45.6%) | |
| Single/divorced/widowed | 67 (40.1%) | 100 (59.9%) | 167 (54.4%) | |
| Alcohol | 0.515 | |||
| Yes | 60 (40.0%) | 90 (60.0%) | 150 (48.7%) | |
| No | 69 (43.7%) | 89 (56.3%) | 158 (51.3%) | |
| Smoking | 0.879 | |||
| Yes | 13 (40.6%) | 19 (59.4%) | 26 (8.4%) | |
| No | 116 (42.0%) | 160 (58.0%) | 276 (89.6%) | |
| History of PTB * | 0.043 | |||
| Yes | 6 (23.1%) | 20 (76.9%) | 26 (8.4%) | |
| No | 123 (43.6%) | 159 (56.4%) | 282 (91.6%) | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ngu, R.C.; Feteh, V.F.; Kika, B.T.; F., E.K.N.; Ayeah, C.M.; Chifor, T.; Njim, T.; Fankem, A.M.; Yengo, F.K.F. Prevalence and Determinants of Antibiotic Self-Medication among Adult Patients with Respiratory Tract Infections in the Mboppi Baptist Hospital, Douala, Cameroon: A Cross-Sectional Study. Diseases 2018, 6, 49. https://doi.org/10.3390/diseases6020049
Ngu RC, Feteh VF, Kika BT, F. EKN, Ayeah CM, Chifor T, Njim T, Fankem AM, Yengo FKF. Prevalence and Determinants of Antibiotic Self-Medication among Adult Patients with Respiratory Tract Infections in the Mboppi Baptist Hospital, Douala, Cameroon: A Cross-Sectional Study. Diseases. 2018; 6(2):49. https://doi.org/10.3390/diseases6020049
Chicago/Turabian StyleNgu, Roland Cheofor, Vitalis Fambombi Feteh, Belmond Tse Kika, Emade Ketchemen Nerice F., Chia Mark Ayeah, Theresia Chifor, Tsi Njim, Alvine Manuela Fankem, and Franklin Kwenti Fai Yengo. 2018. "Prevalence and Determinants of Antibiotic Self-Medication among Adult Patients with Respiratory Tract Infections in the Mboppi Baptist Hospital, Douala, Cameroon: A Cross-Sectional Study" Diseases 6, no. 2: 49. https://doi.org/10.3390/diseases6020049





