Hierarchically Self-Assembled Nanofiber Films from Amylose-Grafted Carboxymethyl Cellulose
Abstract
:1. Introduction
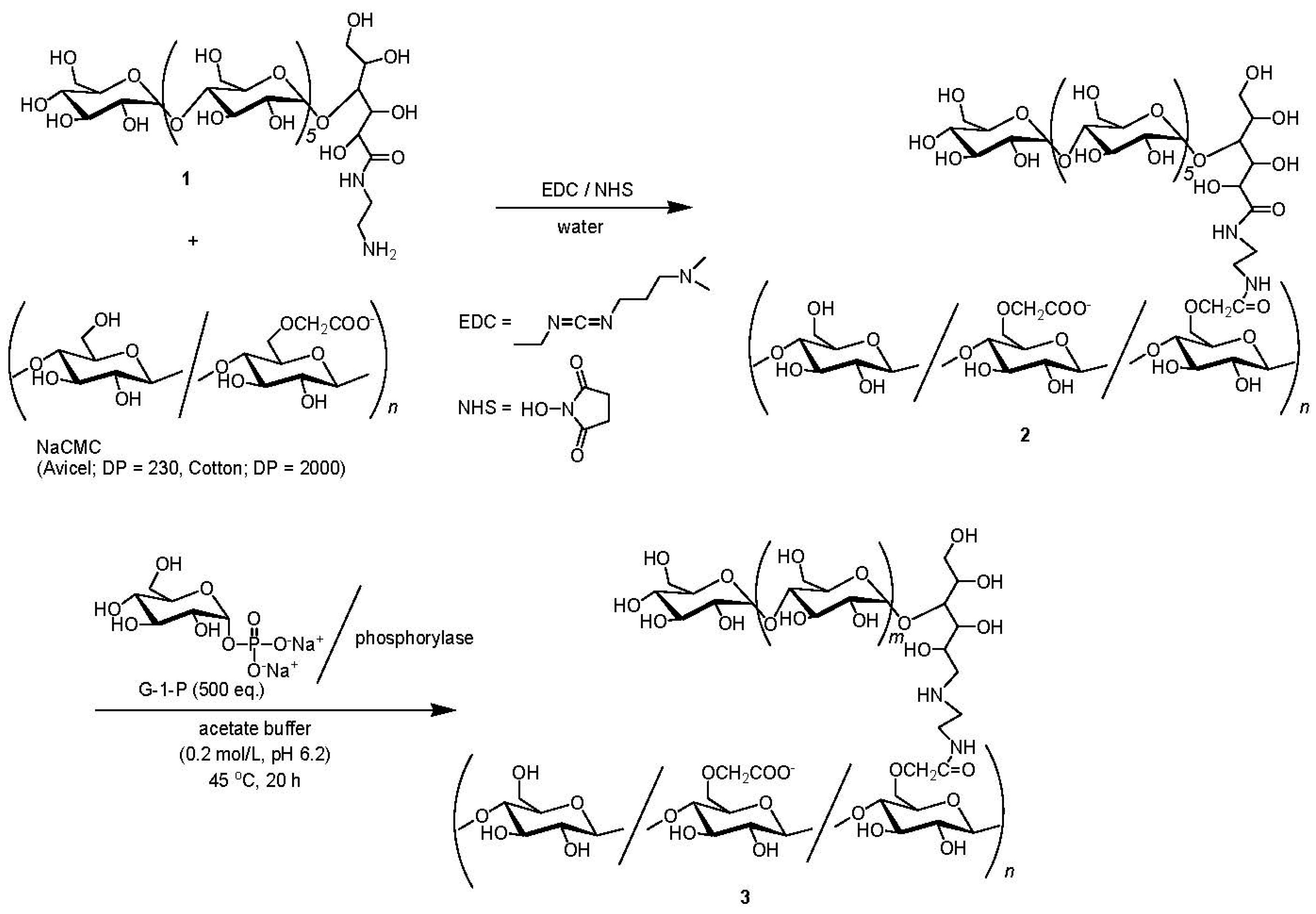
2. Experimental Section
2.1. Materials
2.2. Synthesis of Maltooligosaccharide-Grafted NaCMC (2)
2.3. Synthesis of Amylose-Grafted NaCMC (3)
2.4. Formation of Nanofiber Film from 3
2.5. Measurements
3. Results and Discussion
3.1. Chemoenzymatic Synthesis of Amylose-Grafted NaCMC 3
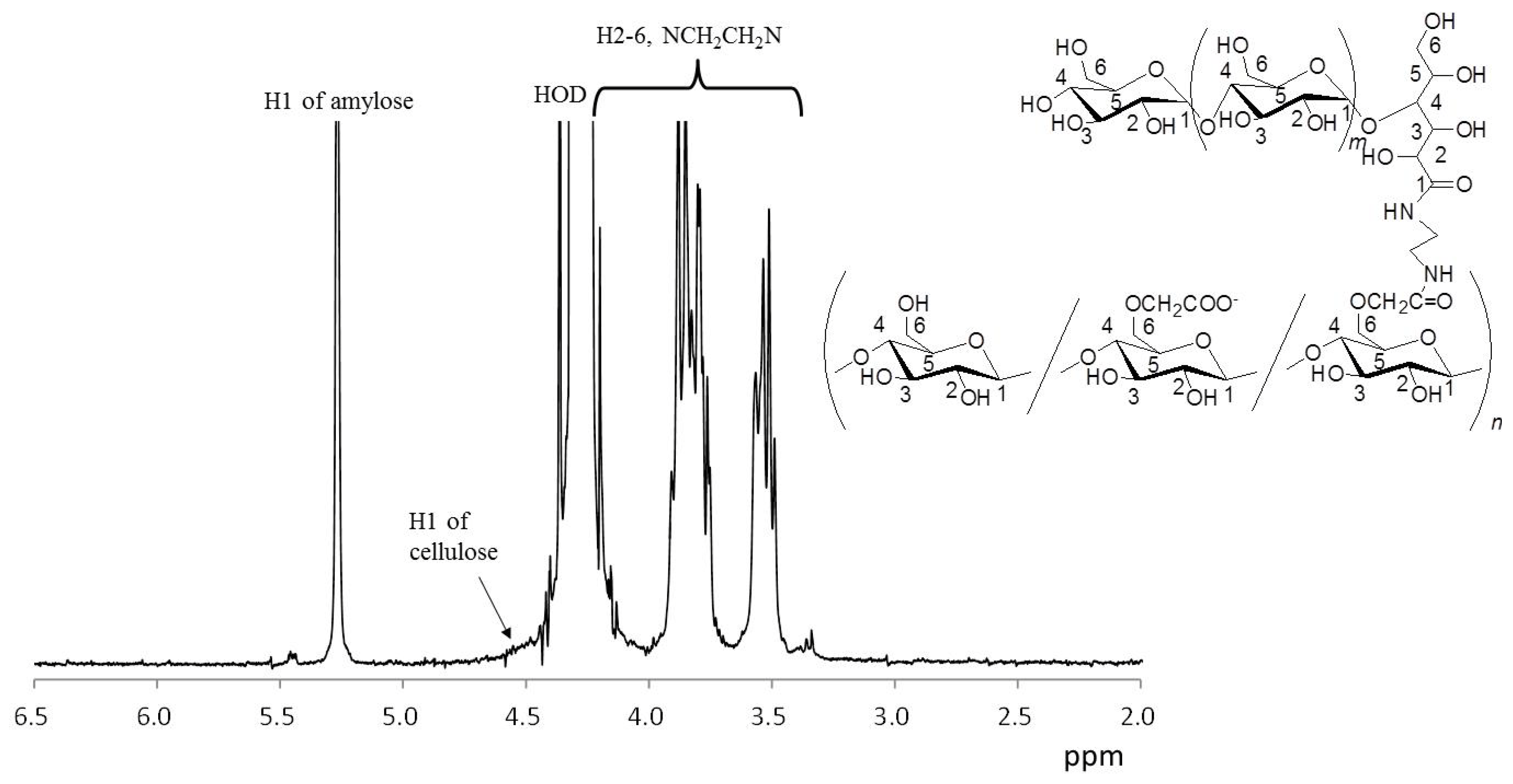
3.2. Formation and Characterization of Self-Assembled Nanofiber Films from 3




4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Klemm, D.; Heublein, B.; Fink, H.-P.; Bohn, A. Cellulose: Fascinating biopolymers and sustainable raw material. Angew. Chem. Int. Ed. 2005, 44, 3358–3393. [Google Scholar] [CrossRef]
- Klemm, D.; Kramer, F.; Moritz, S.; Lindström, T.; Ankerfors, M.; Gray, D.; Dorris, A. Nanocelluloses: A new family of nature-based materials. Angew. Chem. Int. Ed. 2011, 50, 5438–5466. [Google Scholar] [CrossRef]
- Beglou, M.J.; Haghi, A.K. Electrospun biodegdadable and biocompatible natural nanofibers: A detailed review. Cellul. Chem. Technol. 2008, 42, 441–462. [Google Scholar]
- Isogai, A.; Saito, T.; Fukuzumi, H. TEMPO-oxidized cellulose nanofibers. Nanoscale 2011, 3, 71–85. [Google Scholar] [CrossRef]
- Abdul Khalil, H.P.S.; Bhat, A.H.; Ireana Yusra, A.F. Green composites from sustainable cellulose nanofibrils: A review. Carbohydr. Polym. 2012, 87, 963–979. [Google Scholar] [CrossRef]
- Saito, T.; Nishiyama, Y.; Putaux, J.L.; Vignon, M.; Isogai, A. Homogeneous suspensions of individualized microfibrils from TEMPO-catalyzed oxidation of native cellulose. Biomacromolecules 2006, 7, 1687–1691. [Google Scholar] [CrossRef]
- Saito, T.; Kimura, S.; Nishiyama, Y.; Isogai, A. Cellulose nanofibers prepared by TEMPO-mediated oxidation of native cellulose. Biomacromolecules 2007, 8, 2485–2491. [Google Scholar] [CrossRef]
- Abe, K.; Iwamoto, S.; Yano, H. Obtaining cellulose nanofibers with a uniform width of 15 nm from wood. Biomacromolecules 2007, 8, 3276–3278. [Google Scholar] [CrossRef]
- Kadokawa, J.; Arimura, T.; Takemoto, Y.; Yamamoto, K. Self-assembly of amylose-grafted carboxymethyl cellulose. Carbohydr. Polym. 2012, 90, 1371–1377. [Google Scholar] [CrossRef]
- Stephen, A.M.; Philips, G.O.; Williams, P.A. Food Polysaccharides and Their Applications; Taylor & Francis: London, UK, 1995. [Google Scholar]
- Matsuda, S.; Kaneko, Y.; Kadokawa, J. Chemoenzymatic synthesis of amylose-grafted chitosan. Macromol. Rapid Commun. 2007, 28, 863–867. [Google Scholar] [CrossRef]
- Kaneko, Y.; Matsuda, S.; Kadokawa, J. Chemoenzymatic syntheses of amylose-grafted chitin and chitosan. Biomacromolecules 2007, 8, 3959–3964. [Google Scholar] [CrossRef]
- Omagari, Y.; Matsuda, S.; Kaneko, Y.; Kadokawa, J. Chemoenzymatic synthesis of amylose-grafted cellulose. Macromol. Biosci. 2009, 9, 450–455. [Google Scholar] [CrossRef]
- Kaneko, Y.; Kadokawa, J. Chemoenzymatic Synthesis of Amylose-Grafted Polymers. In Handbook of Carbohydrate Polymers: Development, Properties and Applications; Ito, R., Matsuo, Y., Eds.; Nova Science Publishers, Inc.: Hauppauge, NY, USA, 2009; Chapter 23; pp. 671–691. [Google Scholar]
- Omagari, Y.; Kaneko, Y.; Kadokawa, J. Chemoenzymatic synthesis of amylose-grafted alginate and its formation of enzymatic disintegratable beads. Carbohydr. Polym. 2010, 82, 394–400. [Google Scholar] [CrossRef]
- Arimura, T.; Omagari, Y.; Yamamoto, K.; Kadokawa, J. Chemoenzymatic synthesis and hydrogelation of amylose-grafted xanthan gums. Int. J. Biol. Macromol. 2011, 49, 498–503. [Google Scholar] [CrossRef]
- Omagari, Y.; Kadokawa, J. Synthesis of heteropolysaccharides having amylose chains using phosphorylase-catalyzed enzymatic polymerization. Kobunshi Ronbunshu 2011, 68, 242–249. [Google Scholar] [CrossRef]
- Kadokawa, J. Synthesis of Amylose-Grafted Polysaccharide Materials by Phosphorylase-Catalyzed Enzymatic Polymerization. In Biobased Monomers, Polymers, and Materials; Smith, P.B., Gross, R.A., Eds.; ACS Symposium Series: Washington, DC, USA, 2012; Chapter 15; pp. 237–255. [Google Scholar]
- Kadokawa, J.; Kaneko, Y. Engineering of Polysaccharide Materials—By Phosphorylase-Catalyzed Enzymatic Chain-Elongation; Pan Stanford Publishing Pte. Ltd.: Singapore, Singapore, 2013. [Google Scholar]
- Ziegast, G.; Pfannemüller, B. Phosphorolytic syntheses with di-, oligo- and multi-functional primers. Carbohydr. Res. 1987, 160, 185–204. [Google Scholar] [CrossRef]
- Kitaoka, M.; Hayashi, K. Carbohydrate-processing phosphorolytic enzymes. Trends Glycosci. Glycotechnol. 2002, 14, 35–50. [Google Scholar] [CrossRef]
- Fujii, K.; Takata, H.; Yanase, M.; Terada, Y.; Ohdan, K.; Takaha, T.; Okada, S.; Kuriki, T. Bioengineering and application of novel glucose polymers. Biocatal. Biotransform. 2003, 21, 167–172. [Google Scholar]
- Yanase, M.; Takaha, T.; Kuriki, T. α-Glucan phosphorylase and its use in carbohydrate engineering. J. Food Agric. 2006, 86, 1631–1635. [Google Scholar] [CrossRef]
- Ohdan, K.; Fujii, K.; Yanase, M.; Takaha, T.; Kuriki, T. Enzymatic synthesis of amylose. Biocatal. Biotransform. 2006, 24, 77–81. [Google Scholar]
- Seibel, J.; Jördening, H.-J.; Buchholz, K. Glycosylation with activated sugars using glycosyltransferases and transglycosidases. Biocatal. Biotranform. 2006, 24, 311–342. [Google Scholar]
- Kobayashi, S.; Makino, A. Enzymatic polymer synthesis: An opportunity for green polymer chemistry. Chem. Rev. 2009, 109, 5288–5353. [Google Scholar] [CrossRef]
- Kadokawa, J.; Kobayashi, S. Polymer synthesis by enzymatic catalysis. Curr. Opin. Chem. Biol. 2010, 14, 145–153. [Google Scholar] [CrossRef]
- Sasaki, M.; Sekiguchi, G.; Adschiri, T.; Arai, K. Rapid and selective conversion of cellulose to valuable chemical intermediates using supercritical water. In Proceedings of the 6th International Symposium on Supercritical Fluids, 2003, Versailles, France, 28–30 April 2003; Volume 2, pp. 1417–1422.
- Khullar, R.; Varshney, V.K.; Naithani, S.; Heinze, T.; Soni, P.L. Carboxymethylation of cellulosic material (average degree of polymerization 2600) isolated from cotton (Gossypium) linters with respect to degree of substitution and rheological behavior. J. Appl. Polym. Sci. 2005, 96, 1477–1482. [Google Scholar] [CrossRef]
- Olaru, N.; Olaru, L. Influence of organic diluents on cellulose carboxymethylation. Macromol. Chem. Phys. 2001, 202, 207–211. [Google Scholar] [CrossRef]
- Hosokawa, K.; Hanada, N.; Sato, S. Cationized carboxymethyl cellulose sodium salts with good chemical resistance and dispersibility. Japanese Patent 2002-201202; Filed 28 December 2000, Issued 19 July 2002,
- Bhuiyan, S.H.; Rus’d, A.A.; Kitaoka, M.; Hayashi, K. Characterization of a hyperthermostable glycogen phosphorylase from Aquifex aeolicus expressed in Escherichia coli. J. Mol. Catal. B: Enzym. 2003, 22, 173–180. [Google Scholar] [CrossRef]
- Yanase, M.; Takata, H.; Fujii, K.; Takaha, T.; Kuriki, T. Cumulative effect of amino acid replacements results in enhanced thermostability of potato type L α-glucan phosphorylase. Appl. Environ. Microbiol. 2003, 71, 5433–5439. [Google Scholar]
- Zobel, H.F. Starch crystal transformations and their industrial importance. Starch 1988, 40, 1–7. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hatanaka, D.; Takemoto, Y.; Yamamoto, K.; Kadokawa, J.-i. Hierarchically Self-Assembled Nanofiber Films from Amylose-Grafted Carboxymethyl Cellulose. Fibers 2014, 2, 34-44. https://doi.org/10.3390/fib2010034
Hatanaka D, Takemoto Y, Yamamoto K, Kadokawa J-i. Hierarchically Self-Assembled Nanofiber Films from Amylose-Grafted Carboxymethyl Cellulose. Fibers. 2014; 2(1):34-44. https://doi.org/10.3390/fib2010034
Chicago/Turabian StyleHatanaka, Daisuke, Yasutaka Takemoto, Kazuya Yamamoto, and Jun-ichi Kadokawa. 2014. "Hierarchically Self-Assembled Nanofiber Films from Amylose-Grafted Carboxymethyl Cellulose" Fibers 2, no. 1: 34-44. https://doi.org/10.3390/fib2010034
APA StyleHatanaka, D., Takemoto, Y., Yamamoto, K., & Kadokawa, J.-i. (2014). Hierarchically Self-Assembled Nanofiber Films from Amylose-Grafted Carboxymethyl Cellulose. Fibers, 2(1), 34-44. https://doi.org/10.3390/fib2010034





