Thermoplastic Polyurethanes Stemming from Castor Oil: Green Synthesis and Their Application in Wood Bonding
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
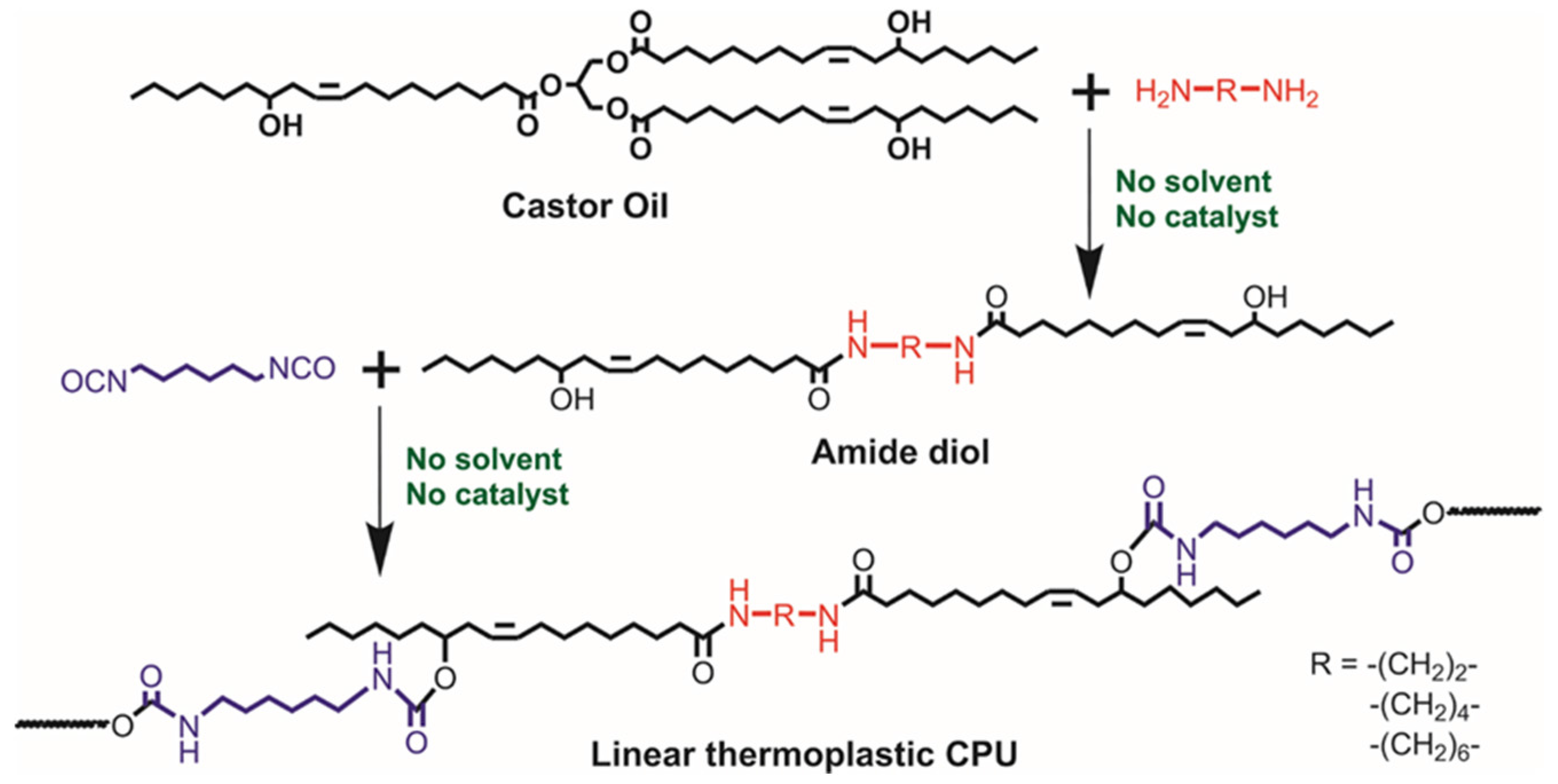
2.2. Synthesis of Precursor Diols from Castor Oil
2.3. Preparation of Castor Oil-Based Thermoplastic PUs
2.4. Characterization Methods
2.5. Wood Bonding and Water-Resistant Testing
3. Results and Discussion
3.1. Synthetisis Procedures
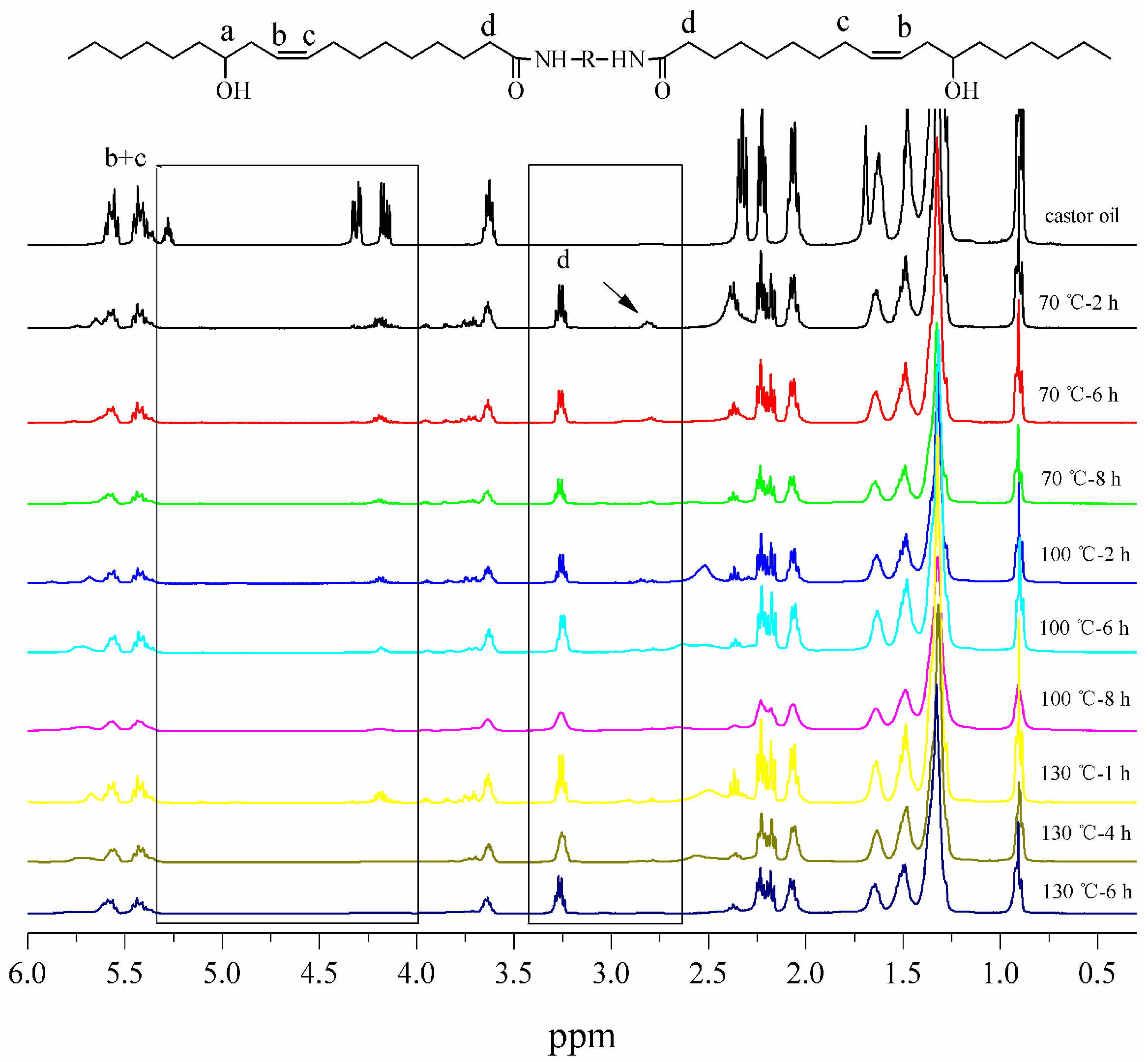
3.2. Structural Characterization
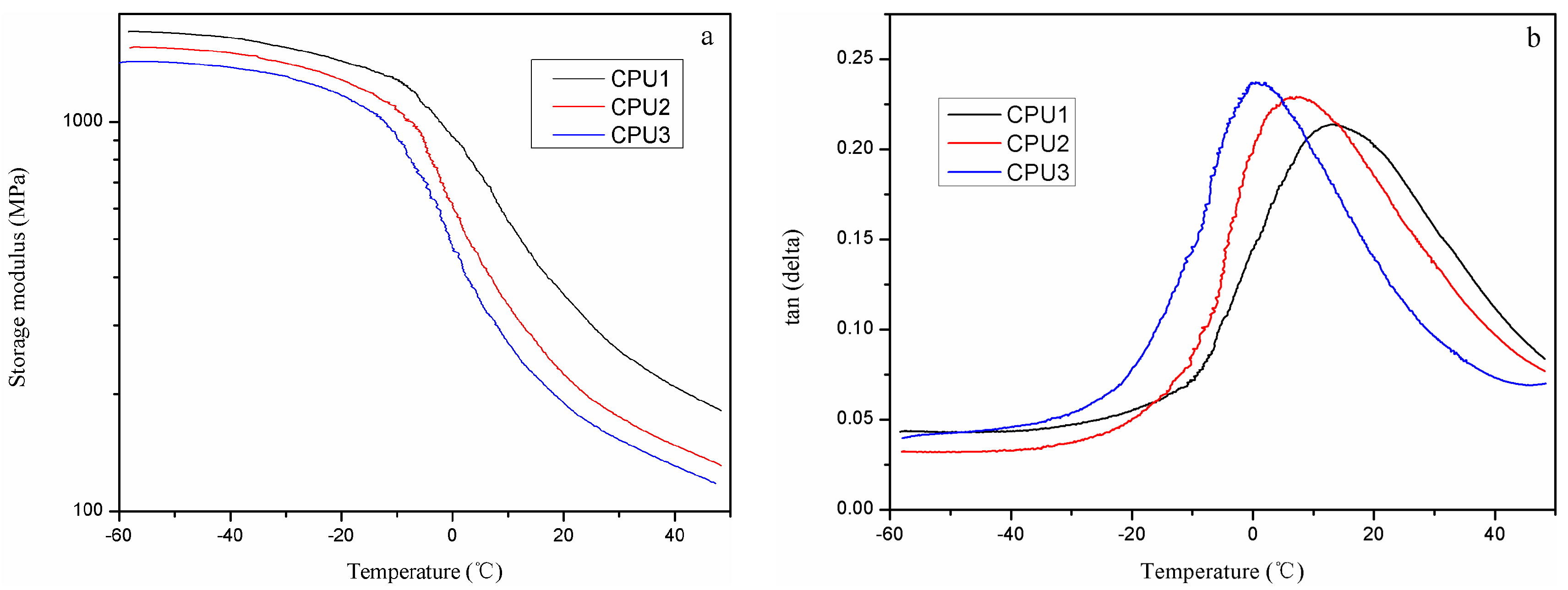
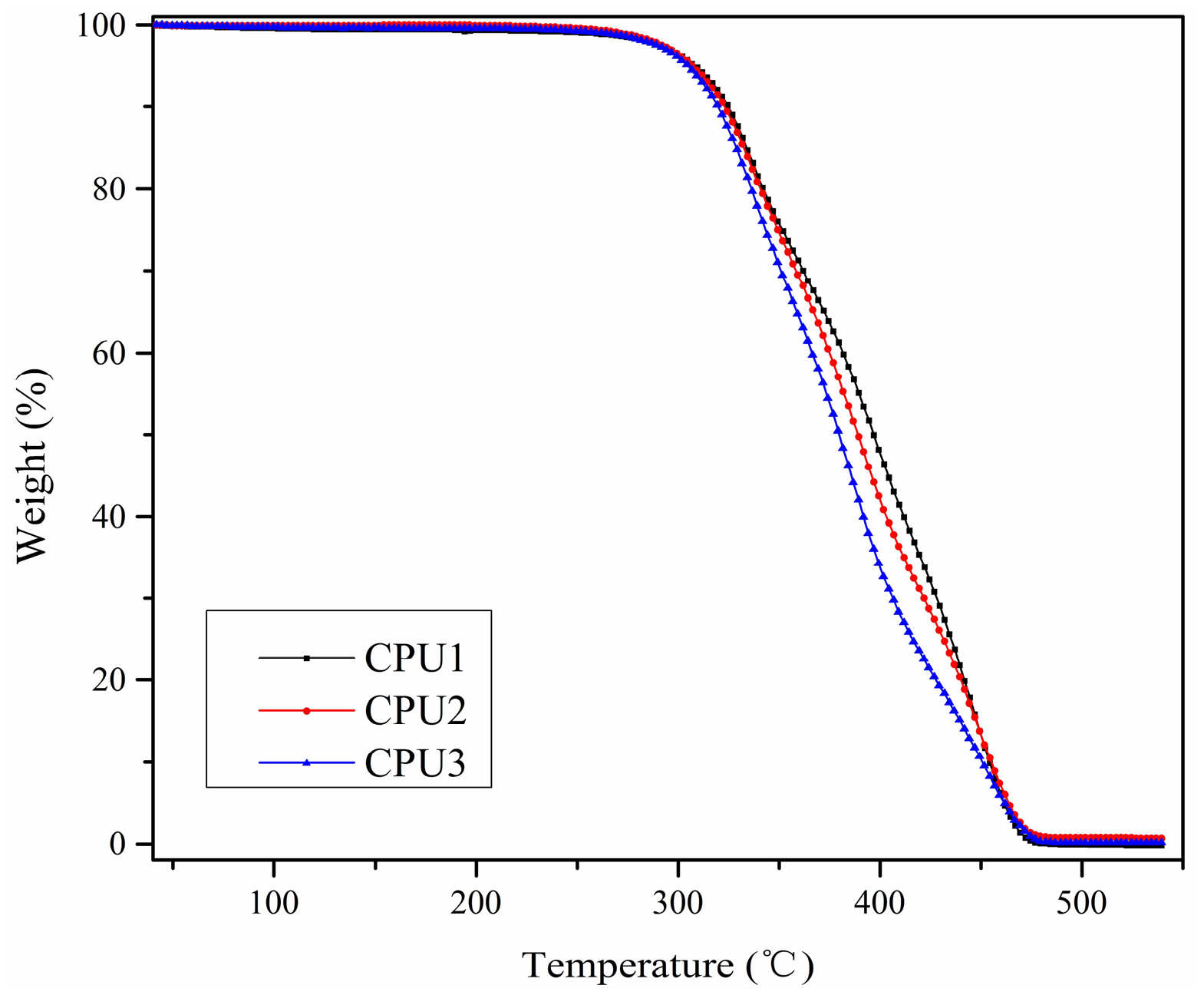
3.3. Thermal Behavior of CPUs
3.4. Wood Bonding and Water-Resistant Properties
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Escouvois, M. From vegetable oils to polyurethanes: Synthetic routes to polyols and main industrial products. Polym. Rev. 2012, 52, 38–79. [Google Scholar]
- Rannard, S.P.; Davis, N.J. The selective reaction of primary amines with carbonyl imidazole containing compounds: Selective amide and carbamate synthesis. Org. Lett. 2000, 2, 2117–2120. [Google Scholar] [CrossRef] [PubMed]
- Grzyb, J.A.; Batey, R.A. Achieving functional group diversity in parallel synthesis: Solution-phase synthesis of a library of ureas, carbamates, thiocarbamates, and amides using carbamoylimidazolium salts. Tetrahedron Lett. 2008, 49, 5279–5282. [Google Scholar] [CrossRef]
- Shimizu, M.; Sodeoka, M. Convenient method for the preparation of carbamates, carbonates, and thiocarbonates. Org. Lett. 2007, 9, 5231–5234. [Google Scholar] [CrossRef] [PubMed]
- Chaturvedi, D. Cheminform abstract: Perspectives on the synthesis of organic carbamates. Cheminform 2012, 43, 15–45. [Google Scholar] [CrossRef]
- Kreye, O.; Mutlu, H.; Meier, M.A.R. Sustainable routes to polyurethane precursors. Green Chem. 2013, 15, 1431–1455. [Google Scholar] [CrossRef]
- Pfister, D.P.; Xia, Y.; Larock, R.C. Recent advances in vegetable oil-based polyurethanes. Chemsuschem 2011, 4, 703–717. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Xia, Y.; Chen, R.; Huh, S.; Johnston, P.A.; Kessler, M.R. Soy-castor oil based polyols prepared using a solvent-free and catalyst-free method and polyurethanes therefrom. Green Chem. 2013, 15, 1477–1484. [Google Scholar] [CrossRef]
- Xia, Y.; Larock, R.C. Vegetable oil-based polymeric materials: Synthesis, properties, and applications. Green Chem. 2010, 12, 1893–1909. [Google Scholar] [CrossRef]
- Ahn, B.K.; Kraft, S.; Wang, D.; Sun, X.S. Thermally stable, transparent, pressure-sensitive adhesives from epoxidized and dihydroxyl soybean oil. Biomacromolecules 2011, 12, 1839–1843. [Google Scholar] [CrossRef] [PubMed]
- Altuna, F.I.; Pettarin, V.; Williams, R.J.J. Self-healable polymer networks based on the cross-linking of epoxidised soybean oil by an aqueous citric acid solution. Green Chem. 2013, 15, 3360–3366. [Google Scholar] [CrossRef]
- Fourcade, D.; Ritter, B.S.; Walter, P.; Schönfeld, R.; Mülhaupt, R. Renewable resource-based epoxy resins derived from multifunctional poly(4-hydroxybenzoates). Green Chem. 2013, 15, 910–918. [Google Scholar] [CrossRef]
- Tsujimoto, T.; Uyama, H. Full biobased polymeric material from plant oil and poly(lactic acid) with a shape memory property. Acs Sustain. Chem. Eng. 2014, 2, 2057–2062. [Google Scholar] [CrossRef]
- Budde, C.F.; Riedel, S.L.; Willis, L.B.; Rha, C.K.; Sinskey, A.J. Production of poly(3-hydroxybutyrate-co-3-hydroxyhexanoate) from plant oil by engineered Ralstonia eutropha strains. Appl. Environ. Microbiol. 2011, 77, 2847–2854. [Google Scholar] [CrossRef] [PubMed]
- Rao, U.; Sridhar, R.; Sehgal, P.K. Biosynthesis and biocompatibility of poly(3-hydroxybutyrate-co-4-hydroxybutyrate) produced by cupriavidus necator from spent palm oil. Biochem. Eng. J. 2010, 49, 13–20. [Google Scholar] [CrossRef]
- Obruca, S.; Marova, I.; Snajdar, O.; Mravcova, L.; Svoboda, Z. Production of poly(3-hydroxybutyrate-co-3-hydroxyvalerate) by cupriavidus necator from waste rapeseed oil using propanol as a precursor of 3-hydroxyvalerate. Biotechnol. Lett. 2010, 32, 1925–1932. [Google Scholar] [CrossRef] [PubMed]
- Ma, F.; Hanna, M.A. Biodiesel production: A review. Bioresour. Technol. 1999, 70, 1–15. [Google Scholar] [CrossRef]
- Espinosa, L.M.D.; Gevers, A.; Woldt, B.; Graß, M.; Meier, M.A.R. Sulfur-containing fatty acid-based plasticizers via thiol-ene addition and oxidation: Synthesis and evaluation in PVC formulations. Green Chem. 2014, 16, 1883–1896. [Google Scholar] [CrossRef]
- Das, G.; Bordoloi, N.K.; Rai, S.K.; Mukherjee, A.K.; Karak, N. Biodegradable and biocompatible epoxidized vegetable oil modified thermostable poly(vinyl chloride): Thermal and performance characteristics post biodegradation with Pseudomonas aeruginosa and Achromobacter sp. J. Hazard. Mater. 2012, 209, 434–442. [Google Scholar] [CrossRef] [PubMed]
- Türünç, O.; Firdaus, M.; Klein, G.; Meier, M.A.R. Fatty acid derived renewable polyamides via thiol-ene additions. Green Chem. 2012, 14, 2577–2583. [Google Scholar] [CrossRef]
- Luo, Q.; Liu, M.; Xu, Y.; Ionescu, M.; Petrović, Z.S. Thermosetting allyl resins derived from soybean oil. J. Appl. Polym. Sci. 2011, 127, 7149–7157. [Google Scholar] [CrossRef]
- Garrison, T.F.; Zhang, Z.; Kim, H.J.; Mitra, D.; Xia, Y.; Pfister, D.P.; Brehm-Stecher, B.F.; Larock, R.C.; Kessler, M.R. Thermo-mechanical and antibacterial properties of soybean oil-based cationic polyurethane coatings: Effects of amine ratio and degree of crosslinking. Macromol. Mater. Eng. 2015, 299, 1042–1051. [Google Scholar] [CrossRef]
- Allauddin, S.; Narayan, R.; Raju, K.V.S.N. Synthesis and properties of alkoxysilane castor oil and their polyurethane/urea-silica hybrid coating films. ACS Sustain. Chem. Eng. 2013, 1, 910–918. [Google Scholar] [CrossRef]
- Yongshang, L.; Larock, R.C. Aqueous cationic polyurethane dispersions from vegetable oils. Chemsuschem 2010, 3, 329–333. [Google Scholar]
- Xia, Y.; Larock, P.R.C. Soybean oil-isosorbide-based waterborne polyurethane-urea dispersions. Chemsuschem 2011, 4, 386–391. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Ding, R.; Kessler, M.R. Reduction of epoxidized vegetable oils: A novel method to prepare bio-based polyols for polyurethanes. Macromol. Rapid Commun. 2014, 35, 1068. [Google Scholar] [CrossRef] [PubMed]
- Pan, X.; Webster, D.C. New biobased high functionality polyols and their use in polyurethane coatings. Chemsuschem 2012, 5, 419–429. [Google Scholar] [CrossRef] [PubMed]
- Garrison, T.F.; Kessler, M.R.; Larock, R.C. Effects of unsaturation and different ring-opening methods on the properties of vegetable oil-based polyurethane coatings. Polymer 2014, 55, 1004–1011. [Google Scholar] [CrossRef]
- Clark, A.J.; Seng, S.H. Copolymers of tetrahydrofuran and epoxidized vegetable oils: Application to elastomeric polyurethanes. Polym. Chem. 2014, 5, 3238–3244. [Google Scholar] [CrossRef]
- Thakur, S.; Karak, N. Castor oil-based hyperbranched polyurethanes as advanced surface coating materials. Prog. Org. Coat. 2013, 76, 157–164. [Google Scholar] [CrossRef]
- Xu, C.L.; Zeng, J.B.; Wang, Y.Z. Sustainable waterborne polyurethane ionomer reinforced poly(vinyl alcohol) composite films. Compos. Sci. Technol. 2014, 96, 109–115. [Google Scholar] [CrossRef]
- Madbouly, S.A.; Xia, Y.; Kessler, M.R. Rheological behavior of environmentally friendly castor oil-based waterborne polyurethane dispersions. Macromolecules 2013, 46, 4606–4616. [Google Scholar] [CrossRef]
- Wang, H.J.; Rong, M.Z.; Zhang, M.Q.; Hu, J.; Chen, H.W.; Czigány, T. Biodegradable foam plastics based on castor oil. Biomacromolecules 2008, 9, 615–623. [Google Scholar] [CrossRef] [PubMed]
- Rao, J.L.; Balakrishna, R.S.; Shirsalkar, M.M. Cathodically electrodepositable novel coating system from castor oil. J. Appl. Polym. Sci. 2010, 44, 1873–1881. [Google Scholar] [CrossRef]
- Roy, T.; Mannari, V.; Raval, D. Two-package polyurethane coatings based on castor oil-modified polyesteramides for industrial maintenance coatings. J. Sci. Ind. Res. 1996, 55, 971–976. [Google Scholar]
- Yadav, S.; Zafar, F.; Hasnat, A.; Ahmad, S. Poly (urethane fatty amide) resin from linseed oil—A renewable resource. Prog. Org. Coat. 2009, 64, 27–32. [Google Scholar] [CrossRef]
- Lluch, C.; Ronda, J.C.; Galià, M.; Lligadas, G.; Cádiz, V. Rapid approach to biobased telechelics through two one-pot thiol-ene click reactions. Biomacromolecules 2010, 11, 1646–1653. [Google Scholar] [CrossRef] [PubMed]
- Lluch, C.; Lligadas, G.; Ronda, J.C.; Galià, M.; Cádiz, V. Thermoplastic polyurethanes from undecylenic acid-based soft segments: Structural features and release properties. Macromol. Biosci. 2013, 13, 614–622. [Google Scholar] [CrossRef] [PubMed]
- Lligadas, G.; Ronda, J.C.; Galià, M.; Cádiz, V. Plant oils as platform chemicals for polyurethane synthesis: Current state-of-the-art. Biomacromolecules 2010, 11, 2825–2835. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Petrovic, Z.; Das, S.; Wilkes, G.L. Morphology and properties of thermoplastic polyurethanes with dangling chains in ricinoleate-based soft segments. Polymer 2008, 49, 4248–4258. [Google Scholar] [CrossRef]
- Petrovic, Z.S.; Cvetkovic, I.; Hong, D.P.; Wan, X.; Zhang, W.; Abraham, T.; Malsam, J. Polyester polyols and polyurethanes from ricinoleic acid. J. Appl. Polym. Sci. 2008, 108, 1184–1190. [Google Scholar] [CrossRef]
- GB/T 7124-2008 ISO 4587:2003 Adhesive Test of Tensile Shear Strength; China National Standardization Management Committee: Beijing, China, 2003.
- Hesp, K.D.; Bergman, R.G.; Ellman, J.A. Expedient Synthesis of N-Acyl Anthranilamides and β-Enamine Amides by the Rh(III)-Catalyzed Amidation of Aryl and Vinyl C–H Bonds with Isocyanates. J. Am. Chem. Soc. 2011, 133, 11430–11433. [Google Scholar] [CrossRef] [PubMed]
- Saunders, J.H. The reactions of isocyanates and isocyanate derivatives at elevated temperatures. Rubber Chem. Technol. 1959, 32, 337–345. [Google Scholar] [CrossRef]
- Momtaz, M.; Razavi-Nouri, M.; Barikani, M. Effect of block ratio and strain amplitude on thermal, structural, and shape memory properties of segmented polycaprolactone-based polyurethanes. J. Mater. Sci. 2014, 49, 7575–7584. [Google Scholar] [CrossRef]
- Zhang, Z.; Wang, G.; Luo, N.; Huang, M.; Jin, M.; Luo, Y. Thermal decomposition of energetic thermoplastic elastomers of poly(glycidyl nitrate). J. Appl. Polym. Sci. 2014, 131, 8558–8572. [Google Scholar] [CrossRef]
- Wei, H.; Ding, D.; Wei, S.; Guo, Z. Anticorrosive conductive polyurethane multiwalled carbon nanotube nanocomposites. J. Mater. Chem. A 2013, 1, 10805–10813. [Google Scholar] [CrossRef]
- Ding, D.; Wei, H.; Zhu, J.; He, Q.; Yan, X.; Wei, S.; Guo, Z. Strain sensitive polyurethane nanocomposites reinforced with multiwalled carbon nanotubes. Energy Environ. Focus 2014, 3, 85–93. [Google Scholar] [CrossRef]
- Poh, A.K.; Sin, L.C.; Cheng, S.F.; Hock, C.C. Polyurethane wood adhesive from palm oil-based polyester polyol. J. Adhes. Sci. Technol. 2014, 28, 1020–1033. [Google Scholar]
- Silva, B.B.R.; Santana, R.M.C.; Forte, M.M.C. A solventless castor oil-based pu adhesive for wood and foam substrates. Int. J. Adhes. Adhes. 2010, 30, 559–565. [Google Scholar] [CrossRef]
- Patel, R.M.; Shukla, M.J.; Patel, K.N.; Patel, H.K. Biomaterial based novel polyurethane adhesives for wood to wood and metal to metal bonding. Mater. Res. 2009, 12, 385–393. [Google Scholar] [CrossRef]


| Sample | Mn (kDa) | Mw (kDa) | Mw/Mn | Tensile strength (TS) (MPa) | Young‘s Modulus (MPa) | ε (%) | Lap Shear Strength (×105 N/m2) |
|---|---|---|---|---|---|---|---|
| CPU1 | 9.53 | 15.7 | 1.7 | 6.2 ± 0.2 | 46.6 ± 3.4 | 52.0 ± 1.1 | 12.0 ± 0.2 |
| CPU2 | 10.9 | 18.1 | 1.7 | 5.0 ± 0.2 | 43.7 ± 1.9 | 48.7 ± 1.8 | 17.2 ± 0.1 |
| CPU3 | 11.0 | 20.0 | 1.8 | 3.6 ± 0.1 | 33.2 ± 0.6 | 69.1 ± 2.5 | 18.9 ± 0.2 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, S.; Xu, C.; Yang, W.; Tang, Q. Thermoplastic Polyurethanes Stemming from Castor Oil: Green Synthesis and Their Application in Wood Bonding. Coatings 2017, 7, 159. https://doi.org/10.3390/coatings7100159
Li S, Xu C, Yang W, Tang Q. Thermoplastic Polyurethanes Stemming from Castor Oil: Green Synthesis and Their Application in Wood Bonding. Coatings. 2017; 7(10):159. https://doi.org/10.3390/coatings7100159
Chicago/Turabian StyleLi, Shaolong, Changlian Xu, Wenfeng Yang, and Qingru Tang. 2017. "Thermoplastic Polyurethanes Stemming from Castor Oil: Green Synthesis and Their Application in Wood Bonding" Coatings 7, no. 10: 159. https://doi.org/10.3390/coatings7100159





