Iron Oxide Nanoparticles for Biomedical Applications: A Perspective on Synthesis, Drugs, Antimicrobial Activity, and Toxicity
Abstract
:1. Introduction
2. Synthesis of IONPs
2.1. Chemical Routes
2.1.1. Co-Precipitation
2.1.2. Microemulsion
2.1.3. Hydrothermal and Solvothermal Syntheses
2.1.4. Thermal Decomposition
2.1.5. Sol-Gel Reaction and Polyol
2.1.6. Sonochemical
2.1.7. Microwave-Assisted Synthesis
2.2. Physical Routes
2.2.1. Pyrolysis Method
2.2.2. Laser Ablation Synthesis in Solution (LASiS)
2.3. Biological Route
Biosynthesis
3. IONPs Coating and Functionalization
3.1. Synthetic and Natural Polymers
3.1.1. Polyethylene Glycol (PEG)
3.1.2. Poly(vinylpyrrolidone) (PVP) e Polyvinyl Alcohool (PVA)
3.1.3. Poly(lactic-co-glycolic acid) (PLGA)
3.1.4. Chitosan (CS)
3.2. Organic Surfactants
3.3. Inorganic Compounds
3.4. Bioactive Molecules
4. Drugs Bound to IONPs
4.1. Anticancer Drugs
4.2. Alternative Drugs
4.3. Immunosuppressives
4.4. Anticonvulsants
4.5. Anti-Inflammatories
4.6. Antibiotics
4.7. Antifungals
5. Antimicrobial Activity of IONPs-Based Nanosystems
6. IONPs Toxicity
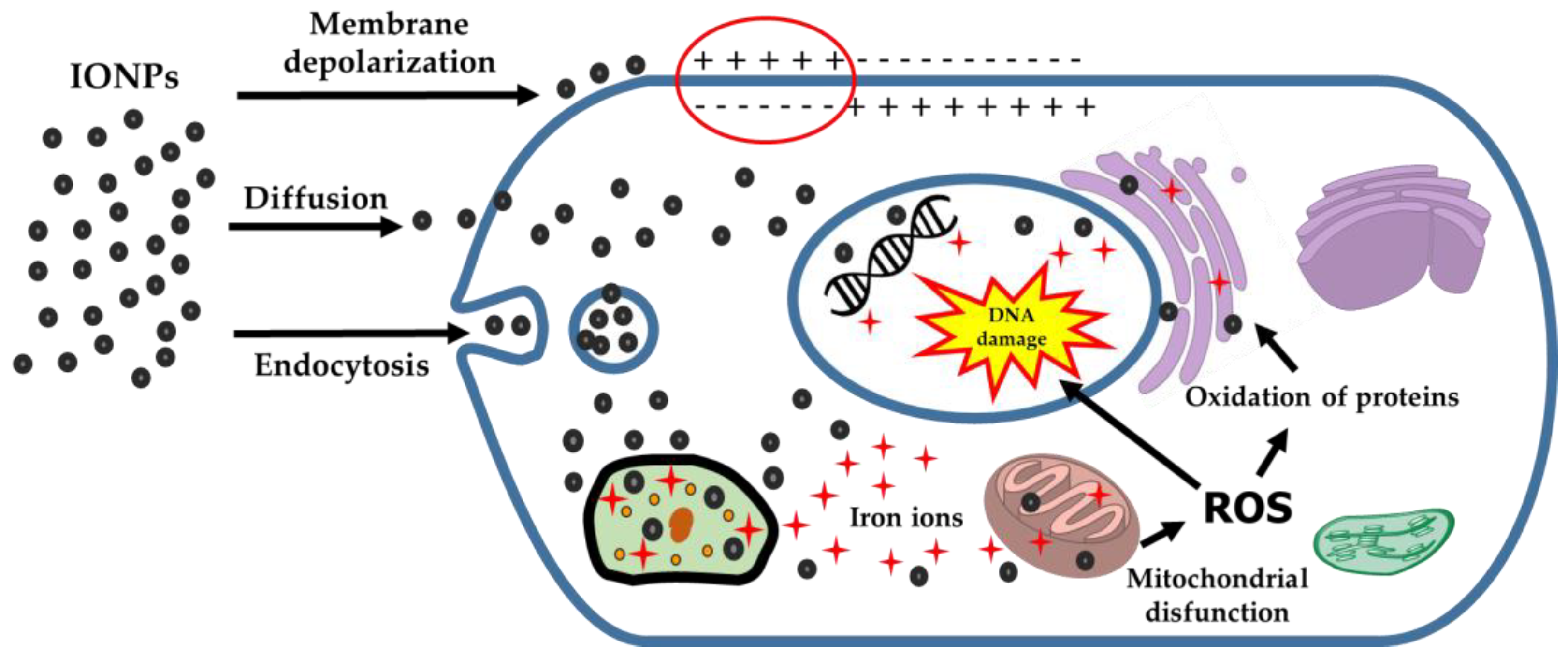
6.1. Mechanisms of IONPs Toxicity
6.2. Influence of Coatings on IONPs Toxicity
7. Conclusions and Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Riviere, C.; Roux, S.; Tillement, O.; Billotey, C.; Perriat, P. Nano-systems for medical applications: Biological detection, drug delivery, diagnostic and therapy. Ann. Chim. Sci. Mater. 2006, 31, 351–367. [Google Scholar] [CrossRef]
- Suri, S.S.; Fenniri, H.; Singh, B. Nanotechnology-based drug delivery systems. J. Occup. Med. Toxicol. 2007, 2, 16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ochekpe, N.A.; Olorunfemi, P.O.; Ngwuluka, N.C. Nanotechnology and drug delivery part 1: Background and applications. Trop. J. Pharm. Res. 2009, 8, 265–274. [Google Scholar] [CrossRef]
- Niemirowicz, K.; Markiewicz, K.H.; Wilczewska, A.Z.; Car, H. Magnetic nanoparticles as new diagnostic tools in medicine. Adv. Med. Sci. 2012, 57, 196–207. [Google Scholar] [CrossRef] [PubMed]
- Wilczewska, A.Z.; Niemirowicz, K.; Markiewicz, K.H.; Car, H. Nanoparticles as drug delivery systems. Pharmacol. Rep. 2012, 64, 1020–1037. [Google Scholar] [CrossRef]
- Mashhadi Malekzadeh, A.; Ramazani, A.; Tabatabaei Rezaei, S.J.; Niknejad, H. Design and construction of multifunctional hyperbranched polymers coated magnetite nanoparticles for both targeting magnetic resonance imaging and cancer therapy. J. Colloid Interface Sci. 2017, 490, 64–73. [Google Scholar] [CrossRef] [PubMed]
- Gu, H.; Xu, K.; Xu, C.; Xu, B. Biofunctional magnetic nanoparticles for protein separation and pathogen detection. Chem. Commun. 2006, 941–949. [Google Scholar] [CrossRef] [PubMed]
- Lin, R.; Li, Y.; MacDonald, T.; Wu, H.; Provenzale, J.; Peng, X.; Huang, J.; Wang, L.; Wang, A.Y.; Yang, J.; et al. Improving sensitivity and specificity of capturing and detecting targeted cancer cells with anti-biofouling polymer coated magnetic iron oxide nanoparticles. Colloids Surf. B Biointerfaces 2017, 150, 261–270. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Z.Q.; Song, S.C. Thermosensitive/superparamagnetic iron oxide nanoparticle-loaded nanocapsule hydrogels for multiple cancer hyperthermia. Biomaterials 2016, 106, 13–23. [Google Scholar] [CrossRef] [PubMed]
- Saikia, C.; Das, M.K.; Ramteke, A.; Maji, T.K. Effect of crosslinker on drug delivery properties of curcumin loaded starch coated iron oxide nanoparticles. Int. J. Biol. Macromol. 2016, 93, 1121–1132. [Google Scholar] [CrossRef] [PubMed]
- Lu, A.H.; Salabas, E.L.; Schüth, F. Magnetic nanoparticles: Synthesis, protection, functionalization, and application. Angew. Chem. Int. Ed. Engl. 2007, 46, 1222–1244. [Google Scholar] [CrossRef] [PubMed]
- Rümenapp, C.; Gleich, B.; Haase, A. Magnetic nanoparticles in magnetic resonance imaging and diagnostics. Pharm. Res. 2012, 29, 1165–1179. [Google Scholar] [CrossRef] [PubMed]
- Anselmo, A.C.; Mitragotri, S. A review of clinical translation of inorganic nanoparticles. AAPS J. 2015, 17, 1041–1054. [Google Scholar] [CrossRef] [PubMed]
- Clinicaltrials.gov. Available online: https://www.clinicaltrials.gov/ct2/results?term=iron+oxide+ nanoparticles (accessed on 7 May 2018).
- Alam, S.R.; Shah, A.S.; Richards, J.; Lang, N.N.; Barnes, G.; Joshi, N.; MacGillivray, T.; McKillop, G.; Mirsadraee, S.; Payne, J.; et al. Ultrasmall superparamagnetic particles of iron oxide in patients with acute myocardial infarction: Early clinical experience. Circ. Cardiovasc. Imaging 2012, 5, 559–565. [Google Scholar] [CrossRef] [PubMed]
- Raghunath, A.; Perumal, E. Metal oxide nanoparticles as antimicrobial agents: A promise for the future. Int. J. Antimicrob. Agents 2017, 49, 137–152. [Google Scholar] [CrossRef] [PubMed]
- Mahmoudi, M.; Sant, S.; Wang, B.; Laurent, S.; Sen, T. Superparamagnetic iron oxide nanoparticles (SPIONs): Development, surface modification and applications in chemotherapy. Adv. Drug Deliv. Rev. 2011, 63, 24–46. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Assa, F.; Jafarizadeh-Malmiri, H.; Ajamein, H.; Anarjan, N.; Vaghari, H.; Sayyar, Z.; Berenjian, A. A biotechnological perspective on the application of iron oxide nanoparticles. Nano Res. 2016, 9, 2203–2225. [Google Scholar] [CrossRef]
- Banerjee, T.; Mitra, S.; Kumar Singh, A.; Kumar Sharma, R.; Maitra, A. Preparation, characterization and biodistribution of ultrafine chitosan nanoparticles. Int. J. Pharm. 2002, 243, 93–105. [Google Scholar] [CrossRef]
- Moghimi, S.M.; Hunter, A.C.; Murray, J.C. Long-circulating and target-specific nanoparticles: Theory to practice. Pharm. Rev. 2001, 52, 283–318. [Google Scholar]
- Park, Y.C.; Smith, J.B.; Pham, T.; Whitaker, R.D.; Sucato, C.A.; Hamilton, J.A.; Bartolak-Suki, E.; Wong, J.Y. Effect of PEG molecular weight on stability, T2 contrast, cytotoxicity, and cellular uptake of superparamagnetic iron oxide nanoparticles (SPIONs). Colloids Surf. B Biointerfaces 2014, 119, 106–114. [Google Scholar] [CrossRef] [PubMed]
- Parsian, M.; Unsoy, G.; Mutlu, P.; Yalcin, S.; Tezcaner, A.; Gunduz, U. Loading of Gemcitabine on chitosan magnetic nanoparticles increases the anti-cancer efficacy of the drug. Eur. J. Pharmacol. 2016, 784, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Silva, A.H.; Lima, E., Jr.; Mansilla, M.V.; Zysler, R.D.; Troiani, H.; Pisciotti, M.L.; Locatelli, C.; Benech, J.C.; Oddone, N.; Zoldan, V.C.; et al. Superparamagnetic iron-oxide nanoparticles mPEG350- and mPEG2000-coated: Cell uptake and biocompatibility evaluation. Nanomedicine 2016, 12, 909–919. [Google Scholar] [CrossRef] [PubMed]
- Luchini, A.; Heenan, R.K.; Paduano, L.; Vitiello, G. Functionalized SPIONs: The surfactant nature modulates the self-assembly and cluster formation. Phys. Chem. Chem. Phys. 2016, 18, 18441–18449. [Google Scholar] [CrossRef] [PubMed]
- Silva, L.H.; da Silva, J.R.; Ferreira, G.A.; Silva, R.C.; Lima, E.C.; Azevedo, R.B.; Oliveira, D.M. Labeling mesenchymal cells with DMSA-coated gold and iron oxide nanoparticles: Assessment of biocompatibility and potential applications. J. Nanobiotechnol. 2016, 14, 59. [Google Scholar] [CrossRef] [PubMed]
- Malvindi, M.A.; De Matteis, V.; Galeone, A.; Brunetti, V.; Anyfantis, G.C.; Athanassiou, A.; Cingolani, R.; Pompa, P.P. Toxicity assessment of silica coated iron oxide nanoparticles and biocompatibility improvement by surface engineering. PLoS ONE 2014, 9, e85835. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.C.; Ren, W.; Zhang, S.; Zhong, T.; Duan, X.C.; Yin, Y.F.; Xu, M.Q.; Hao, Y.L.; Li, Z.T.; Li, H.; et al. The theranostic efficiency of tumor-specific, pH-responsive, peptide-modified, liposome-containing paclitaxel and superparamagnetic iron oxide nanoparticles. Int. J. Nanomed. 2018, 13, 1495–1504. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, W.; He, Q.G.; Jiang, C.Z. Magnetic iron oxide nanoparticles: Synthesis and surface functionalization strategies. Nanoscale Res. Lett. 2008, 3, 397–415. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Wu, Z.; Yu, T.; Jiang, C.; Kim, W.S. Recent progress on magnetic iron oxide nanoparticles: Synthesis, surface functional strategies and biomedical applications. Sci. Technol. Adv. Mater. 2015, 16, 023501. [Google Scholar] [CrossRef] [PubMed]
- Huang, K.S.; Shieh, D.B.; Yeh, C.S.; Wu, P.C.; Cheng, F.Y. Antimicrobial applications of water-dispersible magnetic nanoparticles in biomedicine. Curr. Med. Chem. 2014, 21, 3312–3322. [Google Scholar] [CrossRef] [PubMed]
- Lin, M.M.; Kim, D.K.; El Haj, A.J.; Dobson, J. Development of superparamagnetic iron oxide nanoparticles (SPIONS) for translation to clinical applications. IEEE Trans. Nanobiosci. 2008, 7, 298–305. [Google Scholar] [CrossRef]
- Kansara, K.; Patel, P.; Shukla, R.K.; Pandya, A.; Shanker, R.; Kumar, A.; Dhawan, A. Synthesis of biocompatible iron oxide nanoparticles as a drug delivery vehicle. Int. J. Nanomed. 2018, 13, 79–82. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Drmota, A.; Drofenik, M.; Koselj, J.; Žnidaršič, A. Microemulsion method for synthesis of magnetic oxide nanoparticles. In Microemulsions—An Introduction to Properties and Applications, 1st ed.; Najjar, R., Ed.; InTech: Rijeka, Croatia, 2012; pp. 191–215. ISBN 978-953-51-0247-2. [Google Scholar]
- Levy, L.; Sahoo, Y.; Kim, K.S.; Bergey, E.J.; Prasad, P.N. Nanochemistry: Synthesis and characterization of multifunctional nanoclinics for biological applications. Chem. Mater. 2002, 14, 3715–3721. [Google Scholar] [CrossRef]
- Unsoy, G.; Gunduz, U.; Oprea, O.; Ficai, D.; Sonmez, M.; Radulescu, M.; Alexie, M.; Ficai, A. Magnetite: From synthesis to applications. Curr. Top. Med. Chem. 2015, 15, 1622–1640. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Sun, A.; Zhai, F.; Wang, J.; Xu, W.; Zhang, Q.; Volinsky, A.A. Fe3O4 magnetic nanoparticles synthesis from tailings by ultrasonic chemical co-precipitation. Mater. Lett. 2011, 65, 1882–1884. [Google Scholar] [CrossRef]
- Liu, Y.; Jia, S.; Wu, Q.; Ran, J.; Zhang, W.; Wu, S. Studies of Fe3O4-chitosan nanoparticles prepared by co-precipitation under the magnetic field for lipase immobilization. Catal. Commun. 2011, 12, 717–720. [Google Scholar] [CrossRef]
- Pereira, C.; Pereira, A.M.; Fernandes, C.; Rocha, M.; Mendes, R.; Fernández-García, M.P.; Guedes, A.; Tavares, P.B.; Grenèche, J.M.; Araújo, J.P.; et al. Superparamagnetic MFe2O4 (M=Fe, Co, Mn) nanoparticles: Tuning the particle size and magnetic properties through a novel one-step coprecipitation route. Chem. Mater. 2012, 24, 1496–1504. [Google Scholar] [CrossRef]
- Riaz, S.; Bashir, M.; Naseem, S. Iron oxide nanoparticles prepared by modified co-precipitation method. IEEE Trans. Magn. 2014, 50, 1–4. [Google Scholar] [CrossRef]
- Yan, A.; Liu, Y.; Liu, Y.; Li, X.; Lei, Z.; Liu, P. A NaAc-assisted large-scale coprecipitation synthesis and microwave absorption efficiency of Fe3O4 nanowires. Mater. Lett. 2012, 68, 402–405. [Google Scholar] [CrossRef]
- Wongwailikhit, K.; Horwongsakul, S. The preparation of iron (III) oxide nanoparticles using W/O microemulsion. Mater. Lett. 2011, 65, 2820–2822. [Google Scholar] [CrossRef]
- Malik, M.A.; Wani, M.Y.; Hashim, M.A. Microemulsion method: A novel route to synthesize organic and inorganic nanomaterials. Arab. J. Chem. 2012, 5, 397–417. [Google Scholar] [CrossRef]
- Deng, Y.; Wang, L.; Yang, W.; Fu, S.; Elaïssari, A. Preparation of magnetic polymeric particles via inverse microemulsion polymerization process. J. Magn. Magn. Mater. 2003, 257, 69–78. [Google Scholar] [CrossRef]
- Kaur, G.; Dogra, V.; Kumar, R.; Kumar, S.; Singh, K. Fabrication of iron oxide nanocolloids using metallosurfactant-based microemulsions: Antioxidant activity, cellular, and genotoxicity toward Vitis vinifera. J. Biomol. Struct. Dyn. 2018, 27, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Takami, S.; Sato, T.; Mousavand, T.; Ohara, S.; Umetsu, M.; Adschiri, T. Hydrothermal synthesis of surface-modified iron oxide nanoparticles. Mater. Lett. 2007, 61, 4769–4772. [Google Scholar] [CrossRef]
- Wan, L.; Yan, S.; Wang, X.; Li, Z.; Zou, Z. Solvothermal synthesis of monodisperse iron oxides with various morphologies and their applications in removal of Cr(VI). Cryst. Eng. Comm. 2011, 13, 2727–2733. [Google Scholar] [CrossRef]
- Maity, D.; Ding, J.; Xue, J.M. Synthesis of magnetite nanoparticles by thermal decomposition: Time, temperature, surfactant and solvent effects. Funct. Mater. Lett. 2008, 1, 189–193. [Google Scholar] [CrossRef]
- Maity, D.; Choo, S.G.; Yi, J.; Ding, J.; Xue, J.M. Synthesis of magnetite nanoparticles via a solvent-free thermal decomposition route. J. Magn. Magn. Mater. 2009, 321, 1256–1259. [Google Scholar] [CrossRef]
- Sun, S.; Zeng, H. Size-controlled synthesis of magnetite nanoparticles. J. Am. Chem. Soc. 2002, 124, 8204–8205. [Google Scholar] [CrossRef] [PubMed]
- Yu, W.W.; Falkner, J.C.; Yavuz, C.T.; Colvin, V.L. Synthesis of monodisperse iron oxide nanocrystals by thermal decomposition of iron carboxylate salts. Chem. Commun. 2004, 10, 2306–2307. [Google Scholar] [CrossRef] [PubMed]
- Hatakeyama, M.; Kishi, H.; Kita, Y.; Imai, K.; Nishio, K.; Karasawa, S.; Masaike, Y.; Sakamoto, S.; Sandhu, A.; Tanimoto, A.; et al. A two-step ligand exchange reaction generates highly water-dispersed magnetic nanoparticles for biomedical applications. J. Mater. Chem. 2011, 21, 5959–5966. [Google Scholar] [CrossRef]
- Belaid, S.; Stanici, D.; Vander Elst, L.; Muller, R.N.; Laurent, S. Influence of experimental parameters on iron oxide nanoparticles properties synthesized by thermal decomposition: Size and nuclear magnetic resonance studies. Nanotechnology 2018. [Google Scholar] [CrossRef] [PubMed]
- Pandey, S.; Mishra, S.B. Sol–gel derived organic–inorganic hybrid materials: Synthesis, characterizations and applications. J. Sol-Gel Sci. Technol. 2011, 59, 73–94. [Google Scholar] [CrossRef]
- Xu, J.K.; Zhang, F.F.; Sun, J.J.; Sheng, J.; Wang, F.; Sun, M. Bio and nanomaterials based on Fe3O4. Molecules 2014, 19, 21506–21528. [Google Scholar] [CrossRef] [PubMed]
- Reddy, L.H.; Arias, J.L.; Nicolas, J.; Couvreur, P. Magnetic nanoparticles: Design and characterization, toxicity and biocompatibility, pharmaceutical and biomedical applications. Chem. Rev. 2012, 112, 5818–5878. [Google Scholar] [CrossRef] [PubMed]
- Cai, W.; Wan, J. Facile synthesis of superparamagnetic magnetite nanoparticles in liquid polyols. J. Colloid Interface Sci. 2007, 305, 366–370. [Google Scholar] [CrossRef] [PubMed]
- Hachani, R.; Lowdell, M.; Birchall, M.; Hervault, A.; Mertz, D.; Begin-Colin, S.; Thanh, N.T. Polyol synthesis, functionalisation, and biocompatibility studies of superparamagnetic iron oxide nanoparticles as potential MRI contrast agents. Nanoscale 2016, 8, 3278–3287. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sodipo, B.K.; Aziz, A.A. One minute synthesis of amino-silane functionalized superparamagnetic iron oxide nanoparticles by sonochemical method. Ultrason. Sonochem. 2018, 40, 837–840. [Google Scholar] [CrossRef] [PubMed]
- Dolores, R.; Raquel, S.; Adianez, G.L. Sonochemical synthesis of iron oxide nanoparticles loaded with folate and cisplatin: Effect of ultrasonic frequency. Ultrason. Sonochem. 2015, 23, 391–398. [Google Scholar] [CrossRef] [PubMed]
- Osborne, E.A.; Atkins, T.M.; Gilbert, D.A.; Kauzlarich, S.M.; Liu, K.; Louie, A.Y. Rapid microwave-assisted synthesis of dextran-coated iron oxide nanoparticles for magnetic resonance imaging. Nanotechnology 2012, 23, 215602. [Google Scholar] [CrossRef] [PubMed]
- Bomatí-Miguel, O.; Zhao, X.Q.; Martelli, S.; Di Nunzio, P.E.; Veintemillas-Verdaguer, S. Modeling of the laser pyrolysis process by means of the aerosol theory: Case of iron nanoparticles. J. Appl. Phys. 2010, 107, 014906. [Google Scholar] [CrossRef]
- Thongsuwan, W.; Suparerk, A.; Singjai, P. Preparation of Iron Oxide nanoparticles by a pyrosol technique. Key Eng. Mater. 2007, 353–358, 2175–2178. [Google Scholar] [CrossRef]
- Malumbres, A.; Martínez, G.; Mallada, R.; Hueso, J.L.; Bomatí-Miguel, O.; Santamaría, J. Continuous production of iron-based nanocrystals by laser pyrolysis. Effect of operating variables on size, composition and magnetic response. Nanotechnology 2013, 24, 325603. [Google Scholar] [CrossRef] [PubMed]
- Amendola, V.; Meneghetti, M. What controls the composition and the structure of nanomaterials generated by laser ablation in liquid solution? Phys. Chem. Chem. Phys. 2013, 15, 3027–3046. [Google Scholar] [CrossRef] [PubMed]
- Fracasso, G.; Ghigna, P.; Nodari, L.; Agnoli, S.; Badocco, D.; Pastore, P.; Nicolato, E.; Marzola, P.; Mihajlović, D.; Markovic, M.; et al. Nanoaggregates of iron poly-oxo-clusters obtained by laser ablation in aqueous solution of phosphonates. J. Colloid Interface Sci. 2018, 522, 208–216. [Google Scholar] [CrossRef] [PubMed]
- Fatemi, M.; Mollania, N.; Momeni-Moghaddam, M.; Sadeghifar, F. Extracellular biosynthesis of magnetic iron oxide nanoparticles by Bacillus cereus strain HMH1: Characterization and in vitro cytotoxicity analysis on MCF-7 and 3T3 cell lines. J. Biotechnol. 2018, 270, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.A.; Lovley, D.R.; Tremblay, P.L. Outer cell surface components essential for Fe(III) oxide reduction by Geobacter metallireducens. Appl. Environ. Microbiol. 2013, 79, 901–907. [Google Scholar] [CrossRef] [PubMed]
- Shenton, W.; Mann, S.; Cölfen, H.; Bacher, A.; Fischer, M. Synthesis of nanophase iron oxide in lumazine synthase capsids. Angew. Chem. Int. Ed. Engl. 2001, 40, 442–445. [Google Scholar] [CrossRef]
- Wu, W.; Chen, B.; Cheng, J.; Wang, J.; Xu, W.; Liu, L.; Xia, G.; Wei, H.; Wang, X.; Yang, M.; et al. Biocompatibility of Fe3O4/DNR magnetic nanoparticles in the treatment of hematologic malignancies. Int. J. Nanomed. 2010, 5, 1079–1084. [Google Scholar] [CrossRef]
- Couto, D.; Freitas, M.; Carvalho, F.; Fernandes, E. Iron oxide nanoparticles: An insight into their biomedical applications. Curr. Med. Chem. 2015, 22, 1808–1828. [Google Scholar] [CrossRef] [PubMed]
- Prabhu, S.; Mutalik, S.; Rai, S.; Udupa, N.; Rao, B.S.S. PEGylation of superparamagnetic iron oxide nanoparticle for drug delivery applications with decreased toxicity: An in vivo study. J. Nanopart. Res. 2015, 17, 412. [Google Scholar] [CrossRef]
- Ramírez-Cando, L.J.; De Simone, U.; Coccini, T. Toxicity evaluation of iron oxide (Fe3O4) nanoparticles on human neuroblastoma-derived SH-SY5Y cell line. J. Nanosci. Nanotechnol. 2017, 17, 203–211. [Google Scholar] [CrossRef] [PubMed]
- Chomoucka, J.; Drbohlavova, J.; Huska, D.; Adam, V.; Kizek, R.; Hubalek, J. Magnetic nanoparticles and targeted drug delivering. Pharm. Res. 2010, 62, 144–149. [Google Scholar] [CrossRef] [PubMed]
- Makadia, H.K.; Siegel, S.J. Poly lactic-co-glycolic acid (PLGA) as biodegradable controlled drug delivery carrier. Polymers 2011, 3, 1377–1397. [Google Scholar] [CrossRef] [PubMed]
- Houchin, M.L.; Topp, E.M. Physical properties of PLGA films during polymer degradation. J. Appl. Polym. Sci. 2009, 114, 2848–2854. [Google Scholar] [CrossRef]
- Abdalla, M.O.; Aneja, R.; Dean, D.; Rangari, V.; Russell, A.; Jaynes, J.; Yates, C.; Turner, T. Synthesis and characterization of noscapine loaded magnetic polymeric nanoparticles. J. Magn. Magn. Mater. 2010, 322, 190–196. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Agnihotri, S.A.; Mallikarjuna, N.N.; Aminabhavi, T.M. Recent advances on chitosan-based micro- and nanoparticles in drug delivery. J. Control. Release 2004, 100, 5–28. [Google Scholar] [CrossRef] [PubMed]
- Berscht, P.C.; Nies, B.; Liebendörfer, A.; Kreuter, J. Incorporation of basic fibroblast growth factor into methylpyrrolidinone chitosan fleeces and determination of the in vitro release characteristics. Biomaterials 1994, 15, 593–600. [Google Scholar] [CrossRef]
- Arai, K.; Kinumaki, T.; Fujita, T. Toxicity of chitosan. Bull. Tokai Reg. Fish. Lab. 1968, 43, 89–94. [Google Scholar]
- Nicol, S. Life after death for empty shells: Crustacean fisheries create a mountain of waste shells, made of a strong natural polymer, chitin. Now chemists are helping to put this waste to some surprising uses. New Sci. 1991, 129, 46–48. [Google Scholar]
- Qi, L.; Xu, Z.; Jiang, X.; Hu, C.; Zou, X. Preparation and antibacterial activity of chitosan nanoparticles. Carbohydr. Res. 2004, 339, 2693–2700. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.J.; Shieh, M.J.; Lin, F.H.; Lou, P.J.; Peng, C.L.; Wei, M.F.; Yao, C.J.; Lai, P.S.; Young, T.H. Colorectal cancer cell detection by 5-aminolaevulinic acid-loaded chitosan nano-particles. Cancer Lett. 2009, 273, 210–220. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Chen, Z.L.; Yang, X.X.; Huang, P.; Zhou, X.P.; Du, X.X. Magnetic chitosan nanoparticles as a drug delivery system for targeting photodynamic therapy. Nanotechnology 2009, 20, 135102. [Google Scholar] [CrossRef] [PubMed]
- Mathew, T.V.; Kuriakose, S. Photochemical and antimicrobial properties of silver nanoparticle-encapsulated chitosan functionalized with photoactive groups. Mater. Sci. Eng. C Mater. Biol. Appl. 2013, 33, 4409–4415. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; Wang, Y.; Jian, J.; Song, S. Self-aggregated nanoparticles based on amphiphilic poly(lactic acid)-grafted-chitosan copolymer for ocular delivery of amphotericin B. Int. J. Nanomed. 2013, 8, 3715–3728. [Google Scholar] [CrossRef]
- Radulescu, M.; Ficai, D.; Oprea, O.; Ficai, A.; Andronescu, E.; Holban, A.M. Antimicrobial chitosan based formulations with impact on different biomedical applications. Curr. Pharm. Biotechnol. 2015, 16, 128–136. [Google Scholar] [CrossRef] [PubMed]
- Onnainty, R.; Onida, B.; Páez, P.; Longhi, M.; Barresi, A.; Granero, G. Targeted chitosan-based bionanocomposites for controlled oral mucosal delivery of chlorhexidine. Int. J. Pharm. 2016, 509, 408–418. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Chen, D.; Zhang, Y.; Deng, Z.; Ren, X.; Meng, X.; Tang, F.; Ren, J.; Zhang, L. Magnetic and fluorescent multifunctional chitosan nanoparticles as a smart drug delivery system. Nanotechnology 2007, 18, 405102. [Google Scholar] [CrossRef]
- Soares, P.I.; Machado, D.; Laia, C.; Pereira, L.C.; Coutinho, J.T.; Ferreira, I.M.; Novo, C.M.; Borges, J.P. Thermal and magnetic properties of chitosan-iron oxide nanoparticles. Carbohydr. Polym. 2016, 149, 382–390. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.K.; Gupta, M. Synthesis and surface engineering of iron oxide nanoparticles for biomedical applications. Biomaterials 2005, 26, 3995–4021. [Google Scholar] [CrossRef] [PubMed]
- Prabaharan, M. Review paper: Chitosan derivatives as promising materials for controlled drug delivery. J. Biomater. Appl. 2008, 23, 5–36. [Google Scholar] [CrossRef] [PubMed]
- Zhu, A.; Chan-Park, M.B.; Dai, S.; Li, L. The aggregation behavior of O-carboxymethylchitosan in dilute aqueous solution. Colloids Surf. B Biointerfaces 2005, 43, 143–149. [Google Scholar] [CrossRef] [PubMed]
- Saikia, C.; Hussain, A.; Ramteke, A.; Sharma, H.K.; Maji, T.K. Carboxymethyl starch-chitosan-coated iron oxide magnetic nanoparticles for controlled delivery of isoniazid. J. Microencapsul. 2015, 32, 29–39. [Google Scholar] [CrossRef] [PubMed]
- Soares, P.I.; Lochte, F.; Echeverria, C.; Pereira, L.C.; Coutinho, J.T.; Ferreira, I.M.; Novo, C.M.; Borges, J.P. Thermal and magnetic properties of iron oxide colloids: Influence of surfactants. Nanotechnology 2015, 26, 425704. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wong, J.F.; Teng, X.; Lin, X.Z.; Yang, H. “Pulling” nanoparticles into water: Phase transfer of oleic acid stabilized monodisperse nanoparticles into aqueous solutions of alpha-cyclodextrin. Nano Lett. 2003, 3, 7907–7911. [Google Scholar] [CrossRef]
- Costa, C.; Brandão, F.; Bessa, M.J.; Costa, S.; Valdiglesias, V.; Kiliç, G.; Fernández-Bertólez, N.; Quaresma, P.; Pereira, E.; Pásaro, E.; et al. In vitro cytotoxicity of superparamagnetic iron oxide nanoparticles on neuronal and glial cells. Evaluation of nanoparticle interference with viability tests. J. Appl. Toxicol. 2016, 36, 361–372. [Google Scholar] [CrossRef] [PubMed]
- Toropova, Y.G.; Golovkin, A.S.; Malashicheva, A.B.; Korolev, D.V.; Gorshkov, A.N.; Gareev, K.G.; Afonin, M.V.; Galagudza, M.M. In vitro toxicity of FemOn, FemOn-SiO2 composite, and SiO2-FemOn core-shell magnetic nanoparticles. Int. J. Nanomed. 2017, 12, 593–603. [Google Scholar] [CrossRef] [PubMed]
- Kandibanda, S.R.; Gundeboina, N.; Das, S.; Sunkara, V.M. Synthesis, characterisation, cellular uptake and cytotoxicity of functionalised magnetic ruthenium (II) polypyridine complex core-shell nanocomposite. J. Photochem. Photobiol. B 2018, 178, 270–276. [Google Scholar] [CrossRef] [PubMed]
- Ma, K.; Wang, Q.; Rong, Q.; Zhang, D.; Cui, S.; Yang, J. Preparation of magnetic carbon/Fe3O4 supported zero-valent iron composites and their application in Pb(II) removal from aqueous solutions. Water Sci. Technol. 2017, 76, 2680–2689. [Google Scholar] [CrossRef] [PubMed]
- Ivashchenko, O.; Lewandowski, M.; Peplińska, B.; Jarek, M.; Nowaczyk, G.; Wiesner, M.; Załęski, K.; Babutina, T.; Warowicka, A.; Jurga, S. Synthesis and characterization of magnetite/silver/antibiotic nanocomposites for targeted antimicrobial therapy. Mater. Sci. Eng. C Mater. Biol. Appl. 2015, 55, 343–359. [Google Scholar] [CrossRef] [PubMed]
- Tamer, U.; Gündoğdu, Y.; Boyaci, I.H.; Pekmez, K. Synthesis of magnetic core-shell Fe3O4-Au, nanoparticles for biomolecule immobilization and detection. J. Nanopart. Res. 2010, 12, 1187–1196. [Google Scholar] [CrossRef]
- Bisht, G.; Rayamajhi, S.; Kc, B.; Paudel, S.N.; Karna, D.; Shrestha, B.G. Synthesis, characterization, and study of in vitro cytotoxicity of ZnO-Fe3O4 magnetic composite nanoparticles in human breast cancer cell line (MDA-MB-231) and mouse fibroblast (NIH 3T3). Nanoscale Res. Lett. 2016, 11, 537. [Google Scholar] [CrossRef] [PubMed]
- Rădulescu, M.; Andronescu, E.; Holban, A.M.; Vasile, B.S.; Iordache, F.; Mogoantă, L.; Mogoșanu, G.D.; Grumezescu, A.M.; Georgescu, M.; Chifiriuc, M.C. Antimicrobial nanostructured bioactive coating based on Fe3O4 and patchouli oil for wound dressing. Metals 2016, 6, 103. [Google Scholar] [CrossRef]
- Mu, Q.; Kievit, F.M.; Kant, R.J.; Lin, G.; Jeon, M.; Zhang, M. Anti-HER2/neu peptide-conjugated iron oxide nanoparticles for targeted delivery of paclitaxel to breast cancer cells. Nanoscale 2015, 7, 18010–18014. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jahanban-Esfahlan, A.; Dastmalchi, S.; Davaran, S. A simple improved desolvation method for the rapid preparation of albumin nanoparticles. Int. J. Biol. Macromol. 2016, 91, 703–709. [Google Scholar] [CrossRef] [PubMed]
- Nosrati, H.; Sefidi, N.; Sharafi, A.; Danafar, H.; Kheiri Manjili, H. Bovine Serum Albumin (BSA) coated iron oxide magnetic nanoparticles as biocompatible carriers for curcumin-anticancer drug. Bioorg. Chem. 2018, 76, 501–509. [Google Scholar] [CrossRef] [PubMed]
- Thomas, R.G.; Unnithan, A.R.; Moon, M.J.; Surendran, S.P.; Batgerel, T.; Park, C.H.; Kim, C.S.; Jeong, Y.Y. Electromagnetic manipulation enabled calcium alginate Janus microsphere for targeted delivery of mesenchymal stem cells. Int. J. Biol. Macromol. 2018, 110, 465–471. [Google Scholar] [CrossRef] [PubMed]
- Hans, M.L.; Lowman, A.M. Nanoparticles for Drug Delivery. In Nanomaterials Handbook, 1st ed.; Gogotsi, Y., Ed.; Taylor & Francis: Boca Raton, FL, USA, 2006; pp. 637–664. ISBN 0-8493-2308-8. [Google Scholar]
- Pillai, G. Nanomedicines for cancer therapy: An update of FDA approved and those under various stages of development. SOJ Pharm. Pharm. Sci. 2014, 2, 1–13. [Google Scholar] [CrossRef]
- Ebrahimi, E.; Akbarzadeh, A.; Abbasi, E.; Khandaghi, A.A.; Abasalizadeh, F.; Davaran, S. Novel drug delivery system based on doxorubicin-encapsulated magnetic nanoparticles modified with PLGA-PEG 1000 copolymer. Artif. Cells Nanomed. Biotechnol. 2016, 44, 290–297. [Google Scholar] [CrossRef] [PubMed]
- Nigam, S.; Bahadur, D. Doxorubicin-loaded dendritic-Fe3O4 supramolecular nanoparticles for magnetic drug targeting and tumor regression in spheroid murine melanoma model. Nanomedicine 2018, 14, 759–768. [Google Scholar] [CrossRef] [PubMed]
- Abbasi Aval, N.; Pirayesh Islamian, J.; Hatamian, M.; Arabfirouzjaei, M.; Javadpour, J.; Rashidi, M.R. Doxorubicin loaded large-pore mesoporous hydroxyapatite coated superparamagnetic Fe3O4 nanoparticles for cancer treatment. Int. J. Pharm. 2016, 509, 159–167. [Google Scholar] [CrossRef] [PubMed]
- Liang, P.C.; Chen, Y.C.; Chiang, C.F.; Mo, L.R.; Wei, S.Y.; Hsieh, W.Y.; Lin, W.L. Doxorubicin-modified magnetic nanoparticles as a drug delivery system for magnetic resonance imaging-monitoring magnet-enhancing tumor chemotherapy. Int. J. Nanomed. 2016, 11, 2021–2037. [Google Scholar] [CrossRef]
- Augustin, E.; Czubek, B.; Nowicka, A.M.; Kowalczyk, A.; Stojek, Z.; Mazerska, Z. Improved cytotoxicity and preserved level of cell death induced in colon cancer cells by doxorubicin after its conjugation with iron-oxide magnetic nanoparticles. Toxicol. In Vitro 2016, 33, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Jurado, R.; Frączek, P.; Droetto, M.; Sánchez, P.; Valero, E.; Domínguez-Vera, J.M.; Gálvez, N. Apomaghemite as a doxorubicin carrier for anticancer drug delivery. J. Inorg. Biochem. 2016, 157, 46–51. [Google Scholar] [CrossRef] [PubMed]
- Montha, W.; Maneeprakorn, W.; Buatong, N.; Tang, I.M.; Pon-On, W. Synthesis of doxorubicin-PLGA loaded chitosan stabilized (Mn, Zn)Fe2O4 nanoparticles: Biological activity and pH-responsive drug release. Mater. Sci. Eng. C Mater. Biol. Appl. 2016, 59, 235–240. [Google Scholar] [CrossRef] [PubMed]
- Mallick, N.; Anwar, M.; Asfer, M.; Mehdi, S.H.; Rizvi, M.M.; Panda, A.K.; Talegaonkar, S.; Ahmad, F.J. Chondroitin sulfate-capped super-paramagnetic iron oxide nanoparticles as potential carriers of doxorubicin hydrochloride. Carbohydr. Polym. 2016, 151, 546–556. [Google Scholar] [CrossRef] [PubMed]
- Nadeem, M.; Ahmad, M.; Akhtar, M.S.; Shaari, A.; Riaz, S.; Naseem, S.; Masood, M.; Saeed, M.A. Magnetic properties of polyvinyl alcohol and doxorubicine loaded iron oxide nanoparticles for anticancer drug delivery applications. PLoS ONE 2016, 11, e0158084. [Google Scholar] [CrossRef] [PubMed]
- Tarvirdipour, S.; Vasheghani-Farahani, E.; Soleimani, M.; Bardania, H. Functionalized magnetic dextran-spermine nanocarriers for targeted delivery of doxorubicin to breast cancer cells. Int. J. Pharm. 2016, 501, 331–341. [Google Scholar] [CrossRef] [PubMed]
- Thorat, N.D.; Lemine, O.M.; Bohara, R.A.; Omri, K.; El Mir, L.; Tofail, S.A. Superparamagnetic iron oxide nanocargoes for combined cancer thermotherapy and MRI applications. Phys. Chem. Chem. Phys. 2016, 18, 21331–21339. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhao, R.; Wang, S.; Liu, Z.; Tang, R. In vivo dual-targeted chemotherapy of drug resistant cancer by rationally designed nanocarrier. Biomaterials 2016, 75, 71–81. [Google Scholar] [CrossRef] [PubMed]
- Nehate, C.; Aji Alex, M.R.; Kumar, A.; Koul, V. Combinatorial delivery of superparamagnetic iron oxide nanoparticles (γ Fe2O3) and doxorubicin using folate conjugated redox sensitive multiblock polymeric nanocarriers for enhancing the chemotherapeutic efficacy in cancer cells. Mater. Sci. Eng. C Mater. Biol. Appl. 2017, 75, 1128–1143. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Chen, L.; Liu, F.; Qi, X.; Ge, Y.; Shen, S. Remotely controlled drug release based on iron oxide nanoparticles for specific therapy of cancer. Colloids Surf. B Biointerfaces 2017, 152, 440–448. [Google Scholar] [CrossRef] [PubMed]
- Bekaroglu, M.G.; Isci, Y.; Isci, S. Colloidal properties and in vitro evaluation of Hydroxy ethyl cellulose coated iron oxide particles for targeted drug delivery. Mater. Sci. Eng. C Mater. Biol. Appl. 2017, 78, 847–853. [Google Scholar] [CrossRef] [PubMed]
- Benyettou, F.; Alhashimi, M.; O’Connor, M.; Pasricha, R.; Brandel, J.; Traboulsi, H.; Mazher, J.; Olsen, J.C.; Trabolsi, A. Sequential delivery of doxorubicin and zoledronic acid to breast cancer cells by CB[7]-modified iron oxide nanoparticles. ACS Appl. Mater. Interfaces 2017, 9, 40006–40016. [Google Scholar] [CrossRef] [PubMed]
- Mosafer, J.; Teymouri, M. Comparative study of superparamagnetic iron oxide/doxorubicin co-loaded poly (lactic-co-glycolic acid) nanospheres prepared by different emulsion solvent evaporation methods. Artif. Cells Nanomed. Biotechnol. 2017, 46, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Mosafer, J.; Teymouri, M.; Abnous, K.; Tafaghodi, M.; Ramezani, M. Study and evaluation of nucleolin-targeted delivery of magnetic PLGA-PEG nanospheres loaded with doxorubicin to C6 glioma cells compared with low nucleolin-expressing L929 cells. Mater. Sci. Eng. C Mater. Biol. Appl. 2017, 72, 123–133. [Google Scholar] [CrossRef] [PubMed]
- Prabha, G.; Raj, V. Sodium alginate-polyvinyl alcohol-bovin serum albumin coated Fe3O4 nanoparticles as anticancer drug delivery vehicle: Doxorubicin loading and in vitro release study and cytotoxicity to HepG2 and L02 cells. Mater. Sci. Eng. C Mater. Biol. Appl. 2017, 79, 410–422. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.S.; Haque, P.; Rashid, T.U.; Khan, M.N.; Mallik, A.K.; Khan, M.N.; Khan, M.; Rahman, M.M. Core-shell drug carrier from folate conjugated chitosan obtained from prawn shell for targeted doxorubicin delivery. J. Mater. Sci. Mater. Med. 2017, 28, 55. [Google Scholar] [CrossRef] [PubMed]
- Mu, X.; Zhang, F.; Kong, C.; Zhang, H.; Zhang, W.; Ge, R.; Liu, Y.; Jiang, J. EGFR-targeted delivery of DOX-loaded Fe3O4@ polydopamine multifunctional nanocomposites for MRI and antitumor chemo-photothermal therapy. Int. J. Nanomed. 2017, 12, 2899–2911. [Google Scholar] [CrossRef] [PubMed]
- Arachchige, M.P.; Laha, S.S.; Naik, A.R.; Lewis, K.T.; Naik, R.; Jena, B.P. Functionalized nanoparticles enable tracking the rapid entry and release of doxorubicin in human pancreatic cancer cells. Micron 2017, 92, 25–31. [Google Scholar] [CrossRef] [PubMed]
- Pan, C.; Liu, Y.; Zhou, M.; Wang, W.; Shi, M.; Xing, M.; Liao, W. Theranostic pH-sensitive nanoparticles for highly efficient targeted delivery of doxorubicin for breast tumor treatment. Int. J. Nanomed. 2018, 13, 1119–1137. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dutta, B.; Shetake, N.G.; Barick, B.K.; Barick, K.C.; Pandey, B.N.; Priyadarsini, K.I.; Hassan, P.A. pH sensitive surfactant-stabilized Fe3O4 magnetic nanocarriers for dual drug delivery. Colloids Surf. B Biointerfaces 2018, 162, 163–171. [Google Scholar] [CrossRef] [PubMed]
- Ji, F.; Zhang, K.; Li, J.; Gu, Y.; Zhao, J.; Zhang, J. A Dual pH/magnetic responsive nanocarrier based on PEGylated Fe3O4 nanoparticles for doxorubicin delivery. J. Nanosci. Nanotechnol. 2018, 18, 4464–4470. [Google Scholar] [CrossRef] [PubMed]
- Shelat, R.; Chandra, S.; Khanna, A. Detailed toxicity evaluation of beta-cyclodextrin coated iron oxide nanoparticles for biomedical applications. Int. J. Biol. Macromol. 2018, 110, 357–365. [Google Scholar] [CrossRef] [PubMed]
- Akilo, O.D.; Choonara, Y.E.; Strydom, A.M.; du Toit, L.C.; Kumar, P.; Modi, G.; Pillay, V. An in vitro evaluation of a carmustine-loaded Nano-co-Plex for potential magnetic-targeted intranasal delivery to the brain. Int. J. Pharm. 2016, 500, 196–209. [Google Scholar] [CrossRef] [PubMed]
- Freeman, A.C.; Platt, S.R.; Holmes, S.; Kent, M.; Robinson, K.; Howerth, E.; Eagleson, J.; Bouras, A.; Kaluzova, M.; Hadjipanayis, C.G. Convection-enhanced delivery of cetuximab conjugated iron-oxide nanoparticles for treatment of spontaneous canine intracranial gliomas. J. Neurooncol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Tseng, S.H.; Chou, M.Y.; Chu, I.M. Cetuximab-conjugated iron oxide nanoparticles for cancer imaging and therapy. Int. J. Nanomed. 2015, 10, 3663–3685. [Google Scholar] [CrossRef]
- Kaluzova, M.; Bouras, A.; Machaidze, R.; Hadjipanayis, C.G. Targeted therapy of glioblastoma stem-like cells and tumor non-stem cells using cetuximab-conjugated iron-oxide nanoparticles. Oncotarget 2015, 6, 8788–8806. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shahabadi, N.; Falsafi, H.; Mansouri, K. Improving antiproliferative effect of the anticancer drug cytarabine on human promyelocytic leukemia cells by coating on Fe3O4@SiO2 nanoparticles. Colloids Surf. B Biointerfaces 2016, 141, 213–222. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.C.; Jin, S.F.; Zheng, M.; Wang, Y.; Zhao, P.L.; Tang, D.T.; Chen, J.; Lin, J.Q.; Wang, X.H.; Zhao, P. Daunomycin-loaded superparamagnetic iron oxide nanoparticles: Preparation, magnetic targeting, cell cytotoxicity, and protein delivery research. J. Biomater. Appl. 2016, 31, 261–272. [Google Scholar] [CrossRef] [PubMed]
- Al-Jamal, K.T.; Bai, J.; Wang, J.T.; Protti, A.; Southern, P.; Bogart, L.; Heidari, H.; Li, X.; Cakebread, A.; Asker, D.; et al. Magnetic drug targeting: Preclinical in vivo studies, mathematical modeling, and extrapolation to humans. Nano Lett. 2016, 16, 5652–5660. [Google Scholar] [CrossRef] [PubMed]
- Nagesh, P.K.; Johnson, N.R.; Boya, V.K.; Chowdhury, P.; Othman, S.F.; Khalilzad-Sharghi, V.; Hafeez, B.B.; Ganju, A.; Khan, S.; Behrman, S.W.; et al. PSMA targeted docetaxel-loaded superparamagnetic iron oxide nanoparticles for prostate cancer. Colloids Surf. B Biointerfaces 2016, 144, 8–20. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rao, Y.F.; Chen, W.; Liang, X.G.; Huang, Y.Z.; Miao, J.; Liu, L.; Lou, Y.; Zhang, X.G.; Wang, B.; Tang, R.K.; et al. Epirubicin-loaded superparamagnetic iron-oxide nanoparticles for transdermal delivery: Cancer therapy by circumventing the skin barrier. Small 2015, 11, 239–247. [Google Scholar] [CrossRef] [PubMed]
- Alupei, L.; Peptu, C.A.; Lungan, A.M.; Desbrieres, J.; Chiscan, O.; Radji, S.; Popa, M. New hybrid magnetic nanoparticles based on chitosan-maltose derivative for antitumor drug delivery. Int. J. Biol. Macromol. 2016, 92, 561–572. [Google Scholar] [CrossRef] [PubMed]
- Prabha, G.; Raj, V. Formation and characterization of b-cyclodextrin (b-CD)–polyethyleneglycol (PEG)–polyethyleneimine (PEI) coated Fe3O4 nanoparticles for loading and releasing 5-Fluorouracil drug. Biomed. Pharmacother. 2016, 80, 173–182. [Google Scholar] [CrossRef] [PubMed]
- Esmaelbeygi, E.; Khoei, S.; Khoee, S.; Eynali, S. Role of iron oxide core of polymeric nanoparticles in the thermosensitivity of colon cancer cell line HT-29. Int. J. Hyperth. 2015, 31, 489–497. [Google Scholar] [CrossRef] [PubMed]
- Eynali, S.; Khoei, S.; Khoei, S.; Esmaelbeygi, E. Evaluation of the cytotoxic effects of hyperthermia and 5-fluorouracil loaded magnetic nanoparticles on human colon cancer cell line HT-29. Int. J. Hyperth. 2016, 1–33. [Google Scholar] [CrossRef] [PubMed]
- Hashemi-Moghaddam, H.; Kazemi-Bagsangani, S.; Jamili, M.; Zavareh, S. Evaluation of magnetic nanoparticles coated by 5-fluorouracil imprinted polymer for controlled drug delivery in mouse breast cancer model. Int. J. Pharm. 2016, 497, 228–238. [Google Scholar] [CrossRef] [PubMed]
- Sağir, T.; Huysal, M.; Durmus, Z.; Kurt, B.Z.; Senel, M.; Isık, S. Preparation and in vitro evaluation of 5-flourouracil loaded magnetite–zeolite nanocomposite (5-FU-MZNC) for cancer drug delivery applications. Biomed. Pharmacother. 2016, 77, 182–189. [Google Scholar] [CrossRef] [PubMed]
- Şanlı, O.; Kahraman, A.; Kondolot Solak, E.; Olukman, M. Preparation of magnetite-chitosan/methylcellulose nanospheres by entrapment and adsorption techniques for targeting the anti-cancer drug 5-fluorouracil. Artif. Cells Nanomed. Biotechnol. 2016, 44, 950–959. [Google Scholar] [CrossRef] [PubMed]
- Tuncelli, G.; Ay, A.N.; Zümreoglu-Karan, B. 5-Fluorouracil intercalated iron oxide@layered double hydroxide core-shell nano-composites with isotropic and anisotropic architectures for shape-selective drug delivery applications. Mater. Sci. Eng. C Mater. Biol. Appl. 2015, 55, 562–568. [Google Scholar] [CrossRef] [PubMed]
- Asadi, N.; Annabi, N.; Mostafavi, E.; Anzabi, M.; Khalilov, R.; Saghfi, S.; Mehrizadeh, M.; Akbarzadeh, A. Synthesis, characterization and in vitro evaluation of magnetic nanoparticles modified with PCL-PEG-PCL for controlled delivery of 5FU. Artif. Cells Nanomed. Biotechnol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Mu, Q.; Lin, G.; Patton, V.K.; Wang, K.; Press, O.W.; Zhang, M. Gemcitabine and chlorotoxin conjugated iron oxide nanoparticles for glioblastoma therapy. J. Mater. Chem. B Mater. Biol. Med. 2016, 4, 32–36. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aires, A.; Ocampo, S.M.; Simões, B.M.; Josefa Rodríguez, M.; Cadenas, J.F.; Couleaud, P.; Spence, K.; Latorre, A.; Miranda, R.; Somoza, Á.; et al. Multifunctionalized iron oxide nanoparticles for selective drug delivery to CD44-positive cancer cells. Nanotechnology 2016, 27, 065103. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, R.V.; Martins, T.M.; Goes, A.M.; Fabris, J.D.; Cavalcante, L.C.; Outon, L.E.; Domingues, R.Z. Termosensitive gemcitabine-magnetoliposomes for combined hyperthermia and chemotherapy. Nanotechnology 2016, 27, 085105. [Google Scholar] [CrossRef] [PubMed]
- Hamarat Sanlıer, S.; Yasa, M.; Cihnioglu, A.O.; Abdulhayoglu, M.; Yılmaz, H.; Ak, G. Development of gemcitabine-adsorbed magnetic gelatin nanoparticles for targeted drug delivery in lung cancer. Artif. Cells Nanomed. Biotechnol. 2016, 44, 943–949. [Google Scholar] [CrossRef] [PubMed]
- Jaidev, L.R.; Chellappan, D.R.; Bhavsar, D.V.; Ranganathan, R.; Sivanantham, B.; Subramanian, A.; Sharma, U.; Jagannathan, N.R.; Krishnan, U.M.; Sethuraman, S. Multi-functional nanoparticles as theranostic agents for the treatment & imaging of pancreatic cancer. Acta Biomater. 2017, 49, 422–433. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.; Li, Y.; Li, Y.; Wu, H.; Yu, F.; Zhou, S.; Xie, L.; Luo, F.; Lin, C.; Hou, Z. Drug/dye-loaded, multifunctional PEG-chitosan-iron oxide nanocomposites for methotraxate synergistically self-targeted cancer therapy and dual model imaging. ACS Appl. Mater. Interfaces 2015, 7, 11908–11920. [Google Scholar] [CrossRef] [PubMed]
- Jahangiri, S.; Akbarzadeh, A. Preparation and in vitro evaluation of Methotrexate-loaded magnetic nanoparticles modified with biocompatible copolymers. Artif. Cells Nanomed. Biotechnol. 2016, 44, 1733–1740. [Google Scholar] [CrossRef] [PubMed]
- Nosrati, H.; Salehiabar, M.; Davaran, S.; Danafar, H.; Manjili, H.K. Methotrexate-conjugated L-lysine coated iron oxide magnetic nanoparticles for inhibition of MCF-7 breast cancer cells. Drug Dev. Ind. Pharm. 2017, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Barar, J.; Kafil, V.; Majd, M.H.; Barzegari, A.; Khani, S.; Johari-Ahar, M.; Asgari, D.; Coukos, G.; Omidi, Y. Multifunctional mitoxantrone-conjugated magnetic nanosystem for targeted therapy of folate receptor-overexpressing malignant cells. J. Nanobiotechnol. 2015, 13, 26. [Google Scholar] [CrossRef] [PubMed]
- Cicha, I.; Scheffler, L.; Ebenau, A.; Lyer, S.; Alexiou, C.; Goppelt-Struebe, M. Mitoxantrone-loaded superparamagnetic iron oxide nanoparticles as drug carriers for cancer therapy: Uptake and toxicity in primary human tubular epithelial cells. Nanotoxicology 2016, 10, 557–566. [Google Scholar] [CrossRef] [PubMed]
- Hornung, A.; Poettler, M.; Friedrich, R.P.; Zaloga, J.; Unterweger, H.; Lyer, S.; Nowak, J.; Odenbach, S.; Alexiou, C.; Janko, C. Treatment efficiency of free and nanoparticle-loaded mitoxantrone for magnetic drug targeting in multicellular tumor spheroids. Molecules 2015, 20, 18016–18030. [Google Scholar] [CrossRef] [PubMed]
- Zaloga, J.; Pöttler, M.; Leitinger, G.; Friedrich, R.P.; Almer, G.; Lyer, S.; Baum, E.; Tietze, R.; Heimke-Brinck, R.; Mangge, H.; et al. Pharmaceutical formulation of HSA hybrid coated iron oxide nanoparticles for magnetic drug targeting. Eur. J. Pharm. Biopharm. 2016, 101, 152–162. [Google Scholar] [CrossRef] [PubMed]
- Hsiao, M.H.; Mu, Q.; Stephen, Z.R.; Fang, C.; Zhang, M. Hexanoyl-chitosan-PEG copolymer coated iron oxide nanoparticles for hydrophobic drug delivery. ACS Macro Lett. 2015, 4, 403–407. [Google Scholar] [CrossRef] [PubMed]
- Jeon, H.; Kim, J.; Lee, Y.M.; Kim, J.; Choi, H.W.; Lee, J.; Park, H.; Kang, Y.; Kim, I.S.; Lee, B.H.; et al. Poly-paclitaxel/cyclodextrin-SPION nano-assembly for magnetically guided drug delivery system. J. Control Release 2016, 231, 68–76. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.Y.; Carugo, D.; Crake, C.; Owen, J.; de Saint Victor, M.; Seth, A.; Coussios, C.; Stride, E. Nanoparticle-loaded protein-polymer nanodroplets for improved stability and conversion efficiency in ultrasound imaging and drug delivery. Adv. Mater. 2015, 27, 5484–5492. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, D.H.; Lee, J.S.; Choi, J.H.; Park, K.M.; Lee, Y.; Park, K.D. Hierarchical self-assembly of magnetic nanoclusters for theranostics: Tunable size, enhanced magnetic resonance imagability, and controlled and targeted drug delivery. Acta Biomater. 2016, 35, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Ruggiero, M.R.; Crich, S.G.; Sieni, E.; Sgarbossa, P.; Forzan, M.; Cavallari, E.; Stefania, R.; Dughiero, F.; Aime, S. Magnetic hyperthermia efficiency and (1)H-NMR relaxation properties of iron oxide/paclitaxel-loaded PLGA nanoparticles. Nanotechnology 2016, 27, 285104. [Google Scholar] [CrossRef] [PubMed]
- Swain, A.K.; Pradhan, L.; Bahadur, D. Polymer Stabilized Fe3O4-graphene as an amphiphilic drug carrier for thermo-chemotherapy of cancer. ACS Appl. Mater. Interfaces 2015, 7, 8013–8022. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Yang, Y.; Jia, Y.; Pu, X.; Yang, T.; Wang, Y.; Ma, X.; Chen, Q.; Sun, M.; Wei, D.; et al. Enhanced tumor targeting effects of a novel paclitaxel-loaded polymer: PEG-PCCL-modified magnetic iron oxide nanoparticles. Drug Deliv. 2017, 24, 1284–1294. [Google Scholar] [CrossRef] [PubMed]
- Patra, S.; Roy, E.; Karfa, P.; Kumar, S.; Madhuri, R.; Sharma, P.K. Dual-responsive polymer coated superparamagnetic nanoparticle for targeted drug delivery and hyperthermia treatment. ACS Appl. Mater. Interfaces 2015, 7, 9235–9246. [Google Scholar] [CrossRef] [PubMed]
- Prabha, G.; Raj, V. Preparation and characterization of chitosan-Polyethylene glycol-polyvinylpyrrolidone-coated superparamagnetic iron oxide nanoparticles as carrier system: Drug loading and in vitro drug release study. J. Biomed. Mater. Res. B Appl. Biomater. 2016, 104, 808–816. [Google Scholar] [CrossRef] [PubMed]
- Mancarella, S.; Greco, V.; Baldassarre, F.; Vergara, D.; Maffia, M.; Leporatti, S. Polymer-coated magnetic nanoparticles for curcumin delivery to cancer cells. Macromol. Biosci. 2015, 15, 1365–1374. [Google Scholar] [CrossRef] [PubMed]
- Unterweger, H.; Subatzus, D.; Tietze, R.; Janko, C.; Poettler, M.; Stiegelschmitt, A.; Schuster, M.; Maake, C.; Boccaccini, A.R.; Alexiou, C. Hypericin-bearing magnetic iron oxide nanoparticles for selective drug delivery in photodynamic therapy. Int. J. Nanomed. 2015, 10, 6985–6996. [Google Scholar] [CrossRef] [PubMed]
- Sreeja, S.; Krishnan Nair, C.K. Tumor control by hypoxia-specific chemotargeting of iron-oxide nanoparticle—Berberine complexes in a mouse model. Life Sci. 2018, 195, 71–80. [Google Scholar] [CrossRef] [PubMed]
- Bilcu, M.; Grumezescu, A.M.; Oprea, A.E.; Popescu, R.C.; Mogoșanu, G.D.; Hristu, R.; Stanciu, G.A.; Mihailescu, D.F.; Lazar, V.; Bezirtzoglou, E.; et al. Efficiency of vanilla, patchouli and ylang ylang essential oils stabilized by iron oxide@C14 nanostructures against bacterial adherence and biofilms formed by Staphylococcus aureus and Klebsiella pneumoniae clinical strains. Molecules 2014, 19, 17943–17956. [Google Scholar] [CrossRef] [PubMed]
- Sollinger, H.W. Mycophenolate mofetil for the prevention of acute rejection in primary cadaveric renal allograft recipients. U.S. renal transplant mycophenolate mofetil study group. Transplantation 1995, 60, 225–232. [Google Scholar] [CrossRef] [PubMed]
- Hwang, J.; Lee, E.; Kim, J.; Seo, Y.; Lee, K.H.; Hong, J.W.; Gilad, A.A.; Park, H.; Choi, J. Effective delivery of immunosuppressive drug molecules by silica coated iron oxide nanoparticles. Colloids Surf. B Biointerfaces 2016, 142, 290–296. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Smeets, R.L.; Koenen, H.J.; Vink, P.M.; Wagenaars, J.; Boots, A.M.; Joosten, I. Mycophenolic acid-mediated suppression of human CD4+ T cells: More than mere guanine nucleotide deprivation. Am. J. Transpl. 2011, 11, 439–449. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Yuan, X.; Luo, Y.; He, Y.; Jiang, Y.; Chen, Z.K.; Sun, E. Evaluating the effects of immunosuppressants on human immunity using cytokine profiles of whole blood. Cytokine 2009, 45, 141–147. [Google Scholar] [CrossRef] [PubMed]
- Fu, T.; Kong, Q.; Sheng, H.; Gao, L. Value of functionalized superparamagnetic iron oxide nanoparticles in the diagnosis and treatment of acute temporal lobe epilepsy on MRI. Neural Plast. 2016, 2016, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Rosillo-de la Torre, A.; Zurita-Olvera, L.; Orozco-Suárez, S.; Garcia Casillas, P.E.; Salgado-Ceballos, H.; Luna-Bárcenas, G.; Rocha, L. Phenytoin carried by silica core iron oxide nanoparticles reduces the expression of pharmacoresistant seizures in rats. Nanomedicine 2015, 10, 3563–3577. [Google Scholar] [CrossRef] [PubMed]
- Lazarowski, A.; Ramos, A.J.; García-Rivello, H.; Brusco, A.; Girardi, E. Neuronal and glial expression of the multidrug resistance gene product in an experimental epilepsy model. Cell. Mol. Neurobiol. 2004, 24, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Attia, M.F.; Anton, N.; Khan, I.U.; Serra, C.A.; Messaddeq, N.; Jakhmola, A.; Vecchione, R.; Vandamme, T. One-step synthesis of iron oxide polypyrrole nanoparticles encapsulating ketoprofen as model of hydrophobic drug. Int. J. Pharm. 2016, 508, 61–70. [Google Scholar] [CrossRef] [PubMed]
- Jia, Y.; Fan, M.; Chen, H.; Miao, Y.; Xing, L.; Jiang, B.; Cheng, Q.; Liu, D.; Bao, W.; Qian, B.; et al. Magnetic hyaluronic acid nanospheres via aqueous Diels-Alder chemistry to deliver dexamethasone for adipose tissue engineering. J. Colloid Interface Sci. 2015, 458, 293–299. [Google Scholar] [CrossRef] [PubMed]
- Lafond, J.F.; Shimoji, M.; Ramaswamy, B.; Shukoor, M.I.; Malik, P.; Shapiro, B.; Depireux, D.A. Middle ear histopathology following magnetic delivery to the cochlea of prednisolone-loaded iron oxide nanoparticles in rats. Toxicol. Pathol. 2018, 46, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Hussein-Al-Ali, S.H.; El Zowalaty, M.E.; Hussein, M.Z.; Ismail, M.; Webster, T.J. Synthesis, characterization, controlled release, and antibacterial studies of a novel streptomycin chitosan magnetic nanoantibiotic. Int. J. Nanomed. 2014, 16, 549–557. [Google Scholar] [CrossRef]
- El Zowalaty, M.E.; Hussein-Al-Ali, S.H.; Husseiny, M.I.; Geilich, B.M.; Webster, T.J.; Hussein, M.Z. The ability of streptomycin-loaded chitosan-coated magnetic nanocomposites to possess antimicrobial and antituberculosis activities. Int. J. Nanomed. 2015, 10, 3269–3274. [Google Scholar] [CrossRef] [PubMed]
- Ivashchenko, O.; Jurga-Stopa, J.; Coy, E.; Peplinska, B.; Pietralik, Z.; Jurga, S. Fourier transform infrared and Raman spectroscopy studies on magnetite/Ag/antibiotic nanocomposites. Appl. Surf. Sci. 2016, 364, 400–409. [Google Scholar] [CrossRef]
- Ivashchenko, O.; Coy, E.; Peplinska, B.; Jarek, M.; Lewandowski, M.; Załęski, K.; Warowicka, A.; Wozniak, A.; Babutina, T.; Jurga-Stopa, J.; et al. Influence of silver content on rifampicin adsorptivity for magnetite/Ag/rifampicin nanoparticles. Nanotechnology 2017, 28, 055603. [Google Scholar] [CrossRef] [PubMed]
- Masadeh, M.M.; Karasneh, G.A.; Al-Akhras, M.A.; Albiss, B.A.; Aljarah, K.M.; Al-Azzam, S.I.; Alzoubi, K.H. Cerium oxide and iron oxide nanoparticles abolish the antibacterial activity of ciprofloxacin against gram positive and gram negative biofilm bacteria. Cytotechnology 2015, 67, 427–435. [Google Scholar] [CrossRef] [PubMed]
- Bandara, H.M.; Nguyen, D.; Mogarala, S.; Osiñski, M.; Smyth, H.D. Magnetic fields suppress Pseudomonas aeruginosa biofilms and enhance ciprofloxacin activity. Biofouling 2015, 31, 443–457. [Google Scholar] [CrossRef] [PubMed]
- Istrate, C.M.; Holban, A.M.; Grumezescu, A.M.; Mogoantă, L.; Mogoşanu, G.D.; Savopol, T.; Moisescu, M.; Iordache, M.; Vasile, B.S.; Kovacs, E. Iron oxide nanoparticles modulate the interaction of different antibiotics with cellular membranes. Rom. J. Morphol. Embryol. 2014, 55, 849–856. [Google Scholar] [PubMed]
- Hussein-Al-Ali, S.H.; El Zowalaty, M.E.; Kura, A.U.; Geilich, B.; Fakurazi, S.; Webster, T.J.; Hussein, M.Z. Antimicrobial and controlled release studies of a novel nystatin conjugated iron oxide nanocomposite. Biomed. Res. Int. 2014, 2014, 651831. [Google Scholar] [CrossRef] [PubMed]
- Maltas, E.; Ozmen, M.; Yildirimer, B.; Kucukkolbasi, S.; Yildiz, S. Interaction between ketoconazole and human serum albumin on epoxy modified magnetic nanoparticles for drug delivery. J. Nanosci. Nanotechnol. 2013, 13, 6522–6528. [Google Scholar] [CrossRef] [PubMed]
- Niemirowicz, K.; Durnaś, B.; Tokajuk, G.; Głuszek, K.; Wilczewska, A.Z.; Misztalewska, I.; Mystkowska, J.; Michalak, G.; Sodo, A.; Wątek, M.; et al. Magnetic nanoparticles as a drug delivery system that enhance fungicidal activity of polyene antibiotics. Nanomedicine 2016, 12, 2395–2404. [Google Scholar] [CrossRef] [PubMed]
- Pelgrift, R.Y.; Friedman, A.J. Nanotechnology as a therapeutic tool to combat microbial resistance. Adv. Drug Deliv. Rev. 2013, 65, 1803–1815. [Google Scholar] [CrossRef] [PubMed]
- Pan, X.; Redding, J.E.; Wiley, P.A.; Wen, L.; McConnell, J.S.; Zhang, B. Mutagenicity evaluation of metal oxide nanoparticles by the bacterial reverse mutation assay. Chemosphere 2010, 79, 113–116. [Google Scholar] [CrossRef] [PubMed]
- Saleh, N.B.; Chambers, B.; Aich, N.; Plazas-Tuttle, J.; Phung-Ngoc, H.N.; Kirisits, M.J. Mechanistic lessons learned from studies of planktonic bacteria with metallic nanomaterials: Implications for interactions between nanomaterials and biofilm bacteria. Front. Microbiol. 2015, 6, 677. [Google Scholar] [CrossRef] [PubMed]
- Grumezescu, A.M.; Gestal, M.C.; Holban, A.M.; Grumezescu, V.; Vasile, B.S.; Mogoantă, L.; Iordache, F.; Bleotu, C.; Mogoșanu, G.D. Biocompatible Fe3O4 increases the efficacy of amoxicillin delivery against Gram-positive and Gram-negative bacteria. Molecules 2014, 19, 5013–5027. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Zhang, K.; Zhou, Z.; Li, Q.; Shao, L.; Hao, R.Z.; Xiao, R.; Wang, S. Vancomycin-modified Fe3O4@SiO2@Ag microflowers as effective antimicrobial agents. Int. J. Nanomed. 2017, 12, 3077–3094. [Google Scholar] [CrossRef] [PubMed]
- Shah, S.T.; A Yehya, W.; Saad, O.; Simarani, K.; Chowdhury, Z.; A Alhadi, A.; Al-Ani, L.A. Surface functionalization of iron oxide nanoparticles with gallic acid as potential antioxidant and antimicrobial agents. Nanomaterials 2017, 7, 306. [Google Scholar] [CrossRef] [PubMed]
- Borcherding, J.; Baltrusaitis, J.; Chen, H.; Stebounova, L.; Wu, C.M.; Rubasinghege, G.; Mudunkotuwa, I.A.; Caraballo, J.C.; Zabner, J.; Grassian, V.H.; et al. Iron oxide nanoparticles induce Pseudomonas aeruginosa growth, induce biofilm formation, and inhibit antimicrobial peptide function. Environ. Sci. Nano 2014, 1, 123–132. [Google Scholar] [CrossRef] [PubMed]
- Haney, C.; Rowe, J.J.; Robinson, J.B. Spions increase biofilm formation by Pseudomonas aeruginosa. J. Biomater. Nanobiotechnol. 2012, 3, 508–518. [Google Scholar] [CrossRef]
- Tokajuk, G.; Niemirowicz, K.; Deptula, P.; Piktel, E.; Ciesluk, M.; Wilczewska, A.Z.; Dąbrowski, J.R.; Bucki, R. Use of magnetic nanoparticles as a drug delivery system to improve chlorhexidine antimicrobial activity. Int. J. Nanomed. 2017, 12, 7833–7846. [Google Scholar] [CrossRef] [PubMed]
- Luo, D.; Shahid, S.; Hasan, S.M.; Whiley, R.; Sukhorukov, G.B.; Cattell, M.J. Controlled release of chlorhexidine from a HEMA-UDMA resin using a magnetic field. Dent. Mater. 2018, 34, 764–775. [Google Scholar] [CrossRef] [PubMed]
- Morones, J.R.; Elechiguerra, J.L.; Camacho, A.; Holt, K.; Kouri, J.B.; Ramírez, J.T.; Yacaman, M.J. The bactericidal effect of silver nanoparticles. Nanotechnology 2005, 16, 2346–2353. [Google Scholar] [CrossRef] [PubMed]
- Nel, A.E.; Mädler, L.; Velegol, D.; Xia, T.; Hoek, E.M.; Somasundaran, P.; Klaessig, F.; Castranova, V.; Thompson, M. Understanding biophysicochemical interactions at the nano-bio interface. Nat. Mater. 2009, 8, 543–557. [Google Scholar] [CrossRef] [PubMed]
- Javanbakht, T.; Laurent, S.; Stanicki, D.; Wilkinson, K.J. Relating the surface properties of superparamagnetic iron oxide nanoparticles (SPIONs) to their bactericidal effect towards a biofilm of Streptococcus mutans. PLoS ONE 2016, 11, e0154445. [Google Scholar] [CrossRef] [PubMed]
- Gao, L.; Liu, Y.; Kim, D.; Li, Y.; Hwang, G.; Naha, P.C.; Cormode, D.P.; Koo, H. Nanocatalysts promote Streptococcus mutans biofilm matrix degradation and enhance bacterial killing to suppress dental caries in vivo. Biomaterials 2016, 101, 272–284. [Google Scholar] [CrossRef] [PubMed]
- Valdiglesias, V.; Kiliç, G.; Costa, C.; Fernández-Bertólez, N.; Pásaro, E.; Teixeira, J.P.; Laffon, B. Effects of iron oxide nanoparticles: Cytotoxicity, genotoxicity, developmental toxicity, and neurotoxicity. Environ. Mol. Mutagen. 2015, 56, 125–148. [Google Scholar] [CrossRef] [PubMed]
- Patil, U.S.; Adireddy, S.; Jaiswal, A.; Mandava, S.; Lee, B.R.; Chrisey, D.B. In vitro/In vivo toxicity evaluation and quantification of iron oxide nanoparticles. Int. J. Mol. Sci. 2015, 16, 24417–24450. [Google Scholar] [CrossRef] [PubMed]
- Sonmez, E.; Aydin, E.; Turkez, H.; Özbek, E.; Togar, B.; Meral, K.; Çetin, D.; Cacciatore, I.; Di Stefano, A. Cytotoxicity and genotoxicity of iron oxide nanoparticles: An in vitro biosafety study. Arch. Biol. Sci. 2016, 68, 41–50. [Google Scholar] [CrossRef]
- Xu, H.L.; Mao, K.L.; Huang, Y.P.; Yang, J.J.; Xu, J.; Chen, P.P.; Fan, Z.L.; Zou, S.; Gao, Z.Z.; Yin, J.Y.; et al. Glioma-targeted superparamagnetic iron oxide nanoparticles as drug-carrying vehicles for theranostic effects. Nanoscale 2016, 8, 14222–14236. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Ju, J.E.; Kim, B.I.; Pak, P.J.; Choi, E.K.; Lee, H.S.; Chung, N. Rod-shaped iron oxide nanoparticles are more toxic than sphere-shaped nanoparticles to murine macrophage cells. Environ. Toxicol. Chem. 2014, 33, 2759–2766. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.C.; Lee, J.H.; Lee, J.; Kim, H.Y.; Park, J.Y.; Cho, J.; Lee, J.; Han, D.W. Subtle cytotoxicity and genotoxicity differences in superparamagnetic iron oxide nanoparticles coated with various functional groups. Int. J. Nanomed. 2011, 6, 3219–3231. [Google Scholar] [CrossRef]
- Feng, Q.; Liu, Y.; Huang, J.; Chen, K.; Huang, J.; Xiao, K. Uptake, distribution, clearance, and toxicity of iron oxide nanoparticles with different sizes and coatings. Sci. Rep. 2018, 8, 2082. [Google Scholar] [CrossRef] [PubMed]
- Valdiglesias, V.; Fernández-Bertólez, N.; Kiliç, G.; Costa, C.; Costa, S.; Fraga, S.; Bessa, M.J.; Pásaro, E.; Teixeira, J.P.; Laffon, B. Are iron oxide nanoparticles safe? Current knowledge and future perspectives. J. Trace Elem. Med. Biol. 2016, 38, 53–63. [Google Scholar] [CrossRef] [PubMed]
- Bellusci, M.; La Barbera, A.; Padella, F.; Mancuso, M.; Pasquo, A.; Grollino, M.G.; Leter, G.; Nardi, E.; Cremisini, C.; Giardullo, P.; et al. Biodistribution and acute toxicity of a nanofluid containing manganese iron oxide nanoparticles produced by a mechanochemical process. Int. J. Nanomed. 2014, 9, 1919–1929. [Google Scholar] [CrossRef] [Green Version]
- Hajikarimi, Z.; Khoei, S.; Khoee, S.; Mahdavi, S.R. Evaluation of the cytotoxic effects of PLGA coated iron oxide nanoparticles as a carrier of 5-fluorouracil and mega-voltage X-ray radiation in DU145 prostate cancer cell line. IEEE Trans. Nanobiosci. 2014, 13, 403–408. [Google Scholar] [CrossRef] [PubMed]
- Raji, M.A.; Amara, M.; Amoabediny, G.; Tajik, P.; Barin, A.; Magierowski, S.; Ghafar-Zadeh, E. Cytotoxicity of synthesized iron oxide nanoparticles: Toward novel biomarkers of colon cancer. Conf. Proc. IEEE Eng. Med. Biol. Soc. 2014, 2014, 6179–6182. [Google Scholar] [CrossRef] [PubMed]
- Calero, M.; Gutiérrez, L.; Salas, G.; Luengo, Y.; Lázaro, A.; Acedo, P.; Morales, M.P.; Miranda, R.; Villanueva, A. Efficient and safe internalization of magnetic iron oxide nanoparticles: Two fundamental requirements for biomedical applications. Nanomedicine 2014, 10, 733–743. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sun, B.; Liu, R.; Ye, N.; Xiao, Z.D. Comprehensive evaluation of microRNA expression profiling reveals the neural signaling specific cytotoxicity of superparamagnetic iron oxide nanoparticles (SPIONs) through N-methyl-d-aspartate receptor. PLoS ONE 2015, 10, e0121671. [Google Scholar] [CrossRef] [PubMed]
- Sadeghi, L.; Tanwir, F.; Yousefi Babadi, V. In vitro toxicity of iron oxide nanoparticle: Oxidative damages on Hep G2 cells. Exp. Toxicol. Pathol. 2015, 67, 197–203. [Google Scholar] [CrossRef] [PubMed]
- Yalcin, S.; Khodadust, R.; Unsoy, G.; Garip, I.C.; Mumcuoglu, Z.D.; Gunduz, U. Synthesis and characterization of polyhydroxybutyrate coated magnetic nanoparticles: Toxicity analyses on different cell lines. Synth. React. Inorg. Met.-Org. Nano-Met. Chem. 2015, 45, 700–708. [Google Scholar] [CrossRef]
- Cheng, K.K.; Chan, P.S.; Fan, S.; Kwan, S.M.; Yeung, K.L.; Wang, Y.X.; Chow, A.H.; Wu, E.X.; Baum, L. Curcumin-conjugated magnetic nanoparticles for detecting amyloid plaques in Alzheimer’s disease mice using magnetic resonance imaging (MRI). Biomaterials 2015, 44, 155–172. [Google Scholar] [CrossRef] [PubMed]
- Comănescu, M.V.; Mocanu, M.A.; Anghelache, L.; Marinescu, B.; Dumitrache, F.; Bădoi, A.D.; Manda, G. Toxicity of L-DOPA coated iron oxide nanoparticles in intraperitoneal delivery setting—Preliminary preclinical study. Rom. J. Morphol. Embryol. 2015, 56, 691–696. [Google Scholar] [PubMed]
- Couto, D.; Sousa, R.; Andrade, L.; Leander, M.; Lopez-Quintela, M.A.; Rivas, J.; Freitas, P.; Lima, M.; Porto, G.; Porto, B.; et al. Polyacrylic acid coated and non-coated iron oxide nanoparticles are not genotoxic to human T lymphocytes. Toxicol. Lett. 2015, 234, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Ghasempour, S.; Shokrgozar, M.A.; Ghasempour, R.; Alipour, M. Investigating the cytotoxicity of iron oxide nanoparticles in in vivo and in vitro studies. Exp. Toxicol. Pathol. 2015, 67, 509–515. [Google Scholar] [CrossRef] [PubMed]
- Liao, S.H.; Liu, C.H.; Bastakoti, B.P.; Suzuki, N.; Chang, Y.; Yamauchi, Y.; Lin, F.H.; Wu, K.C. Functionalized magnetic iron oxide/alginate core-shell nanoparticles for targeting hyperthermia. Int. J. Nanomed. 2015, 10, 3315–3328. [Google Scholar] [CrossRef]
- Magdolenova, Z.; Drlickova, M.; Henjum, K.; Rundén-Pran, E.; Tulinska, J.; Bilanicova, D.; Pojana, G.; Kazimirova, A.; Barancokova, M.; Kuricova, M.; et al. Coating-dependent induction of cytotoxicity and genotoxicity of iron oxide nanoparticles. Nanotoxicology 2015, 9, 44–56. [Google Scholar] [CrossRef] [PubMed]
- Venkatesan, K.; Rajan Babu, D.; Kavya Bai, M.P.; Supriya, R.; Vidya, R.; Madeswaran, S.; Anandan, P.; Arivanandhan, M.; Hayakawa, Y. Structural and magnetic properties of cobalt-doped iron oxide nanoparticles prepared by solution combustion method for biomedical applications. Int. J. Nanomed. 2015, 10, 189–198. [Google Scholar] [CrossRef]
- Paolini, A.; Guarch, C.P.; Ramos-López, D.; de Lapuente, J.; Lascialfari, A.; Guari, Y.; Larionova, J.; Long, J.; Nano, R. Rhamnose-coated superparamagnetic iron-oxide nanoparticles: An evaluation of their in vitro cytotoxicity, genotoxicity and carcinogenicity. J. Appl. Toxicol. 2016, 36, 510–520. [Google Scholar] [CrossRef] [PubMed]
- Bhandari, R.; Gupta, P.; Dziubla, T.; Hilt, J.Z. Single step synthesis, characterization and applications of curcumin functionalized iron oxide magnetic nanoparticles. Mater. Sci. Eng. C Mater. Biol. Appl. 2016, 67, 59–64. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Friedrich, R.P.; Zaloga, J.; Schreiber, E.; Tóth, I.Y.; Tombácz, E.; Lyer, S.; Alexiou, C. Tissue plasminogen activator binding to superparamagnetic iron oxide nanoparticle-covalent versus adsorptive approach. Nanoscale Res. Lett. 2016, 11, 297. [Google Scholar] [CrossRef] [PubMed]
- Park, J.; Kadasala, N.R.; Abouelmagd, S.A.; Castanares, M.A.; Collins, D.S.; Wei, A.; Yeo, Y. Polymer-iron oxide composite nanoparticles for EPR-independent drug delivery. Biomaterials 2016, 101, 285–295. [Google Scholar] [CrossRef] [PubMed]
- Rajiv, S.; Jerobin, J.; Saranya, V.; Nainawat, M.; Sharma, A.; Makwana, P.; Gayathri, C.; Bharath, L.; Singh, M.; Kumar, M.; et al. Comparative cytotoxicity and genotoxicity of cobalt (II, III) oxide, iron (III) oxide, silicon dioxide, and aluminum oxide nanoparticles on human lymphocytes in vitro. Hum. Exp. Toxicol. 2016, 35, 170–183. [Google Scholar] [CrossRef] [PubMed]
- Jarockyte, G.; Daugelaite, E.; Stasys, M.; Statkute, U.; Poderys, V.; Tseng, T.C.; Hsu, S.H.; Karabanovas, V.; Rotomskis, R. Accumulation and toxicity of superparamagnetic iron oxide nanoparticles in cells and experimental animals. Int. J. Mol. Sci. 2016, 17, 1193. [Google Scholar] [CrossRef] [PubMed]
- Song, X.; Gu, X.; Sun, H.; Fu, C.; Zhang, Y.; Dong, P. Biomimetic modification and in vivo safety assessment of superparamagnetic iron oxide nanoparticles. J. Nanosci. Nanotechnol. 2016, 16, 4100–4107. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Mao, K.; Zhang, B.; Zhao, Y. Superparamagnetic iron oxide nanoparticles conjugated with folic acid for dual target-specific drug delivery and MRI in cancer theranostics. Mater. Sci. Eng. C Mater. Biol. Appl. 2017, 70, 763–771. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Xia, J.; Pang, X.; Zhao, M.; Wang, B.; Yang, L.; Wan, H.; Wu, J.; Fu, S. Magnetic nanoparticle-loaded electrospun polymeric nanofibers for tissue engineering. Mater. Sci. Eng. C Mater. Biol. Appl. 2017, 73, 537–543. [Google Scholar] [CrossRef] [PubMed]
- Sohrabijam, Z.; Saeidifar, M.; Zamanian, A. Enhancement of magnetofection efficiency using chitosan coated superparamagnetic iron oxide nanoparticles and calf thymus DNA. Colloids Surf. B Biointerfaces 2017, 152, 169–175. [Google Scholar] [CrossRef] [PubMed]
- Moise, S.; Céspedes, E.; Soukup, D.; Byrne, J.M.; El Haj, A.J.; Telling, N.D. The cellular magnetic response and biocompatibility of biogenic zinc- and cobalt-doped magnetite nanoparticles. Sci. Rep. 2017, 7, 39922. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Patel, S.; Jana, S.; Chetty, R.; Thakore, S.; Singh, M.; Devkar, R. Toxicity evaluation of magnetic iron oxide nanoparticles reveals neuronal loss in chicken embryo. Drug Chem. Toxicol. 2017, 27, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Salimi, M.; Sarkar, S.; Fathi, S.; Alizadeh, A.M.; Saber, R.; Moradi, F.; Delavari, H. Biodistribution, pharmacokinetics, and toxicity of dendrimer-coated iron oxide nanoparticles in BALB/c mice. Int. J. Nanomed. 2018, 13, 1483–1493. [Google Scholar] [CrossRef] [PubMed]
- Shevtsov, M.; Nikolaev, B.; Marchenko, Y.; Yakovleva, L.; Skvortsov, N.; Mazur, A.; Tolstoy, P.; Ryzhov, V.; Multhoff, G. Targeting experimental orthotopic glioblastoma with chitosan-based superparamagnetic iron oxide nanoparticles (CS-DX-SPIONs). Int. J. Nanomed. 2018, 13, 1471–1482. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Apopa, P.L.; Qian, Y.; Shao, R.; Guo, N.L.; Schwegler-Berry, D.; Pacurari, M.; Porter, D.; Shi, X.; Vallyathan, V.; Castranova, V.; et al. Iron oxide nanoparticles induce human microvascular endothelial cell permeability through reactive oxygen species production and microtubule remodeling. Part. Fibre Toxicol. 2009, 6, 1. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Singh, N.; Jenkins, G.J.; Asadi, R.; Doak, S.H. Potential toxicity of superparamagnetic iron oxide nanoparticles (SPION). Nano Rev. 2010, 1, 5358. [Google Scholar] [CrossRef] [PubMed]
- Yarjanli, Z.; Ghaedi, K.; Esmaeili, A.; Rahgozar, S.; Zarrabi, A. Iron oxide nanoparticles may damage to the neural tissue through iron accumulation, oxidative stress, and protein aggregation. BMC Neurosci. 2017, 18, 51. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dwivedi, S.; Siddiqui, M.A.; Farshori, N.N.; Ahamed, M.; Musarrat, J.; Al-Khedhairy, A.A. Synthesis, characterization and toxicological evaluation of iron oxide nanoparticles in human lung alveolar epithelial cells. Colloids Surf. B Biointerfaces 2014, 122, 209–215. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Zhao, M.; Yang, F.; Mao, Y.; Xie, H.; Zhou, Q. Iron overload by superparamagnetic iron oxide nanoparticles is a high risk factor in cirrhosis by a systems toxicology assessment. Sci. Rep. 2016, 6, 29110. [Google Scholar] [CrossRef] [PubMed]
- Mahmoudi, M.; Simchi, A.; Imani, M.; Milani, A.S.; Stroeve, P. An in vitro study of bare and poly(ethylene glycol)-co-fumaratecoated superparamagnetic iron oxide nanoparticles: A new toxicity identification procedure. Nanotechnology 2009, 20, 225104. [Google Scholar] [CrossRef] [PubMed]
- Mahmoudi, M.; Simchi, A.; Imani, M.; Shokrgozar, M.A.; Milani, A.S.; Häfeli, U.O.; Stroeve, P. A new approach for the in vitro identification of the cytotoxicity of superparamagnetic iron oxide nanoparticles. Colloids Surf. B Biointerfaces 2010, 75, 300–309. [Google Scholar] [CrossRef] [PubMed]
- Sanganeria, P.; Sachar, S.; Chandra, S.; Bahadur, D.; Ray, P.; Khanna, A. Cellular internalization and detailed toxicity analysis of protein-immobilized iron oxide nanoparticles. J. Biomed. Mater. Res. B Appl. Biomater. 2015, 103, 125–134. [Google Scholar] [CrossRef] [PubMed]
- Pöttler, M.; Staicu, A.; Zaloga, J.; Unterweger, H.; Weigel, B.; Schreiber, E.; Hofmann, S.; Wiest, I.; Jeschke, U.; Alexiou, C.; et al. Genotoxicity of superparamagnetic iron oxide nanoparticles in granulosa cells. Int. J. Mol. Sci. 2015, 16, 26280–26290. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Zhang, H.; Liang, X.; Zhang, J.; Tao, W.; Zhu, X.; Chang, D.; Zeng, X.; Liu, G.; Mei, L. Iron oxide nanoparticles induce autophagosome accumulation through multiple mechanisms: Lysosome impairment, mitochondrial damage, and ER stress. Mol. Pharm. 2016, 13, 2578–2587. [Google Scholar] [CrossRef] [PubMed]
- Pongrac, I.M.; Pavičić, I.; Milić, M.; Brkić Ahmed, L.; Babič, M.; Horák, D.; Vinković Vrček, I.; Gajović, S. Oxidative stress response in neural stem cells exposed to different superparamagnetic iron oxide nanoparticles. Int. J. Nanomed. 2016, 11, 1701–1715. [Google Scholar] [CrossRef]



| Reference | Year | Coating | Concentration | Cell Line or in Vivo Model | Study Model | Toxicity |
|---|---|---|---|---|---|---|
| [221] | 2014 | Manganese (Mn) | 5, 10, 20, 50 and 100 mg/L (in vitro); 150 µmol/kg Fe/kg body weight (in vivo) | Murine Balb/3T3 fibroblasts (in vitro) and CD1 female mice (8 weeks old) (in vivo) | In vitro/In vivo | Dose-dependent toxicity |
| [222] | 2014 | Poly(lactic-co-glycolic acid) (PLGA) + 5-Fluorouracil | 50, 100 and 200 µΜ | Human prostate cancer cell line DU145 | In vitro | Non toxic |
| [26] | 2014 | Silica shell (Fe3O4/SiO2 NPs) | 0.5, 1, 2.5 and 5 nM | A549 and HeLa cells | In vitro | Silica coating diminished Fe3O4 cytotoxic and genotoxic effects |
| [21] | 2014 | Poly-(ethylene glycol) (PEG) (PEGylated IONPs) | 100 and 500 ppm Fe | Bovine vascular smooth muscle cells (VSMCs) | In vitro | PEGylated IONPs showed less cytotoxicity than uncoated IONPs |
| [223] | 2014 | - | 1, 3 and 5 mg/mL | Epithelial cell and cancer cell lines; ECR 116 (NCBI code: C570) | In vitro | Non toxic |
| [224] | 2014 | Aminodextran (AD), 3-aminopropyltriethoxysilane (APS) and dimercaptosuccinic acid (DMSA) | 0.05, 0.1, and 0.5 mg/mL | HeLa (human cervical adenocarcinoma) | In vitro | Non toxic |
| [225] | 2015 | - | 214 mg/L | Neuronal cell line (Rat pheochromocytoma-PC12 cells) | In vitro | Neurocytotoxic |
| [226] | 2015 | - | 25, 50, 75 and 100 mg/L | Human hepatoma cells (Hep G2) | In vitro | Toxic (reduced cell viability with oxidative damage) |
| [227] | 2015 | Polyhydroxybutyrate (PHB) | 29–500 μM | MCF-7, SKBR-3 and HeLa human breast and ovarian cancer cell lines | In vitro | Non toxic |
| [228] | 2015 | Curcumin (Cur) | IONPs: 120 mg/L Cur: 40 mg/L | Wild type MDKC and human neuroblastoma cells | In vitro | Non toxic |
| [229] | 2015 | L-DOPA (L-3,4-dihydroxyphenylalanine) | 0–0.05 mg/mL (in vitro) and 2.5 mg/mouse (approximately 125 g/kg body weight) (in vivo) | Normal mouse L929 fibroblasts/C57BL/6 mice | In vitro/in vivo | Non toxic |
| [230] | 2015 | Polyacrylic acid (PAA) and non-coated | 4, 20 and 100 mg/L | Human T lymphocytes | In vitro | Non genotoxic |
| [231] | 2015 | - | 200 and 400 mg/L (in vitro), and 200 mg/L (in vivo) | Mouse fibroblast cell (in vitro) and wistar rat’s liver and kidney (in vivo) | In vitro/In vivo | Non toxic |
| [232] | 2015 | Alginate (Alg)/Alg + D-galactosamine (GA) | 0–1000 mg/L | Liver cancer/hepatocellular carcinoma (HepG2) cell line | In vitro | Non toxic |
| [233] | 2015 | Uncoated (U-Fe3O4) and oleate-coated Fe3O4 (OC-Fe3O4) | 10.8, 21.6 and 108 mg/L | Human lymphoblastoid TK6 cells and primary human blood cells | In vitro | U-Fe3O4 was not toxic; OC-Fe3O4 was cytotoxic in a dose-dependent manner and genotoxic |
| [71] | 2015 | Bare (uncoated) SPION (BS) and PEG (PEG-SPION (PS)) | 50.8 mg/kg b w for PS and 16.3 mg/kg b w for BS | BALB/c Swiss Albino mice | In vivo | PEGylation reduced the toxicity of BS (Low toxicity) |
| [234] | 2015 | Cobalt | 75, 150, 250, 500, 750 and 1,000 mg/L | MCF-7 cell lines | In vitro | Moderate toxicity to cancer cells |
| [235] | 2016 | Rhamnose | 0,1,2, 5, 10, 25, 50 and 100 µg Fe mL−1 for cancer cell lines, and 15.63 to 1000 µgFemL−1 for fibroblasts cell lines | Human glioblastoma cell lines (T98G and U251MG) and the human urinary bladder carcinoma cell line (ECV304), mouse fibroblast (BALB/3T3) cell line and its clone (A31-1-1). | In vitro | Moderate toxicity to tumoral cell lines and non toxic to fibroblast cells |
| [96] | 2016 | Silica and oleic acid | 5–300 mg/L | Human neuroblastoma SHSY5Y and glioblastoma A172 | In vitro | Low citotoxicity/oleic acid-coated IONPs with less citotoxicity than silica-coated IONPs |
| [163] | 2016 | Mitoxantrone (MTO) | 0.0001–0.1 mg/L | Human primary tubular epithelial cells (hTEC) | In vitro | Moderate toxicity (depends on the drug loaded to the SPION) |
| [25] | 2016 | 2,3-dimercaptosuccinic acid (DMSA) | 15, 30, 60 e 80 mg/L (IONPs) | human mesenchymal stem cells from dental pulp tissues | In vitro | Non toxic |
| [236] | 2016 | No coating and curcumin-coating | 1–1000 mg/L | Human umbilical vein endothelial cells (HUVECs) | In vitro | Curcumin-coated IONPS were less toxic than uncoated IONPs |
| [237] | 2016 | Polyacrylic acid-co-maleic acid (PAM) + tissue plasminogen activator (tPA) | 30 μg Fe/mL | Human umbilical vein endothelial cells (HUVECs) | In vitro | Low toxicity |
| [238] | 2016 | Polymer (converted from Poly(lactic-co-glycolic acid) nanoparticles) | 0.005–0.32 mg/mL | SKOV3 human ovarian cancer cells and NIH/3T3 murine fibroblasts | In vitro | Low toxicity |
| [22] | 2016 | Chitosan + Gemcitabine | IC50 for SKBR-3 (4.8 µM) and MCF-7 (1.5 µM) | SKBR-3 and MCF-7 breast cancer cells | In vitro | More cytotoxic to the tested breast cancer cell lines than free gemcitabine |
| [239] | 2016 | - | 10, 25, 50, 75, and 100 mg/L | Human peripheral lymphocytes | In vitro | Moderate toxicity |
| [215] | 2016 | - | 0–1000 mg/L | Human whole blood cultures | In vitro | Dose-dependent toxicity |
| [240] | 2016 | - | 65 ng/mL (in vitro), and 520 µg Fe3O4/kg and 20.8 µg Fe3O4/kg (in vivo) | Mouse embryonic fibroblasts NIH3T3 (in vitro) and Wistar rats (in vivo) | In vitro/In vivo | Non toxic at a desirable concentration |
| [23] | 2016 | PEG350 and PEG2000 | 50–200 mg/L (in vitro)/12.5, 25 and 50 mg/kg/day (in vivo) | Monkey kidney ephitelium (Vero), dog kidney fibroblasts (MDKC) and mouse embryonic fibroblast (NIH-3 T3) (in vitro) and Swiss albino male mice (in vivo) | In vitro/In vivo | SPION-PEG2000 showed no toxicity in vitro, but lead to liver and kidney injury in vivo. In vitro, SPION-PEG350 showed no toxicity up to 100 µg/mL |
| [241] | 2016 | c(RGDyK) + dopamine | 1.50, 2.07, 2.87, 3.97, 5.49, 7.59 and 8.50 g/kg | Kunming mice of SPF grade | In vivo | Non toxic |
| [242] | 2017 | Poly-(ethylene glycol) (PEG) and polyethylenimine (PEI) polymers + folic acid (FA-IONPs) Doxorubicin + FA IONPs (DOX@FA-IONPs) | 0.2–10 mg/L | MCF7 cells | In vitro | FA-IONPs show low cytotoxicity and DOX@FA-IONPs is more cytotoxic than free DOX |
| [243] | 2017 | Tri-block copolymer: poly(ε-caprolactone)-poly(ethylene glycol)-poly(ε-caprolactone) (PCL-PEG-PCL, PCEC) | 0, 0.5, 1, 2, 5 and 10% | NIH 3T3 cells | In vitro | The PCEC coating reduced Fe3O4 NPs toxicity |
| [97] | 2017 | SiO2 (FemOn-SiO2 composite and SiO2-FemOn core-shell IONPs) | 0.7, 7.0 and 70.0 µg | Human umbilical vein endothelial cell culture (cultured HUVECs) | In vitro | Dose-dependent toxicity in the presence of silica. Bare IONPs were less toxic |
| [244] | 2017 | Chitosan (CS) + calf-thymus DNA (DNA) | - | Human foreskin fibroblast cell line (HFFF2) | In vitro | Non toxic |
| [245] | 2017 | Zinc/Cobalt | 10, 100, 250 and 500 μM | Primary human bone marrow-derived mesenchymal stem cells (hMSCs) and human osteosarcoma-derived cells (MG-63) | In vitro | The levels of toxicity do not compromise the biocompatibility |
| [246] | 2017 | - | 10, 25, 50, 100, and 200 mg/mL (0.3 mL/egg in airspace) | Fertilized eggs of White leghorn (Gallus gallus domesticus) | In vivo | Neurotoxic in lower doses and 100% mortality at 200 mg/mL dose |
| [72] | 2017 | Polyvinylpirrolidone | 1, 10, 25, 50 and 100 μg/mL | Human neuroblastoma (SH-SY5Y cell line) | In vitro | Dose-dependent toxicity |
| [98] | 2018 | Luminescent ruthenium (II) complex encapsulated with silica shell + amine group (APTMS)-Fe3O4@SiO2@[Ru(Phen)3] 2+@SiO2@NH2 | 10, 50 and 100 μg/mL | Cancer cell (B16F10) and normal cell (CHO) | In vitro | Low cytotoxicity |
| [66] | 2018 | - | 0.1, 0.5, 1, 2.5, 5 and 7.5 mg/mL | MCF7 and 3T3 cell lines | In vitro | Dose-dependent toxicity |
| [219] | 2018 | Polyethylenimine (PEI) and polyethylene glycol (PEG) | 3.125–100 µg/mL (in vitro)/Up to 5mg/kg (in vivo) | RAW264.7 macrophages and non-phagocytic SKOV-3 ovarian cancer cells (in vitro)/SKOV-3 tumor bearing nude mice and BALB/c mice (in vivo) | In vitro/In vivo | PEI-coated-IONPs were toxic in vitro with dose-dependent toxicity in vivo/PEG-coated-IONPs presented low toxicity |
| [247] | 2018 | Polyamidoamine (PAMAM) dendrimer (Fourth generation—G4) | Acute toxicity: 25, 50 and 100 mg/kg Chronic toxicity: 0.5, 1, 5 and 10 mg/Kg | BALB/c mice | In vivo | Acceptable toxicity |
| [248] | 2018 | Chitosan (CS)-dextran (DX) | 1, 5, 10, 50, and 150 µg/mL | Rat C6 glioma, human U87 glioma, and human cervix carcinoma HeLa cells | In vitro | Dose and time-dependent toxicity |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Arias, L.S.; Pessan, J.P.; Vieira, A.P.M.; Lima, T.M.T.d.; Delbem, A.C.B.; Monteiro, D.R. Iron Oxide Nanoparticles for Biomedical Applications: A Perspective on Synthesis, Drugs, Antimicrobial Activity, and Toxicity. Antibiotics 2018, 7, 46. https://doi.org/10.3390/antibiotics7020046
Arias LS, Pessan JP, Vieira APM, Lima TMTd, Delbem ACB, Monteiro DR. Iron Oxide Nanoparticles for Biomedical Applications: A Perspective on Synthesis, Drugs, Antimicrobial Activity, and Toxicity. Antibiotics. 2018; 7(2):46. https://doi.org/10.3390/antibiotics7020046
Chicago/Turabian StyleArias, Laís Salomão, Juliano Pelim Pessan, Ana Paula Miranda Vieira, Taynara Maria Toito de Lima, Alberto Carlos Botazzo Delbem, and Douglas Roberto Monteiro. 2018. "Iron Oxide Nanoparticles for Biomedical Applications: A Perspective on Synthesis, Drugs, Antimicrobial Activity, and Toxicity" Antibiotics 7, no. 2: 46. https://doi.org/10.3390/antibiotics7020046





