Exploring the Effect of Phage Therapy in Preventing Vibrio anguillarum Infections in Cod and Turbot Larvae
Abstract
:1. Introduction
2. Results
2.1. Phage Effect on Turbot Mortality in Vibrio Challenge Trials
2.1.1. Turbot Challenge Trial 1
2.1.2. Turbot Challenge Trial 2
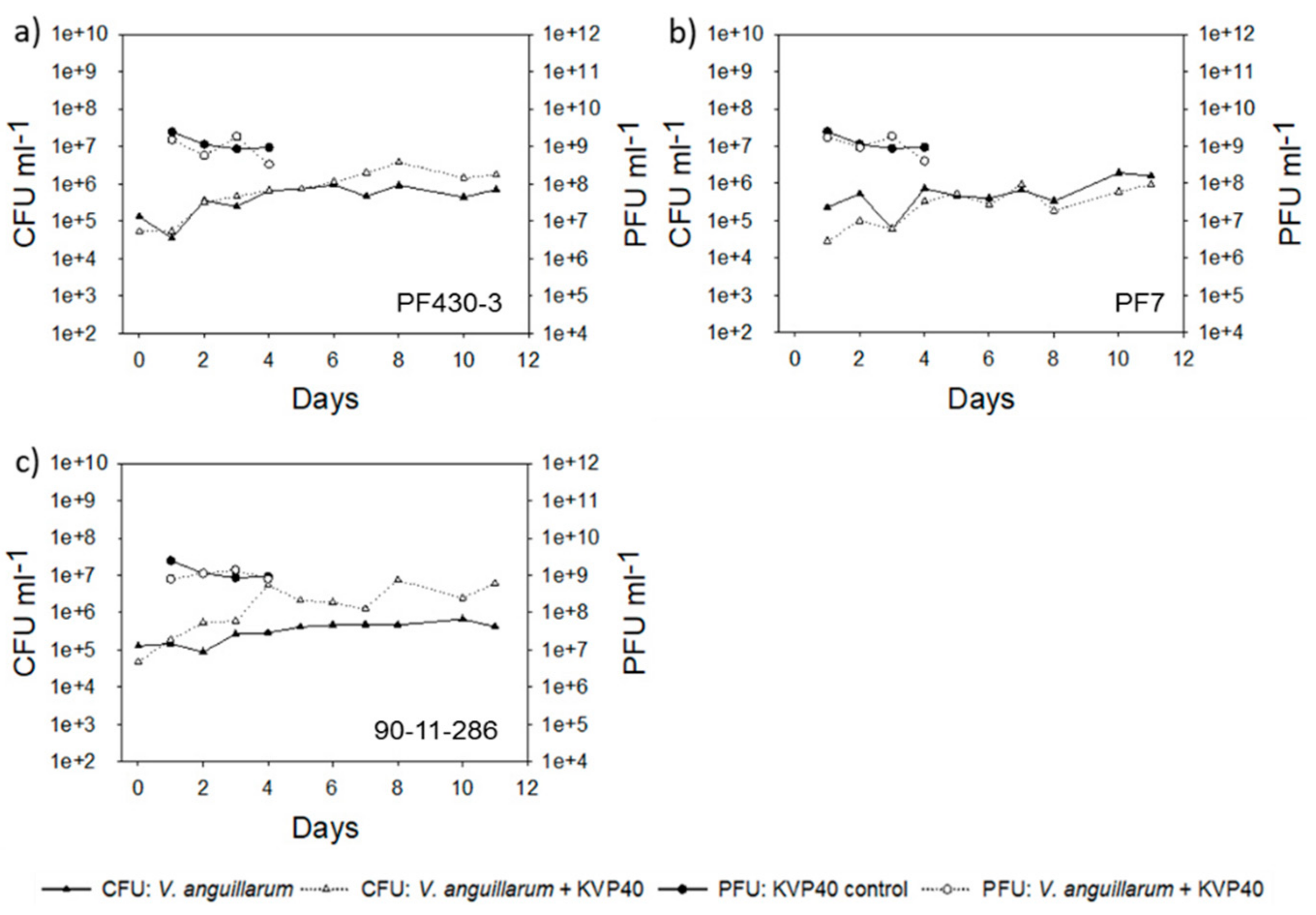
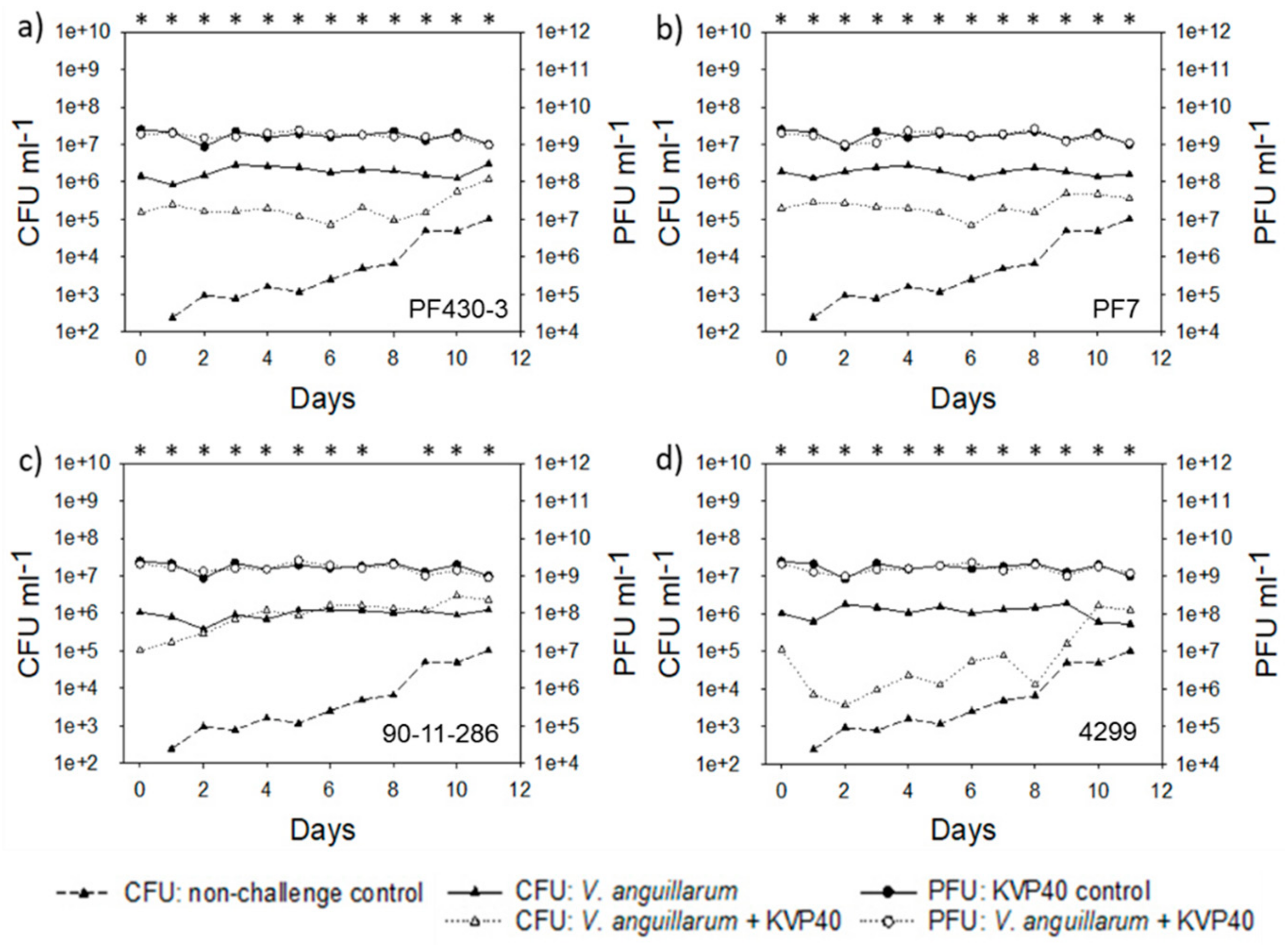
2.2. Abundance of Bacteria and Phages in Turbot Challenge Trial 2
2.3. Phage Effect on Cod Mortality in Vibrio Challenge Trials
2.3.1. Cod Challenge Trial 1
2.3.2. Cod Challenge Trial 2
2.4. Abundance of Bacteria and Phages in Cod Challenge Trials
2.4.1. Cod Challenge Trial 1
2.4.2. Cod Challenge Trial 2
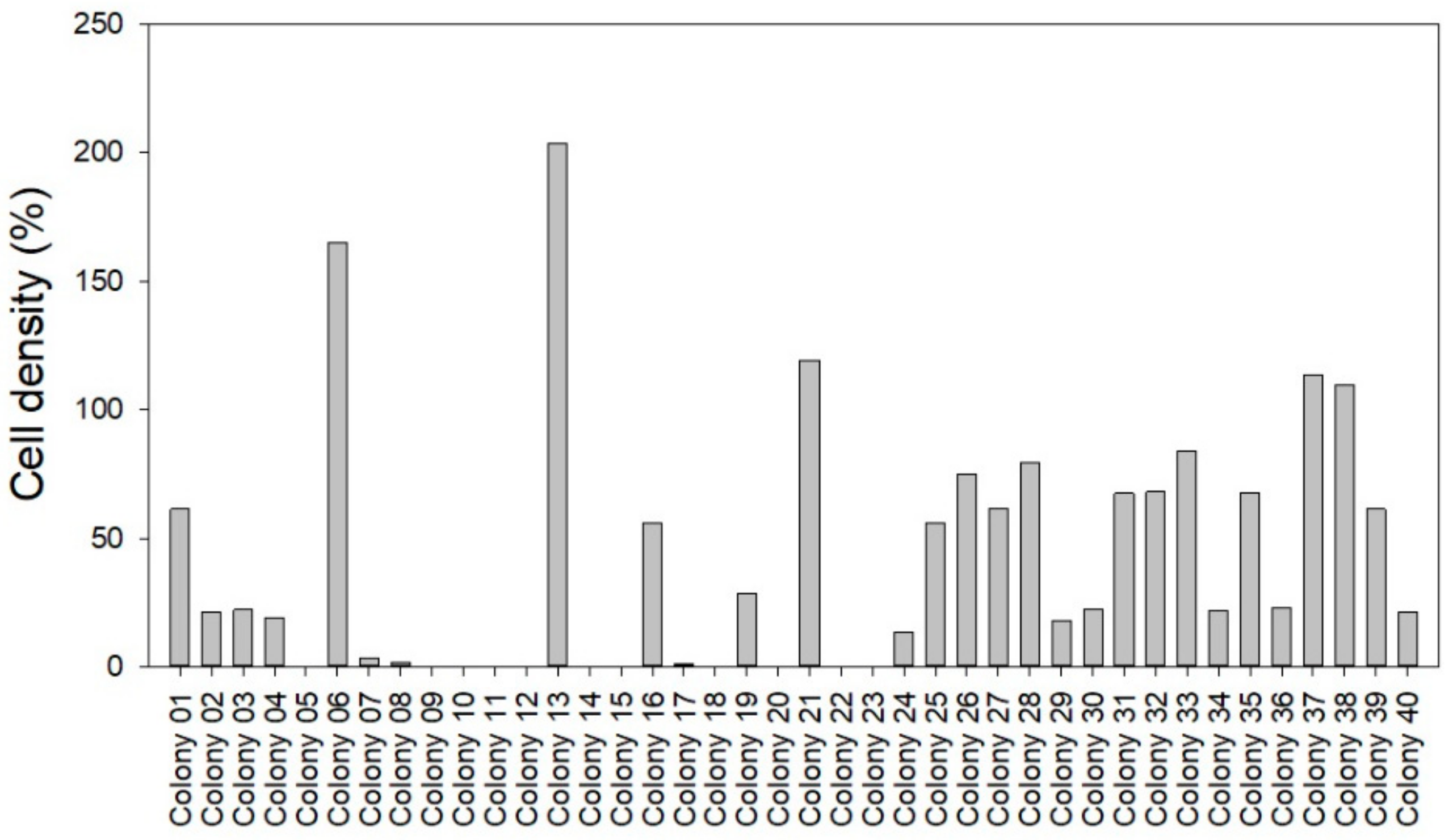
2.5. Abundance and Phage KVP40 Susceptibility of Bacterial Background Communities Associated with the Turbot Eggs
3. Discussion
4. Materials and Methods
4.1. Bacterial Strains and Growth Conditions
4.2. Phage Infectivity and Production
4.3. Eggs and Larvae
4.4. Phage Therapy Assays
4.5. Bacterial Background Community and Susceptibility Assays
4.6. Statistical Analysis
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Thompson, F.L.; Iida, T.; Swings, J. Biodiversity of Vibrios. Microbiol. Mol. Biol. Rev. 2004, 68, 403–431. [Google Scholar] [CrossRef] [PubMed]
- Actis, L.A.; Tolmasky, M.E.; Crosa, J.H. Vibriosis. In Fish Diseases and Disorders; Woo, P.T.K., Bruno, D.W., Eds.; CAB International: Oxfordshire, UK, 2011; pp. 570–605. [Google Scholar]
- Frans, I.; Michiels, C.W.; Bossier, P.; Willems, K.A.; Lievens, B.; Rediers, H. Vibrio anguillarum as a fish pathogen: Virulence factors, diagnosis and prevention. J. Fish Dis. 2011, 34, 643–661. [Google Scholar] [CrossRef] [PubMed]
- Pereira, C.; Silva, Y.J.; Santos, A.L.; Cunha, A.; Gomes, N.C.M.; Almeida, A. Bacteriophages with potential for inactivation of fish pathogenic bacteria: Survival, host specificity and effect on bacterial community structure. Mar. Drugs 2011, 9, 2236–2255. [Google Scholar] [CrossRef] [PubMed]
- Karunasagar, I.; Pai, R.; Malathi, G.R.; Karunasagar, I. Mass mortality of Penaeus monodon larvae due to antibiotic-resistant Vibrio harveyi infection. Aquaculture 1994, 128, 203–209. [Google Scholar] [CrossRef]
- Bricknell, I.R.; Bowden, T.J.; Verner-Jeffreys, D.W.; Bruno, D.W.; Shields, R.J.; Ellis, A.A.E. Susceptibility of juvenile and sub-adult Atlantic halibut (Hippoglossus hippoglossus L.) to infection by Vibrio anguillarum and efficacy of protection induced by vaccination. Fish Shellfish Immunol. 2000, 10, 319–327. [Google Scholar] [CrossRef] [PubMed]
- Mikkelsen, H.; Lund, V.; Larsen, R.; Seppola, M. Vibriosis vaccines based on various sero-subgroups of Vibrio anguillarum O2 induce specific protection in Atlantic cod (Gadus morhua L.) juveniles. Fish Shellfish Immunol. 2011. [Google Scholar] [CrossRef] [PubMed]
- Vinod, M.G.; Shivu, M.M.; Umesha, K.R.; Rajeeva, B.C.; Krohne, G.; Karunasagar, I.; Karunasagar, I. Isolation of Vibrio harveyi bacteriophage with a potential for biocontrol of luminous vibriosis in hatchery environments. Aquaculture 2006. [Google Scholar] [CrossRef]
- Karunasagar, I.; Shivu, M.M.; Girisha, S.K.; Krohne, G.; Karunasagar, I. Biocontrol of pathogens in shrimp hatcheries using bacteriophages. Aquaculture 2007, 268, 288–292. [Google Scholar] [CrossRef]
- Higuera, G.; Bastías, R.; Tsertsvadze, G.; Romero, J.; Espejo, R.T. Recently discovered Vibrio anguillarum phages can protect against experimentally induced vibriosis in Atlantic salmon, Salmo salar. Aquaculture 2013, 392, 128–133. [Google Scholar] [CrossRef]
- Mateus, L.; Costa, L.; Silva, Y.J.; Pereira, C.; Cunha, A.; Almeida, A. Efficiency of phage cocktails in the inactivation of Vibrio in aquaculture. Aquaculture 2014, 424, 167–173. [Google Scholar] [CrossRef]
- Silva, Y.J.; Costa, L.; Pereira, C.; Mateus, C.; Cunha, Â.; Calado, R.; Gomes, N.C.M.; Pardo, M.A.; Hernandez, I.; Almeida, A. Phage therapy as an approach to prevent Vibrio anguillarum infections in fish larvae production. PLoS ONE 2014. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.; Gram, L.; Middelboe, M. Vibriophages and their interactions with the fish pathogen vibrio anguillarum. Appl. Environ. Microbiol. 2014, 80, 3128–3140. [Google Scholar] [CrossRef] [PubMed]
- Stenholm, A.R.; Dalsgaard, I.; Middelboe, M. Isolation and characterization of bacteriophages infecting the fish pathogen Flavobacterium psychrophilum. Appl. Environ. Microbiol. 2008, 74, 4070–4078. [Google Scholar] [CrossRef] [PubMed]
- Letchumanan, V.; Chan, K.G.; Pusparajah, P.; Saokaew, S.; Duangjai, A.; Goh, B.H.; Ab Mutalib, N.S.; Lee, L.H. Insights into bacteriophage application in controlling vibrio species. Front. Microbiol. 2016, 7. [Google Scholar] [CrossRef] [PubMed]
- Matsuzaki, S.; Tanaka, S.; Koga, T.; Kawata, T. A broad-host-range vibriophage, KVP40, isolated from sea water. Microbiol. Immunol. 1992. [Google Scholar] [CrossRef]
- Inoue, T.; Matsuzaki, S.; Tanaka, S. A 26-kDa outer membrane protein, OmpK, common to Vibrio species is the receptor for a broad-host-range vibriophage, KVP40. FEMS Microbiol. Lett. 1995, 125, 101–105. [Google Scholar] [CrossRef] [PubMed]
- Labrie, S.J.; Samson, J.E.; Moineau, S. Bacteriophage resistance mechanisms. Nat. Rev. Microbiol. 2010, 8, 317–327. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.; Svenningsen, S.L.; Middelboe, M. Quorum sensing determines the choice of antiphage defense strategy in Vibrio anguillarum. MBio 2015, 6, e00627. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.; Dahl, A.; Middelboe, M. Vibriophages differentially influence biofilm formation by Vibrio anguillarum strains. Appl. Environ. Microbiol. 2015, 81, 4489–4497. [Google Scholar] [CrossRef] [PubMed]
- Kalatzis, P.G.; Bastías, R.; Kokkari, C.; Katharios, P. Isolation and characterization of two lytic bacteriophages, φst2 and φgrn1; Phage therapy application for biological control of vibrio alginolyticus in aquaculture live feeds. PLoS ONE 2016, 11. [Google Scholar] [CrossRef] [PubMed]
- Hansen, G.H.; Olafsen, J.A. Bacterial colonization of cod (Gadus morhua L.) and halibut (Hippoglossus hippoglossus) eggs in marine aquaculture. Appl. Environ. Microbiol. 1989, 55, 1435–1446. [Google Scholar] [PubMed]
- Austin, B. Taxonomy of bacteria isolated from a coastal marine fish-rearing unit. J. Appl. Bacteriol. 1982, 53, 253–268. [Google Scholar] [CrossRef]
- Goodridge, L.D. Designing phage therapeutics. Curr. Pharm. Biotechnol. 2010, 11, 15–27. [Google Scholar] [CrossRef] [PubMed]
- Khemayan, K.; Prachumwat, A.; Sonthayanon, B.; Intaraprasong, A.; Sriurairatana, S.; Flegel, T.W. Complete genome sequence of virulence-enhancing siphophage VHS1 from Vibrio harveyi. Appl. Environ. Microbiol. 2012, 78, 2790–2796. [Google Scholar] [CrossRef] [PubMed]
- Imbeault, S.; Parent, S.; Lagacé, M.; Carl, F.; Blais, J. Using bacteriophages to prevent furunculosis caused by Aeromonas salmonicida in farmed brook trout. J. Aquat. Anim. Health 2006, 18, 203–214. [Google Scholar] [CrossRef]
- Verner–Jeffreys, D.W.; Algoet, M.; Pond, M.J.; Virdee, H.K.; Bagwell, N.J.; Roberts, E.G. Furunculosis in Atlantic salmon (Salmo salar L.) is not readily controllable by bacteriophage therapy. Aquaculture 2007, 270, 475–484. [Google Scholar] [CrossRef]
- Hansen, G.H.; Olafsen, J.A. Bacterial interactions in early life stages of marine cold water fish. Microb. Ecol. 1999, 38, 1–26. [Google Scholar] [CrossRef] [PubMed]
- Skov, M.N.; Pedersen, K.; Larsen, J.L. Comparison of pulsed-field gel electrophoresis, ribotyping, and plasmid profiling for typing of Vibrio anguillarum serovar O1. Appl. Environ. Microbiol. 1995, 61, 1540–1545. [Google Scholar] [PubMed]
- Mikkelsen, H.; Schrøder, M.B.; Lund, V. Vibriosis and atypical furunculosis vaccines; efficacy, specificity and side effects in Atlantic cod, Gadus morhua L. Aquaculture 2004. [Google Scholar] [CrossRef]
- Ranganathan, P.; Pramesh, C.; Aggarwal, R. Common pitfalls in statistical analysis: Absolute risk reduction, relative risk reduction, and number needed to treat. Perspect. Clin. Res. 2016, 7. [Google Scholar] [CrossRef] [PubMed]
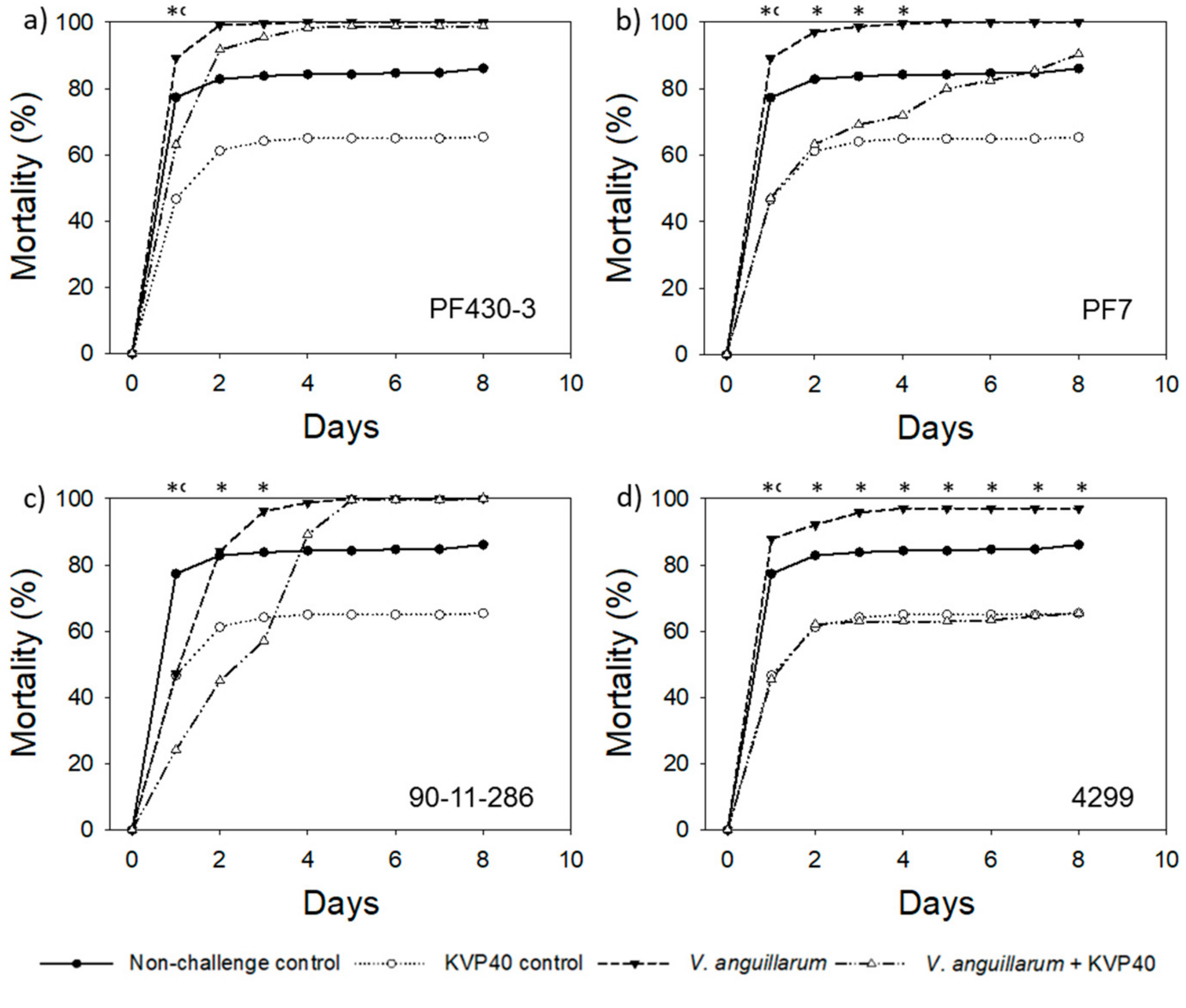
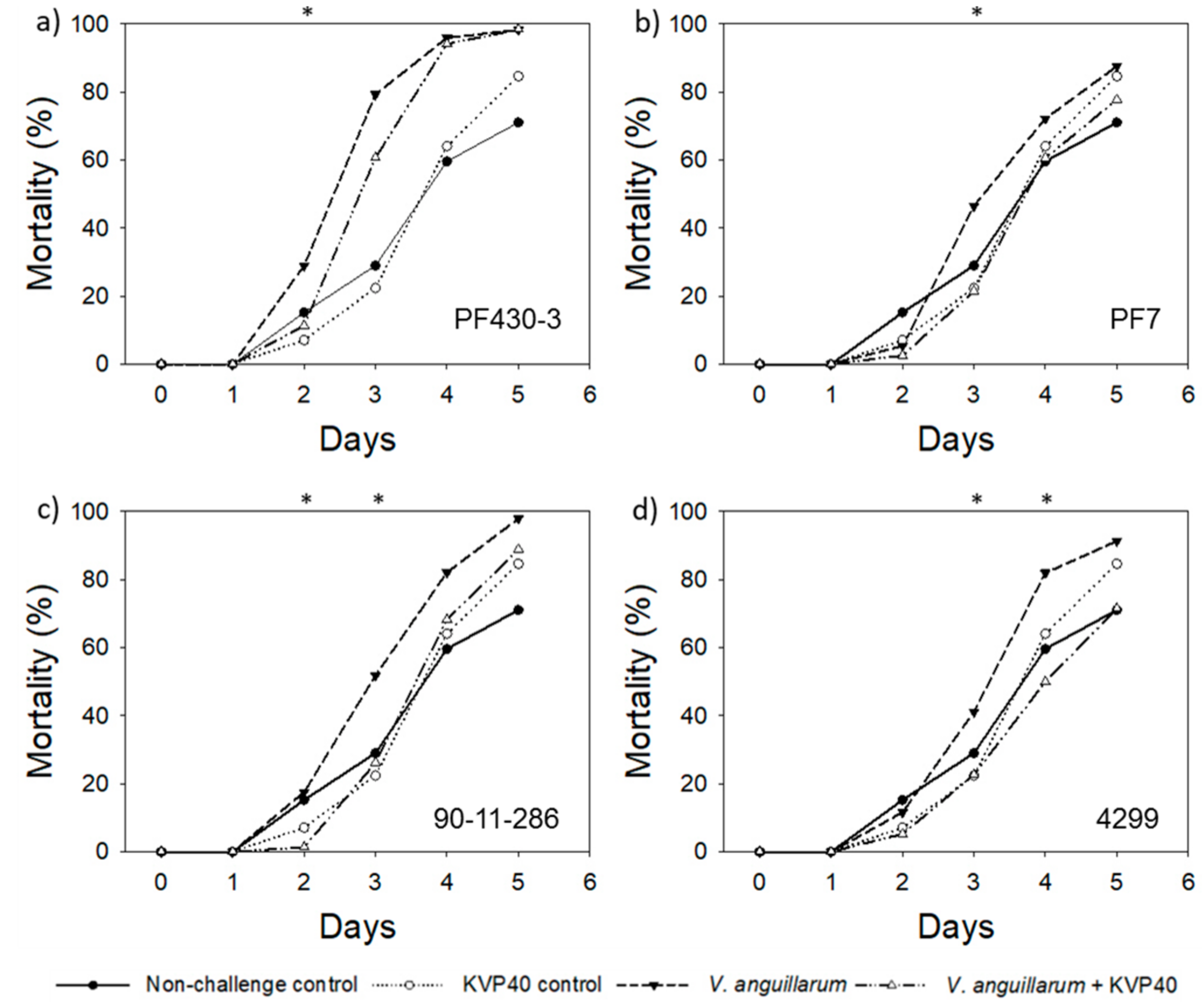

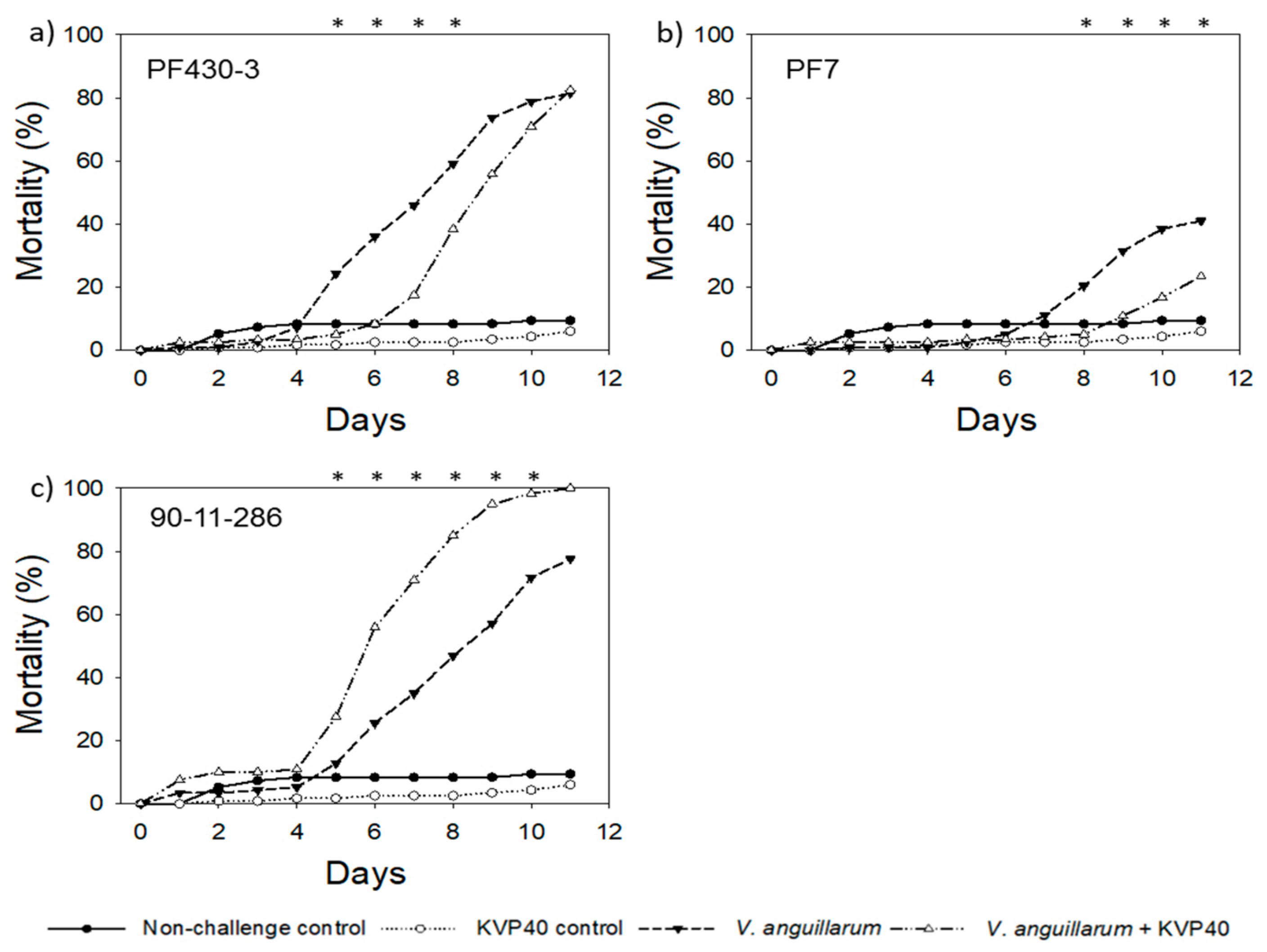
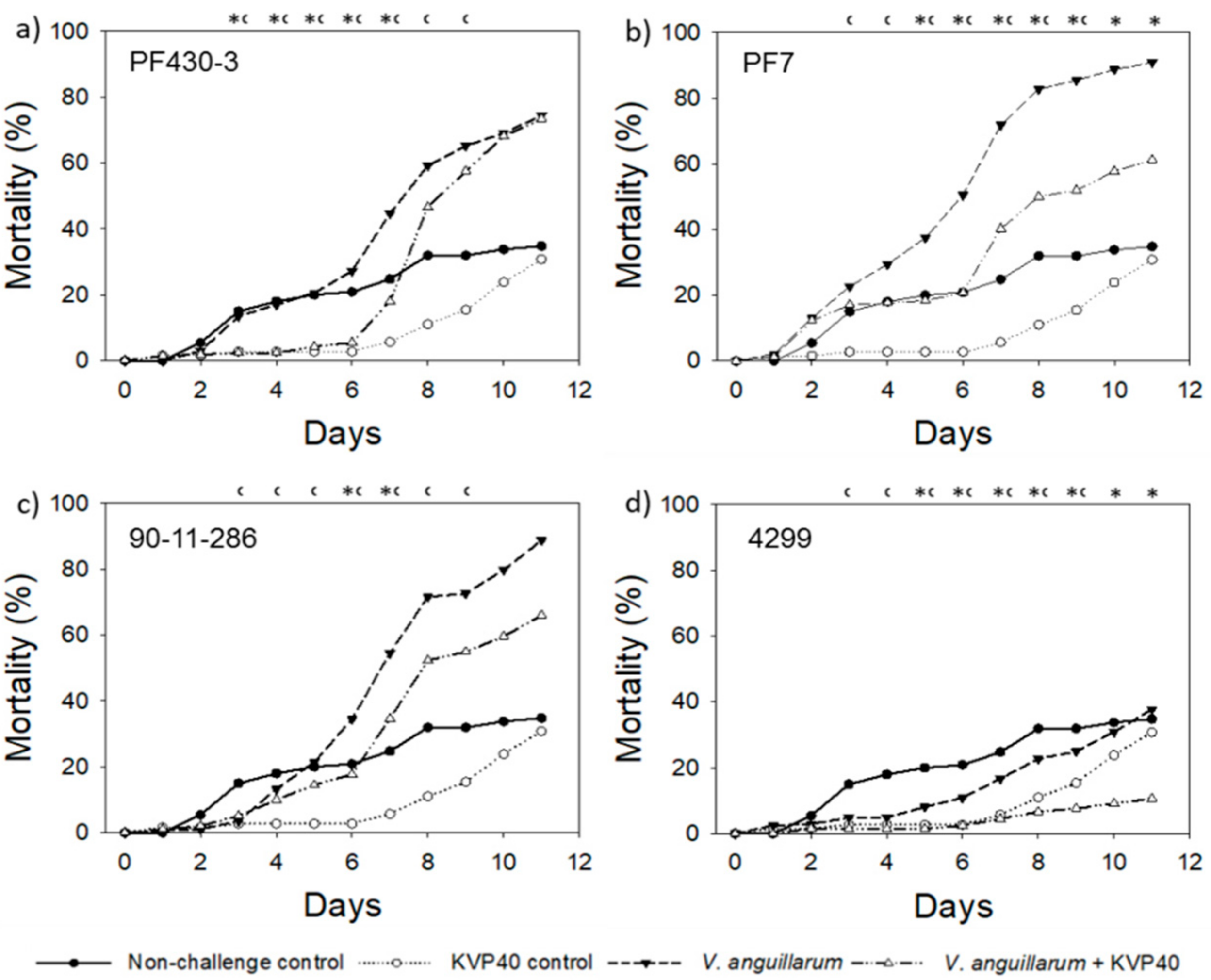
| Relative Reduction * in Larval Mortality in the Presence of Phages (%) | ||||||||
|---|---|---|---|---|---|---|---|---|
| V. anguillarum Strains | Turbot Challenge Trial | Cod Challenge Trial | ||||||
| 1 | 2 | 1 | 2 | |||||
| Max. | Final | Max. | Final | Max. | Final | Max. | Final | |
| PF430-3 | 29 | N/S 1 | 60 | N/S 1 | 79 | N/S 1 | 86 | N/ 1 |
| PF7 | 47 | N/S 1 | 53 | N/S 1 | 75 | 43 | 59 | 32 |
| 90-11-286 | 47 | N/S 1 | 92 | N/S 1 | −119 | N/S 1 | 49 | N/S 1 |
| 4299 | 48 | 33 | 45 | N/S 1 | N/D 2 | N/D 2 | 82 | 72 |
| Growth Substrate | Day 0 (CFU mL−1) | Day 11 (CFU mL−1) |
|---|---|---|
| LB media | 2 × 107 | 9.39 × 106 |
| TCBS media | 2 × 106 | 1.5 × 108 |
| Marine agar | N/D 1 | 2.89 × 108 |
| Group | Treatment | V. anguillarum (CFU mL−1) | Phage KVP40 (PFU mL−1) | Replicate Wells |
|---|---|---|---|---|
| 1 | V. anguillarum only | 0.5–1 × 106 | - | 5 × 24 wells × 4 strains |
| 2 | V. anguillarum + phage KVP40 | 0.5–1 × 106 | 0.5–12 × 108 | 5 × 24 wells × 4 strains |
| 3 | Nonchallenge control | - | - | 5 × 24 wells |
| 4 | Phage KVP40 control | - | 0.5–12 × 108 | 5 × 24 wells |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rørbo, N.; Rønneseth, A.; Kalatzis, P.G.; Rasmussen, B.B.; Engell-Sørensen, K.; Kleppen, H.P.; Wergeland, H.I.; Gram, L.; Middelboe, M. Exploring the Effect of Phage Therapy in Preventing Vibrio anguillarum Infections in Cod and Turbot Larvae. Antibiotics 2018, 7, 42. https://doi.org/10.3390/antibiotics7020042
Rørbo N, Rønneseth A, Kalatzis PG, Rasmussen BB, Engell-Sørensen K, Kleppen HP, Wergeland HI, Gram L, Middelboe M. Exploring the Effect of Phage Therapy in Preventing Vibrio anguillarum Infections in Cod and Turbot Larvae. Antibiotics. 2018; 7(2):42. https://doi.org/10.3390/antibiotics7020042
Chicago/Turabian StyleRørbo, Nanna, Anita Rønneseth, Panos G. Kalatzis, Bastian Barker Rasmussen, Kirsten Engell-Sørensen, Hans Petter Kleppen, Heidrun Inger Wergeland, Lone Gram, and Mathias Middelboe. 2018. "Exploring the Effect of Phage Therapy in Preventing Vibrio anguillarum Infections in Cod and Turbot Larvae" Antibiotics 7, no. 2: 42. https://doi.org/10.3390/antibiotics7020042





