Clinical Efficacy of a Single Two Gram Dose of Azithromycin Extended Release for Male Patients with Urethritis
Abstract
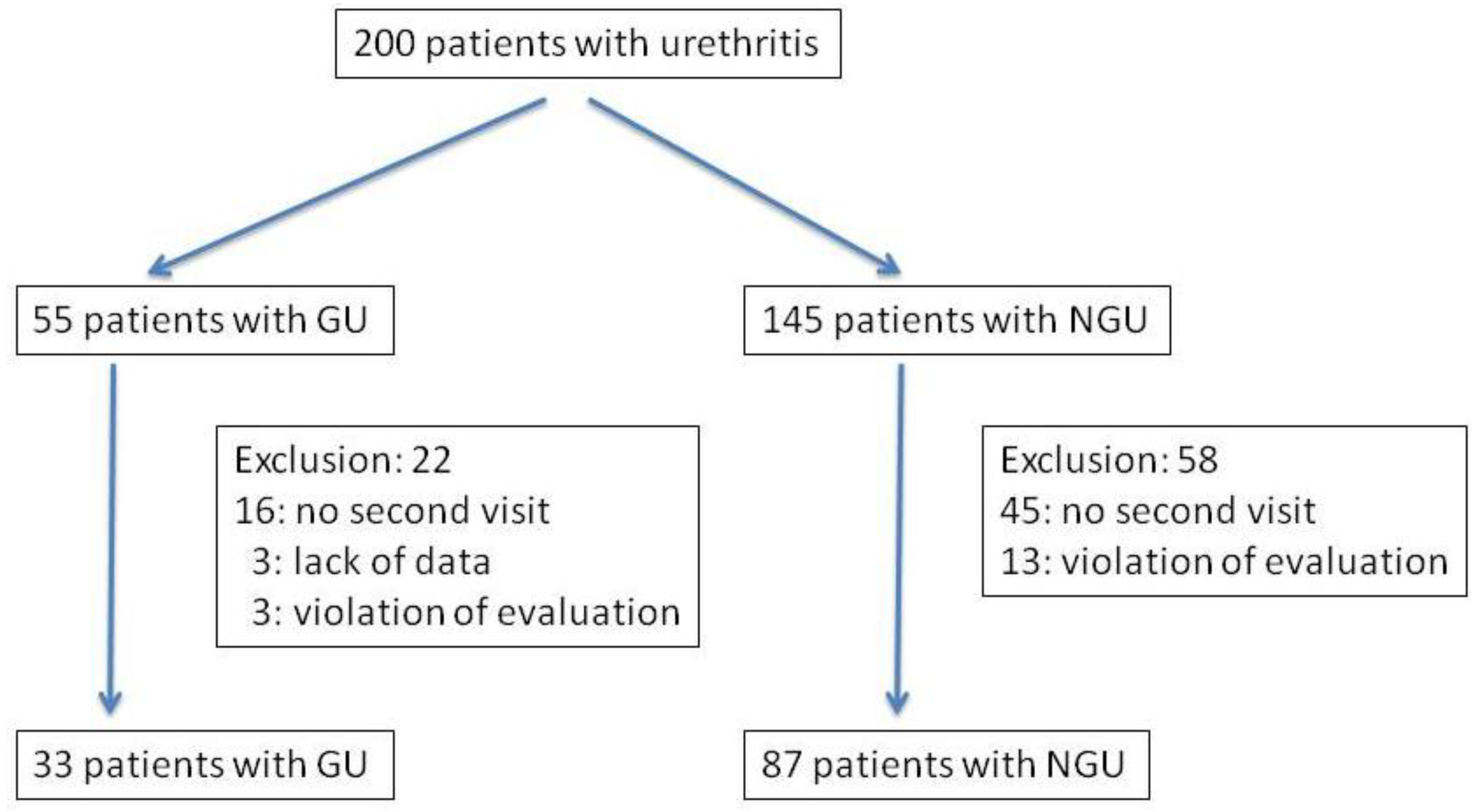
:1. Introduction
2. Patients and Methods
2.1. Study Design
2.2. Patients
2.3. Procedures for Detection of Pathogens
2.4. Antimicrobial Susceptibility Testing
2.5. Outcome
2.6. Assessment of Adverse Events
2.7. Ethical Considerations
3. Results
| Antimicrobial agents | MICs (µg/mL) | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ≦0.001 | 0.002 | 0.004 | 0.008 | 0.015 | 0.03 | 0.06 | 0.12 | 0.25 | 0.5 | 1 | 2 | 4 | 8 | 16 | 32 | 64 | |
| PCG | - | - | - | - | 2 | - | 2 | 9 | 7 | 9 | 6 | 6 | 1 | - | - | 1 | - |
| CTRX | 2 | 4 | 6 | 6 | 5 | 11 | 7 | 2 | - | - | - | - | - | - | - | - | - |
| CDZM | - | 1 | 3 | 8 | 2 | 9 | 11 | 8 | 1 | - | - | - | - | - | - | - | - |
| CFIX | - | 1 | 1 | 6 | 5 | 2 | 9 | 14 | 3 | 2 | - | - | - | - | - | - | - |
| SPCM | - | - | - | - | - | - | - | - | - | - | - | - | - | 8 | 22 | 13 | - |
| AZM | - | - | - | - | 1 | - | 6 | 5 | 4 | 9 | 11 | 5 | 1 | - | 1 | - | - |
| CPFX | - | - | 3 | 4 | 2 | - | - | - | - | - | - | 1 | - | 4 | 8 | 17 | 4 |
| LVFX | - | - | 3 | 4 | 2 | - | - | - | - | - | 1 | 1 | 4 | 18 | 9 | 1 | - |
| Antimicrobial agents | Outcome | MICs | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ≦0.001 | 0.002 | 0.004 | 0.008 | 0.015 | 0.03 | 0.06 | 0.12 | 0.25 | 0.5 | 1 | 2 | 4 | 8 | 16 | 32 | 64 | ||
| PCG | Eradication | - | - | - | - | 2 | - | 1 | 5 | 5 | 4 | 4 | 2 | - | - | - | - | - |
| Failure | 1 | 1 | 1 | |||||||||||||||
| CTRX | Eradication | 1 | 3 | 2 | 3 | 4 | 2 | 6 | 2 | - | - | - | - | - | - | - | - | - |
| Failure | 1 | 1 | 1 | |||||||||||||||
| CDZM | Eradication | - | 1 | 2 | 4 | - | 3 | 7 | 5 | 1 | - | - | - | - | - | - | - | - |
| Failure | 1 | 2 | ||||||||||||||||
| CFIX | Eradication | - | 1 | 1 | 3 | 2 | - | 3 | 8 | 3 | 2 | - | - | - | - | - | - | - |
| Failure | 1 | 1 | 1 | |||||||||||||||
| SPCM | Eradication | - | - | - | - | - | - | - | - | - | - | - | - | - | 3 | 14 | 6 | - |
| Failure | 1 | 2 | ||||||||||||||||
| AZM | Eradication | - | - | - | - | - | - | 4 | 3 | 3 | 6 | 6 | 1 | - | - | - | - | - |
| Failure | 1 | 1 | 1 | |||||||||||||||
| CPFX | Eradication | - | - | 1 | 2 | 2 | - | - | - | - | - | - | 1 | - | 4 | 4 | 8 | 1 |
| Failure | 3 | |||||||||||||||||
| LVFX | Eradication | - | - | 1 | 2 | 2 | - | - | - | - | - | 1 | 1 | 4 | 8 | 4 | - | - |
| Failure | 3 | |||||||||||||||||
| Urethritis | Pathogen coinfection | Number | Eradication (Number) | Cure (Number) | Pathogen of treatment failure and number |
| GU | None | 23 | 30 | 28 | M. genitalium in 1 |
| C. trachomatis | 8 | ||||
| M. genitalium | 1 | ||||
| U. urealyticum | 1 | ||||
| Urethritis | Pathogen infection | Number | Eradication | Cure | Pathogen in failure and number |
| NGU | C. trachomatis | 35 | 31 | 34 | C. trachomatis in 4 |
| M. genitalium | 3 | 2 | 3 | ||
| U. urealyticum | 5 | 5 | 5 | ||
| C. trachomatis and U. urealyticum | 4 | 4 | 4 | ||
| M. genitalium and U. urealyticum | 3 | 3 | 3 | ||
| Without any microbial detection | 37 | - | 33 |
4. Discussion
5. Conclusions
Author Contributions
Conflicts of Interest
References
- Wetmore, C.M.; Manhart, L.E.; Lowens, M.S.; Golden, M.R.; Whittington, W.L.; Xet-Mull, A.M.; Astete, S.G.; McFarland, N.L.; McDougal, S.J.; Totten, P.A. Demographic, behavioral, and clinical characteristics of men with nongonococcal urethritis differ by etiology: A case-comparison study. Sex. Transm. Dis. 2011, 38, 180–186. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. Update to CDC’s sexually transmitted diseases treatment guidelines, 2010: Oral cephalosporins no longer a recommended treatment for gonococcal infections. Morb. Mortal. Wkly. Rep. 2012, 10, 590–594. [Google Scholar]
- Workowski, K.A.; Berman, S.; Centers for Disease Control and Prevention. Sexually transmitted diseases treatment guidelines, 2010. Morb. Mortal. Wkly. Rep. 2010, 59, 1–110. [Google Scholar]
- Chisholm, S.A.; Neal, T.J.; Alawattegama, A.B.; Birley, H.D.; Howe, R.A.; Ison, C.A. Emergence of high-level azithromycin resistance in Neisseria gonorrhoeae in England and Wales. J. Antimicrob. Chemother. 2009, 64, 353–358. [Google Scholar] [CrossRef]
- Galarza, P.G.; Alcala, B.; Salcedo, C.; Canigia, L.F.; Buscemi, L.; Pagano, I.; Oviedo, C.; Vázquez, J.A. Emergence of high level azithromycin-resistant Neisseria gonorrhoeae strain isolated in Argentina. Sex. Transm. Dis. 2009, 36, 787–788. [Google Scholar] [CrossRef]
- Starnino, S.; Stefanelli, P. Neisseria gonorrhoeae Italian Study G. Azithromycin-resistant Neisseria gonorrhoeae strains recently isolated in Italy. J. Antimicrob. Chemother. 2009, 63, 1200–1204. [Google Scholar] [CrossRef]
- Bradshaw, C.S.; Jensen, J.S.; Tabrizi, S.N.; Read, T.R.; Garland, S.M.; Hopkins, C.A.; Moss, L.M.; Fairley, C.K. Azithromycin failure in Mycoplasma genitalium urethritis. Emerg. Infect. Dis. 2006, 12, 1149–1152. [Google Scholar] [CrossRef]
- Jensen, J.S.; Bradshaw, C.S.; Tabrizi, S.N.; Fairley, C.K.; Hamasuna, R. Azithromycin treatment failure in Mycoplasma genitalium-positive patients with nongonococcal urethritis is associated with induced macrolide resistance. Clin. Infect. Dis. 2008, 47, 1546–1553. [Google Scholar] [CrossRef]
- Manhart, L.E.; Gillespie, C.W.; Lowens, M.S.; Khosropour, C.M.; Colombara, D.V.; Golden, M.R.; Hakhu, N.R.; Thomas, K.K.; Hughes, J.P.; Jensen, N.L.; et al. Standard treatment regimens for nongonococcal urethritis have similar but declining cure rates: A randomized controlled trial. Clin. Infect. Dis. 2013, 56, 934–942. [Google Scholar] [CrossRef]
- Jain, R.; Danziger, L.H. The macrolide antibiotics: A pharmacokinetic and pharmacodynamic overview. Curr. Pharm. Des. 2004, 10, 3045–3053. [Google Scholar] [CrossRef]
- Handsfield, H.H.; Dalu, Z.A.; Martin, D.H.; Douglas, J.M., Jr.; McCarty, J.M.; Schlossberg, D.; Azithromycin Gonorrhea Study Group. Multicenter trial of single-dose azithromycin vs. ceftriaxone in the treatment of uncomplicated gonorrhea. Sex Transm. Dis. 1994, 21, 107–111. [Google Scholar] [CrossRef]
- Chandra, R.; Liu, P.; Breen, J.D.; Fisher, J.; Xie, C.; LaBadie, R.; Benner, R.J.; Benincosa, L.J.; Sharma, A. Clinical pharmacokinetics and gastrointestinal tolerability of a novel extended-release microsphere formulation of azithromycin. Clin. Pharm. 2007, 46, 247–259. [Google Scholar] [CrossRef]
- Yasuda, M.; Takahashi, S.; Kiyota, H.; Ishikawa, K.; Takahashi, A.; Yamamoto, S.; Arakawa, S.; Monden, K.; Muratani, T.; Hamasuna, R.; et al. Japanese guideline for clinical research of antimicrobial agents on urogenital infections: The first edition. J. Infect. Chemother. 2011, 17, 579–594. [Google Scholar] [CrossRef]
- Takahashi, S.; Takeyama, K.; Kunishima, Y.; Takeda, K.; Suzuki, N.; Nishimura, M.; Furuya, R.; Tsukamoto, T. Analysis of clinical manifestations of male patients with urethritis. J. Infect. Chemother. 2006, 12, 283–286. [Google Scholar] [CrossRef]
- Saika, T.; Nishiyama, T.; Kanayama, A.; Kobayashi, I.; Nakayama, H.; Tanaka, M.; Naito, S. Comparison of Neisseria gonorrhoeae isolates from the genital tract and pharynx of two gonorrhea patients. J. Infect. Chemother. 2001, 7, 175–179. [Google Scholar] [CrossRef]
- Yoshida, T.; Maeda, S.; Deguchi, T.; Ishiko, H. Phylogeny-based rapid identification of mycoplasmas and ureaplasmas from urethritis patients. J. Clin. Microbiol. 2002, 40, 105–110. [Google Scholar] [CrossRef]
- The European Committee on Antimicrobial Susceptibility Testing—EUCAST. Available online: http://www.escmid.org/reseach_projects/eucast/ (accessed on 19 March 2014).
- Institutional Review Board of Sapporo Medical University Hospital. Available online: http://web.sapmed.ac.jp/byoin/chiken/irb.html/ (accessed on 19 March 2014).
- University Hospital Medical Information Network Clinical Trial Registry (UMIN-CTR). Available online: http://www.umin.ac.jp/ctr/index-j.htm/ (accessed on 19 March 2014).
- Bolan, G.A.; Sparling, P.F.; Wasserheit, J.N. The emerging threat of untreatable gonococcal infection. N. Engl. J. Med. 2012, 366, 485–487. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. Cephalosporin susceptibility among Neisseria gonorrhoeae isolates—United States, 2000–2010. Morb. Mortal. Wkly. Rep. 2011, 60, 873–877. [Google Scholar]
- Ohnishi, M.; Saika, T.; Hoshina, S.; Iwasaku, K.; Nakayama, S.; Watanabe, H.; Kitawaki, J. Ceftriaxone-resistant Neisseria gonorrhoeae, Japan. Emerg. Infect. Dis. 2011, 17, 148–149. [Google Scholar] [CrossRef]
- Takahashi, S.; Matsukawa, M.; Kurimura, Y.; Takeyama, K.; Kunishima, Y.; Iwasawa, A.; Koroku, M.; Tanda, H.; Suzuki, N.; Takagi, Y.; et al. Clinical efficacy of azithromycin for male nongonococcal urethritis. J. Infect. Chemother. 2008, 14, 409–412. [Google Scholar] [CrossRef]
- Khaki, P.; Bhalla, P.; Sharma, A.; Kumar, V. Correlation between in vitro susceptibility and treatment outcome with azithromycin in gonorrhoea: A prospective study. Ind. J. Med. Microbiol. 2007, 25, 354–357. [Google Scholar] [CrossRef]
- Endo, K.; Onodera, S.; Kiyota, H.; Suzuki, H.; Hosobe, T.; Naruoka, T.; Sanuki, K. Drug-susceptibilities of Neisseria gonorrhoeae strains isolated from male patients with gonococcal urethritis against antimicrobial agents—Comparisons from 2006 to 2010. Jpn. J. Chemother. 2011, 59, 308–312. [Google Scholar]
- Hamasuna, R.; Yasuda, M.; Ishikawa, K.; Uehara, S.; Takahashi, S.; Hayami, H.; Yamamoto, S.; Matsumoto, T.; Minamitani, S.; Watanabe, A.; et al. Nationwide surveillance of the antimicrobial susceptibility of Neisseria gonorrhoeae from male urethritis in Japan. J. Infect. Chemother. 2013, 19, 571–578. [Google Scholar] [CrossRef]
- Katz, A.R.; Komeya, A.Y.; Soge, O.O.; Kiaha, M.I.; Lee, M.V.; Wasserman, G.M.; Maningas, E.V.; Whelen, A.C.; Kirkcaldy, R.D.; Shapiro, S.J.; et al. Neisseria gonorrhoeae with high-level resistance to azithromycin: Case report of the first isolate identified in the United States. Clin. Infect. Dis. 2012, 54, 841–843. [Google Scholar] [CrossRef]
- Takahashi, S.; Ichihara, K.; Hashimoto, J.; Kurimura, Y.; Iwasawa, A.; Hayashi, K.; Sunaoshi, K.; Takeda, K.; Suzuki, N.; Satoh, T.; et al. Clinical efficacy of levofloxacin 500 mg once daily for 7 days for patients with non-gonococcal urethritis. J. Infect. Chemother. 2011, 17, 392–396. [Google Scholar]
- Ito, S.; Yasuda, M.; Seike, K.; Sugawara, T.; Tsuchiya, T.; Yokoi, S.; Nakano, M.; Deguchi, T. Clinical and microbiological outcomes in treatment of men with non-gonococcal urethritis with a 100-mg twice-daily dose regimen of sitafloxacin. J. Infect. Chemother. 2012, 18, 414–418. [Google Scholar] [CrossRef]
- Takahashi, S.; Hamasuna, R.; Yasuda, M.; Ito, S.; Ito, K.; Kawai, S.; Yamaguchi, T.; Satoh, T.; Sunaoshi, K.; Takeda, K.; et al. Clinical efficacy of sitafloxacin 100 mg twice daily for 7 days for patients with non-gonococcal urethritis. J. Infect. Chemother. 2013, 19, 941–945. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Takahashi, S.; Kiyota, H.; Ito, S.; Iwasawa, A.; Hiyama, Y.; Uehara, T.; Ichihara, K.; Hashimoto, J.; Masumori, N.; Sunaoshi, K.; et al. Clinical Efficacy of a Single Two Gram Dose of Azithromycin Extended Release for Male Patients with Urethritis. Antibiotics 2014, 3, 109-120. https://doi.org/10.3390/antibiotics3020109
Takahashi S, Kiyota H, Ito S, Iwasawa A, Hiyama Y, Uehara T, Ichihara K, Hashimoto J, Masumori N, Sunaoshi K, et al. Clinical Efficacy of a Single Two Gram Dose of Azithromycin Extended Release for Male Patients with Urethritis. Antibiotics. 2014; 3(2):109-120. https://doi.org/10.3390/antibiotics3020109
Chicago/Turabian StyleTakahashi, Satoshi, Hiroshi Kiyota, Shin Ito, Akihiko Iwasawa, Yoshiki Hiyama, Teruhisa Uehara, Koji Ichihara, Jiro Hashimoto, Naoya Masumori, Kenichi Sunaoshi, and et al. 2014. "Clinical Efficacy of a Single Two Gram Dose of Azithromycin Extended Release for Male Patients with Urethritis" Antibiotics 3, no. 2: 109-120. https://doi.org/10.3390/antibiotics3020109




