The Effects of Two Novel Copper-Based Formulations on Helicobacter pylori
Abstract
:1. Introduction
2. Results and Discussion
2.1. Kill Curves of 5 NCTC/Clinical Isolates


2.2. Activity of CuAL42 and CuPC33 against 57 Clinical Isolates
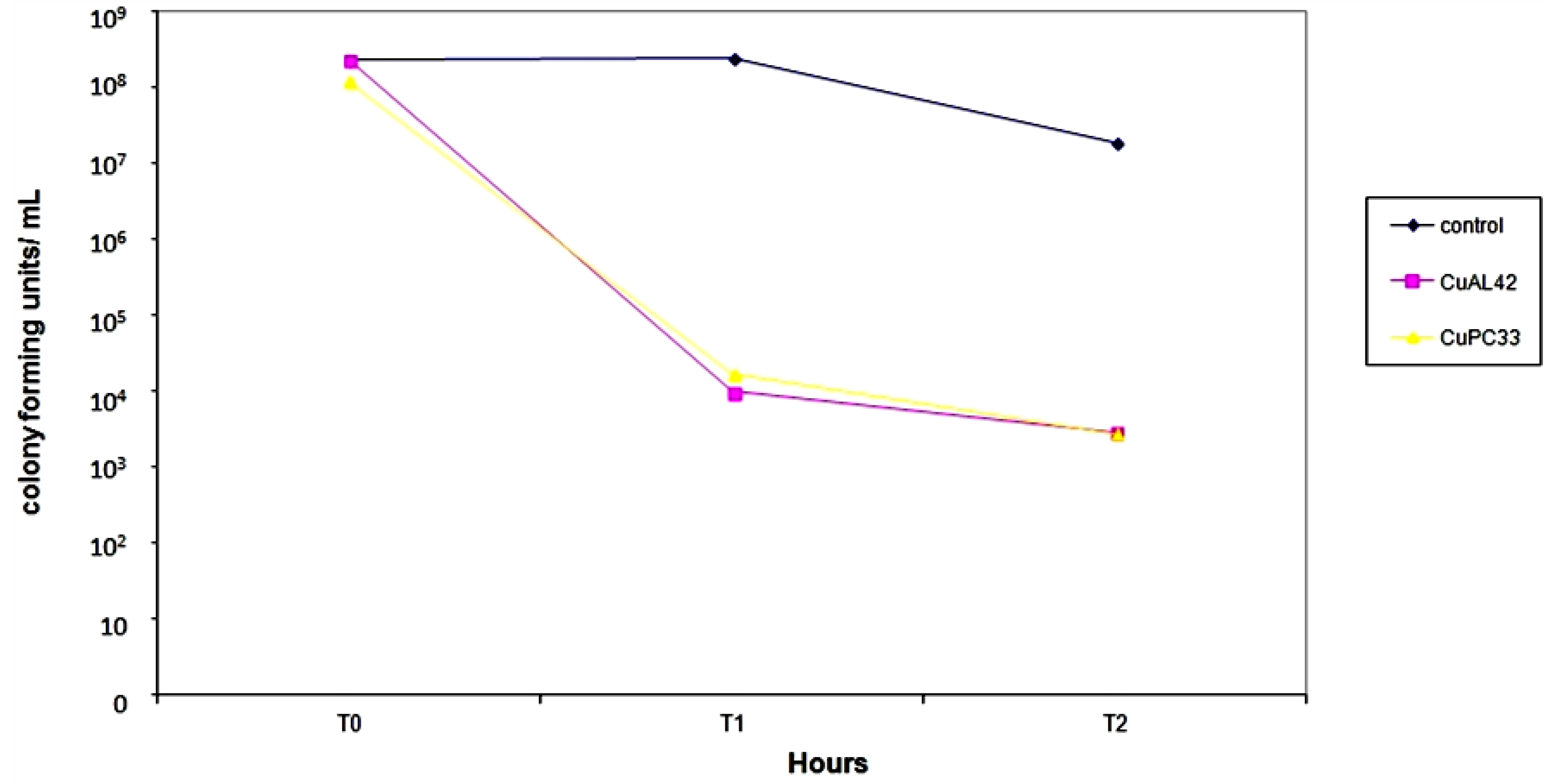
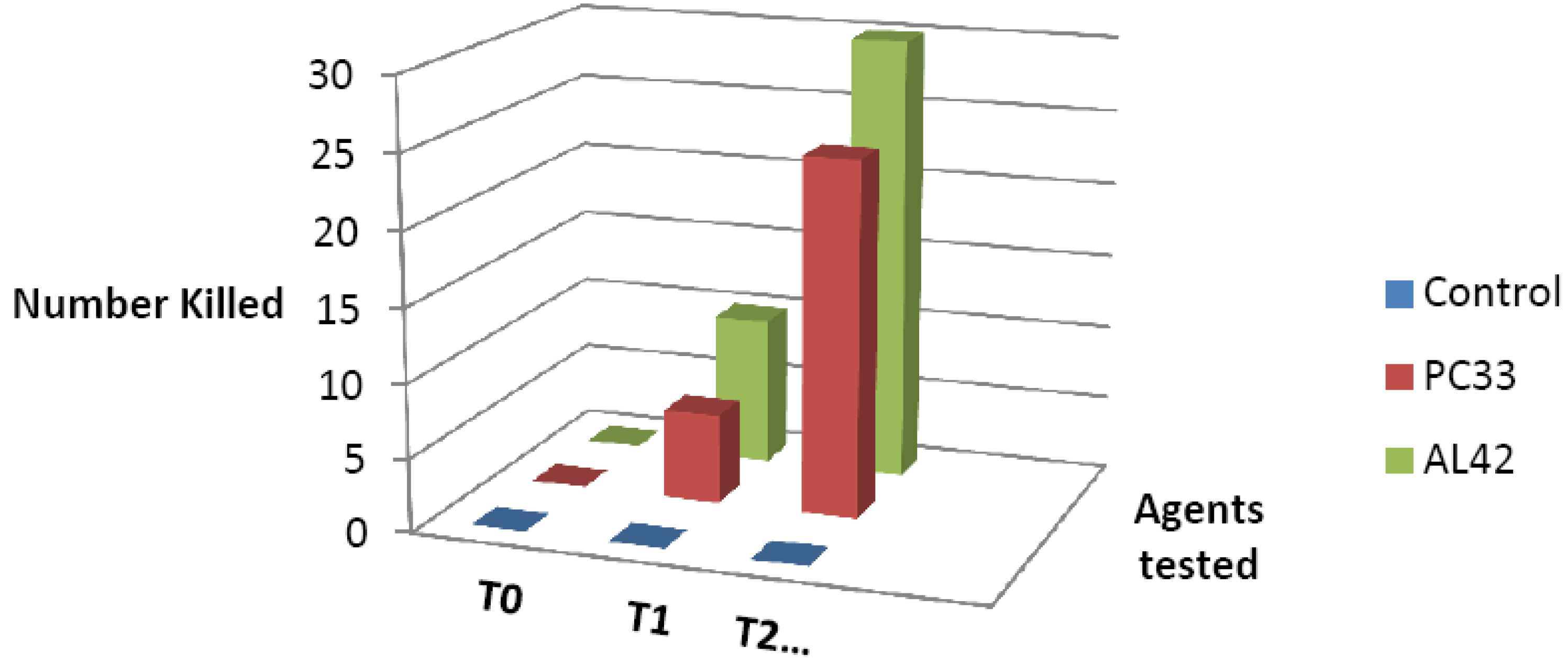
2.3. Discussion
3. Experimental
3.1. Materials
3.2. Determination of Antibiotic Sensitivity
3.3. Determination of Activity of CuAL42 and CuPC33
3.4. Statistics
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Warren, J.R.; Marshall, B.J. Unidentified curved bacilli on gastric epithelium in active chronic gastritis. Lancet 1983, I, 1273–1275. [Google Scholar]
- Sung, J.J.; Kuipers, E.J.; El-Serag, H.B. The global incidence and prevalence of PUD. Aliment. Pharmacol. Ther. 2009, 29, 938–946. [Google Scholar] [CrossRef]
- Atherton, J.C. The clinical relevance of strain types of Helicobacter pylori. Gut 1997, 40, 701–703. [Google Scholar] [CrossRef]
- IARC Working Group on the Evaluation of Carcinogenic Risks to Humans, Schistosomes, Liver Flukes and Helicobacter pylori; World Health Organization, International Agency for Research on Cancer: Lyon, France, 1994; Volume 61, pp. 1–241.
- Santacroce, L.; Cagiano, R.; del Prete, D.; Bottalico, L.; Sabatini, R.; Carlaio, R.G.; Prejbeanu, R.; vermesan, H.; Dragulescu, S.I.; Vermesan, D.; et al. Helicobacter pylori infection and gastric MALToma: An up to date and therapeutic highlight. Clin. Ter. 2008, 159, 457–462. [Google Scholar]
- O’Connor, A.; Gisbert, J.P.; McNamara, D.; O’Morain, C.A. Treatment of Helicobacter pylori infection 2010. Helicobacter 2010, 15, 46–52. [Google Scholar]
- Megraud, F. H pylori antibiotic resistance: Prevalence, importance and advances in testing. Gut 2004, 53, 1374–1384. [Google Scholar] [CrossRef]
- Koletzko, S.; Richy, F.; Bontems, P.; Crone, J.; Lalach, N.; Monteiro, M.L.; Gottrand, F.; Celinska-Cedro, D.; Roma-Giannikou, E.; Oderda, G.; et al. Prospective multicentre study on antibiotic resistance of Helicobacter pylori strains obtained from children living in Europe. Gut 2006, 55, 1711–1716. [Google Scholar] [CrossRef] [Green Version]
- Ramond, J.; Lamarque, D.; Kalach, N.; Chaussade, S.; Burucoa, C. High levels of antimicrobial resistance in French Helicobacter pylori isolates. Helicobacter 2010, 15, 21–27. [Google Scholar]
- Agudo, S.; Alarcon, T.; Cibrelus, L.; Urruzuno, P.; Martinez, M.J.; Lopez-Brea, M. High percentage of clarithromycin and metronidazole resistance in Helicobacter pylori clinical isolates obtained from Spanish children. Rev. Esp. Quimioter. 2009, 22, 88–92. [Google Scholar]
- Sasaki, M.; Ogasawara, N.; Utsumi, K.; Kawamura, N.; Kamiya, T.; Kataoka, H.; Tanida, S.; Mizoshita, T.; Kasugai, K.; Joh, T. Changes in 12 year first line eradication rate of Helicobacter pylori based on triple therapy with proton pump inhibitor, amoxicillin and clarithromycin. J. Clin. Biochem. Nutr. 2010, 47, 53–58. [Google Scholar] [CrossRef]
- Gisbert, J.P.; Calvet, X.; O’Connor, A.; Megraud, F.; O’Morain, C. A Sequential therapy for Helicobacter pylori eradication: A critical review. J. Clin. Gastroenterol. 2010, 44, 313–325. [Google Scholar]
- Jafri, N.S.; Hornung, C.A.; Howden, C.W. Meta analysis: Sequential therapy appears superior to standard therapy for Helicobacter pylori infection in patients naïve to treatment. Ann. Int. Med. 2008, 148, 23–31. [Google Scholar]
- Graham, D.Y.; Lu, H.; Yamaoka, Y. A report card to grade Helicobacter therapy. Helicobacter 2007, 12, 275–278. [Google Scholar] [CrossRef]
- Graham, D.Y.; Lu, H.; Yamaoka, Y. Therapy for Helicobacter pylori infection can be improved: Sequential therapy and beyond. Drugs 2008, 68, 725–736. [Google Scholar] [CrossRef]
- Graham, D.Y. Efficient identification and evaluation of effective Helicobacter pylori therapies. Clin. Gastroenterol. Hepatol. 2009, 7, 145–148. [Google Scholar] [CrossRef]
- Rotramel, A.; Poritz, L.S.; Messaris, E.; Berg, A.; Stewart, D.B. PPI therapy and albumen are better predictors of recurrent Clostridium difficile colitis than choice of antibiotics. J. Gastrointest. Surg. 2012, 16, 2267–2273. [Google Scholar]
- Younson, J.; O’Mahony, R.; Liu, H.; Basset, C.; Grant, S.; Campion, C.; Jennings, L.; Vaira, D.; Kelly, C.G.; Roitt, I.M.; et al. Human domain antibody and Lewisb-glycoconjugate that inhibit binding of Helicobacter pylori to Lewisb receptor and adhesion to human gastric epithelium. J. Infect. Dis. 2009, 200, 1574–1582. [Google Scholar] [CrossRef]
- Bedwell, J.; Holton, J.; Vaira, D.; MacRobert, A.J.; Bown, S.G. In vitro killing of Helicobacter pylori with photodynamic therapy. Lancet 1990, 335, 1287. [Google Scholar] [CrossRef]
- O’Mahony, R.; Al-Khtheeri, H.; Weerasekera, D.; Fernando, N.; Vaira, D.; Holton, J.; Basset, C. Bactericidal and Anti-adhesive properties of culinary and medicinal plants against Helicobacter pylori. World J. Gastroenterol. 2005, 47, 7499–7507. [Google Scholar]
- Boschian, A.; Jensen, C.S.; Rasmussen, L. Antibiotic effect of anti-malarials on H. pylori infection in vivo. Helicobacter 2010, 15, 335, Abstract. [Google Scholar]
- Menghini, L.; Leporini, L.; Tirillini, B.; Epifano, F.; Genovese, S. Chemical composition and inhibitory activity against Helicobacter pylori of the essential oil of Apium nodiflorum (Apiaceae). J. Med. Food 2010, 13, 228–230. [Google Scholar] [CrossRef]
- Lawal, T.O.; Adeniyi, B.A.; Moody, J.O.; Mahady, G.B. Combination studies of Eucalyptus torelliana (F MUELL) leaf extract and clarithromycin on H. pylori. Helicobacter 2010, 15, 336, Abstract. [Google Scholar]
- Gant, V.A.; Wren, M.W.; Rollins, M.S.; Jeanes, A.; Hickok, S.S.; Hall, T.J. Three novel highly charged copper based biocides: Safety and efficacy against healthcare associated organisms. J. Antimicrob. Chemother. 2007, 60, 294–299. [Google Scholar] [CrossRef]
- Shaikh, A.R.; Yadav, M.; Giridhar, R. Antibiotic metal complexes—An approach for Helicobacter therapy. Helicobacter 2010, 15, 341, Abstract. [Google Scholar]
- Cervantes, C.; Gutierrez-Corona, F. Copper resistance mechanisms in bacteria and fungi. FEMS Microbiol. Rev. 1994, 14, 121–138. [Google Scholar] [CrossRef]
- Sagripanti, J.L.; Routson, L.B.; Lytle, C.D. Virus inactivation by copper or iron ions alone and in the presence of peroxide. Appl. Environ. Microbiol. 1993, 59, 4374–4376. [Google Scholar]
- Kim, J.H.; Cho, H.; Ryu, S.E.; Choi, M.U. Effects of metal ions on the activity of protein tyrosine phosphatise VHR: Highly potent and reversible oxidative inactivation by Cu2+ ion. Arch. Biochem. Biophys. 2000, 382, 72–80. [Google Scholar]
- Hall, T.J.; Wren, M.W.; Jeanes, A.; Gant, V.A. A comparisonof the antibacterial efficacy and cytotoxicity to cultured human skin cells of 7 commercial hand rubs and Xgel, a new copper-based biocidal hand rub. Am. J. Infect. Control. 2009, 37, 322–326. [Google Scholar] [CrossRef]
- Koch, K.A.; Pena, O.; Thiele, D.J. Copper binding motifs in catalysis, transport, detoxification and signalling. Chem. Biol. 1997, 4, 549–560. [Google Scholar] [CrossRef]
- Silen, W.; Ito, S. Mechanisms for rapid re-epithelialization of the gastric mucosal surface. Annu. Rev. Physiol. 1985, 47, 217–229. [Google Scholar] [CrossRef]
- Wallace, J.L. Mechanisms of protection and healing: Current knowledge and future research. Am. J. Med. 2001, 110, 19S–23S. [Google Scholar]
- Konturek, S.J.; Dembinski, A.; Warzecha, Z.; Brzozoski, T.; Gregory, H. Role of epidermal growth factor in healing of chronic gastroduodenal ulcers in rats. Gastroenterology 1992, 94, 1300–1307. [Google Scholar]
- Borkow, G.; Gabbay, J.; Dardik, R.; Eidelman, A.I.; Lavie, Y.; Grunfeld, Y.; Ikher, S.; Huszar, M.; Zatcoff, R.C.; Marikovsky, M. Molecular mechanisms of enhanced wound healing by copper-oxide impregnated dressings. Wound Repair Regen. 2010, 18, 266–275. [Google Scholar] [CrossRef]
- D’Andrea, L.D.; Romanelli, A.; Di Stasi, R.; Pedone, C. Bioinorganic aspects of angiogenesis. Dalton Trans. 2010, 39, 7625–7636. [Google Scholar] [CrossRef]
- Megraud, F.; Lehors, P. Helicobacter pylori detection and antimicrobial susceptibility testing. Clin. Microbiol. Rev. 2007, 20, 280–322. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Saracino, I.M.; Zaccaro, C.; Re, G.L.; Vaira, D.; Holton, J. The Effects of Two Novel Copper-Based Formulations on Helicobacter pylori. Antibiotics 2013, 2, 265-273. https://doi.org/10.3390/antibiotics2020265
Saracino IM, Zaccaro C, Re GL, Vaira D, Holton J. The Effects of Two Novel Copper-Based Formulations on Helicobacter pylori. Antibiotics. 2013; 2(2):265-273. https://doi.org/10.3390/antibiotics2020265
Chicago/Turabian StyleSaracino, Ilaria M., Cristina Zaccaro, Giovanna Lo Re, Dino Vaira, and John Holton. 2013. "The Effects of Two Novel Copper-Based Formulations on Helicobacter pylori" Antibiotics 2, no. 2: 265-273. https://doi.org/10.3390/antibiotics2020265



