Application of Paper-Supported Printed Gold Electrodes for Impedimetric Immunosensor Development
Abstract
:1. Introduction
2. Experimental Section
2.1. Print Substrate
2.2. Synthesis and Characterization of the Gold Nanoparticles
2.3. Inkjet Printing of Gold Electrodes
2.4. Monothiols and Proteins
2.5. Formation of Supramolecular Protein Layers on Printed Gold Electrodes
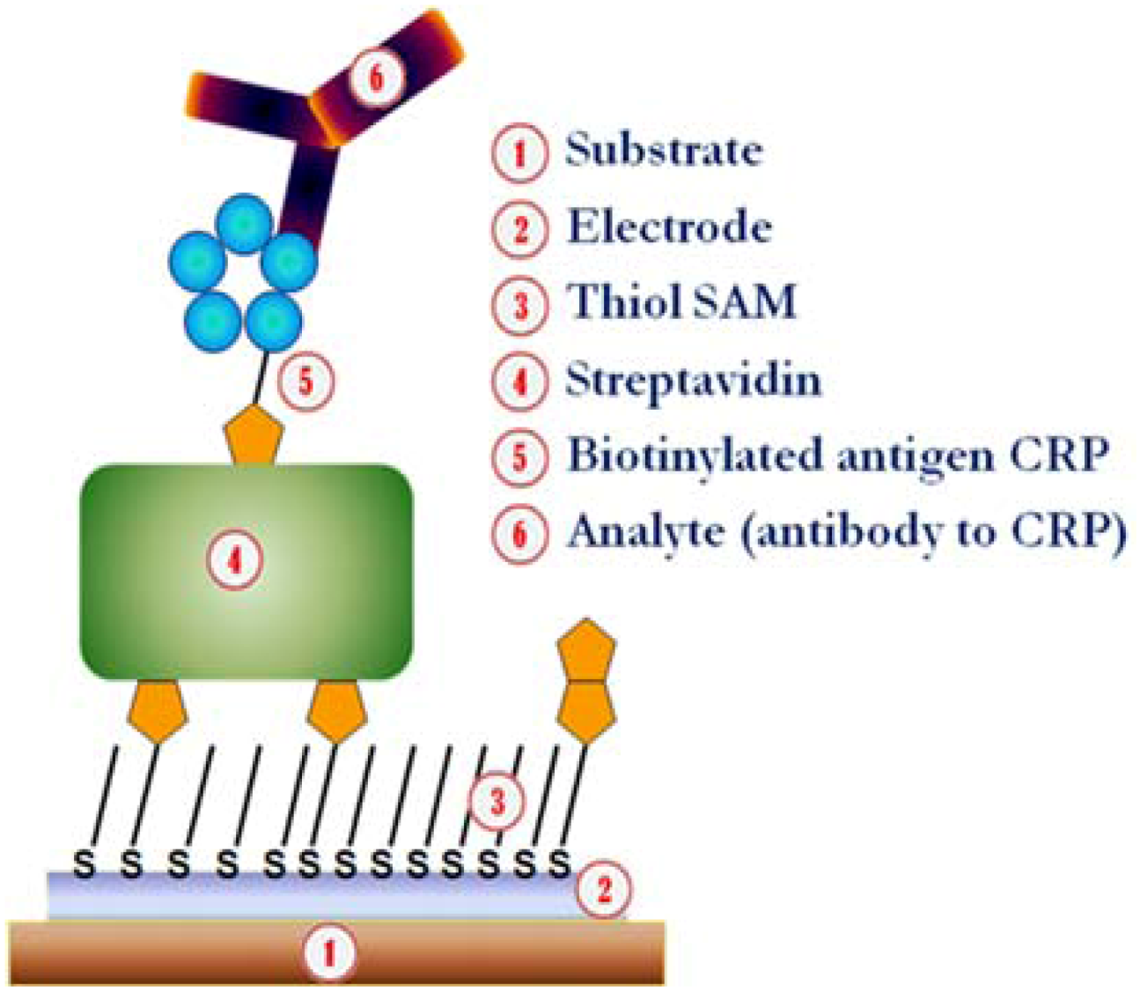
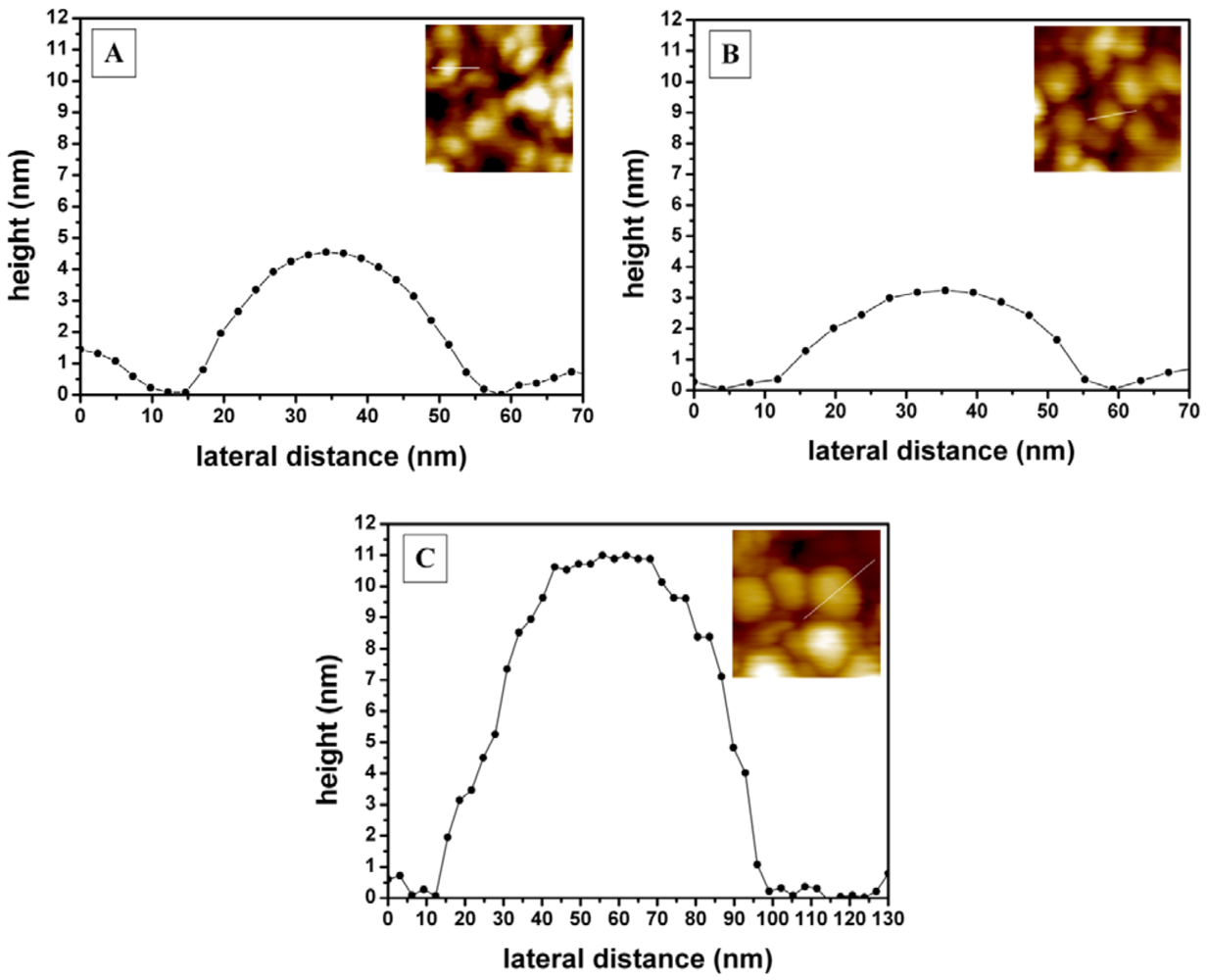
2.6. Scanning Probe Microscopy
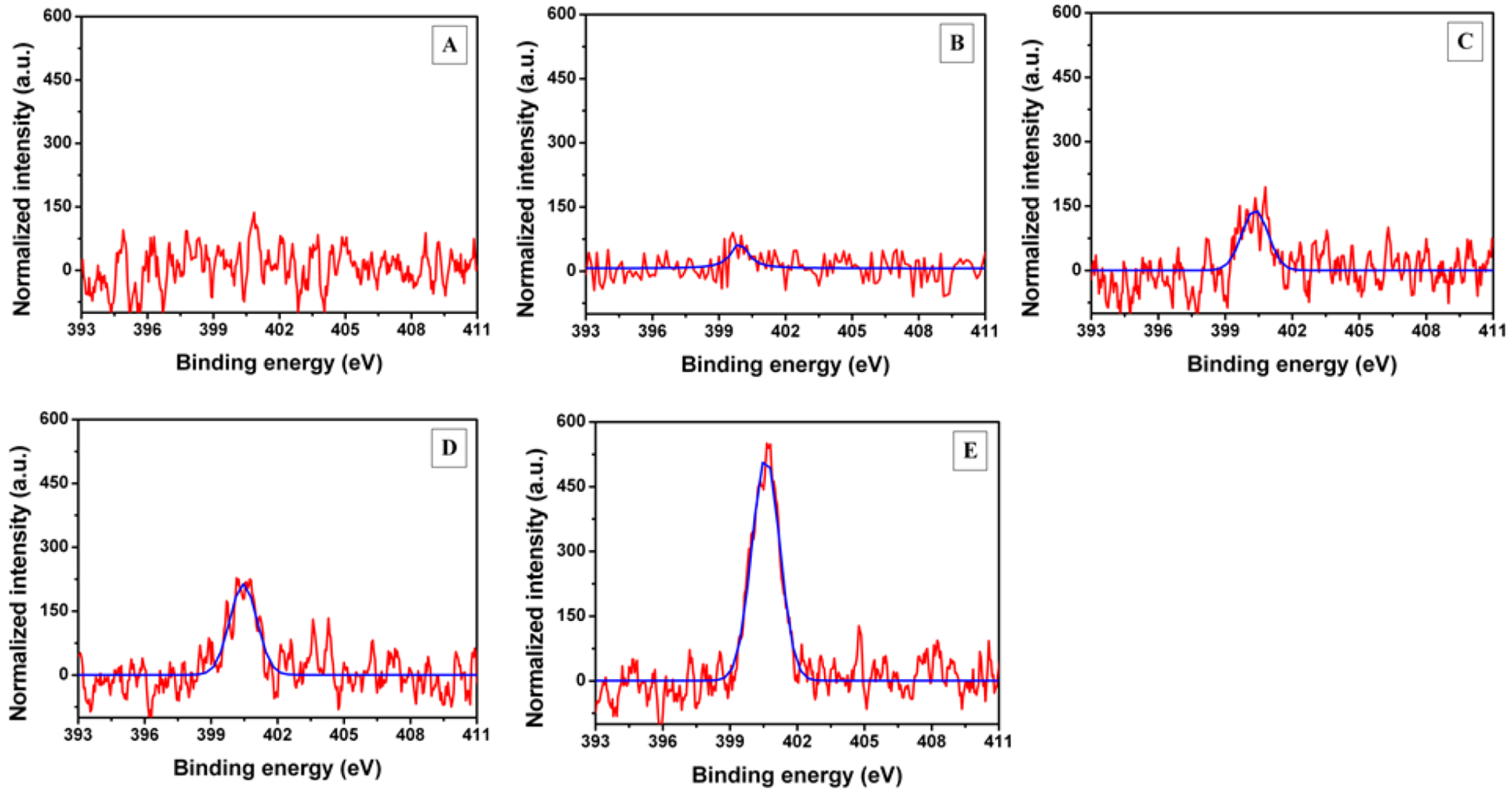
2.7. X-Ray Photoelectron Spectroscopy
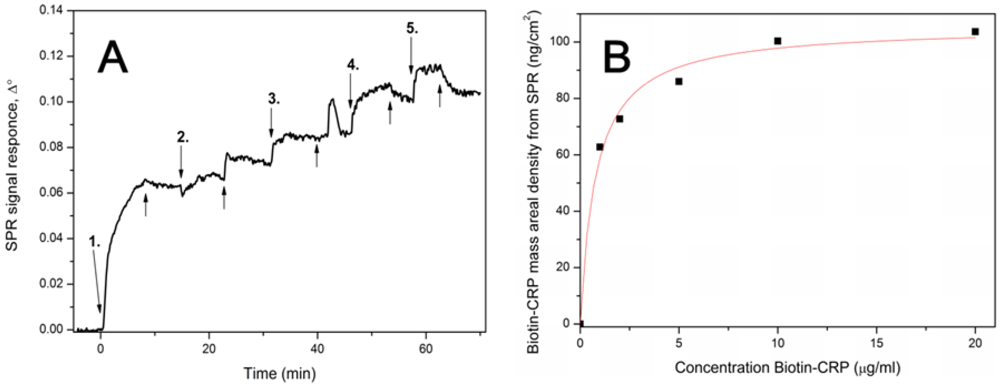
2.8. Surface Plasmon Resonance

2.9. Impedance Measurements
3. Results and Discussion
3.1. Binding Capacity of Biofunctional Layers Determined by SPR
3.2. Topographical and Chemical Characterization of the Immobilized Layers on Paper-Supported Printed Gold Electrodes
| XPS element | MuOH:Biotin-PEG-thiol (85:15 mol%) SAM | Streptavidin | bio-CRP antigen | anti-CRP antibody |
|---|---|---|---|---|
| C1s | 67.8 ± 4.0% | 69.3 ± 3.4% | 62.9 ± 3.0% | 67.7 ± 4.2% |
| Au4f | 15.6 ± 0.6% | 10.2 ± 1.0% | 9.0± 0.8% | 7.0 ± 1.0% |
| N1s | 0.7 ± 0.4% | 2.0 ± 0.3% | 4.0 ± 0.8% | 7.8 ± 0.5% |
| S2p | 0.3 ± 0.2% | 0.2 ± 0.2% | 0.1 ± 0.1% | - |
| O1s | 15.8 ± 4.1% | 20.3 ± 2.0% | 21.2 ± 1.7% | 17.5 ± 1.3% |

| Layer | Sdr ± SD [%] | σ ± SD [nm] |
|---|---|---|
| gold electrode | 1.6 ± 0.2 | 2.1 ± 0.3 |
| MuOH:Biotin-PEG-thiol (85:15 mol%) SAM | 1.4 ± 0.3 | 2.2 ± 0.5 |
| Streptavidin | 3.1 ± 0.5 | 2.5 ± 0.4 |
| bio-CRP antigen | 2.4 ± 0.5 | 3.3 ± 0.5 |
| Anti-CRP antibody | 4.7 ± 0.8 | 5.4 ± 1.0 |
3.3. Impedimetric Analysis of the Immobilized Layers


4. Conclusions
Acknowledgments
References
- Borisov, S.M.; Wolfbeis, O.S. Optical biosensors. Chem. Rev. 2008, 60, 307–318. [Google Scholar]
- Sjöwall, C.; Wetterö, J. Pathogenic implications for autoantibodies against C-reactive protein and other acute phase proteins. Clin. Chim. Acta 2007, 378, 13–23. [Google Scholar] [CrossRef]
- Berggren, C.; Bjarnason, B.; Johansson, G. Capacitive biosensors. Electroanalysis 2001, 13, 173–180. [Google Scholar] [CrossRef]
- Katz, E.; Willner, I. Probing biomolecular interactions at conductive and semiconductive surfaces by impedance spectroscopy: Routes to impedimetric immunosensors, DNA-sensors and enzyme biosensors. Electroanalysis 2003, 15, 913–947. [Google Scholar] [CrossRef]
- Guan, J.G.; Miao, Y.Q.; Zhang, Q.J. Impedimetric biosensors. J. Biosens. Bioeng. 2004, 97, 219–226. [Google Scholar]
- K’Owino, I.O.; Sadik, O.A. Impedance spectroscopy: A powerful tool for rapid, biomolecular screening and cell culturing monitoring. Electroanalysis 2005, 17, 2101–2113. [Google Scholar] [CrossRef]
- Daniels, J.S.; Pourmand, N. Label-free impedance biosensors: Opportunities and challenges. Electroanalysis 2007, 19, 1239–1257. [Google Scholar] [CrossRef]
- Prodromidis, M.I. Impedimetric immunosensors—A review. Electrochim. Acta 2010, 55, 4227–4233. [Google Scholar] [CrossRef]
- Tobjörk, D.; Österbacka, R. Paper Electronics. Adv. Mater. 2011, 23, 1935–1961. [Google Scholar]
- Nie, Z.; Nijhuis, C.A.; Gong, J.; Chen, X.; Kumachev, A.; Martinez, A.W.; Narovlyansky, M.; Whitesides, G.M. Electrochemical sensing in paper-based microfluidic devices. Lab Chip 2010, 10, 477–483. [Google Scholar] [CrossRef]
- Zang, D.; Ge, L.; Yan, M.; Song, X.; Yu, J. Electrochemical immunoassay on a 3D microfluidic paper-based device. Chem. Commun. 2012, 48, 4683–4685. [Google Scholar]
- Dungchai, W.; Chailapakul, O.; Henry, C.S. Electrochemical detection for paper-based microfluidics. Anal. Chem. 2009, 81, 5821–5826. [Google Scholar]
- Määttänen, A.; Vanamo, U.; Ihalainen, P.; Pulkkinen, P.; Tenhu, H.; Bobacka, J.; Peltonen, J. A low-cost paper-based inkjet-printed platform for electrochemical analyses. Sens. Actuator. B Chem. 2012. [Google Scholar] [CrossRef]
- Bollström, R.; Määttänen, A.; Ihalainen, P.; Toivakka, M. Method for Creating a Substrate for Printed or Coated Functionality, Substrate, Functional Device and Its Use. WO 2010/086511, PCT/FI2010/050056, August 2010. [Google Scholar]
- Bollström, R.; Määttänen, A.; Tobjörk, D.; Ihalainen, P.; Kaihovirta, N.; Österbacka, R.; Peltonen, J.; Toivakka, M. A multilayer coated fiber-based substrate suitable for printed functionality. Org. Electron. 2009, 10, 1020–1023. [Google Scholar]
- Määttänen, A.; Ihalainen, P.; Bollström, R; Toivakka, M.; Peltonen, J. Wetting and print quality study of an inkjet-printed poly(3-hexylthiophene) on pigment coated papers. Colloid. Surface. A 2010, 367, 76–84. [Google Scholar]
- Ihalainen, P.; Määttänen, A.; Mattinen, U.; Stepien, S.; Bollström, R.; Toivakka, M.; Bobacka, J.; Peltonen, J. Electrodeposition on PEDOT-Cl film on a fully printed Ag/polyaniline electrode on paper. Thin Solid Films 2011, 519, 2172–2175. [Google Scholar]
- Tobjörk, D.; Aarnio, H.; Pulkkinen, P.; Bollström, R.; Määttänen, A.; Ihalainen, P.; Mäkelä, T.; Peltonen, J.; Toivakka, M.; Tenhu, H.; Österbacka, R. IR-sintering of ink-jet printed metal-nanoparticles on paper. Thin Solid Films 2012, 520, 2949–2955. [Google Scholar]
- Määttänen, A.; Ihalainen, P.; Pulkkinen, P.; Wang, S.; Tenhu, H.; Peltonen, J. Inkjet-printed gold electrodes on paper: Characterization and functionalization. ACS Appl. Mater. Interfaces 2012, 4, 955–964. [Google Scholar]
- Ihalainen, P.; Majumdar, H.; Määttänen, A.; Wang, S.; Österbacka, R.; Peltonen, J. Versatile characterization of thiol-functionalized printed metal electrodes on flexible substrates for cheap diagnostic applications. BBA-Gen. Subjects 2012. [Google Scholar] [CrossRef]
- Hostetler, M.J.; Wingate, J.E.; Zhong, C.-J.; Harris, J.E.; Vachet, R.W.; Clark, M.R.; Londono, J.D.; Green, S.J.; Stokes, J.J.; Wignall, G.D.; Glish, G.L.; Porter, M.D.; Evans, N.D.; Murray, R.W. Alkanethiolate gold cluster molecules with core diameters from 1.5 to 5.2 nm: Core and monolayer properties as a function of core size. Langmuir 1998, 14, 17–30. [Google Scholar]
- Määttänen, A.; Fors, D.; Wang, S.; Valtakari, D.; Ihalainen, P.; Peltonen, J. Paper-based planar reaction arrays for printed diagnostics. Sens. Actuator. B Chem. 2011, 160, 1404–1412. [Google Scholar] [CrossRef]
- Reimhult, E.; Larsson, C.; Kasemo, B.; Höök, F. Simultaneous surface plasmon resonance and quartz crystal microbalance with dissipation monitoring measurements of biomolecular adsorption events involving structural transformations and variations in coupled water. Anal. Chem. 2004, 76, 7211–7220. [Google Scholar] [CrossRef]
- Su, X.; Wu, Y.; Robelek, R.; Knoll, W. Surface plasmon resonance spectroscopy and quartz crystal microbalance study of streptavidin film structure effects on biotinylated DNA assembly and target DNA hybridization. Langmuir 2005, 21, 348–353. [Google Scholar] [CrossRef]
- Yang, N.; Su, X.; Tjong, V.; Knoll, W. Evaluation of 2-D and 3-D streptavidin chips for study DNA-DNA and protein-DNA interactions. Biosens. Bioeletron. 2009, 24, 2522–2527. [Google Scholar] [CrossRef]
- Motie, M.; Brockmeier, S.; Potempa, L.A. Binding of model soluble immune complexes to modified C-reactive protein. J. Immunol. 1996, 156, 4435–4441. [Google Scholar]
- Shirve, A.K.; Cheetham, G.M.T.; Holden, D.; Myles, D.A.; Turnell, W.G.; Volanakis, J.E.; Pepys, M.B.; Bloomer, A.C.; Greenhough, T.J. Three dimensional structure of human C-reactive protein. Nat. Struct. Biol. 1996, 3, 346–354. [Google Scholar] [CrossRef]
- Thompson, D.; Pepys, M.B.; Wood, S.P. The physiological structure of human C-reactive protein and its complex with phosphocholine. Structure 1999, 7, 167–177. [Google Scholar] [CrossRef]
- Lin, S.; Lee, C.-K.; Wang, Y.-M.; Huang, L.-S.; Lin, Y.-H.; Lee, S.-Y.; Sheu, B.-C.; Hsu, S.-M. Measurement of dimensions of pentagonal doughnut-shaped C-reactive protein using an atomic force microscope and a dual polarisation interferometric biosensor. Biosens. Bioeletron. 2005, 22, 323–327. [Google Scholar]
- Lee, S.-K.; Kim, H.-C.; Cho, S.-J.; Jeong, S.W.; Jeon, W.B. Binding behavior of CRP and anti-CRP antibody analyzed with SPR and AFM measurement. Ultramicroscopy 2008, 108, 1374–1378. [Google Scholar] [CrossRef]
- Lindberg, B.; Maripuu, R.; Siegbahn, K.; Larsson, R.; Golander, C.C.; Eriksson, J.C. ESCA studies of heparinized and related surfaces. J. Colloid Interface Sci. 1983, 95, 308–321. [Google Scholar] [CrossRef]
- Moulder, J.F.; Stickle, W.F.; Sobol, P.E.; Bomben, K.D. Handbook of X-Ray Photoelectron Spectroscopy; Perkin-Elmer Corporation: Eden Prairie, MN, USA, 1992. [Google Scholar]
- Sousa, S.R.; Moradas-Ferreira, P.; Saramago, B.; Viseu Melo, L.; Barbosa, M.A. Human serum albumin adsorption on TiO2 from single protein solutions and from plasma. Langmuir 2004, 20, 9745–9754. [Google Scholar]
- Weber, P.C.; Ohlendorf, D.H.; Wendoloski, J.J.; Salemme, F.R. Structural origins of high-affinity biotin binding to streptavidin. Science 1989, 243, 85–88. [Google Scholar]
- Silverton, E.W.; Navia, M.A.; Davies, D.R. Three-dimensional structure of an intact human immunoglobulin. Proc. Natl. Acad. Sci. USA 1977, 74, 5140–5144. [Google Scholar] [CrossRef]
- Stelzle, M.; Weissmueller, G.; Sackmann, E. On the application of supported bilayers as receptive layers for biosensors with electrical detection. J. Phys. Chem. 1993, 97, 2974–2981. [Google Scholar]
- Pei, R.; Cheng, Z.; Wang, E.; Yang, X. Amplification of antigen-antibody interactions based on biotin labeled protein-streptavidin network complex using impedance spectroscopy. Biosens. Bioeletron. 2001, 16, 355–361. [Google Scholar]
Appendix

© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ihalainen, P.; Majumdar, H.; Viitala, T.; Törngren, B.; Närjeoja, T.; Määttänen, A.; Sarfraz, J.; Härmä, H.; Yliperttula, M.; Österbacka, R.; et al. Application of Paper-Supported Printed Gold Electrodes for Impedimetric Immunosensor Development. Biosensors 2013, 3, 1-17. https://doi.org/10.3390/bios3010001
Ihalainen P, Majumdar H, Viitala T, Törngren B, Närjeoja T, Määttänen A, Sarfraz J, Härmä H, Yliperttula M, Österbacka R, et al. Application of Paper-Supported Printed Gold Electrodes for Impedimetric Immunosensor Development. Biosensors. 2013; 3(1):1-17. https://doi.org/10.3390/bios3010001
Chicago/Turabian StyleIhalainen, Petri, Himadri Majumdar, Tapani Viitala, Björn Törngren, Tuomas Närjeoja, Anni Määttänen, Jawad Sarfraz, Harri Härmä, Marjo Yliperttula, Ronald Österbacka, and et al. 2013. "Application of Paper-Supported Printed Gold Electrodes for Impedimetric Immunosensor Development" Biosensors 3, no. 1: 1-17. https://doi.org/10.3390/bios3010001





