Controlling the 3D Electromagnetic Coupling in Co-Sputtered Ag–SiO2 Nanomace Arrays by Lateral Sizes
Abstract
:1. Introduction
2. Experimental Section
2.1. Materials
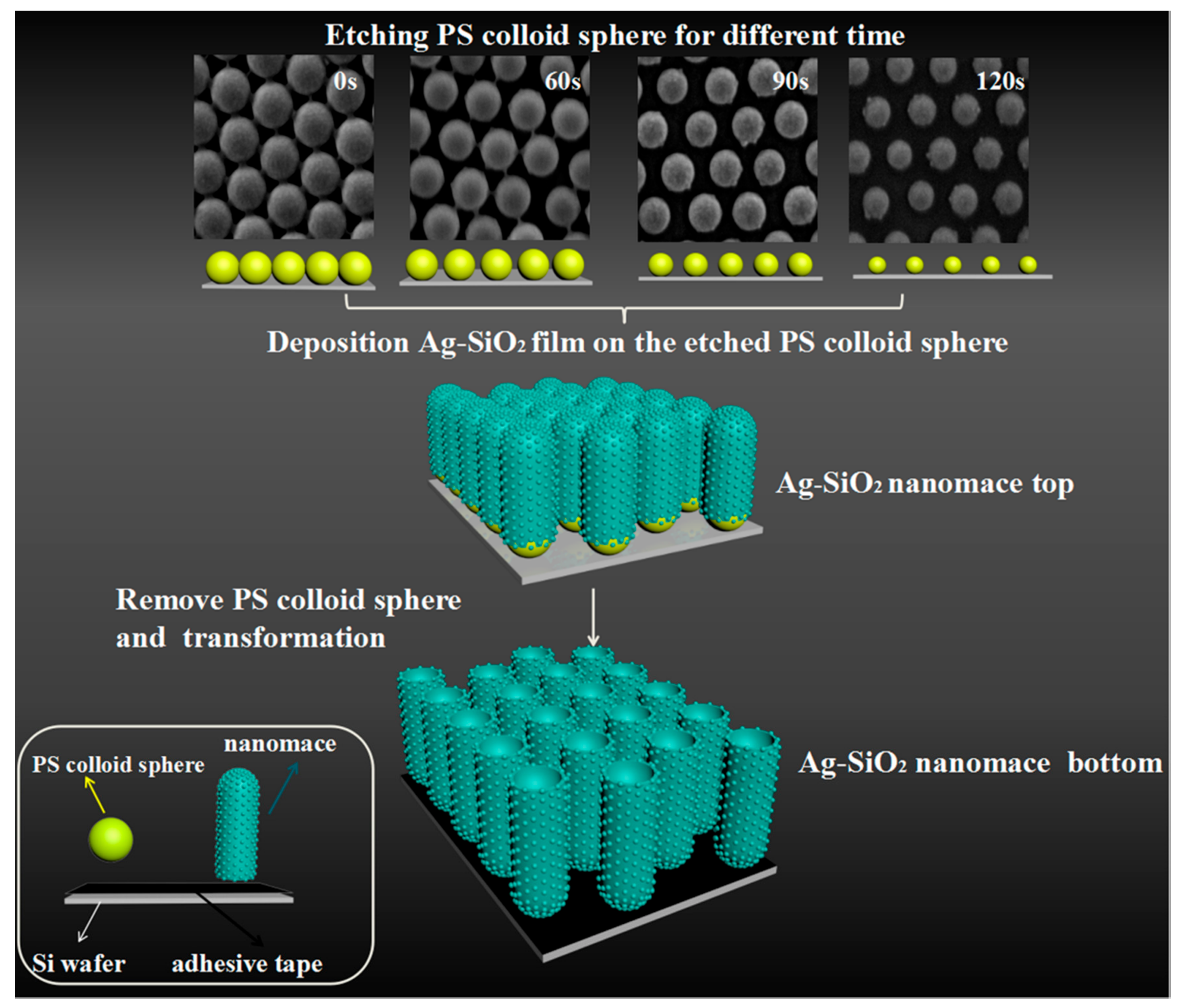
2.2. Assemble of PS Sphere Arrays
2.3. Preparation of Ag–SiO2 Nanomace Arrays and Probe Molecule Absorption
2.4. Characterization of Substrates and SERS
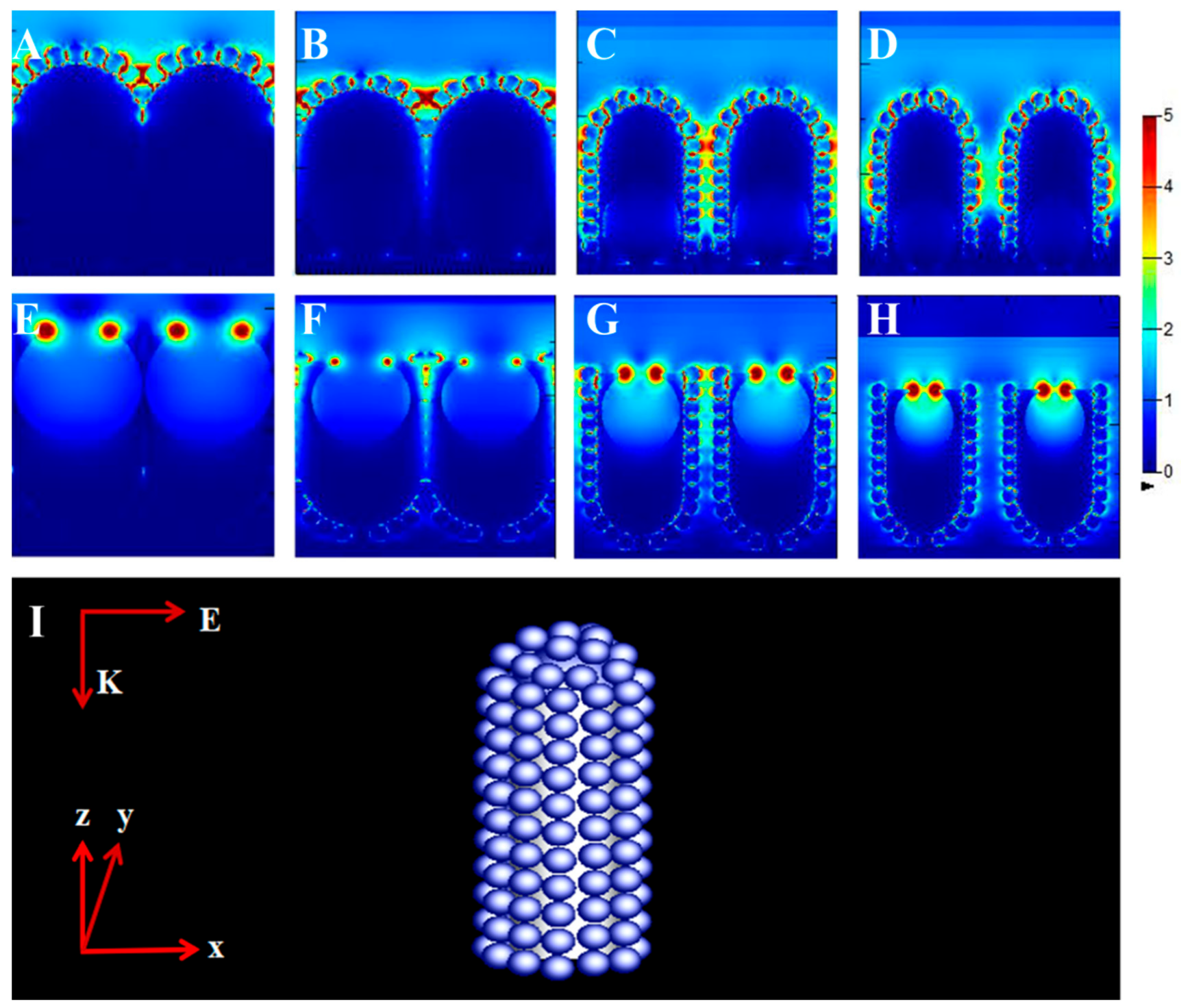
2.5. Finite-Difference Time-Domain (FDTD) Simulations
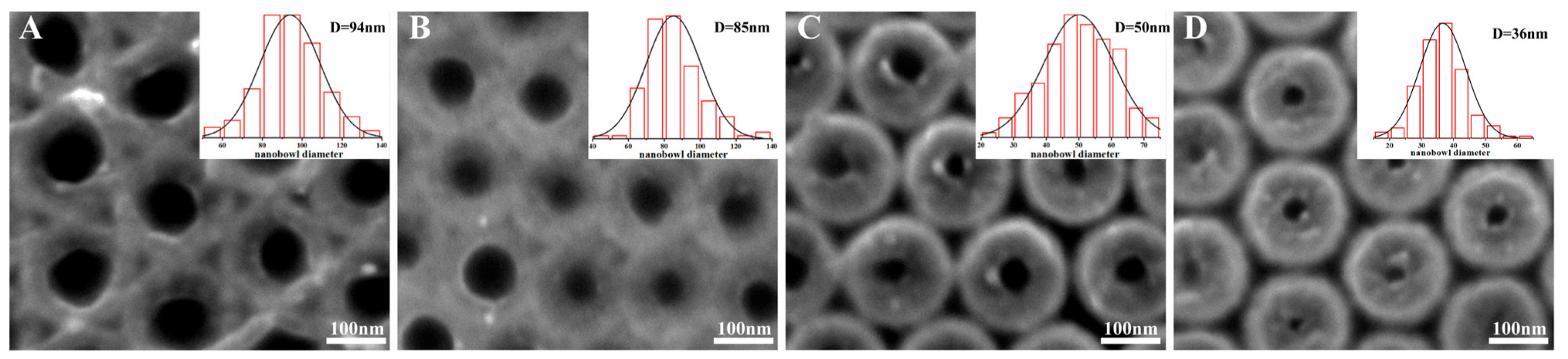
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wang, Z.Z.; Wen, X.; Feng, Z.H.; Lin, L.; Liu, R.H.; Huang, P.P.; Chen, G.L.; Huang, F.; Zheng, Z.Q. Highly ordered Au-Ag alloy arrays with tunable morphologies for surface enhanced Raman spectroscopy. Chen. Eng. J. 2018, 345, 389–394. [Google Scholar] [CrossRef]
- Chikkaraddy, R.; De Nijs, B.; Benz, F.; Barrow, S.J.; Scherman, O.A.; Rosta, E.; Demetriadou, A.; Fox, P.; Hess, O.; Baumberg, J.J. Single-molecule strong coupling at room temperature in plasmonic nanocavities. Nature 2016, 535, 127–130. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gao, R.X.; Zhang, Y.Y.; Zhang, F.; Guo, S.; Wang, Y.X.; Chen, L.; Yang, J.H. SERS Polarization-dependent Effects for an Ordered 3D Plasmonic Tilted Silver Nanorod Array. Nanoscale 2018, 10, 8106–8114. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Lin, W.H.; Zhao, H.F.; Wang, P.J.; Sun, M.T. The nature of plasmon-exciton codriven surface catalytic reaction. J. Raman Spectrosc. 2017, 49, 383–387. [Google Scholar] [CrossRef]
- Lu, B.; Zhan, F.Y.; Gong, G.D.; Cao, Y.L.; Zhen, Q.; Hu, P.F. Room-temperature mechanochemical synthesis of silver nanoparticle homojunction assemblies for the surface-enhanced raman scattering substrate. RSC Adv. 2016, 6, 74662–74669. [Google Scholar] [CrossRef]
- Zrimsek, A.B.; Chiang, N.H.; Mattei, M.; Zaleski, S.; McAnally, M.O.; Chapman, C.T.; Henry, A.I.; Schatz, G.C.; Van Duyne, R.P. Single-Molecule Chemistry with Surface- and Tip-Enhanced Raman Spectroscopy. Chem. Rev. 2017, 117, 7583–7613. [Google Scholar] [CrossRef] [PubMed]
- Ilkhani, H.; Hughes, T.; Li, J.; Zhong, C.J.; Hepel, M. Nanostructured SERS-electrochemical biosensors for testing of anticancer drug interactions with DNA. Biosens. Bioelectron. 2016, 80, 257–264. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Jiang, S.Z.; Yang, C.; Li, C.H.; Huo, Y.Y.; Liu, X.Y.; Liu, A.H.; Wei, Q.; Gao, S.S.; Gao, X.G.; et al. Gold@silver bimetal nanoparticles/pyramidal silicon 3D substrate with high reproducibility for high-performance SERS. Sci. Rep. 2016, 6, 25243. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Millstone, J.E.; Hurst, S.J.; Métraux, G.S.; Cutler, J.I.; Mirkin, C.A. Colloidal Gold and Silver Triangular Nanoprisms. Small 2009, 5, 646–664. [Google Scholar] [CrossRef] [PubMed]
- Wu, K.Y.; Li, T.; Schmidt, M.S.; Rindzevicius, T.; Boisen, A.; Ndoni, S. Gold Nanoparticles Sliding on Recyclable Nanohoodoos-Engineered for Surface-Enhanced Raman Spectroscopy. Adv. Funct. Mater. 2018, 28, 1704818. [Google Scholar] [CrossRef]
- Talley, C.E.; Jackson, J.B.; Oubre, C.; Grady, N.K.; Hollars, C.W.; Lane, S.M.; Huser, T.R.; Nordlander, P.; Halas, N.J. Surface-Enhanced Raman Scattering from Individual Au Nanoparticles and Nanoparticle Dimer Substrates. Nano Lett. 2005, 5, 1569–1574. [Google Scholar] [CrossRef] [PubMed]
- Li, W.Y.; Camargo Pedro, H.C.; Lu, X.M.; Xia, Y.N. Dimers of Silver Nanospheres: Facile Synthesis and Their Use as Hot Spots for Surface-Enhanced Raman Scattering. Nano Lett. 2009, 9, 485–490. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, G.; Wang, Y.; Yang, M.X.; Xu, J.; Goh, S.J.; Pan, M.; Chen, H.Y. Measuring Ensemble-Averaged Surface-Enhanced Raman Scattering in the Hotspots of Colloidal Nanoparticle Dimers and Trimers. J. Am. Chem. Soc. 2010, 132, 3644–3645. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Wang, H.; Chen, G.; Wang, Y.; Feng, Y.H.; Teo, W.S.; Wu, T.; Chen, H.Y. Hotspot-Induced Transformation of Surface-Enhanced Raman Scattering Fingerprints. ACS Nano 2010, 4, 3087–3094. [Google Scholar] [CrossRef] [PubMed]
- Li, S.Z.; Pedano, M.L.; Chang, S.H.; Mirkin, C.A.; Schatz, G.C. Gap Structure Effects on Surface-Enhanced Raman Scattering Intensities for Gold Gapped Rods. Nano Lett. 2010, 10, 1722–1727. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.Y.; Zheng, Y.H.; Liu, X.; Lu, W.; Dai, J.Y.; Lei, D.Y.; MacFarlane, D.R. Hierarchical Porous Plasmonic Metamaterials for Reproducible Ultrasensitive Surface-Enhanced Raman Spectroscopy. Adv. Mater. 2015, 27, 1090–1096. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.K.; Yang, N.; You, T.T.; Jiang, L.; Yin, P.G. Ultra-thin Au tip structure: a novel SERS substrate for in situ observation of a p-aminothiophenol surface-catalytic reaction. RSC Adv. 2017, 7, 4541–4546. [Google Scholar] [CrossRef]
- Shi, Y.E.; Wang, W.S.; Zhan, J.H. A positively charged silver nanowire membrane for rapid on-site swabbing extraction and detection of trace inorganic explosives using a portable Raman spectrometer. Nano Res. 2016, 9, 2487. [Google Scholar] [CrossRef]
- Li, N.; Feng, L.; Teng, F.; Lu, N. Fabrication of Plasmonic Cavity Arrays for SERS Analysis. Nanotechnology 2017, 28, 185301. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Liu, C.; Cao, X.W.; Wang, Y.; Dong, J.; Qian, W.P. Determination of Carcinoembryonic Antigen by Surface-Enhanced Raman Spectroscopy Using Gold Nanobowl Arrays. Anal. Lett. 2017, 50, 982–998. [Google Scholar] [CrossRef]
- Banaee, M.G.; Crozier, K.B. Gold nanorings as substrates for surface-enhanced Raman scattering. Opt. Lett. 2010, 35, 760–762. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Liu, G.L.; Kim, J.; Mejia, Y.X.; Lee, L.P. Nanophotonic Crescent Moon Structures with Sharp Edge for Ultrasensitive Biomolecular Detection by Local Electromagnetic Field Enhancement Effect. Nano Lett. 2005, 5, 119–124. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.Y.; Ross, B.M.; Lee, L.P. Optical Properties of the Crescent-Shaped Nanohole Antenna. Nano Lett. 2009, 9, 1956–1961. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Polavarapu, L.; Mourdikoudis, S.; Pastoriza-Santos, I.; Pérez-Juste, J. Nanocrystal engineering of noble metals and metal chalcogenides: Controlling the morphology, composition and crystallinity. CrystEngComm 2015, 17, 3727–3762. [Google Scholar] [CrossRef]
- Gómez-Graña, S.; Fernández-López, C.; Polavarapu, L.; Salmon, J.B.; Leng, J.; Pastoriza-Santos, I.; Perez-Juste, J. Gold nanooctahedra with tunable size and Microfluidic-Induced 3D assembly for Highly Uniform SERS-active Supercrystals. Chem. Mater. 2015, 27, 8310–8317. [Google Scholar] [CrossRef]
- Zhang, Y.J.; Sun, H.H.; Gao, R.X.; Zhang, F.; Zhua, A.N.; Chen, L.; Wang, Y.X. Facile SERS-active chip (PS@Ag/SiO2/Ag) for the determination of HCC biomarker. Sens. Actuat. B-Chem. 2018, 272, 34–42. [Google Scholar] [CrossRef]
- Zhang, X.F.; Du, X.Z. Carbon Nanodot-Decorated Ag@SiO2 Nanoparticles for Fluorescence and Surface-Enhanced Raman Scattering Immunoassays. ACS Appl. Mater. Interfaces 2016, 8, 1033–1040. [Google Scholar] [CrossRef] [PubMed]
- Mo, A.H.; Landon, P.B.; Gomez, K.S.; Kang, H.; Lee, J.; Zhang, C.; Janetanakit, W.; Sant, V.; Lu, Y.; Colburn, D.A.; et al. Magnetically-responsive silica-gold nanobowls for targeted delivery and SERS-based sensing. Nanoscale 2016, 8, 11840–11850. [Google Scholar] [CrossRef] [PubMed]
- Li, J.F.; Zhang, Y.J.; Ding, S.Y.; Panneerselvam, R.; Tian, Z.Q. Core–Shell Nanoparticle-Enhanced Raman Spectroscopy. Chem. Rev. 2017, 117, 5002–5069. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.X.; Zhao, X.Y.; Chen, L.; Chen, S.; Wei, M.; Gao, M.; Zhao, Y.; Wang, C.; Qu, X.; Zhang, Y.Y.; et al. Ordered Nanocap Array Composed of SiO2-Isolated Ag Islands as SERS Platform. Langmuir 2014, 30, 15285–15291. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Sun, H.H.; Zhao, Y.; Gao, R.X.; Wang, Y.X.; Liu, Y.; Zhang, Y.J.; Hua, Z.; Yang, J.H. Iron Layer-Dependent Surface-Enhanced Raman Scattering of Hierarchical Nanocap Arrays. Appl. Surf. Sci. 2017, 423, 1124–1133. [Google Scholar] [CrossRef]
- Zhang, F.; Wang, Y.X.; Zhang, Y.J.; Chen, L.; Liu, Y.; Yang, J.H. Ag Nanotwin-Assisted Grain Growth-Induced by Stress in SiO2/Ag/SiO2 Nanocap Arrays. Nanomaterials 2018, 8, 436. [Google Scholar] [CrossRef] [PubMed]
- Muehlethaler, C.; Considine, C.R.; Menon, V.; Lin, W.C.; Lee, Y.H.; Lombard, J.R. Ultrahigh Raman Enhancement on Monolayer MoS2. ACS Photonics 2016, 3, 1164–1169. [Google Scholar] [CrossRef]
- Wang, Y.; Ji, W.; Sui, H.M.; Kitahama, Y.; Ruan, W.D.; Ozaki, Y.; Zhao, B. Exploring the Effect of Intermolecular H-Bonding: A Study on Charge-Transfer Contribution to Surface-Enhanced Raman Scattering of p-Mercaptobenzoic Acid. J. Phys. Chem. C 2014, 118, 10191–10197. [Google Scholar] [CrossRef]
- Guerrini, L.; Pazos, E.; Penas, C.; Vazquez, M.E.; Mascareñas, J.L.; Alvarez-Puebla, R.A. Highly Sensitive SERS Quantification of the Oncogenic Protein c-Jun in Cellular Extracts. J. Am. Chem. Soc. 2013, 135, 10314–10317. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.Y.; Wen, J.H.; Zhang, M.N.; Wang, D.H.; Wang, Y.X.; Chen, L.; Zhang, Y.J.; Yang, J.H.; Du, Y.W. Design of Hybrid Nanostructural Arrays to Manipulate SERS-Active Substrates by Nanosphere Lithography. ACS Appl. Mater. Interfaces 2017, 9, 7710–7716. [Google Scholar] [CrossRef] [PubMed]


© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, F.; Guo, S.; Liu, Y.; Chen, L.; Wang, Y.; Gao, R.; Zhu, A.; Zhang, X.; Zhang, Y. Controlling the 3D Electromagnetic Coupling in Co-Sputtered Ag–SiO2 Nanomace Arrays by Lateral Sizes. Nanomaterials 2018, 8, 493. https://doi.org/10.3390/nano8070493
Zhang F, Guo S, Liu Y, Chen L, Wang Y, Gao R, Zhu A, Zhang X, Zhang Y. Controlling the 3D Electromagnetic Coupling in Co-Sputtered Ag–SiO2 Nanomace Arrays by Lateral Sizes. Nanomaterials. 2018; 8(7):493. https://doi.org/10.3390/nano8070493
Chicago/Turabian StyleZhang, Fan, Shuang Guo, Yang Liu, Lei Chen, Yaxin Wang, Renxian Gao, Aonan Zhu, Xiaolong Zhang, and Yongjun Zhang. 2018. "Controlling the 3D Electromagnetic Coupling in Co-Sputtered Ag–SiO2 Nanomace Arrays by Lateral Sizes" Nanomaterials 8, no. 7: 493. https://doi.org/10.3390/nano8070493
APA StyleZhang, F., Guo, S., Liu, Y., Chen, L., Wang, Y., Gao, R., Zhu, A., Zhang, X., & Zhang, Y. (2018). Controlling the 3D Electromagnetic Coupling in Co-Sputtered Ag–SiO2 Nanomace Arrays by Lateral Sizes. Nanomaterials, 8(7), 493. https://doi.org/10.3390/nano8070493




