Incorporation of Levodopa into Biopolymer Coatings Based on Carboxylated Carbon Nanotubes for pH-Dependent Sustained Release Drug Delivery
Abstract
:1. Introduction
2. Materials and Methods
2.1. Instruments and Measurements
2.2. Preparation of Drug Loading onto Carbon Nanotubes
2.3. Preparation of Biopolymer Wrapping onto Drug-Loaded Carbon Nanotubes
2.4. In Vitro Drug Release of LD
2.5. Cell Culture
2.6. Cell Viability Assay
2.7. Statistical Analysis
3. Results
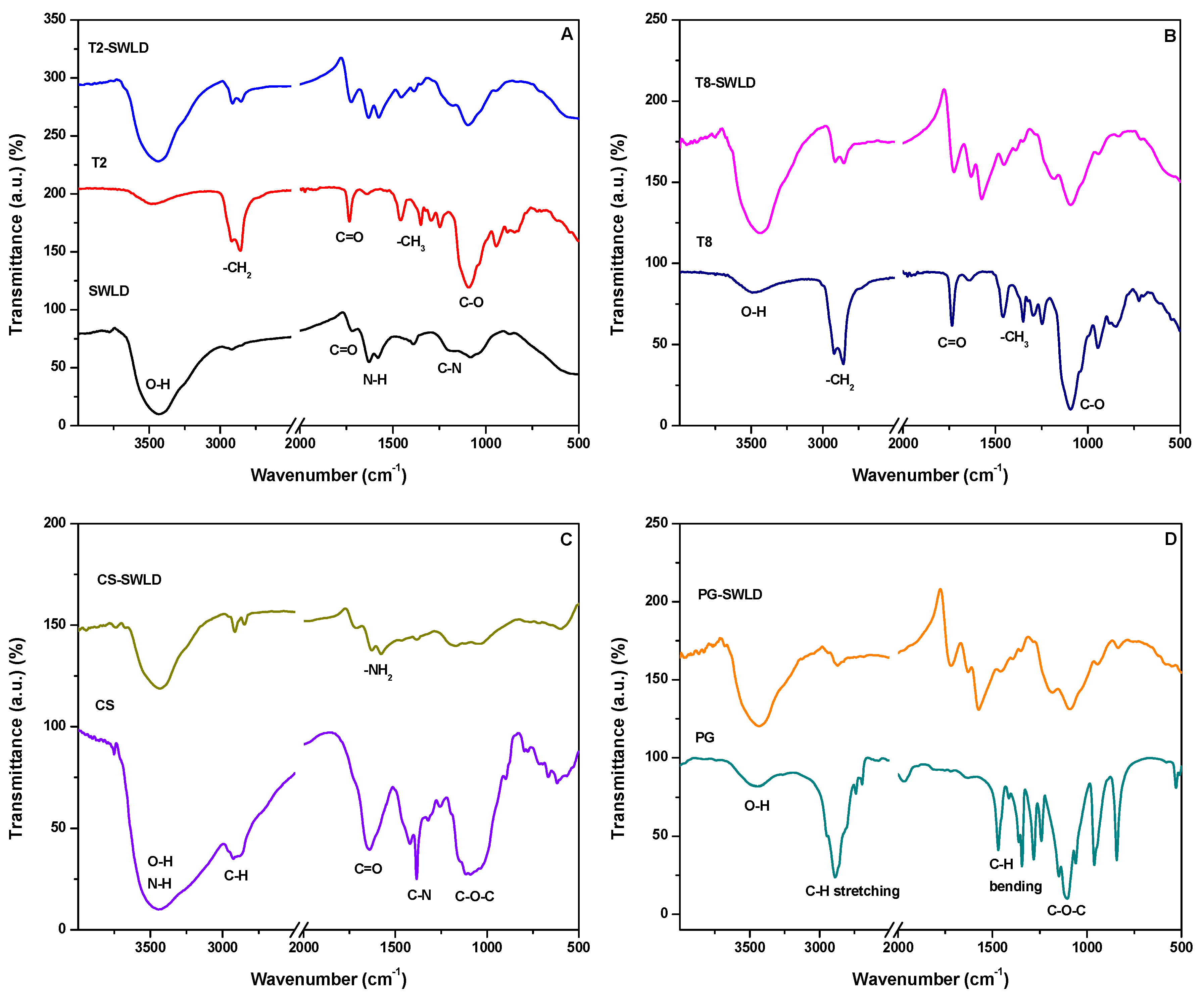
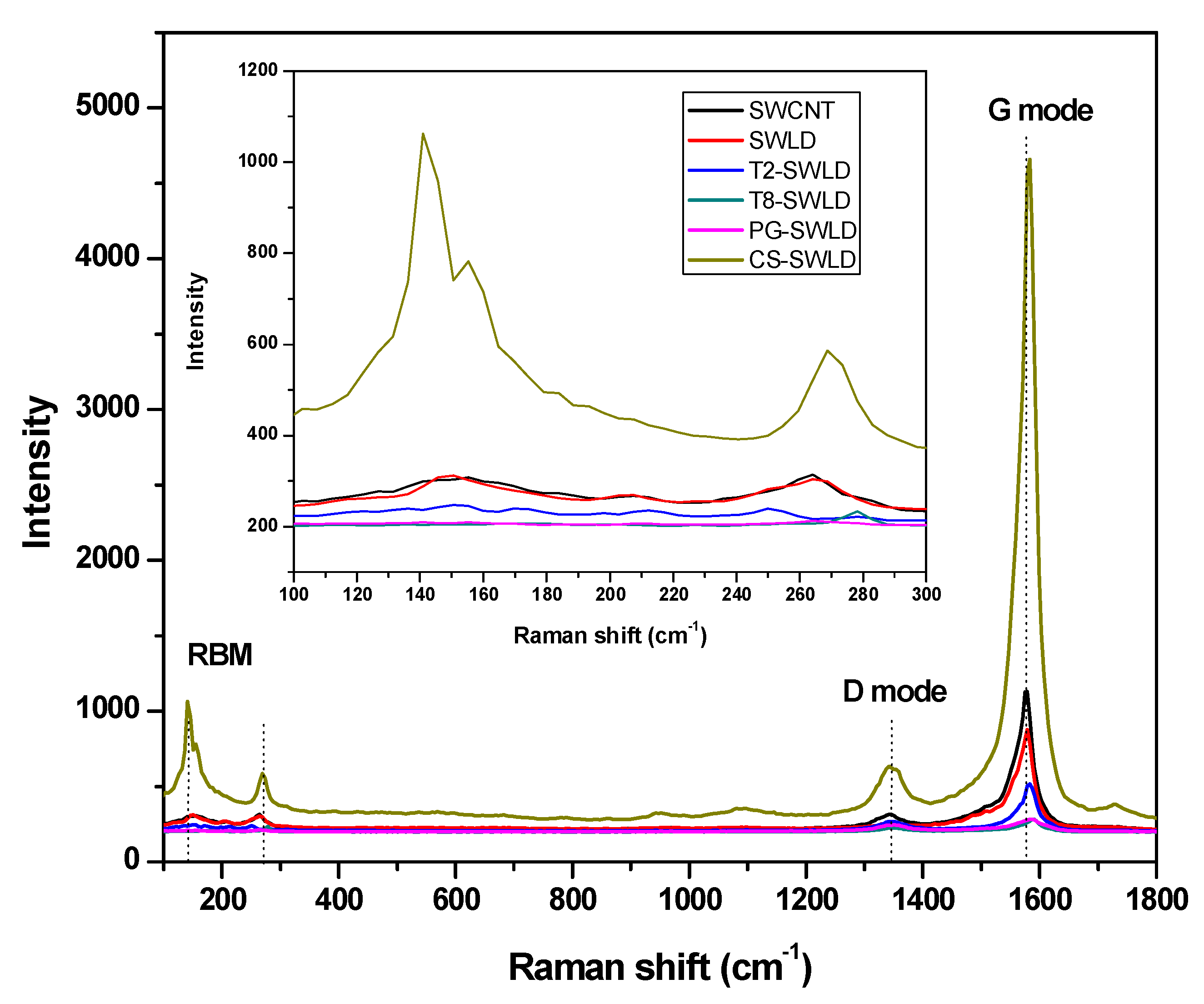
3.1. Characterization
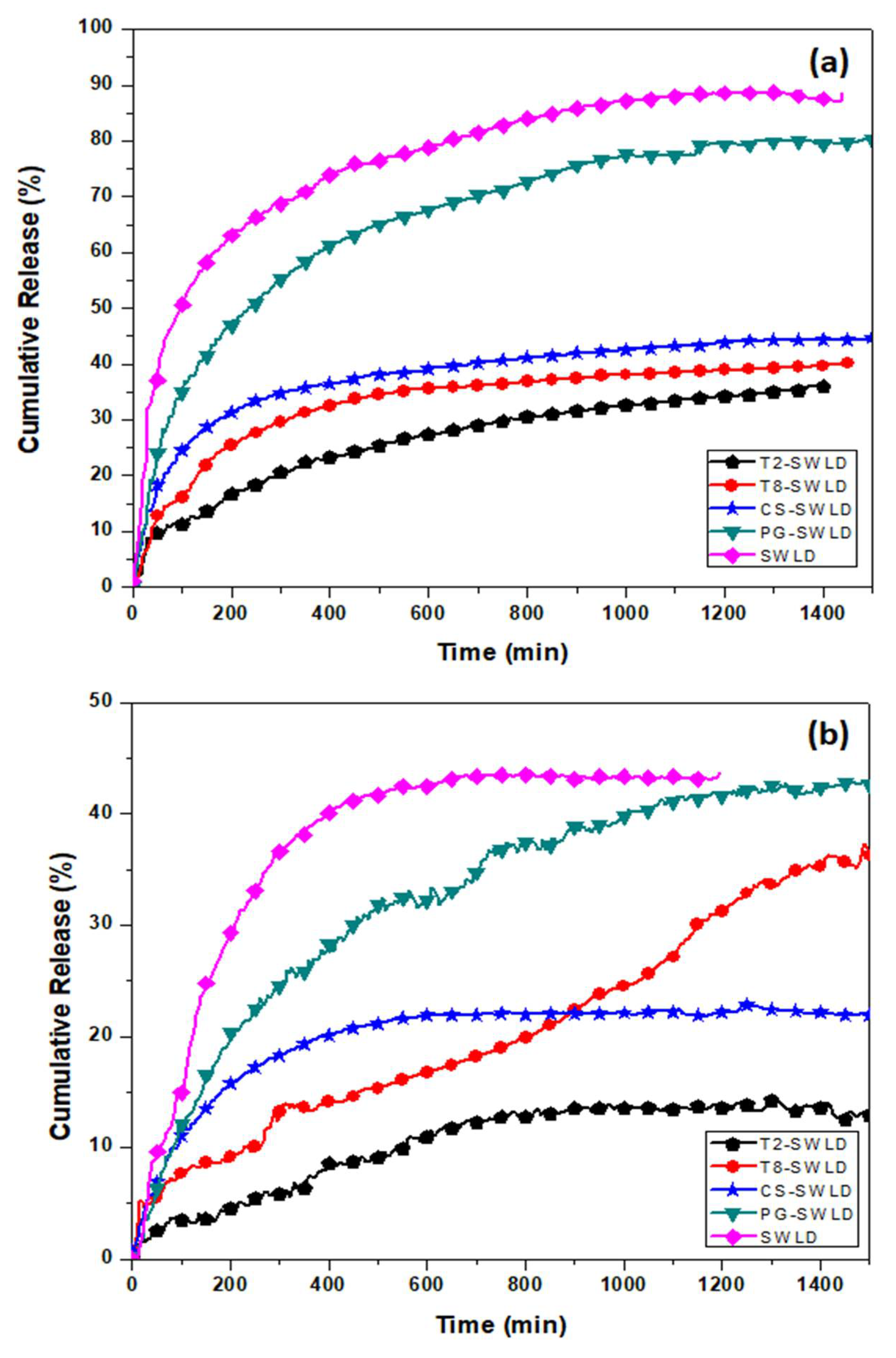
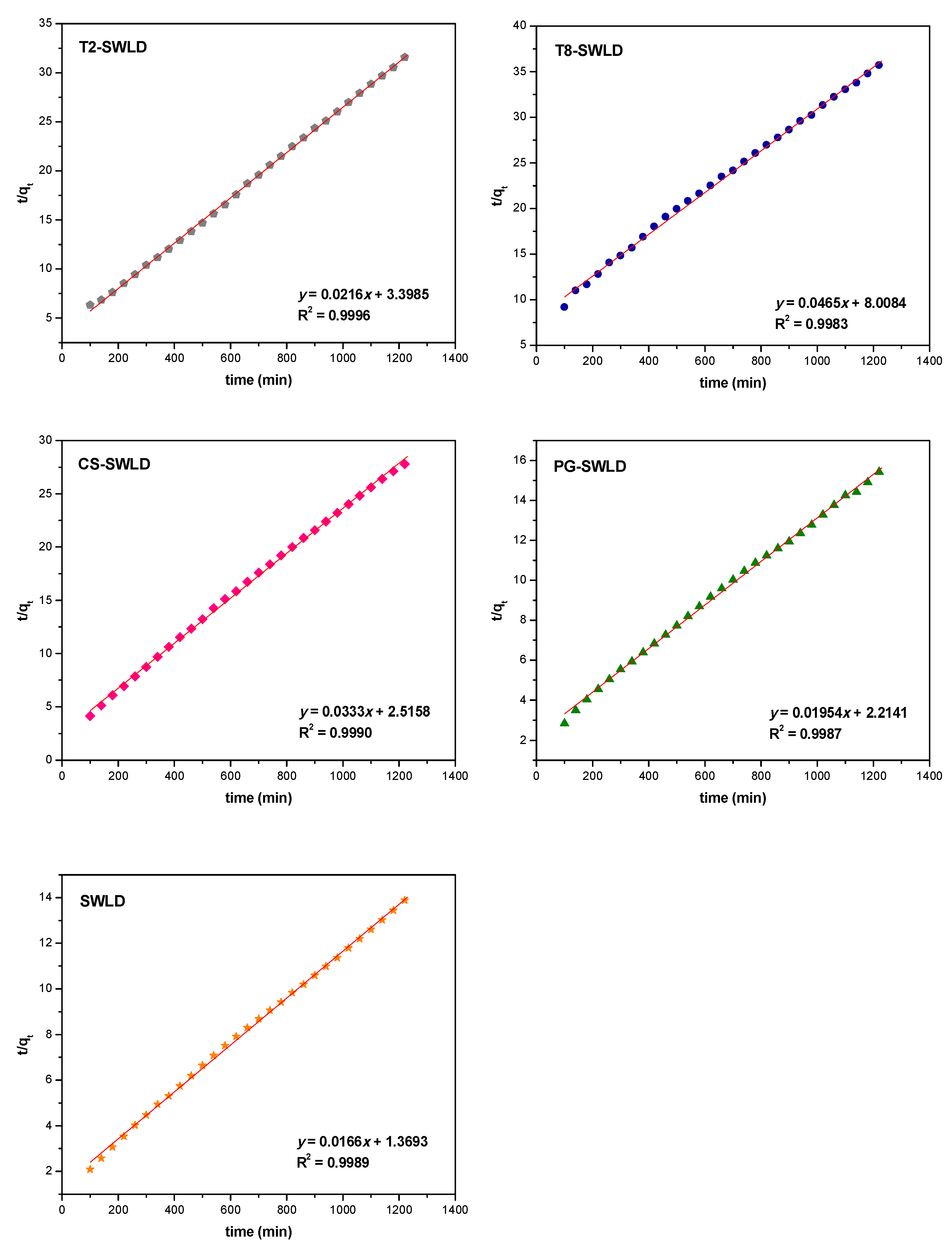
3.2. In Vitro Drug Release of LD
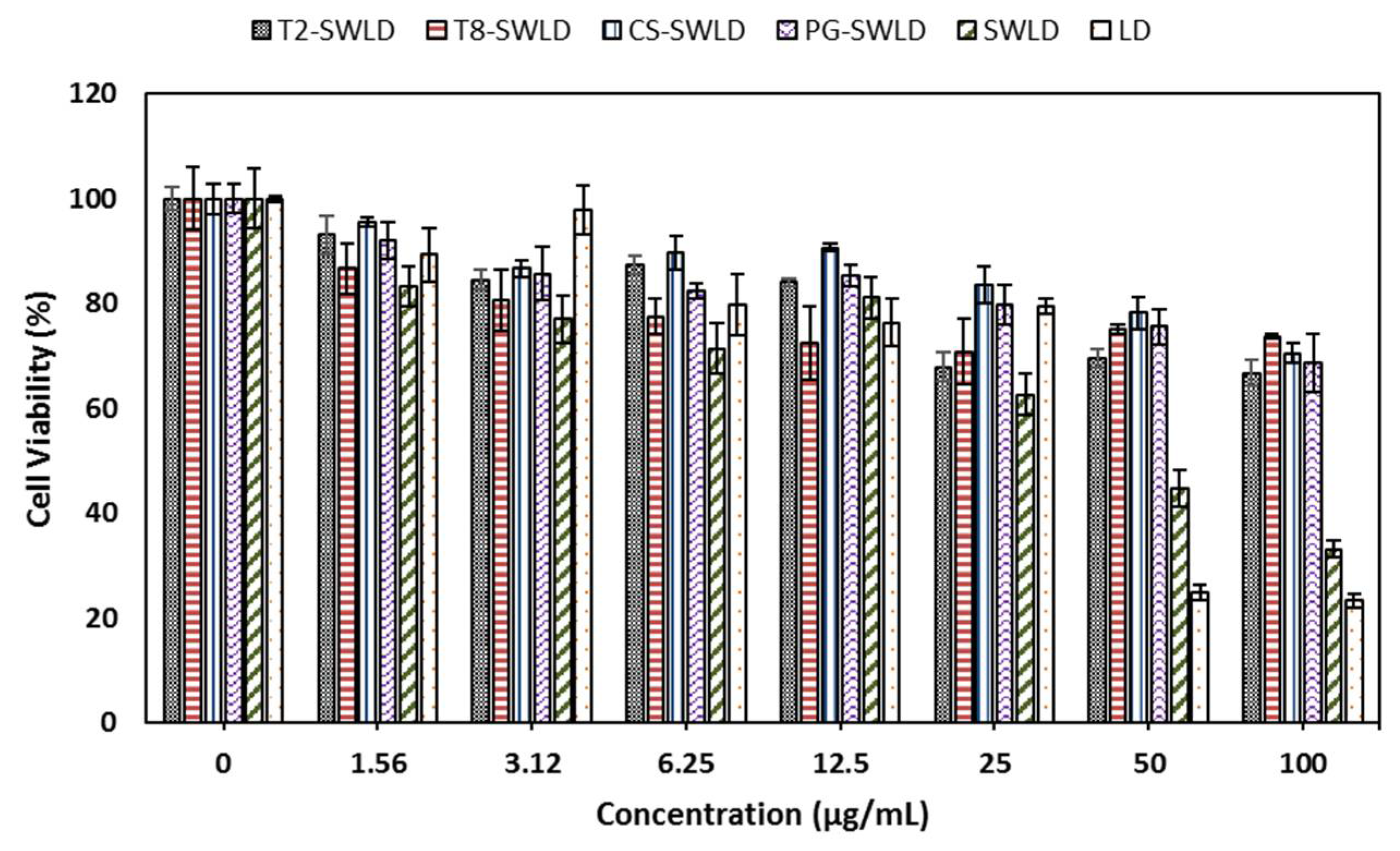
3.3. In Vitro Cytotoxicity Assay
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- De Rijk, M.C.; Launer, L.J.; Berger, K.; Breteler, M.M.; Dartigues, J.F.; Baldereschi, M.; Fratiglioni, L.; Lobo, A.; Martinez-Lage, J.; Trenkwalder, C.; et al. Prevalence of Parkinson’s disease in Europe: A collaborative study of population-based cohorts. Neurologic diseases in the elderly research group. Neurology 2000, 54, S21–23. [Google Scholar] [PubMed]
- LeWitt, P.A. Levodopa therapeutics for Parkinson’s disease: New developments. Parkinsonism Relat. Disord. 2009, 15, S31–S34. [Google Scholar] [CrossRef]
- Brodell, D.W.; Stanford, N.T.; Jacobson, C.E.; Schmidt, P.; Okun, M.S. Carbidopa/levodopa dose elevation and safety concerns in Parkinson’s patients: A cross-sectional and cohort design. BMJ Open 2012, 2, e001971. [Google Scholar] [CrossRef] [PubMed]
- Fahn, S. Does levodopa slow or hasten the rate of progression of Parkinson’s disease? J. Neurol. 2005, 252, iv37–iv42. [Google Scholar] [CrossRef] [PubMed]
- Kura, A.U.; Hussein-Al-Ali, S.H.; Hussein, M.Z.; Fakurazi, S. Preparation of tween 80-Zn/Al-levodopa-layered double hydroxides nanocomposite for drug delivery system. Sci. World J. 2014, 104246, 10. [Google Scholar] [CrossRef] [PubMed]
- Mahringer, A.; Ott, M.; Reimold, I.; Reichel, V.; Fricker, G. The ABC of the blood-brain barrier—Regulation of drug efflux pumps. Curr. Pharm. Des. 2011, 17, 2762–2770. [Google Scholar] [CrossRef] [PubMed]
- Haddad, F.; Sawalha, M.; Khawaja, Y.; Najjar, A.; Karaman, R. Dopamine and levodopa prodrugs for the treatment of Parkinson’s disease. Molecules 2018, 23, 40. [Google Scholar] [CrossRef] [PubMed]
- Lawther, B.K.; Kumar, S.; Krovvidi, H. Blood-brain barrier. BJA Educ. 2011, 11, 128–132. [Google Scholar] [CrossRef]
- Dwibhashyam, V.S.N.M.; Nagappa, A.N. Strategies for enhanced drug delivery to the central nervous system. Indian J. Pharm. Sci. 2008, 70, 145–153. [Google Scholar] [PubMed]
- Wilczewska, A.Z.; Niemirowicz, K.; Markiewicz, K.H.; Car, H. Nanoparticles as drug delivery systems. Pharmacol. Rep. 2012, 64, 1020–1037. [Google Scholar] [CrossRef]
- Park, S.; Shin, J.; Lee, J.; Cha, M. Manipulation of NIH3T3 cells with functionalized single-walled carbon nanotubes under a magnetic field. Mater. Lett. 2012, 68, 378–381. [Google Scholar] [CrossRef]
- Abbott, N.J. Blood-brain barrier structure and function and the challenges for CNS drug delivery. J. Inherit. Metab. Dis. 2013, 36, 437–449. [Google Scholar] [CrossRef] [PubMed]
- Bardi, G.; Nunes, A.; Gherardini, L.; Bates, K.; Al-Jamal, K.T.; Gaillard, C.; Prato, M.; Bianco, A.; Pizzorusso, T.; Kostarelos, K. Functionalized carbon nanotubes in the brain: Cellular internalization and neuroinflammatory response. PLoS ONE 2013, 8, e80964. [Google Scholar] [CrossRef] [PubMed]
- Kafa, H.; Wang, J.T.; Rubio, N.; Venner, K.; Anderson, G.; Pach, E.; Ballesteros, B.; Preston, J.E.; Abbott, N.J.; Al-Jamal, K.T. The interaction of carbon nanotubes with an in vitro blood-brain barrier model and mouse brain in vivo. Biomaterials 2015, 53, 437–452. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, L.; Alizadeh, D.; Badie, B. Carbon nanotube uptake and toxicity in the brain. Methods Mol. Biol. 2010, 625, 55–65. [Google Scholar] [PubMed]
- Serpell, C.J.; Kostarelos, K.; Davis, B.G. Can carbon nanotubes deliver on their promise in biology? Harnessing unique properties for unparalleled applications. ACS Cent. Sci. 2016, 2, 190–200. [Google Scholar] [CrossRef] [PubMed]
- Vitale, F.; Summerson, S.R.; Aazhang, B.; Kemere, C.; Pasquali, M. Neural stimulation and recording with bidirectional, soft carbon nanotube fiber microelectrodes. ACS Nano 2015, 9, 4465–4474. [Google Scholar] [CrossRef] [PubMed]
- Tan, J.M.; Arulselvan, P.; Fakurazi, S.; Ithnin, H.; Hussein, M.Z. A review on characterizations and biocompatibility of functionalized carbon nanotubes in drug delivery design. J. Nanomater. 2014, 917024. [Google Scholar] [CrossRef]
- Villa, C.H.; Dao, T.; Ahearn, I.; Fehrenbacher, N.; Casey, E.; Rey, D.A.; Korontsvit, T.; Zakhaleva, V.; Batt, C.A.; Philips, M.R.; et al. Single-walled carbon nanotubes delivery peptide antigen into dendritic cells and enhance IgG responses to tumor-associated antigens. ACS Nano 2011, 5, 5300–5311. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.P.; Lin, I.J.; Chen, C.C.; Hsu, Y.C.; Chang, C.C.; Lee, M.J. Delivery of small interfering RNAs in human cervical cancer cells by polyethylenimine-functionalized carbon nanotubes. Nanoscale Res. Lett. 2013, 8, 267–277. [Google Scholar] [CrossRef] [PubMed]
- Yu, B.; Tan, L.; Zheng, R.; Tan, H.; Zheng, L. Targeted delivery and controlled release of paclitaxel for the treatment of lung cancer using single-walled carbon nanotubes. Mater. Sci. Eng. C Mater. Biol. Appl. 2016, 68, 579–584. [Google Scholar] [CrossRef] [PubMed]
- Tominaga, M.; Watanabe, N.; Yatsugi, Y. In situ Raman spectroelectrochemical study of potential-induced molecular encapsulation of β-carotene inside single-walled carbon nanotubes. J. Electroanal. Chem. 2017, 800, 156–161. [Google Scholar] [CrossRef]
- Porter, A.E.; Gass, M.; Muller, K.; Skepper, J.N.; Midgley, P.A.; Welland, M. Direct imaging of single-walled carbon nanotubes in cells. Nat. Nanotechnol. 2007, 2, 713–717. [Google Scholar] [CrossRef] [PubMed]
- López, M.L.; Bernal, E.; Moyá, M.L.; Sanchez, F.; Cornejo, P.L. Study of ionic surfactants interactions with carboxylated single-walled carbon nanotubes by using ion-selective electrodes. Electrochem. Commun. 2016, 67, 31–34. [Google Scholar] [CrossRef]
- Arnold, M.S.; Green, A.A.; Hulvat, J.F.; Stupp, S.I.; Hersam, M.C. Sorting carbon nanotubes by electronic structure using density differentiation. Nat. Nanotechnol. 2006, 1, 60–65. [Google Scholar] [CrossRef] [PubMed]
- Jos, A.; Pichardo, S.; Puerto, M.; Sánchez, E.; Grilo, A.; Cameán, A.M. Cytotoxicity of carboxylic acid functionalized single wall carbon nanotubes on the human intestinal cell line Caco-2. Toxicol. In Vitro 2009, 23, 1491–1496. [Google Scholar] [CrossRef] [PubMed]
- Vinardell, M.P.; Infante, M.R. The relationship between the chain length of non-ionic surfactants and their haemolytic action on human erythrocytes. Comp. Biochem. Physiol. C 1999, 124, 117–120. [Google Scholar] [PubMed]
- Hoffman, W.L.; Jump, A.A. Tween 20 removes antibodies and other proteins from nitrocellulose. J. Immunol. Methods 1986, 94, 191–196. [Google Scholar] [CrossRef]
- Gelperina, S.; Maksimenko, O.; Khalansky, A.; Vanchugova, L.; Shipulo, E.; Abbasova, K.; Berdiev, R.; Wohlfart, S.; Chepurnova, N.; Kreuter, J. Drug delivery to the brain using surfactant-coated poly(lactide-co-glycolide) nanoparticles: Influence of the formulation parameters. Eur. J. Pharm. Biopharm. 2010, 74, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Alyautdin, R.N.; Petrov, V.E.; Langer, K.; Berthold, A.; Kharkevich, D.A.; Kreuter, J. Delivery of loperamide across the blood-brain barrier with polysorbate 80-coated poly-butylcyanoacrylate nanoparticles. Pharm. Res. 1997, 14, 325–328. [Google Scholar] [CrossRef] [PubMed]
- Barnabas, W.; Malay, K.S.; Kumaraswamy, S.; Sampath, K.K.P.; Paramakrishnan, N.; Suresh, B. Targeted delivery of tacrine into the brain with polysorbate 80-coated poly(n-butylcyanoacrylate) nanoparticles. Eur. J. Pharm. Biopharm. 2008, 70, 75–84. [Google Scholar]
- Alyautdin, R.N.; Tezikov, E.B.; Ramge, P.; Kharkevich, D.A.; Begley, D.J.; Kreuter, J. Significant entry of tubocurarine into the brain of rats by absorption to polysorbate 80-coated polybutylcyanoacrylate nanoparticles: An in situ brain perfusion study. J. Microencapsul. 1998, 15, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Alyautdin, R.; Gothier, D.; Petrov, V.; Kharkevich, D.; Kreuter, J. Analgesic activity of the hexapeptide dalargin adsorbed on the surface of polysorbate 80-coated poly(butyl cyanoacrylate) nanoparticles. Eur. J. Pharm. Biopharm. 1995, 41, 44–48. [Google Scholar]
- Kerwin, B.A. Polysorbates 20 and 80 used in the formulation of protein biotherapeutics: Structure and degradation pathways. J. Pharm. Sci. 2008, 97, 2924–2935. [Google Scholar] [CrossRef] [PubMed]
- Mohammadi, Z.A.; Aghamiri, S.F.; Zarrabi, A.; Talaie, M.R. A comparative study on non-covalent functionalisation of carbon nanotubes by chitosan and its derivatives for delivery of doxorubicin. Chem. Phys. Lett. 2015, 642, 22–28. [Google Scholar] [CrossRef]
- Li, C.; Yang, K.; Zhang, Y.; Tang, H.; Yan, F.; Tan, L.; Xie, Q.; Yao, S. Highly biocompatible multi-walled carbon nanotube-chitosan nanoparticle hybrids as protein carriers. Acta Biomater. 2011, 7, 3070–3077. [Google Scholar] [CrossRef] [PubMed]
- Saraiva, C.; Praca, C.; Ferreira, R.; Santos, T.; Ferreira, L.; Bernardino, L. Nanoparticle-mediated brain drug delivery: Overcoming blood-brain barrier to treat neurodegenerative diseases. J. Controll. Release 2016, 235, 34–47. [Google Scholar] [CrossRef] [PubMed]
- Leuner, C.; Dressman, J. Improving drug solubility for oral delivery using solid dispersions. Eur. J. Pharm. Biopharm. 2000, 50, 47–60. [Google Scholar] [CrossRef]
- Moghimi, S.M.; Szebeni, J. Stealth liposomes and long circulating nanoparticles: Critical issues in pharmacokinetics, opsonisation and protein-binding properties. Prog. Lipid Res. 2003, 42, 463–478. [Google Scholar] [CrossRef]
- Yusuf, B.; Gopurappilly, R.; Dadheech, N.; Gupta, S.; Bhonde, R.; Pal, R. Embryonic fibroblasts represent a connecting link between mesenchymal and embryonic stem cells. Dev. Growth Differ. 2013, 55, 330–340. [Google Scholar] [CrossRef] [PubMed]
- Tan, J.M.; Foo, J.B.; Fakurazi, S.; Hussein, M.Z. Release behaviour and toxicity evaluation of levodopa from carboxylated single-walled carbon nanotubes. Beilstein J. Nanotechnol. 2015, 6, 243–253. [Google Scholar] [CrossRef] [PubMed]
- Naumenko, A.P.; Korniyenko, N.E.; Yashchuk, V.M.; Singamaneni, S.; Bliznyuk, V.N. Raman Spectroscopy for Nanomaterials Characterization; Springer: Berlin/Heidelberg, Germany, 2012; pp. 137–165. [Google Scholar]
- Sneddon, G.; Ganin, A.Y.; Yiu, H.H.P. Sustainable CO2 adsorbents prepared by coating chitosan onto mesoporous silicas for large-scale carbon capture technology. Energy Technol. 2015, 3, 249–258. [Google Scholar] [CrossRef] [Green Version]
- Dittmer, S.; Olofsson, N.; Weis, J.E.; Nerushev, O.A.; Gromov, A.V.; Campbell, E.E.B. In situ Raman studies of single-walled carbon nanotubes grown by local catalyst heating. Chem. Phys. Lett. 2008, 457, 206–210. [Google Scholar] [CrossRef]
- Huang, X.; Brazel, C.S. On the importance and mechanisms of burst release in matrix-controlled drug delivery systems. J. Controll. Release 2001, 73, 121–136. [Google Scholar] [CrossRef]
- Santiago, A.J.; Factor, S.A. Handbook of Parkinson’s Disease; CRC Press: Boca Raton, FL, USA, 2003; pp. 381–406. [Google Scholar]
- Bai, H.; Li, C.; Wang, X.; Shi, G. A pH-sensitive graphene oxide composite hydrogel. Chem. Commun. (Camb.) 2010, 46, 2376–2378. [Google Scholar] [CrossRef] [PubMed]
- Ito, Y.; Venkatesan, N.; Hirako, N.; Sugioka, N.; Takada, K. Effect of fiber length of carbon nanotubes on the absorption of erythroprotein from rat small intestine. Int. J. Pharm. 2007, 337, 357–360. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Zhang, Y.; Yang, Y.; Sun, L.; Han, D.; Li, H.; Wang, C. Pharmacological and toxicological target organelles and safe use of single-walled carbon nanotubes as drug carriers in treating Alzheimer disease. Nanomedicine 2010, 6, 427–441. [Google Scholar] [CrossRef] [PubMed]
- Azimirad, V.; Hosseinpour, M.; Shahabi, P.; Alimohammadi, M.; Sadighi, M.; Hatami, H. Effects of injection of carbon nanotubes on EEG and results of a behavioral test in rats. Neurophysiology 2015, 47, 198–204. [Google Scholar] [CrossRef]
- Ren, J.; Shen, S.; Wang, D.; Xi, Z.; Guo, L.; Pang, Z.; Qian, Y.; Sun, X.; Jiang, X. The targeted delivery of anticancer drugs to brain glioma by PEGylated oxidized multi-walled carbon nanotubes modified with angiopep-2. Biomaterials 2012, 33, 3324–3333. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Davis, C.; Cai, W.; He, L.; Chen, X.; Dai, H. Circulation and long-term fate of functionalized, biocompatible single-walled carbon nanotubes in mice probed by Raman spectroscopy. Proc. Natl. Acad. Sci. USA 2008, 105, 1410–1415. [Google Scholar] [CrossRef] [PubMed]
- Sasahara, K.; Nitanai, T.; Habara, T.; Kojima, T.; Kawahara, Y.; Morioka, T.; Nakajima, E. Dosage form design for improvement of levodopa IV: Possible causes of low bioavailability of oral levodopa in dogs. J. Pharm. Sci. 1981, 70, 730–733. [Google Scholar] [CrossRef] [PubMed]
- Benyettou, F.; Hardouin, J.; Lecouvey, M.; Jouni, H.; Mottle, L. PEGylated versus non-PEGylated γFe2O3@alendronate nanoparticles. J. Bioanal. Biomed. 2012, 4, 039–045. [Google Scholar] [CrossRef]
- Muzzalupo, R.; Tavano, L.; Cassano, R.; Trombino, S.; Cilea, A.; Picci, N. Colon-specific devices based on methacrylic functionalized Tween monomer networks: Swelling studies and in vitro drug release. Eur. Polym. J. 2010, 46, 209–216. [Google Scholar] [CrossRef]
- Kumar, K.V.; Khaddour, I.A.; Gupta, V.K. A pseudo second-order kinetic expression for dissolution kinetic profiles of solids in solutions. Ind. Eng. Chem. Res. 2010, 49, 7257–7262. [Google Scholar] [CrossRef]
- Ho, Y.S.; McKay, G. Pseudo-second order model for sorption process. Process Biochem. 1999, 34, 451–465. [Google Scholar] [CrossRef]
- Barahuie, F.; Saifullah, B.; Dorniani, D.; Fakurazi, S.; Karthivashan, G.; Hussein, M.Z.; Elfghi, F.M. Graphene oxide as a nanocarrier for controlled release and targeted delivery of an anticancer active agent, chlorogenic acid. Mater. Sci. Eng. C Mater. Biol. Appl. 2017, 74, 177–185. [Google Scholar] [CrossRef] [PubMed]
- Mannerström, M.; Zou, J.; Toimela, T.; Pyykkö, I.; Heinonen, T. The applicability of conventional cytotoxicity assays to predict safety/toxicity of mesoporous silica nanoparticles, silver and gold nanoparticles and multi-walled carbon nanotubes. Toxicol. In Vitro 2016, 37, 113–120. [Google Scholar] [CrossRef] [PubMed]
- Skowroń, J.; Zapór, L. Cytotoxicity of resorcinol under short- and long-term exposure in vitro. Int. J. Occup. Saf. Ergon. (JOSE) 2004, 10, 147–156. [Google Scholar] [CrossRef] [PubMed]
- Kura, A.U.; Hussein Al Ali, S.H.; Hussein, M.Z.; Fakurazi, S.; Arulselvan, P. Development of a controlled-release anti-parkinsonian nanodelivery system using levodopa as the active agent. Int. J. Nanomed. 2013, 8, 1103–1110. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wörle-Knirsch, J.M.; Pulskamp, K.; Krug, H.F. Oops they did it again! Carbon nanotubes hoax scientists in viability assays. Nano Lett. 2006, 6, 1261–1268. [Google Scholar] [CrossRef] [PubMed]
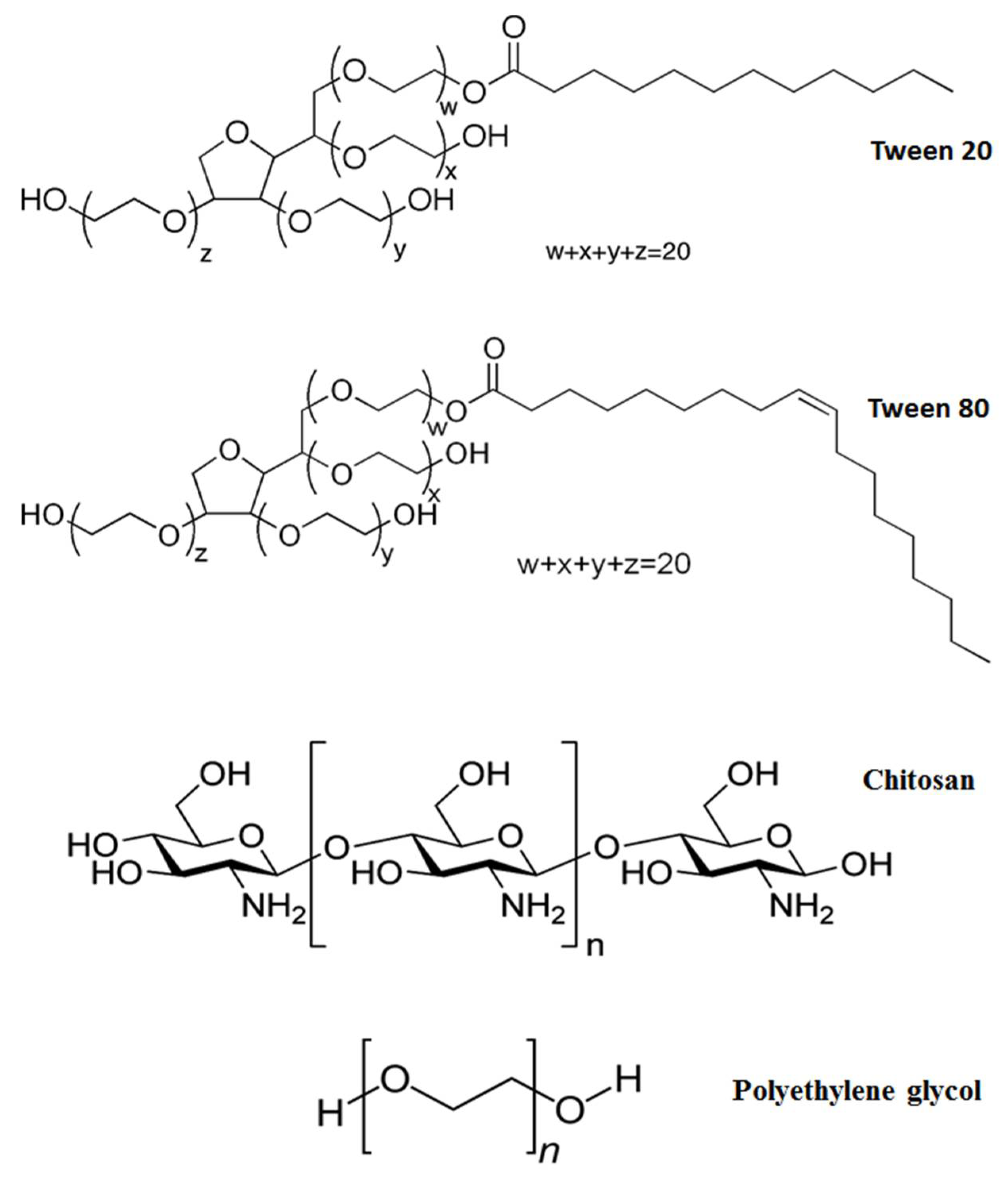

| Type | OD (nm) | Length (μm) | Functional Group Content (wt %) | Production Method |
|---|---|---|---|---|
| Short SWCNT | 1–2 | 1–3 | 2.73 | Chemical vapor deposition |
| Samples | D Mode (cm−1) | G Mode (cm−1) | ID/IG |
|---|---|---|---|
| SWCNT (without coating and LD) | 1346 | 1579 | 0.279 |
| SWLD (without coating) | 1350 | 1579 | 0.292 |
| T2-SWLD | 1342 | 1583 | 0.520 |
| T8-SWLD | 1350 | 1591 | 0.804 |
| CS-SWLD | 1342 | 1583 | 0.136 |
| PG-SWLD | 1350 | 1587 | 0.863 |
| Formulation | Correlation Coefficient, R2 | ||||
|---|---|---|---|---|---|
| Zeroth order (qt = q0 + k0t) | Pseudo-first order (ln(qe − qt) = ln qe − k1t) | Pseudo-second order () | Korsmeyer–Peppas model ( | Higuchi model () | |
| PBS solution at pH 7.4 | |||||
| SWLD | 0.8980 | 0.9503 | 0.9989 | 0.9868 | 0.9651 |
| T2-SWLD | 0.7748 | 0.9847 | 0.9996 | 0.9065 | 0.8762 |
| T8-SWLD | 0.9255 | 0.9917 | 0.9983 | 0.9844 | 0.9811 |
| CS-SWLD | 0.8777 | 0.9735 | 0.9990 | 0.9755 | 0.9485 |
| PG-SWLD | 0.8960 | 0.9899 | 0.9987 | 0.9812 | 0.9637 |
| PBS solution at pH 4.8 | |||||
| SWLD | 0.5839 | 0.9944 | 0.9953 | 0.7854 | 0.7134 |
| T2-SWLD | 0.9071 | 0.9552 | 0.9123 | 0.9650 | 0.9487 |
| T8-SWLD | 0.9802 | 0.9480 | 0.8508 | 0.9656 | 0.9427 |
| CS-SWLD | 0.6551 | 0.9392 | 0.9971 | 0.8516 | 0.7756 |
| PG-SWLD | 0.9249 | 0.9818 | 0.9953 | 0.9786 | 0.9761 |
| Concentration (μg/mL) | Cell Viability (%) | |||||
|---|---|---|---|---|---|---|
| T2-SWLD | T8-SWLD | CS-SWLD | PG-SWLD | SWLD | LD | |
| 1.56 | 93.1 ± 3.6 a | 86.7 ± 4.9 a | 95.6 ± 0.8 a | 92.1 ± 3.5 a | 83.3 ± 3.8 a | 89.4 ± 5.1 b |
| 3.12 | 84.5 ± 1.9 c | 80.7 ± 5.9 b | 86.7 ± 1.6 b,c | 85.7 ± 5.1 b | 77.1 ± 4.5 a,b | 97.8 ± 4.7 a |
| 6.25 | 87.4 ± 1.7 b | 77.5 ± 3.3 b,c | 89.6 ± 3.2 b | 82.4 ± 1.4 b,c | 71.5 ± 4.8 b,c | 79.8 ± 5.8 c |
| 12.50 | 84.1 ± 0.8 c | 72.5 ± 6.9 c,d | 90.7 ± 0.8 a,b | 85.2 ± 2.0 b | 81.1 ± 3.9 a | 76.4 ± 4.5 c |
| 25.00 | 67.9 ± 2.7 d,e | 70.8 ± 6.3 d | 83.6 ± 3.5 c | 79.9 ±3.7 c | 62.7 ± 3.9 c | 79.5 ± 1.5 c |
| 50.00 | 69.5 ± 1.8 d | 75.1 ± 0.9 c,d | 78.3 ± 3.0 d | 75.6 ± 3.3 d | 44.6 ± 3.5 d | 24.9 ± 1.6 d |
| 100.00 | 66.8 ± 2.6 e | 73.7 ± 0.5 c,d | 70.5 ± 1.9 e | 68.6 ± 5.5 e | 33.1 ± 1.6 e | 23.3 ± 1.3 d |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tan, J.M.; Saifullah, B.; Kura, A.U.; Fakurazi, S.; Hussein, M.Z. Incorporation of Levodopa into Biopolymer Coatings Based on Carboxylated Carbon Nanotubes for pH-Dependent Sustained Release Drug Delivery. Nanomaterials 2018, 8, 389. https://doi.org/10.3390/nano8060389
Tan JM, Saifullah B, Kura AU, Fakurazi S, Hussein MZ. Incorporation of Levodopa into Biopolymer Coatings Based on Carboxylated Carbon Nanotubes for pH-Dependent Sustained Release Drug Delivery. Nanomaterials. 2018; 8(6):389. https://doi.org/10.3390/nano8060389
Chicago/Turabian StyleTan, Julia Meihua, Bullo Saifullah, Aminu Umar Kura, Sharida Fakurazi, and Mohd Zobir Hussein. 2018. "Incorporation of Levodopa into Biopolymer Coatings Based on Carboxylated Carbon Nanotubes for pH-Dependent Sustained Release Drug Delivery" Nanomaterials 8, no. 6: 389. https://doi.org/10.3390/nano8060389





