The Effects of Carbon Dots on Immune System Biomarkers, Using the Murine Macrophage Cell Line RAW 264.7 and Human Whole Blood Cell Cultures
Abstract
:1. Introduction
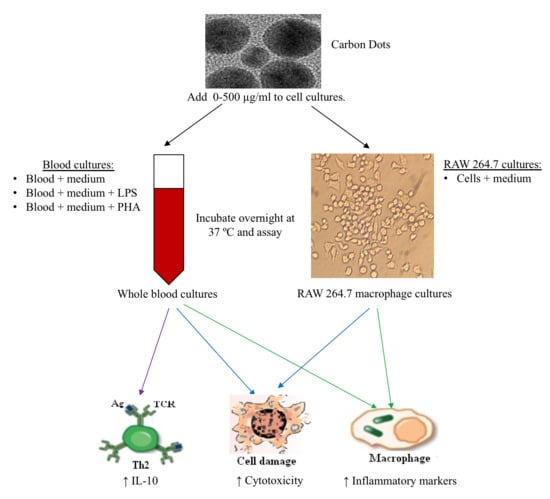
2. Materials and Methods
2.1. Synthesis and Characterization of Carbon Dots (CDs)
2.2. Preparation of CD Stock Solutions
2.3. RAW 264.7 Cells
2.3.1. RAW 264.7 Macrophage Assay
2.3.2. Cytotoxicity Assay
2.3.3. NO Determination
2.3.4. Mouse IL-6 Double Antibody Sandwich (DAS) Enzyme Linked Immunosorbent Assay (ELISA)
2.3.5. Mouse MIPs (MIP-1α, MIP-1β and MIP-2) DAS ELISAs
2.3.6. Mouse Proteome Profiling Assay
2.3.7. Quantification of Pixel Density for Cytokine and Chemokine Membranes
2.4. Whole Blood Cell (WBC) Culture
2.4.1. Blood Collection
2.4.2. Cell Culture
2.4.3. Cytotoxicity Assay
2.4.4. Cytokine Analysis using DAS ELISAs
2.4.5. Human MIP-1β DAS ELISA
2.4.6. Human Proteome Profiling
2.5. Statistical Analysis
3. Results
3.1. The Effects of CDs on RAW 264.7 Cells
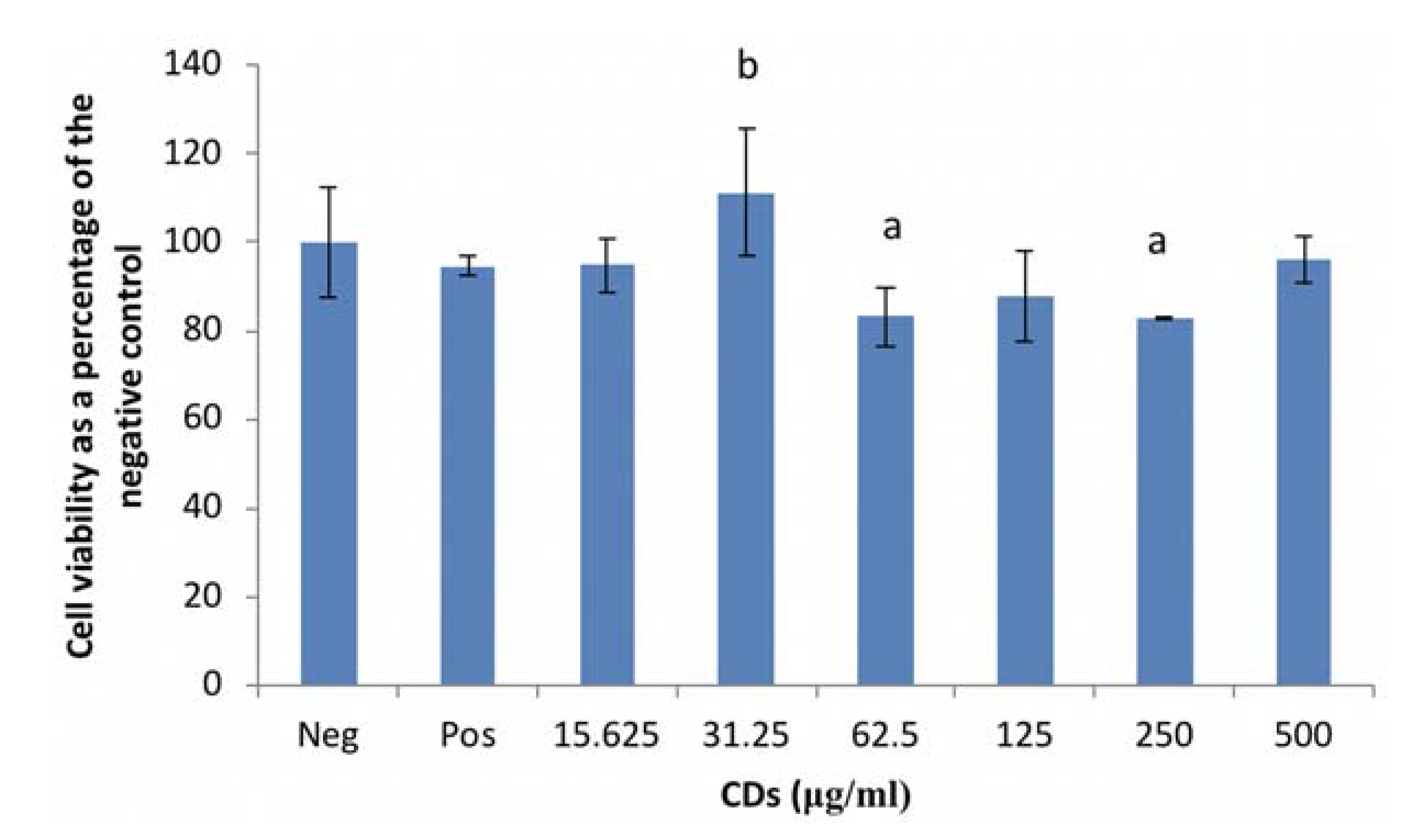
3.1.1. The Effects of CDs on the Viability of RAW 264.7 Cells
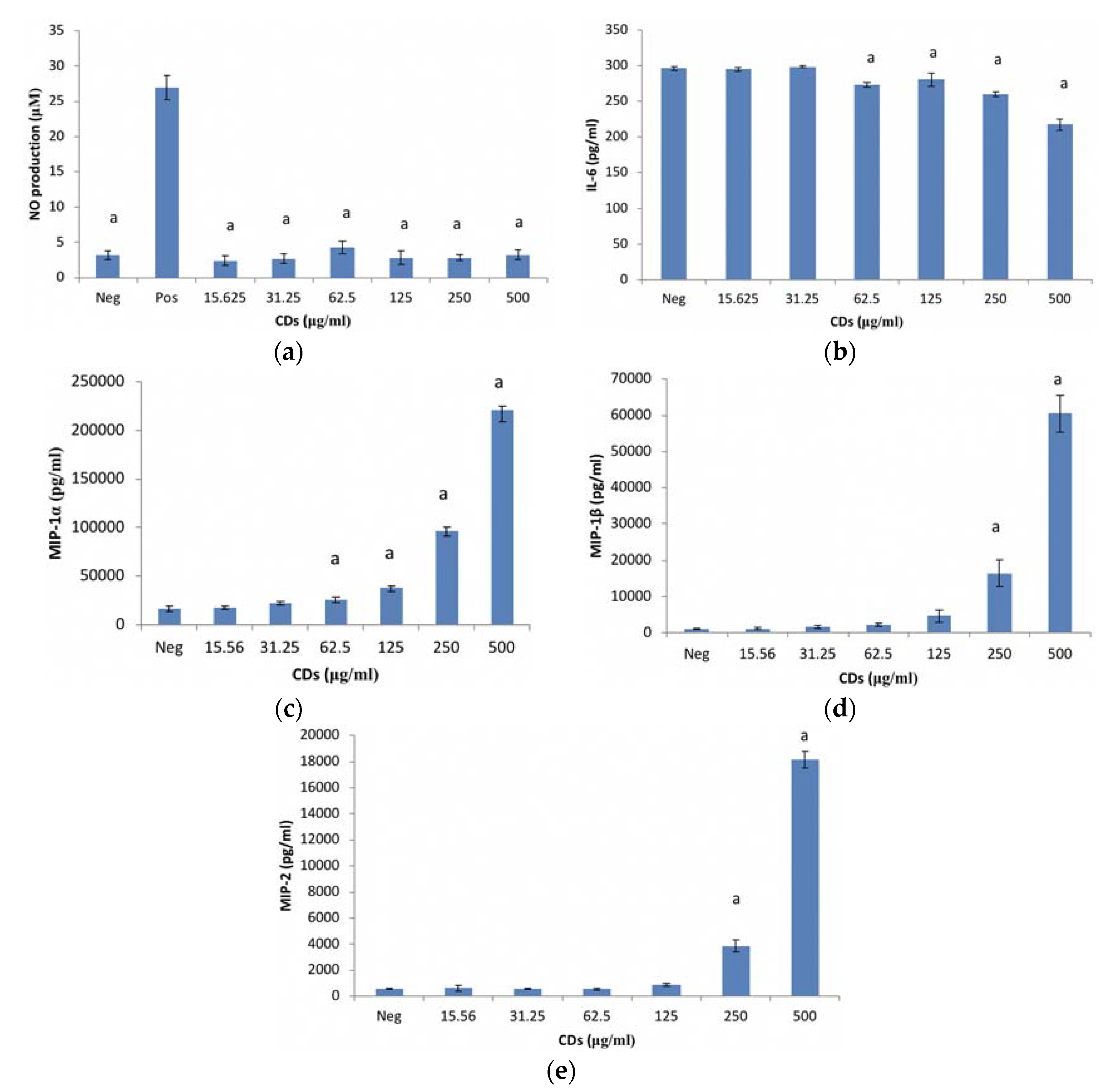
3.1.2. The Effects of CDs on the Inflammatory Biomarker NO Using RAW 264.7 Cells
3.1.3. The Effects of CDs on the Inflammatory Biomarker IL-6 Using RAW 264.7 Cells
3.1.4. The Effects of CDs on MIP-1α Using RAW 264.7 Cells
3.1.5. The Effects of CDs on MIP-1β Using RAW 264.7 Cells
3.1.6. The Effects of CDs on MIP-2 Using RAW 264.7 Cells
3.2. The Effects of CDs on the Secretory Cytokine and Chemokine Profile of RAW 264.7 Cells
3.3. The Effects of CDs on WBCs
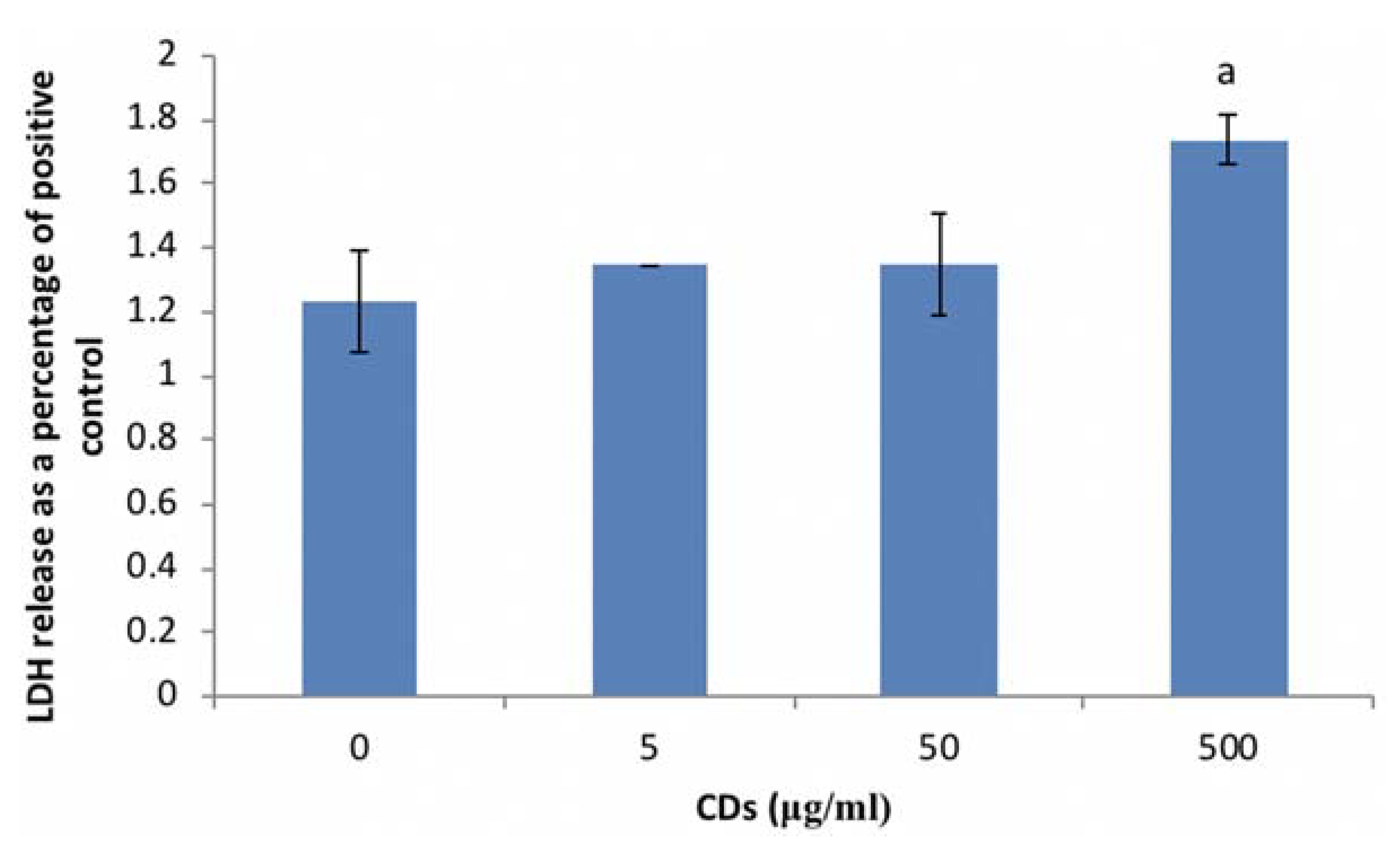
3.3.1. The Effects of CDs on Viability of WBCs
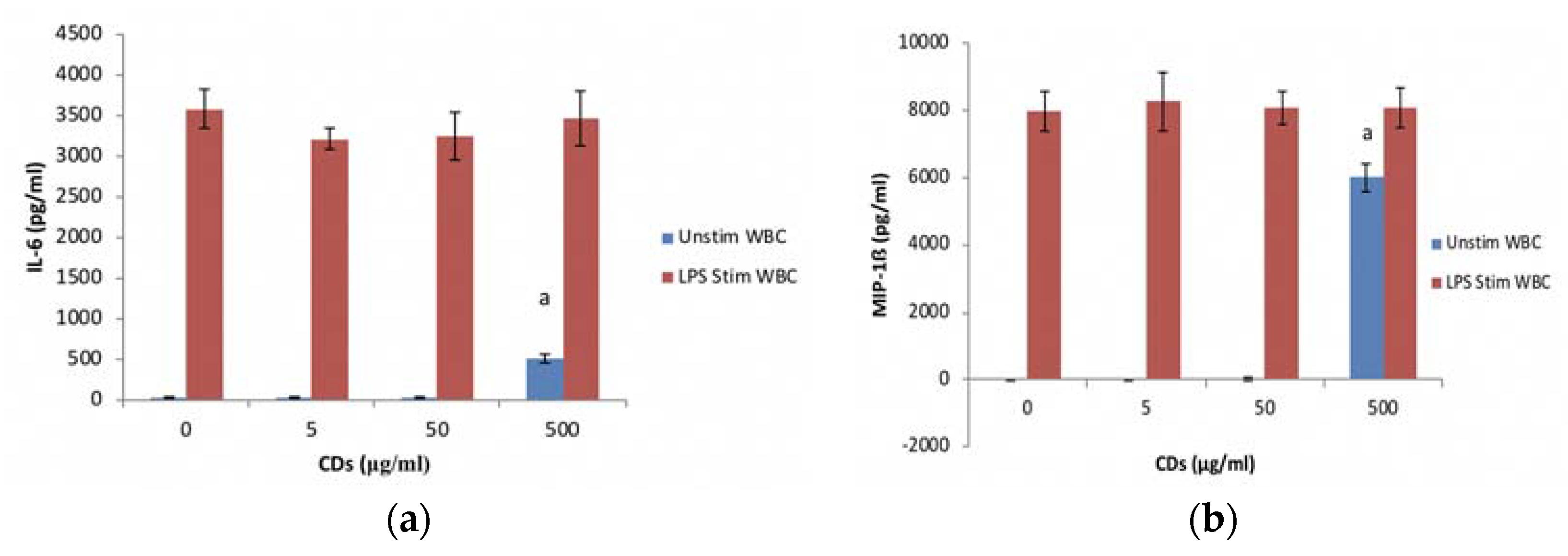
3.3.2. The Effects of CDs on the Inflammatory System Biomarker IL-6 Using WBCs
3.3.3. The Effects of CDs on the Inflammatory Chemokine, MIP-1β Using WBCs
3.3.4. The Effects of CDs on the Humoral Immune System Biomarker IL-10 Using WBCs
3.3.5. The Effects of CDs on the Cell Mediated Immune System Biomarker IFNγ Using WBCs
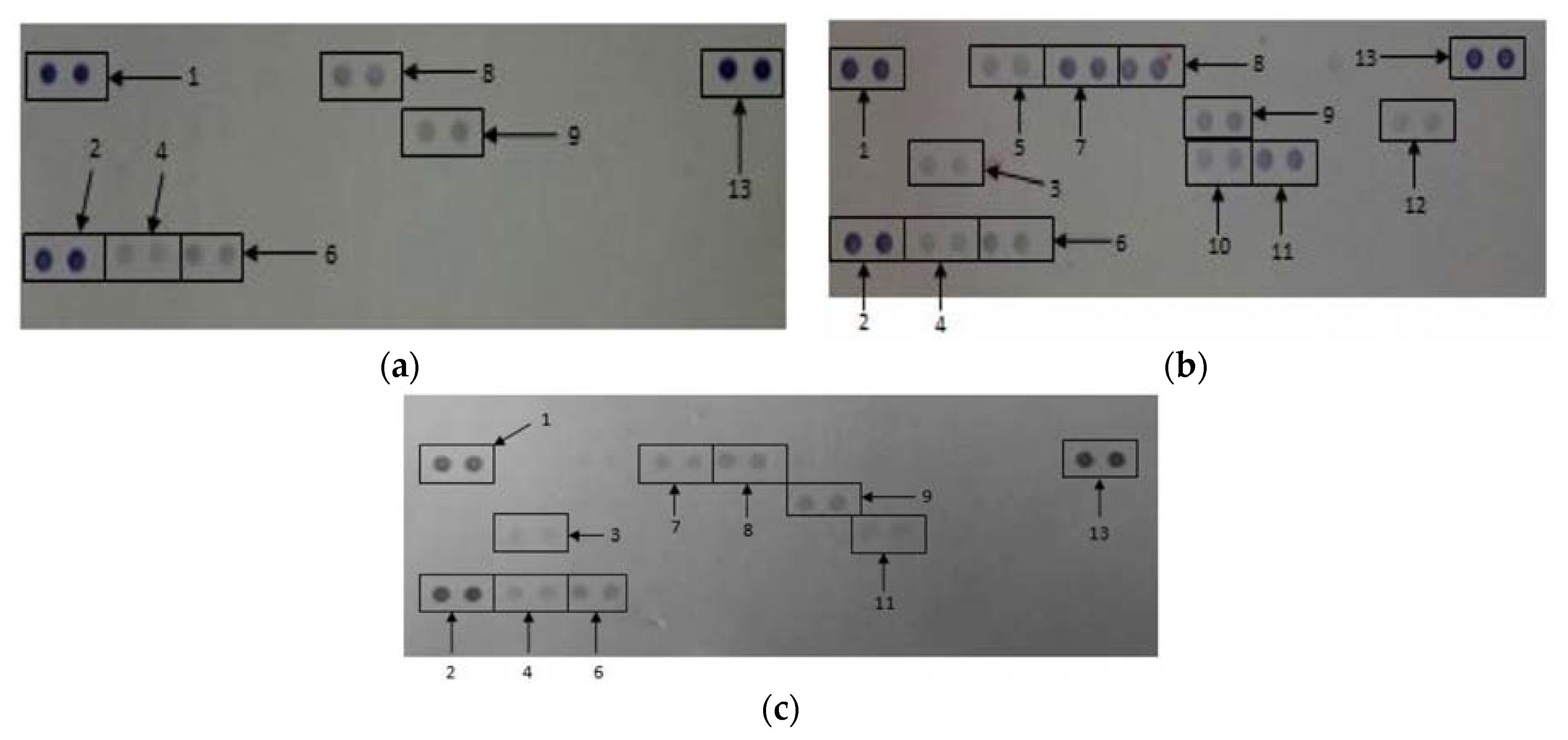
3.4. The Effects of CDs on the Secretory Cytokine and Chemokine Profile of WBCs
4. Discussion
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
Appendix A


| pH | Average | Standard Deviation |
|---|---|---|
| 3.04 | −1.42 | 0.87 |
| 5.25 | −11.40 | 4.35 |
| 6.99 | −16.83 | 1.40 |
| 10.12 | −35.07 | 2.74 |
References
- Peng, Z.; Han, X.; Li, S.; Al-Youbi, A.O.; Bashammakh, A.S.; El-Shahawi, M.S.; Leblanc, R.M. Carbon Dots: Biomacromolecule Interaction, Bioimaging and Nanomedicine. Coord. Chem. Rev. 2017, 343, 256–277. [Google Scholar] [CrossRef]
- Bayati, M.; Dai, J.; Zambrana, A.; Rees, C.; de Cortalezzi, M.F. Effect of water chemistry on the aggregation and photoluminescence behavior of carbon dots. J. Environ. Sci. 2017, 65, 223–235. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Lei, Y. Fluorescent carbon dots and their sensing applications. TrAC Trends Anal. Chem. 2017, 89, 163–180. [Google Scholar] [CrossRef]
- Tuerhong, M.; Xu, Y.; Yin, X.-B. Review on Carbon Dots and Their Applications. Chin. J. Anal. Chem. 2017, 45, 139–150. [Google Scholar] [CrossRef]
- Tao, S.; Zhu, S.; Feng, T.; Xia, C.; Song, Y.; Yang, B. The polymeric characteristics and photoluminescence mechanism in polymer carbon dots: A review. Mater. Today Chem. 2017, 6, 13–25. [Google Scholar] [CrossRef]
- Pierrat, P.; Wang, R.; Kereselidze, D.; Lux, M.; Didier, P.; Kichler, A.; Pons, F.; Lebeau, L. Efficient in vitro and in vivo pulmonary delivery of nucleic acid by carbon dot-based nanocarriers. Biomaterials 2015, 51, 290–302. [Google Scholar] [CrossRef] [PubMed]
- Kudr, J.; Richtera, L.; Xhaxhiu, K.; Hynek, D.; Heger, Z.; Zitka, O.; Adam, V. Carbon dots based FRET for the detection of DNA damage. Biosens. Bioelectron. 2017, 92, 133–139. [Google Scholar] [CrossRef] [PubMed]
- Pandey, S.; Gedda, G.R.; Thakur, M.; Bhaisare, M.L.; Talib, A.; Khan, M.S.; Wu, S.-M.; Wu, H.-F. Theranostic carbon dots ‘clathrate-like’nanostructures for targeted photo-chemotherapy and bioimaging of cancer. J. Ind. Eng. Chem. 2017, 56, 62–73. [Google Scholar] [CrossRef]
- Wang, J.; Li, Q.; Zhou, J.; Wang, Y.; Yu, L.; Peng, H.; Zhu, J. Synthesis, characterization and cells and tissues imaging of carbon quantum dots. Opt. Mater. 2017, 72, 15–19. [Google Scholar] [CrossRef]
- Havrdova, M.; Hola, K.; Skopalik, J.; Tomankova, K.; Petr, M.; Cepe, K.; Polakova, K.; Tucek, J.; Bourlinos, A.B.; Zboril, R. Toxicity of carbon dots–Effect of surface functionalization on the cell viability, reactive oxygen species generation and cell cycle. Carbon 2016, 99, 238–248. [Google Scholar] [CrossRef]
- Yuan, Y.; Guo, B.; Hao, L.; Liu, N.; Lin, Y.; Guo, W.; Li, X.; Gu, B. Doxorubicin-loaded environmentally friendly carbon dots as a novel drug delivery system for nucleus targeted cancer therapy. Colloids Surf. B Biointerfaces 2017, 159, 349–359. [Google Scholar] [CrossRef] [PubMed]
- Inoue, K.-I.; Koike, E.; Yanagisawa, R.; Hirano, S.; Nishikawa, M.; Takano, H. Effects of multi-walled carbon nanotubes on a murine allergic airway inflammation model. Toxicol. Appl. Pharmacol. 2009, 237, 306–316. [Google Scholar] [CrossRef] [PubMed]
- Kayat, J.; Gajbhiye, V.; Tekade, R.K.; Jain, N.K. Pulmonary toxicity of carbon nanotubes: A systematic report. Nanomed. Nanotechnol. Biol. Med. 2011, 7, 40–49. [Google Scholar] [CrossRef] [PubMed]
- Qu, C.; Wang, L.; He, J.; Tan, J.; Liu, W.; Zhang, S.; Zhang, C.; Wang, Z.; Jiao, S.; Liu, S. Carbon nanotubes provoke inflammation by inducing the pro-inflammatory genes IL-1β and IL-6. Gene 2012, 493, 9–12. [Google Scholar] [CrossRef] [PubMed]
- Granger, D.L.; Taintor, R.R.; Boockvar, K.S.; Hibbs, J.B. Measurement of nitrate and nitrite in biological samples using nitrate reductase and Griess reaction. Methods Enzymol. 1996, 268, 142–151. [Google Scholar] [PubMed]
- Wang, Y.; Anilkumar, P.; Cao, L.; Liu, J.-H.; Luo, P.G.; Tackett, K.N.; Sahu, S.; Wang, P.; Wang, X.; Sun, Y.-P. Carbon dots of different composition and surface functionalization: Cytotoxicity issues relevant to fluorescence cell imaging. Exp. Biol. Med. 2011, 236, 1231–1238. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.-T.; Wang, X.; Wang, H.; Lu, F.; Luo, P.G.; Cao, L.; Meziani, M.J.; Liu, J.-H.; Liu, Y.; Chen, M. Carbon dots as nontoxic and high-performance fluorescence imaging agents. J. Phys. Chem. C 2009, 113, 18110–18114. [Google Scholar] [CrossRef] [PubMed]
- Kagan, V.; Tyurina, Y.; Tyurin, V.; Konduru, N.; Potapovich, A.; Osipov, A.; Kisin, E.; Schwegler-Berry, D.; Mercer, R.; Castranova, V. Direct and indirect effects of single walled carbon nanotubes on RAW 264.7 macrophages: Role of iron. Toxicol. Lett. 2006, 165, 88–100. [Google Scholar] [CrossRef] [PubMed]
- Crouzier, D.; Follot, S.; Gentilhomme, E.; Flahaut, E.; Arnaud, R.; Dabouis, V.; Castellarin, C.; Debouzy, J.-C. Carbon nanotubes induce inflammation but decrease the production of reactive oxygen species in lung. Toxicology 2010, 272, 39–45. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Murray, A.; Kisin, E.; Leonard, S.; Young, S.; Kommineni, C.; Kagan, V.; Castranova, V.; Shvedova, A. Oxidative stress and inflammatory response in dermal toxicity of single-walled carbon nanotubes. Toxicology 2009, 257, 161–171. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Tang, M.; Kong, L.; Li, H.; Zhang, T.; Zhang, S.; Xue, Y.; Pu, Y. Comparison of cytotoxic and inflammatory responses of pristine and functionalized multi-walled carbon nanotubes in RAW 264.7 mouse macrophages. J. Hazard. Mater. 2012, 219, 203–212. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.-H.; Yang, S.-T.; Chen, X.-X.; Wang, H. Fluorescent carbon dots and nanodiamonds for biological imaging: Preparation, application, pharmacokinetics and toxicity. Curr. Drug Metab. 2012, 13, 1046–1056. [Google Scholar] [CrossRef] [PubMed]
- De Jong, W.H.; Van Loveren, H. Screening of xenobiotics for direct immunotoxicity in an animal study. Methods 2007, 41, 3–8. [Google Scholar] [CrossRef] [PubMed]
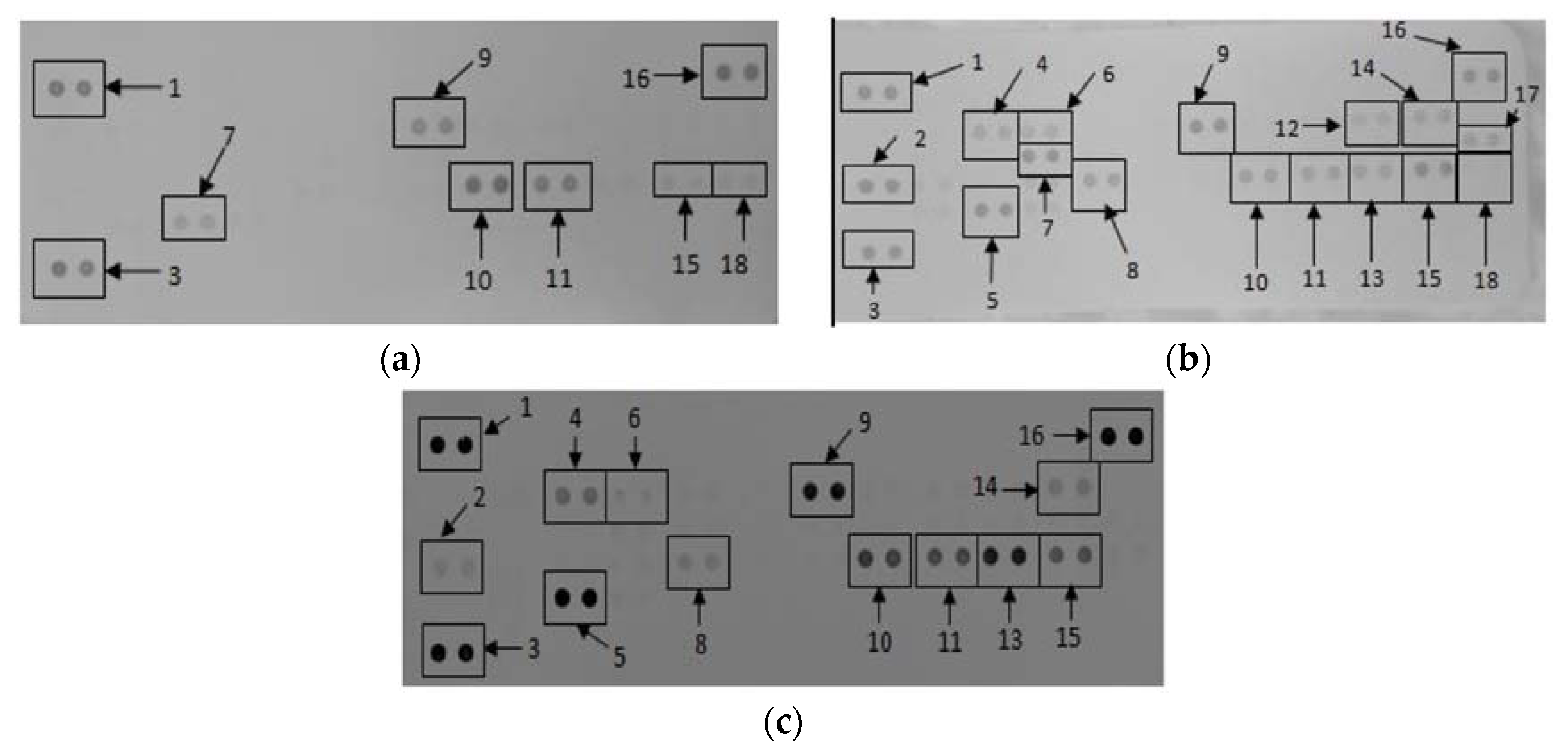

| Cytokines and Chemokines | Positive Control | Negative Control | 500 μg/mL CDs |
|---|---|---|---|
| Reference Spot | 100 ± 8.62 | 100 ± 10.49 | 100 ± 15.19 |
| IP-10 | 114.74 ± 5.24 | 0 ± 0 | 8.20 ± 0.11 a,b |
| G-CSF | 58.27 ± 2.55 | 0 ± 0 | 26.76 ± 0.51 a,b |
| TNF-α | 102.01 ± 5.91 | 17.96 ± 0.13 | 101.00 ± 0.60 a |
| GM-CSF | 47.32 ± 3.02 | 0 ± 0 | 3.72 ± 0.03 a,b |
| IL-6 | 91.94 ± 3.60 | 0 ± 0 | 0 ± 0 b |
| JE | 78.97 ± 4.48 | 0 ± 0 | 10.47 ± 0.01 a,b |
| sICAM | 109.05 ± 6.00 | 34.76 ± 0.02 | 78.65 ± 0.98 a,b |
| MIP-1α | 72.87 ± 4.22 | 57.00 ± 1.94 | 64.87 ± 1.87 a,b |
| MIP-1β | 62.77 ± 3.35 | 37.68 ± 0.33 | 45.40 ± 0.81 a,b |
| MIP-2 | 55.51 ± 2.75 | 0 ± 0 | 76.63 ± 0.06 a,b |
| RANTES | 100.57 ± 4.42 | 17.93 ± 0.83 | 36.09 ± 0.78 a,b |
| SDF-1 | 16.41 ± 1.61 | 21.75 ± 1.01 | 0 ± 0 a,b |
| IL-27 | 54.97 ± 4.05 | 0 ± 0 | 0 ± 0 b |
| IL-1ra | 55.84 ± 1.66 | 0 ± 0 | 14.96 ± 0.21 a,b |
| IL-1β | 39.16 ± 3.31 | 0 ± 0 | 0 ± 0 b |
| Cytokines and Chemokines | Positive Control | Negative Control | 500 μg/mL CDs |
|---|---|---|---|
| Reference Spot | 100 ± 7.28 | 100 ± 5.57 | 100 ± 11.35 |
| IL-1ra | 16.92 ± 1.46 | 0 ± 0 | 12.57 ± 1.27 a,b |
| MIF | 17.56 ± 1.51 | 10.57 ± 1.91 | 20.41 ± 4.00 a |
| MCP-1 | 26.26 ± 2.54 | 0 ± 0 | 0 ± 0 b |
| Serpin E1 | 32.40 ± 2.49 | 24.26 ± 2.26 | 31.81 ± 4.38 |
| MIP-1α/β | 70.22 ± 4.11 | 0 ± 0 | 25.84 ± 2.17 a,b |
| RANTES | 62.94 ± 2.55 | 34.28 ± 1.37 | 32.95 ± 2.75 b |
| sICAM | 37.15 ± 0.59 | 35.98 ± 2.19 | 32.38 ± 4.52 |
| IL-6 | 29.31 ± 4.81 | 0 ± 0 | 0 ± 0 b |
| IL-8 | 50.27 ± 8.41 | 0 ± 0 | 13.56 ± 2.53 a,b |
| IL-1β | 20.29 ± 0.99 | 0 ± 0 | 0 ± 0 b |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lategan, K.; Fowler, J.; Bayati, M.; Fidalgo de Cortalezzi, M.; Pool, E. The Effects of Carbon Dots on Immune System Biomarkers, Using the Murine Macrophage Cell Line RAW 264.7 and Human Whole Blood Cell Cultures. Nanomaterials 2018, 8, 388. https://doi.org/10.3390/nano8060388
Lategan K, Fowler J, Bayati M, Fidalgo de Cortalezzi M, Pool E. The Effects of Carbon Dots on Immune System Biomarkers, Using the Murine Macrophage Cell Line RAW 264.7 and Human Whole Blood Cell Cultures. Nanomaterials. 2018; 8(6):388. https://doi.org/10.3390/nano8060388
Chicago/Turabian StyleLategan, Kim, Jodi Fowler, Mohamed Bayati, Maria Fidalgo de Cortalezzi, and Edmund Pool. 2018. "The Effects of Carbon Dots on Immune System Biomarkers, Using the Murine Macrophage Cell Line RAW 264.7 and Human Whole Blood Cell Cultures" Nanomaterials 8, no. 6: 388. https://doi.org/10.3390/nano8060388
APA StyleLategan, K., Fowler, J., Bayati, M., Fidalgo de Cortalezzi, M., & Pool, E. (2018). The Effects of Carbon Dots on Immune System Biomarkers, Using the Murine Macrophage Cell Line RAW 264.7 and Human Whole Blood Cell Cultures. Nanomaterials, 8(6), 388. https://doi.org/10.3390/nano8060388