1. Introduction
Green chemistry seeks to reduce or eliminate the generation and use of hazardous substances, and is built on a set of practices and principles in pursuit of that aim [
1,
2]. Industrial and academic research has, in recent years, shown increased interest in the fabrication of ‘green’ polymers from natural resources. More effort has gone into the replacement of polymers derived from petroleum with sustainable natural biopolymers that are biodegradable, environment-friendly, and require the consumption of less energy for their renewal [
3,
4]. Green electrospun materials may provide new end uses, such as protection against microorganisms by using solvents that are environmentally friendly, non-toxic, and low cost. Water-based solvents and materials provide a good example.
Electrospun nanofibres are generally made by electrospinning organic polymers [
5], and possess high porosity, good stability, and a large surface area to volume ratio [
6]. The end uses of electrospun nanofibres are wide-ranging, such as composites, catalysis, filters, tissue engineering, drug delivery, and wound healing [
7,
8,
9,
10,
11,
12]. In less than 10 years, we have seen progress in developing modified electrospinning methods, such as: Two-stream electrospinning; co-axial electrospinning; reactive electrospinning; and layer-by-layer electrospinning (LBL)—enhancing the end use spectrum [
13,
14,
15,
16]. Although we have produced nanofibres from more than 100 individual polymers [
17], developing a green electrospinning process remains a challenge, largely because most polymers have poor solubility in water and there is a very limited supply of neutral polymers that are soluble in water—charged polyelectrolytes form the majority of water-soluble polymers. Since like charges repel each other, the viscosities of polyelectrolyte solutions where salt is absent are a good deal higher than those of neutral polymer solutions with similar polymer concentration. Almost all reported electrospun polyelectrolytes, whether neutral or salt, use toxic solvents in spinning and may need cross-linking agents to achieve chemically stable fibre mats [
18,
19,
20,
21]. The cytotoxicity of cross-linking agents and solvents has prevented extensive uses of fibre mats based on water-soluble polymers.
Chitosan (CHT) is a biodegradable polymer comprising of randomly distributed b-(1-4)-linked
d-glucosamine (deacetylated) and
N-acetyl-
d-glucosamine (acetylated) units, which contribute to a cationic nature, as well as other properties. It is the cationic property that gives CHT the ability to penetrate mucous layers in biomedical applications, perform an antimicrobial function in food preservatives, and trap dyes and metals in waste water [
22,
23,
24,
25,
26]. It is also being used in technical applications such as packaging and decontamination, because of its physico-chemical properties, such as hydrophobicity, thermal stability, and mechanical performances [
27,
28,
29]. These properties, unique to CHT, combined with high porosity and surface area by its nanosize, render it important for a range of different functional systems. Nevertheless, a number of factors increase the difficulty in electrospinning CHT, such as: Poor solubility in most organic solvents; crystal formation; high molecular weight; polycationic characteristics in solution; H-bonding in the
d-glucosamine unit; and a wide distribution of molecular weights [
30]. Polyvinyl alcohol (PVA) can be added to reduce CHT viscosity. With a semi-crystalline molecular structure, PVA is a biodegradable and biocompatible synthetic polymer combining solubility in water with a nontoxic nature, rendering it widely usable in biomedical and biochemical applications. PVA is easy to spin, easy to coagulate, easy to orient, and easy to cross-link [
31]. Attempts have been made to produce a high surface area nanofibre from a blend of CHT and PVA by electrospinning [
32,
33,
34], but involved in the process were expensive and toxic chemicals including trifluoroacetic acid (TFA), 1,1,1,3,3,3-hexafluoro-2-propanol, dichloromethane, chloroform, and acetic acid. Any residues of these chemicals can have undesirable long-term environmental impacts and be a health hazard, so these nanofibres are not considered green. Therefore, the use of functional fibres of this sort is difficult to justify in applications demanding non-toxicity and environmental friendliness.
Many factors dictate electrospun nanofibres’ diameter: Solution parameters; process parameters; and ambient parameters [
35]. Rigorous monitoring of these parameters is required to obtain the desired properties. The traditional approach has been to change only one parameter at a time, keeping all others fixed [
36]. We have used the Taguchi experimental design approach to optimise our experiments [
37,
38].
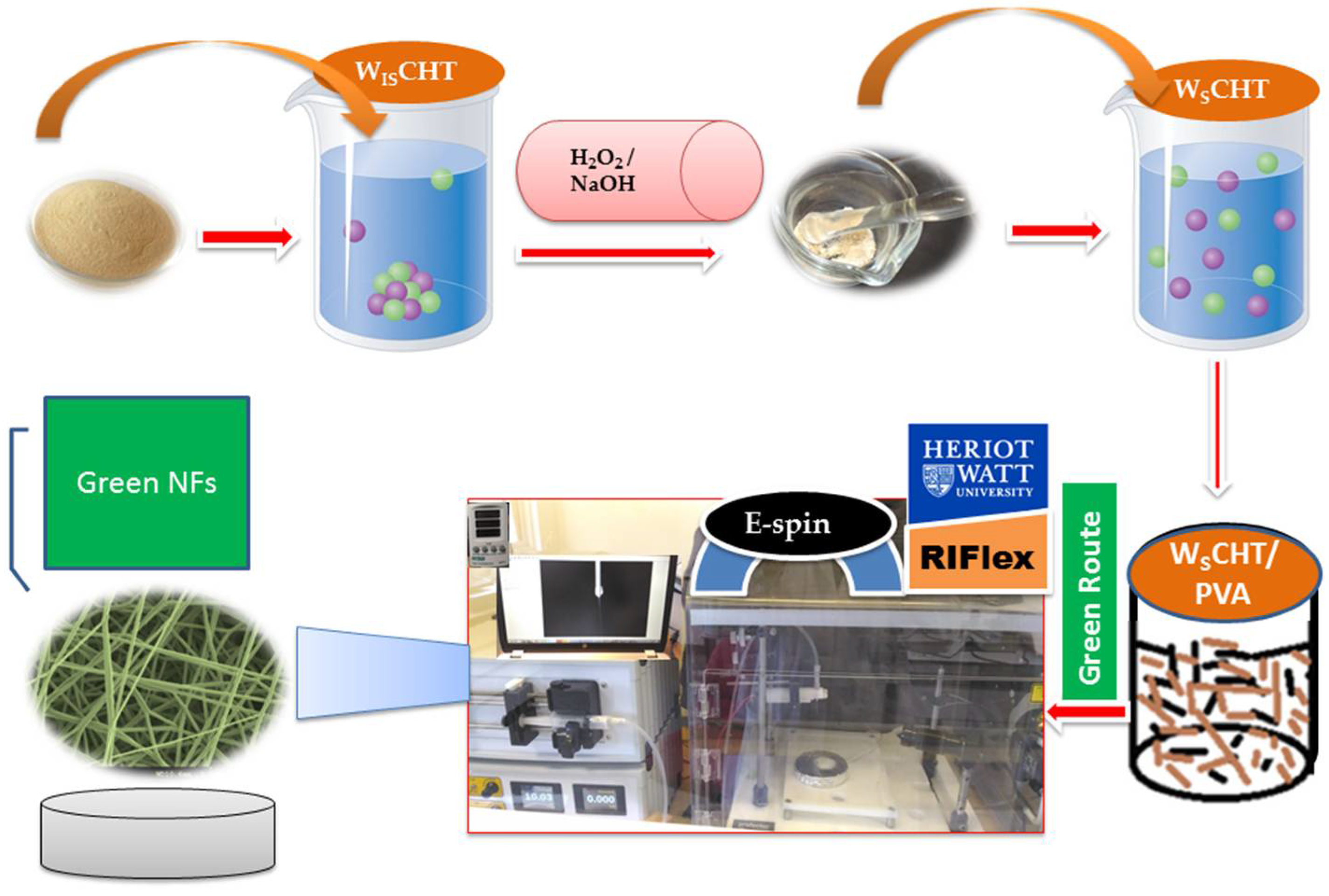
Therefore, our broad aim in this work is to replace organic solvent electrospinning of water-insoluble polymers with water-based formulations i.e., green electrospinning. To that effect, water-soluble chitosan (WSCHT) was synthesised under mild and heterogeneous reaction conditions (H2O2-assisted) and prepared in an all ‘green’ approach to fabricate WSCHT/PVA-blended nanofibres using water as a non-toxic solvent for the very first time in literature. Another aim is to optimise the electrospinning process parameters to get a finer fibre diameter range using the Taguchi approach.
2. Experimental
2.1. Materials
High molecular weight chitosan (degree of deacetylation 65%), was purchased from (Sigma–Aldrich, Dorset, UK). PVA (Mw: 115,000) was purchased from (BDH Ltd Poole, UK). The following chemicals were all obtained and used as reagents: Sodium hydroxide, hydrogen peroxide, hydrochloric acid, and ethanol. All the chemicals were used without further purification.
2.2. Synthesis of Water-Soluble Chitosan (WSCHT)
The following synthesis protocol was based on a previously reported method [
39] with a number of modifications, as described further. CHT was completely dissolved in 1% (
v/
v) aqueous hydrochloric acid to a concentration of 1% (
w/
v), then 30% (
v/
v) H
2O
2 aqueous solution was added and the solution was then maintained in a thermostatic water bath at 65 °C for 40 min. The final solution was then filtered using Whatman filter paper to expel polluting influences, and to promote decontamination. The final product was acquired after dialysis using a Slide-a-Lyzer dialysis cassette (Pierce, MWCO 2000) (molecular weight cut-off) in deionised (DI) water for 12 h at room temperature. Then the pH of the solution was brought to 7 by adding few drops of 5 M NaOH solution, and precipitation was obtained by adding ethanol. This step was followed by another rinsing with hot water to ensure complete removal of NaOH. Then a solid was collected after drying the precipitate in a vacuum, which was then washed with ice cold water in order to remove the excess traces of acid and gently cast onto polystyrene dishes and dried at 25 °C to obtain pure W
SCHT.
Table 1 shows the properties of W
ISCHT and W
SCHT.
2.3. Preparation of WSCHT-PVA Nanofibre
For preparing water-based electrospinning, solutions of W
SCHT-PVA were made in various concentrations, as listed
Table 2, by using water as a common solvent. Firstly, different W
SCHT concentration solutions were prepared by dissolving the required amount of W
SCHT powder in water (milli-Q grade) with gentle magnetic stirring for 56 h at room temperature to form a homogeneous solution. Different concentrations of PVA solutions were prepared by dissolving PVA in double-distilled water under magnetic stirring for 4 h at 75–80 °C, and then cooled to room temperature. Finally, the two polymer solutions were mixed at different weight ratios ultrasonically for 4 h to ensure a well-distribution of blended solutions before being subjected to the electrospinning process. The electrospinning experiment was carried out using an electrostatic spinning instrument (Research Institute for Flexible Materials, Heriot-Watt University, Edinburgh, UK), as shown in
Scheme 1. The prepared blend solution was drawn into a 5 ml disposable plastic syringe equipped with a metallic needle (inner diameter gauge of 22). The flow rate was monitored by a syringe pump (TS2-60, Longer Precision Pump Co. Ltd., Baoding, China). A high voltage power supply was applied to the solution in order to generate electrostatic field between the grounded collector (copper collector plate measuring 15 cm × 15 cm) and the syringe needle tip. Aluminum foil covered the grounded collector plate to collect the electrospun nanofibres. Finally, the collected nanofibres were dried at 25 °C under vacuum for 24 h to remove the residual solvent. All experiments were conducted at room temperature with a relative humidity of less than 40%.
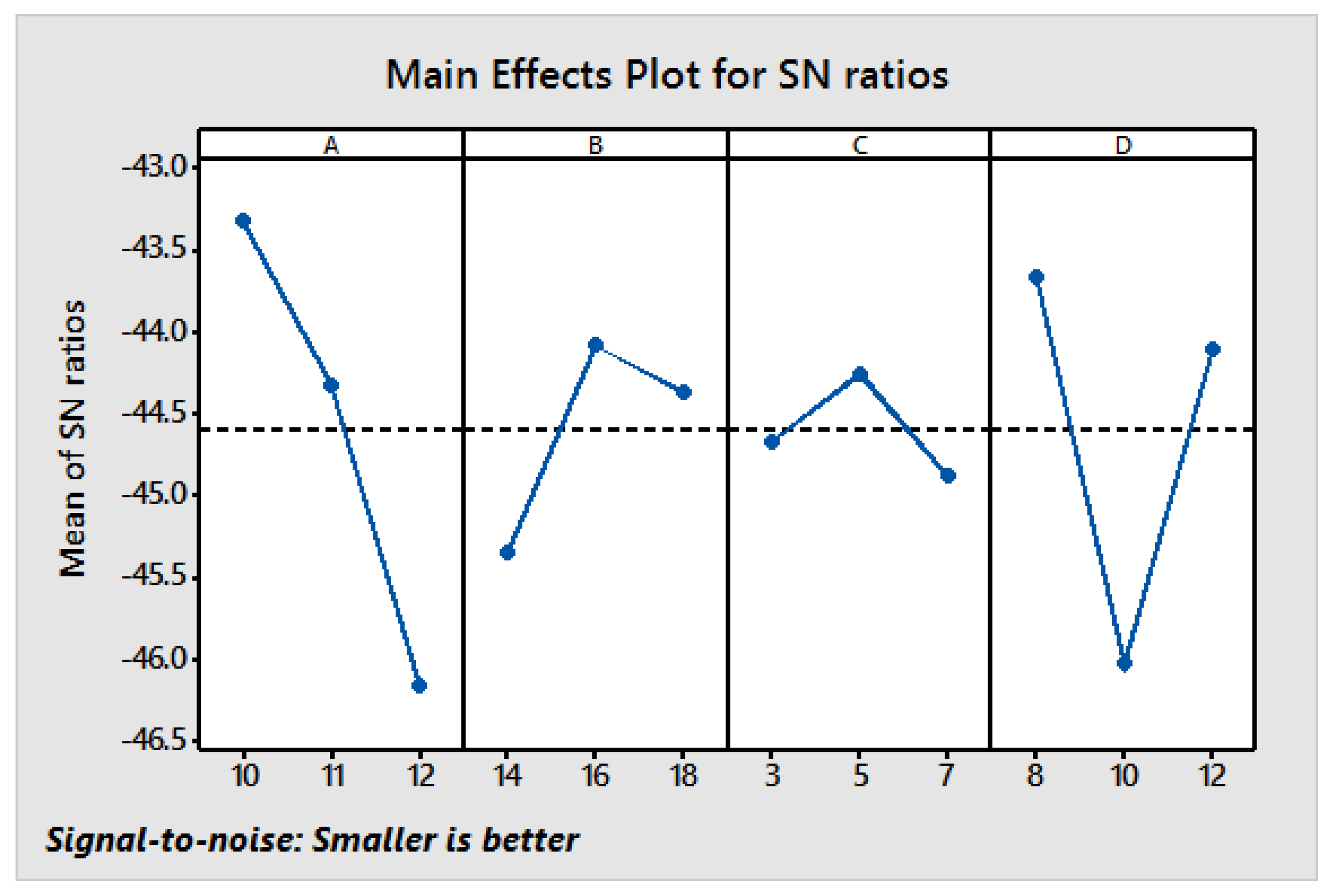
2.4. Taguchi Design of Experiment (DoE)
The work has been designed in a sequence of experimental steps to ensure that the collection of data satisfies statistical rules. This DoE and Taguchi-based DoE are simple methods of using orthogonal arrays to simulate a variety of experimental conditions. It is used to reduce experimental errors, process variation and for enhancing efficiency resulting in optimised process parameters and experimental reproducibility, whilst reducing time and cost of experimentation [
37]. Work time and cost are reduced in the processes [
39]. The method works through the conversion of target features to a signal to-noise (S/N) ratio, which allows the level of controlling parameters in contradiction to these parameters to be measured. S/N is the favourite signal ratio for random noise and assesses the quality of the experimental values. Three dissimilar functions were used as the target: ‘Larger is better’, ‘nominal is best’, and ‘smaller is better’. Using the S/N ratios allowed achievement of the optimum electrospinning condition. Then optimum electrospinning conditions were determined by the Taguchi optimisation method to validate the optimisation [
40,
41].
Table 3 shows the four parameters selected at each of the three levels: Larger, nominal, and smaller. Factors with different levels were combined in a manner matching the L
9 type orthogonal array,
Table 4. A ‘smaller is better’ characteristic formula determines the optimum combination of factors that would produce the smallest diameter electrospun nanofibres in accordance with Equation (1):
where S/N is the signal-to-noise ratio,
n is the number of observations, and
y is the diameter of nanofibres measured. Software Minitab 17 was used for optimisation of the electrospinning process by the Taguchi approach.
2.5. Measurements and Characterisation
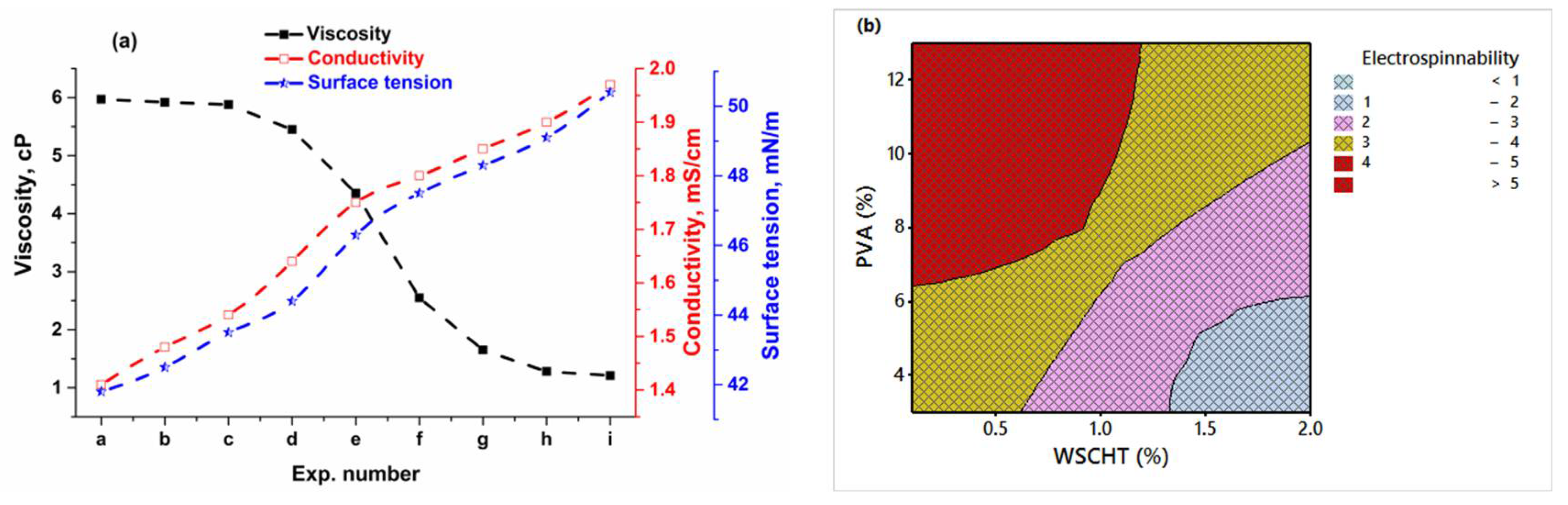
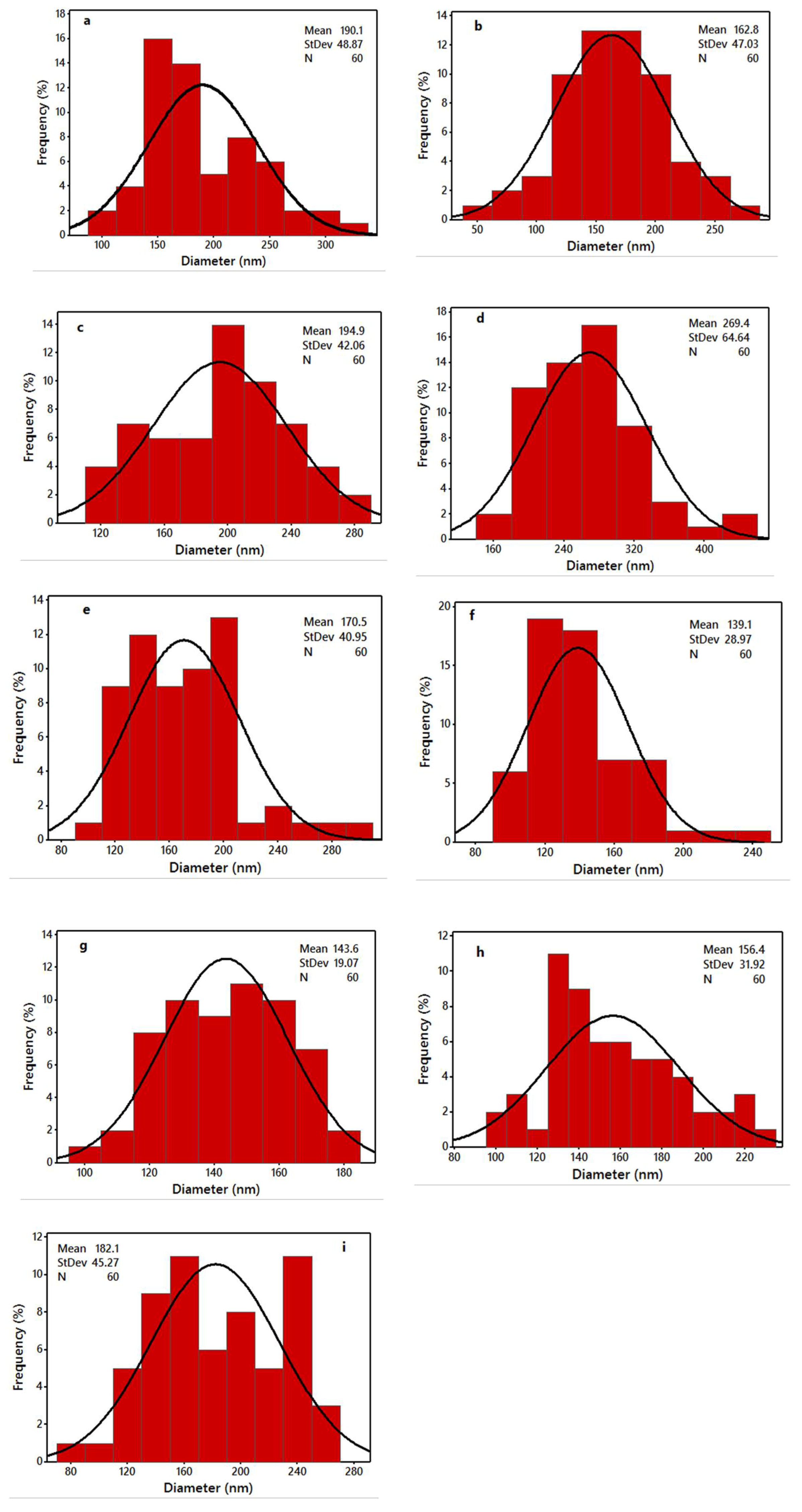
The viscosity of the solutions was measured using a viscometer (DV-II, Brookfield Co., Stoughton, MA, USA). The surface tension of the solutions was tested by a surface tensiometer (KRÜSS GmbH, Hamburg, Germany). The conductivity of the solutions was measured by a conductivity meter (HANNA HI8733, Leighton Buzzard, UK). All measurements were carried out at room temperature. The surface morphologies of the electrospun nanofibres were examined on a scanning electron microscopy (SEM; Hitachi, S-4300, Tokyo, Japan) at an accelerating voltage of 5 kV, and a working distance of 14 cm. Prior to the observation, the samples were sputter-coated with gold–palladium to keep away from charge gathering for 3 min in a Denton Desk II Sputter Coating Unit (Denton Vacuum, Moorestown, NJ, USA) for better conductivity during imaging. The diameters of nanofibres were measured by using an image analyser (Image J, National Institutes of Health, Bethesda, MD, USA).
4. Conclusions
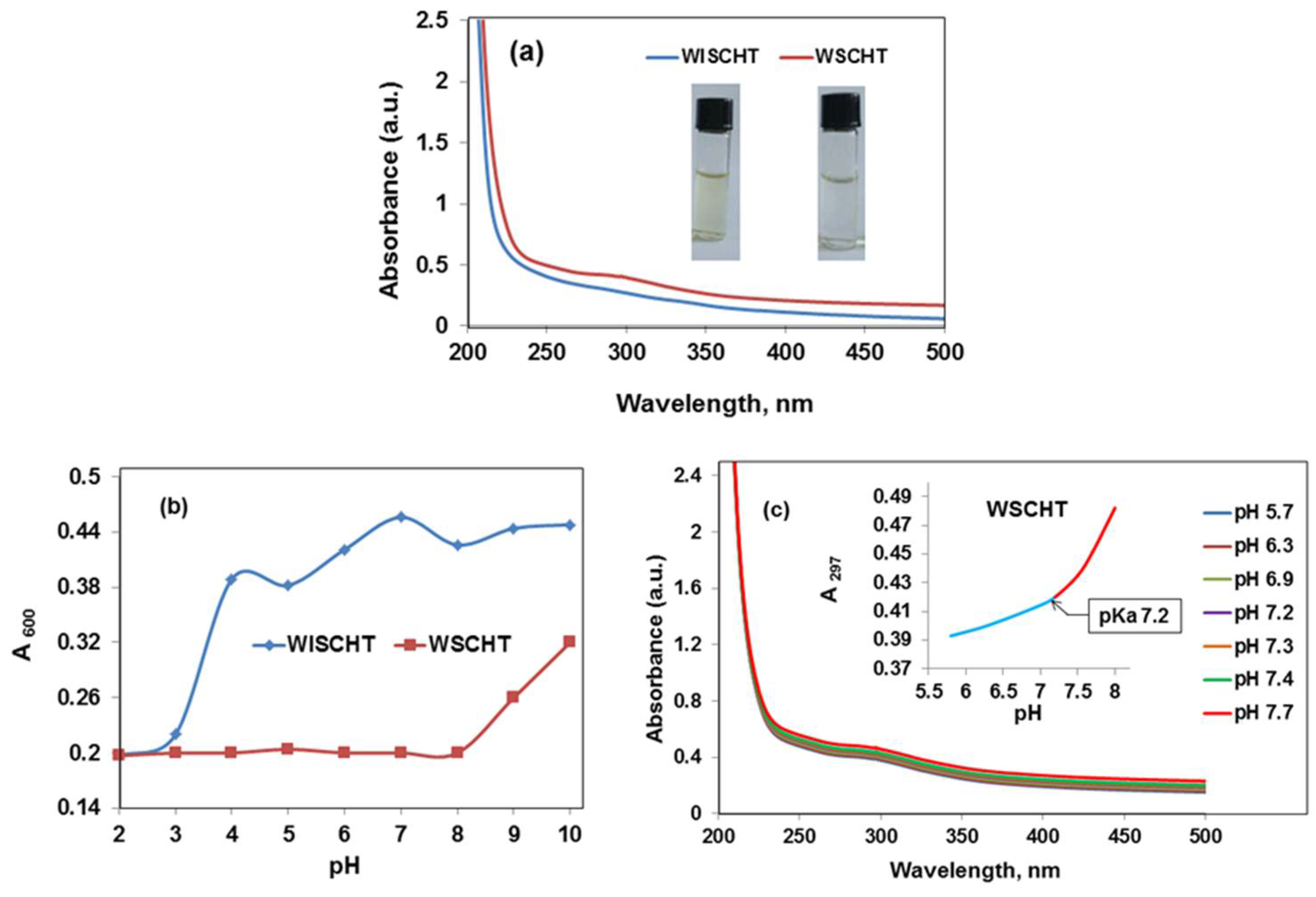
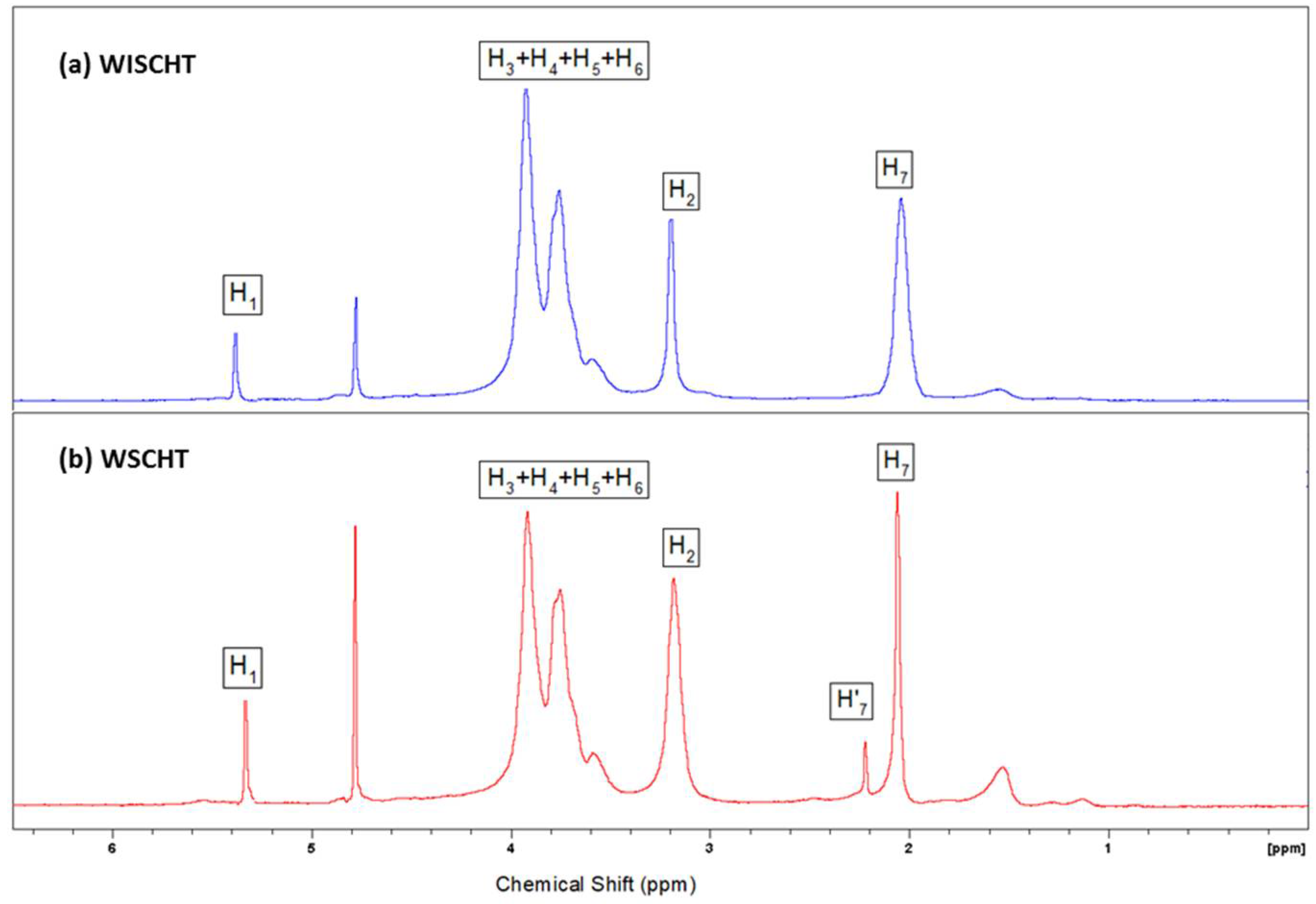
In this study, WSCHT has been synthesised through heterogeneous based reaction and used for fabrication of nanofibres containing PVA as guest polymer (WSCHT/PVA), via a green electrospinning method with water as the solvent. First, WSCHT has been produced via H2O2 medium, and from optical micrographs it was shown that particles of WSCHT exhibited round shaped structure, as further confirmed by UV-Vis spectra analysis, and by the proton (1H) NMR spectra showing the corresponding shift of peaks. The physical properties of the WSCHT/PVA solution blend were obtained as reported, and morphological analysis using SEM has confirmed the uniformity of the electrospun fiber with standard distribution.
Furthermore, the process parameters were optimised using the Taguchi method, and conditions affecting the morphology of the produced nanofibres were established. From ANOVA, it is clearly identified that the solution concentration has the highest influence (49.31%) on diameter, followed by spinning distance (37.77%), voltage (10.54%), and flow rate (2.38%). In addition, the obtained optimum combination was validated by conducting a confirmatory experiment with improvement of S/N ratios, and proving the application of Taguchi’s method for electrospinning optimisation. This research shows how environmentally sustainable nano materials may be developed for many diverse end-uses e.g., biomedical science (drug carriers, antimalarial products, antiviral, etc.), entomology (to control mosquitoes and ticks) and for environmental applications.