Citrate-Linked Keto- and Aldo-Hexose Monosaccharide Cellulose Conjugates Demonstrate Selective Human Neutrophil Elastase-Lowering Activity in Cotton Dressings
Abstract
:1. Introduction
2. Results and Discussion
2.1. Preparation and Analysis of Keto- and Aldo-Hexose Conjugates of Cellulose
2.1.1. Preparation of Monosaccharide-Cellulose Conjugates
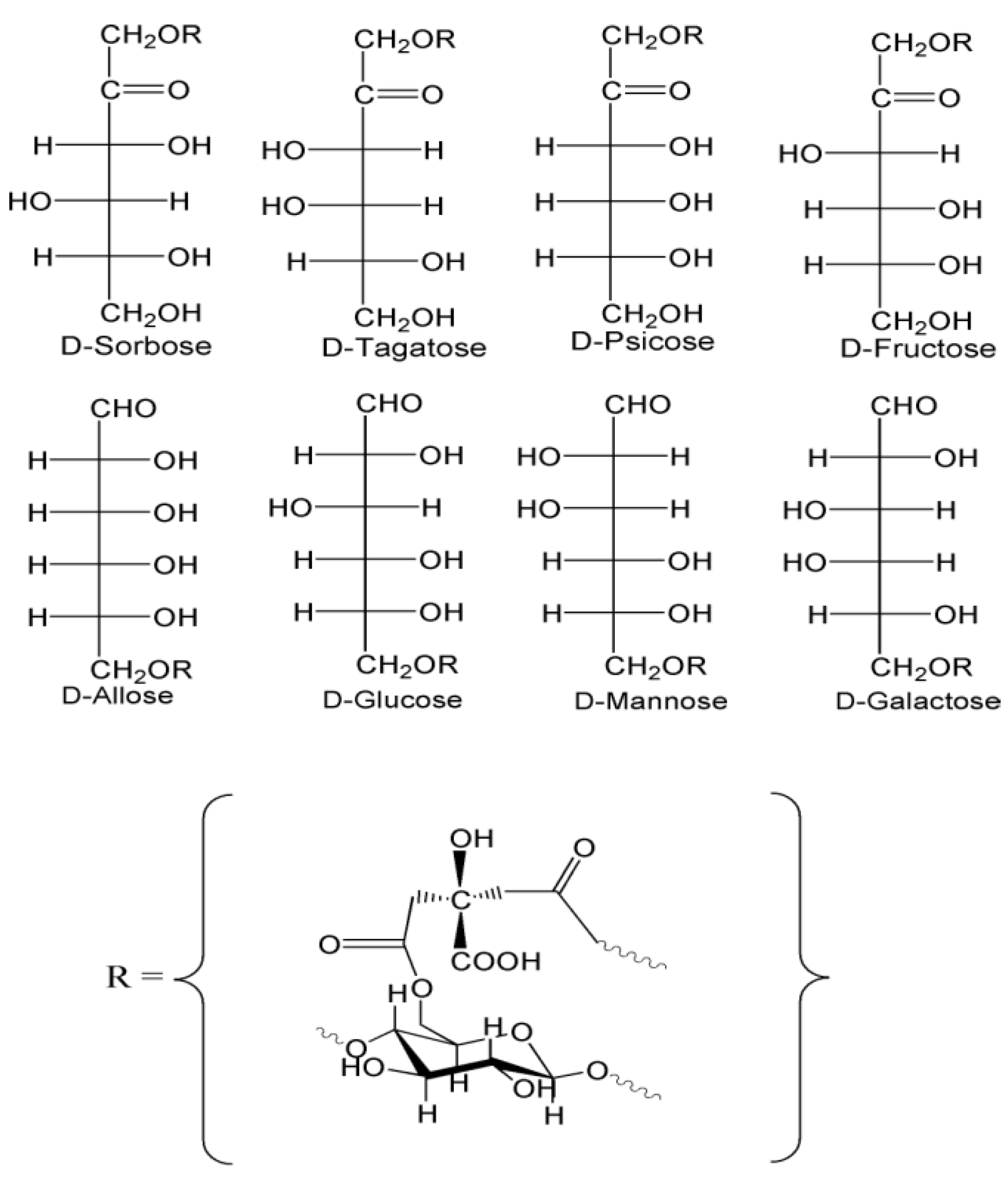
2.1.2. Characterization of Monosaccharide-Cellulose Conjugates
2.1.3. HPLC Analysis of Monosaccharides

| Monosaccharides | Concentration of Aldo- & Keto-Hexose in ppms Determined in HPLC Eluant * | Levels of Monosaccharide Linked to Cotton Gauze ** |
|---|---|---|
| Allose | 405 | 4.0 |
| Fructose | 121 | 1.2 |
| Mannose | 210 | 2.1 |
| Galactose | 200 | 2.0 |
| Glucose | 419 | 4.1 |
| Psicose | 405 | 4.1 |
| Sorbose | 205 | 2.1 |
| Tagatose | 201 | 2.0 |
2.1.4. FTIR Characterization of Cellulose Citrate Link

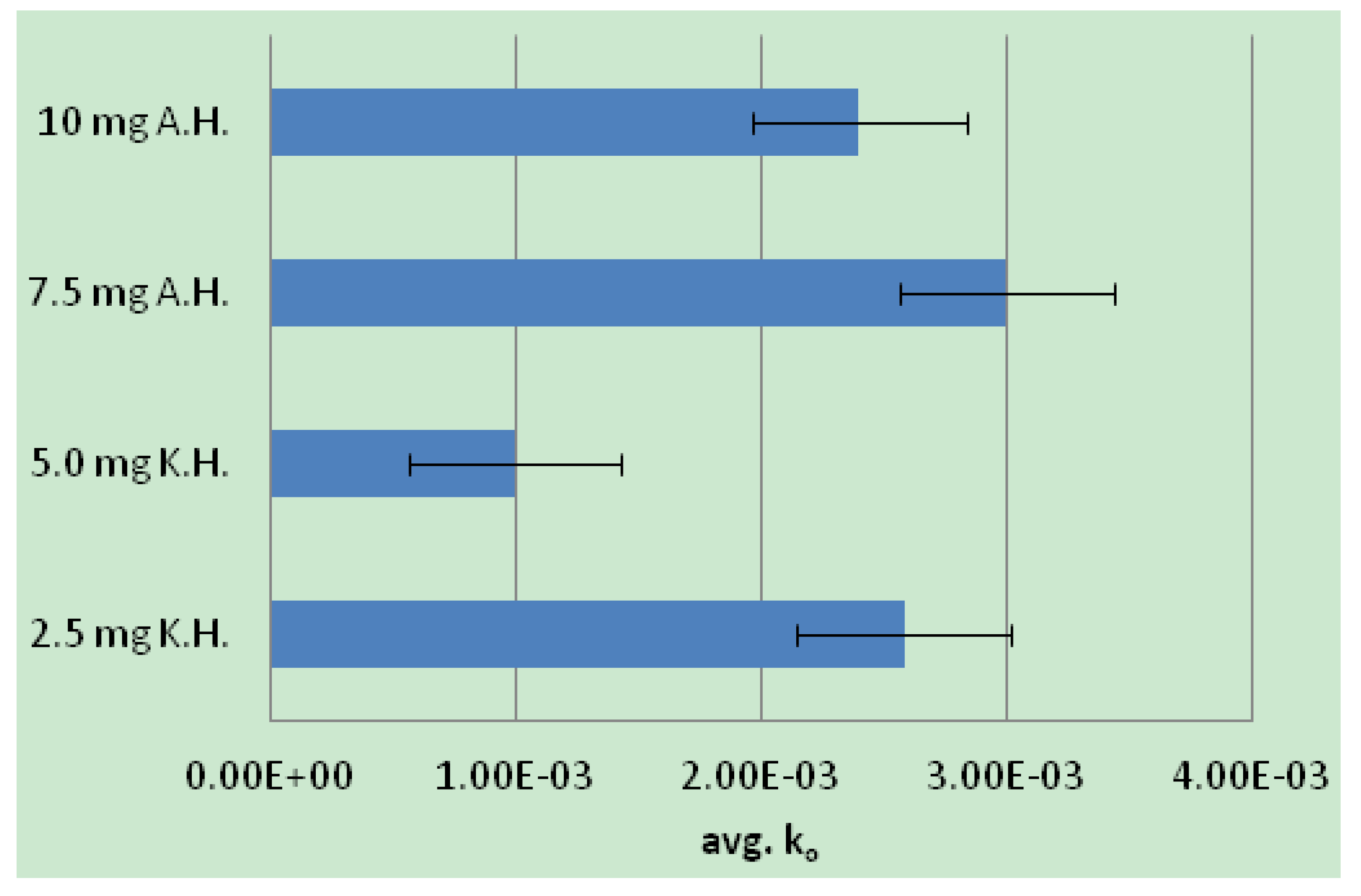
2.2. Human Neutrophil Elastase Sequestration by Modified Analogs
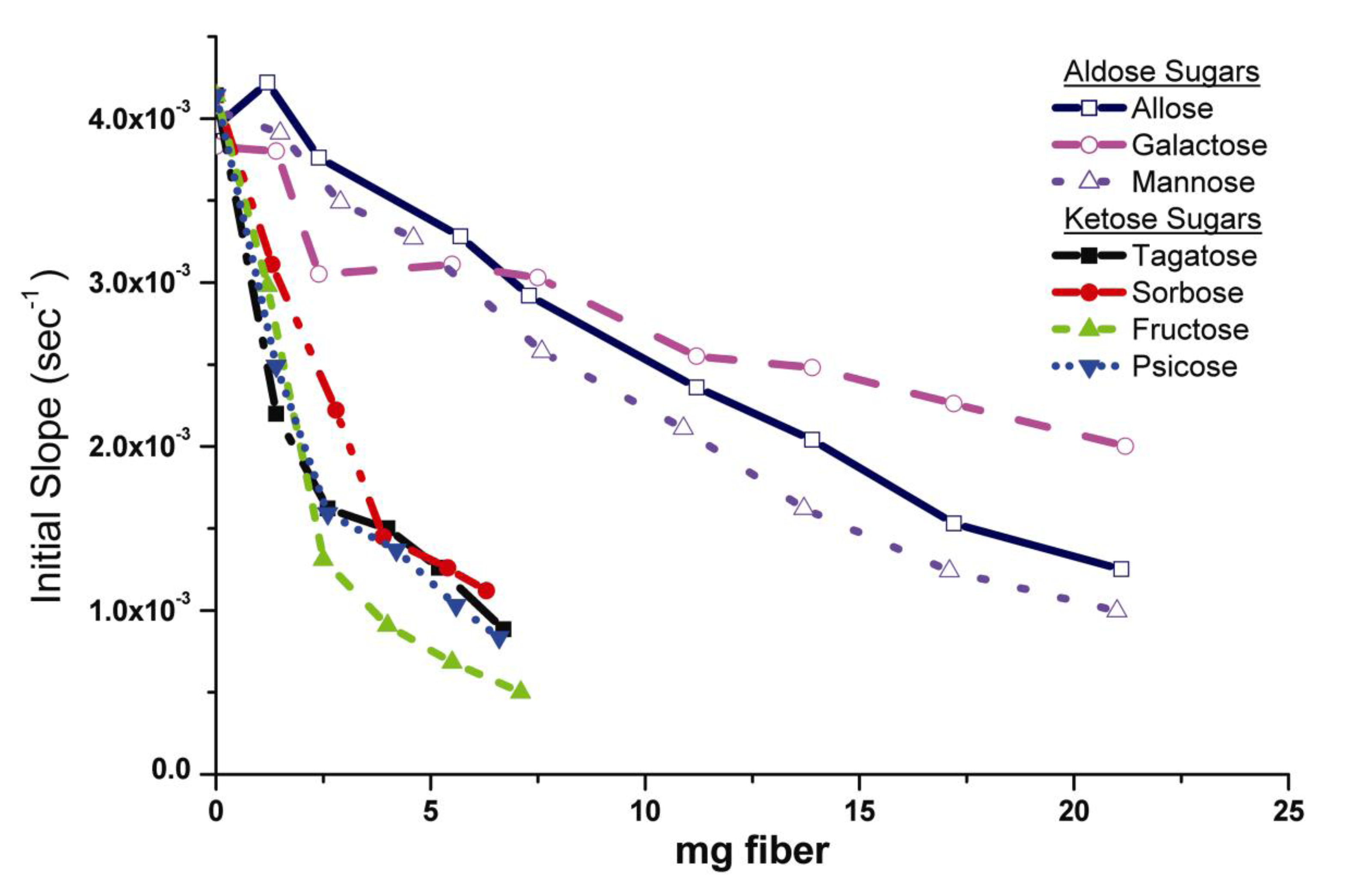
2.2.1. Mechanism of HNE Binding to Analogs
2.2.2. Monosaccharide Carbonyl Binding
2.2.3. Elastase/Monosaccharide-Cellulose Subsite Binding Motifs
3. Experimental Section
3.1. Preparation of Crosslinked Cotton with Monosaccharide-Cellulose Conjugates
3.2. Chromatographic Analysis of Monosaccharides on Cotton Gauze
3.3. Fourier Transform Infrared Spectroscopic Measurements
3.4. Enzyme Assay
4. Conclusions
References
- Falanga, V. Occlusive wound dressings. Why, when, which? Arch. Dermatol. 1988, 124, 872–877. [Google Scholar] [CrossRef]
- Hilton, J.R.; William, D.T.; Beuker, B.; Miller, D.R.; Harding, K.G. Wound dressings in diabetic foot disease. Clin. Infect. Dis. 2004, 39, S100–S103. [Google Scholar] [CrossRef]
- Miraftab, M.; Qlao, Q.; Kennedy, J.F.; Anand, S.C.; Groocock, M.R. Fibres for wound dressings based on mixed carbohydrate polymer fibres. Carbohydr. Polym. 2003, 53, 225–231. [Google Scholar] [CrossRef]
- Van der Weyden, E.A. Treatment of a venous leg ulcer with a honey alginate dressing. Br. J. Community Nurs. 2005, 10, S21–S27. [Google Scholar]
- Kirker, K.R.; Luo, Y.; Nielson, J.H.; Shelby, J.; Prestweich, G.D. Glycosaminoglycan hydrogel films as bio-interactive dressings for wound healing. Biomaterials 2002, 23, 3661–3671. [Google Scholar] [CrossRef]
- Garg, T.; Singh, O.; Arora, S.; Murthy, R.S.R. Scaffold: A novel carrier for cell and drug delivery. Crit. Rev. Ther. Drug Carrier Syst. 2012, 29, 1–63. [Google Scholar] [CrossRef]
- Voigt, J.; Driver, V.R. Hyaluronic acid derivatives and their healing effect on burns, epithelial surgical wounds, and chronic wounds: A systematic review and meta-analysis of randomized controlled trials. Wound Repair Regen. 2012, 20, 317–331. [Google Scholar] [CrossRef]
- Edwards, J.V. Future Structure and Properties of Mechanism-Based Wound Dressings. In Modified Fibers with Medical and Specialty Applications; Edwards, J.V., Buschle-Diller, G., Goheen, S.C., Eds.; Springer: Dordrecht, The Netherlands, 2006; pp. 11–33. [Google Scholar]
- Delatte, S.J.; Evans, J.; Hebra, A.; Adamson, W.; Othersen, H.B.; Tagge, E.P.; Hardin, W.; Priebe, C.; Winthrop, A. Effectiveness of beta-glucan collagen for treatment of partial-thickness burns in children. J. Pediatr. Surg. 2001, 36, 113–118. [Google Scholar] [CrossRef]
- Rathinamoorthy, R.; Sasikala, L. Polysaccharide fibers in wound management. Int. J. Pharm. Pharm. Sci. 2011, 3, 38–44. [Google Scholar]
- Backdahl, H.; Helenlus, G.; Bodin, A.; Nannmark, U.; Johansson, B.R.; Risber, B.; Gatenholm, P. Mechanical properties of bacterial cellulose and interactions with smooth muscle cells. Biomaterials 2006, 27, 2141–2149. [Google Scholar]
- Wiegand, C.; Elsner, P.; Hipler, U.-C.; Klemm, D. Protease and ROS activities influenced by a composite of bacterial cellulose and collagen type I in vitro. Cellulose 2006, 13, 689–696. [Google Scholar] [CrossRef]
- Cullen, B.; Smith, R.; McCulloch, E.; Silcock, D.; Morrison, L. Mechanism of action of PROMOGRAN, a protease modulating matrix, for the treatment of diabetic foot ulcers. Wound Repair Regen. 2002, 10, 16–25. [Google Scholar] [CrossRef]
- Szczesniak, M.; Kubis, A. The influence of hydrophilizing agents on gel formation rate of cellulose derivatives. Part 3: Effect of hydrophilizing agents and of polymer type on the release rate of hydrocortisone form xerogel dressing. Pharmazie 1993, 48, 926–927. [Google Scholar]
- Costache, M.C.; Qu, H.; Ducheyne, P.; Devore, D.I. Polymer-xerogel composites for controlled release wound dressings. Biomaterials 2010, 31, 6336–6343. [Google Scholar] [CrossRef]
- Wollina, U.; Abdel-Naser, M.B.; Verma, S. Skin physiology and textiles – Consideratoin of basic interactions. Curr. Probl. Dermatol. 2006, 33, 1–16. [Google Scholar] [CrossRef]
- Thomas, S. Wound Management and Dressings; The Pharmaceutical Press: London, UK, 1990; pp. 1–197. [Google Scholar]
- Hashimoto, T.; Suzuki, Y.; Tanihara, M.; Kakimaru, Y.; Suzuki, K. Development of alginate wound dressings with hybrid peptides derived from lamin and elastin. Biomaterials 2004, 25, 1407–1414. [Google Scholar] [CrossRef]
- Edwards, J.V.; Bopp, A.F.; Batiste, S.L.; Goynes, W.R. Human Neutrophil Elastase Innhibition with a Novel Cotton-Alginate Wound Dressing Formulation. J. Biomed. Mater. Res. Part A 2003, 66, 433–440. [Google Scholar]
- Dumville, J.C.; Deshpande, S.; O’Meara, S.; Speak, K. Alginate dressings for healing diabetic foot ulcers. Chochrane Database of Syst. Rev. 2012, 2. [Google Scholar] [CrossRef]
- Kato, Y.; Onishi, H.; Machida, Y. Application of chitin and chitosan derivatives in the pharmaceutical field. Curr. Pharm. Biotechnol. 2003, 4, 303–309. [Google Scholar] [CrossRef]
- Jayakumar, R.; Prabaharan, M.; Sudheesh Kumar, P.T.; Nair, S.V.; Tamura, H. Biomaterials based on chitin and chitosan in wound dressing applications. Biotechnol. Adv. 2011, 29, 322–337. [Google Scholar] [CrossRef]
- Madhumathi, K.; Sudheesh Kumar, P.T.; Abhilash, S.; Sreeja, V.; Tamura, H.; Manzoor, K.; Nair, S.V.; Jayakumar, R. Development of novel chitin/nanosilver composite scaffolds for wound dressing applications. J. Mater. Sci: Mater. Med. 2010, 21, 807–813. [Google Scholar] [CrossRef]
- Kashyab, N.; Kumar, N.; Kumar, M.N.V.R. Hydrogels for pharmaceutical and biomedical applications. Crit. Rev. TherDrug Carrier Syst. 2005, 22, 107–149. [Google Scholar] [CrossRef]
- Peng, H.T.; Shek, P.N. Novel wound sealants: Biomaterials and applications. Expert Rev. Med. Devices 2010, 7, 639–659. [Google Scholar] [CrossRef]
- Chaby, G.; Senet, P.; Vaneau, M.; Martel, P.; Guillaume, J.-C.; Meaume, S.; Teot, L.; Debure, C.; Dompmartin, A.; Bachelet, H.; et al. Dressing for acute and chronic wounds: A systematic review. Arch Dermatol. 2007, 143, 1297–1304. [Google Scholar] [CrossRef]
- Edwards, J.V.; Yager, D.R.; Cohen, I.K.; Diegelmann, R.F.; Montante, S.; Bertoniere, N.; Bopp, A.F. Modified cotton gauze dressings that selectively absorb neutrophil elastase activity in solution. Wound Repair Regen. 2001, 9, 50–58. [Google Scholar] [CrossRef]
- Edwards, J.V.; Eggleston, G.; Yager, D.R.; Cohen, I.K.; Diegelmann, R.F.; Bopp, A.F. Design, preparation and assessment of citrate-linked monosaccharide cellulose conjugates with elastase-lowering activity. Carbohydr. Polym. 2002, 50, 305–314. [Google Scholar] [CrossRef]
- Edwards, J.V.; Howley, P.S. Human neutrophil elastase and collagenase sequestration with phosphorylated cotton wound dressings. J. Biomed. Mater. Res. Part A 2007, 82, 446–454. [Google Scholar] [CrossRef]
- Yager, D.; Nwomeh, B. The proteolytic environment of chronic wounds. Wound Repair Regen. 1999, 7, 433–441. [Google Scholar] [CrossRef]
- Barros, S.C.; Martins, J.A.; Marcos, J.C.; Cavaco-Paulo, A. Characterization of potential elastase inhibitor-peptides regulated by a molecular switch for wound dressing applications. Enzyme Microb. Technol. 2012, 50, 107–114. [Google Scholar] [CrossRef] [Green Version]
- Edwards, J.V.; Batiste, S.L.; Gibbins, E.M.; Goheen, S.C. Synthesis and activity of NH2- and COOH-terminal elastase recognition sequences on cotton. J. Pept. Res. 1999, 54, 536–543. [Google Scholar] [CrossRef]
- Wiegand, C.; Abel, M.; Ruth, P.; Hipler, U.C. Superabsorbent polymer-containing wound dressings have a beneficial effect on wound healing by reducing PMN elastase concentration and inhibiting microbial growth. J. Mater. Sci. Mater. Med. 2011, 22, 2583–2590. [Google Scholar] [CrossRef]
- Meyer-Ingold, W.; Eichner, W.; Ettner, N.; Schink, M. Wound coverings for removal of interfering factors from wound fluid. U.S. Patent 6156334, 5 December 2000. [Google Scholar]
- Rayment, E.A.; Dargaville, T.R.; Shooter, G.K.; George, G.A.; Upton, Z. Attenuation of protease activity in chronic wound fluid with bisphosphonate-functionalised hydrogels. Biomaterials 2008, 29, 1785–1795. [Google Scholar] [Green Version]
- Vachon, D.J.; Yager, D.R. Novel sulfonated hydrogel composite with the ability to inhibit proteases and bacterial growth. J Biomed. Mater. Res. Part A 2006, 76, 35–43. [Google Scholar] [CrossRef]
- Eming, S.A.; Smola-Hess, S.; Kurschat, P.; Hirche, D.; Krieg, T.; Smola, H. A novel property of povidon-iodine: Inhibition of excessive protease levels in chronic non-healing wounds. J. Investig. Dermatol. 2006, 126, 2731–2733. [Google Scholar] [CrossRef]
- Edwards, J.V.; Bopp, A.F.; Batiste, S.; Ullah, A.J.; Cohen, I.K.; Diegelmann, R.F.; Montante, S.J. Inhibition of elastase by a synthetic cotton-bound serine protease inhibitor: In vitro kinetics and inhibitor release. Wound Repair Regen. 1999, 7, 106–108. [Google Scholar]
- Edwards, J.V.; Howley, P.; Davis, R.; Mashchak, A.; Goheen, S.C. Protease inhibition by oleic acid transfer from chronic wound dressing to albumin. Int. J. Pharm. 2007, 340, 42–51. [Google Scholar] [CrossRef]
- Wright, J.B.; Lam, K.; Buret, A.G.; Olson, M.E.; Burrell, R.E. Early healing events in a procine model of contaminated wounds: effects of nanocrystalline silver on matrix metalloproteinases, cell apoptosis, and healing. Wound Repair Regen. 2002, 10, 141–151. [Google Scholar] [CrossRef]
- Adhiraan, N.; Shanmugasundaram, N.; Babu, M. Gelatin microspheres cross-linked with EDC as a drug delivery system for doxycyline: development and characterization. J. Microencapsul. 2007, 24, 659–671. [Google Scholar] [CrossRef]
- Yang, C.Q. Characterizing ester crosslinkages in cotton cellulose with FT-IR photoacoustic spectroscopy. Text. Res. J. 1991, 61, 298–305. [Google Scholar] [CrossRef]
- Bode, W.; Wei, A.-Z.; Huber, R.; Meyer, E.; Travis, J.; Neumann, S. X-ray crystal structure of the complex of human leukocyte elastase (PMN elastase) and the third domain of the turkey ovomucoid inhibitor. EMBO J. 1986, 5, 2453–2458. [Google Scholar]
- He, X.-P.; Wang, X.-W.; Jin, X.-P.; Zhou, H.; Shi, X.-X.; Chen, G.-R.; Long, Y.-T. Epimeric monosaccharide-quinone hybrids on gold electrodes toward the electrochemical probing of specific carbohydrate-protein recognitions. J. Am. Chem. Soc. 2011, 133, 3649–3657. [Google Scholar]
- Toone, E.J. Structure and energetics of protein carbohydrate complexes. Curr. Opin. Struct. Biol. 1994, 4, 719–728. [Google Scholar] [CrossRef]
- Ledl, F.; Schleicher, E. New aspects of the Maillard reaction in foods and in the human body. Angew. Chem. 1990, 29, 565–594. [Google Scholar] [CrossRef]
- Blow, D.M. Structure and Mechanism of Chymotrypsin. Acc. Chem. Res. 1976, 9, 145–152. [Google Scholar] [CrossRef]
- Bode, W.; Meyer, E.; Powers, J.C. Human leukocyte and porcine pancreatic elastase: X-ray crystal structures, mechanism, substrate specificity and mechanism-based inhibitors. Biochemistry 1989, 28, 1951–1963. [Google Scholar] [CrossRef]
- Levit, D.A.S.; Schechter, I.; Berger, A. On the active site of elastase: Partial mapping by means of specific peptide substrates. FEBS Lett. 1970, 11, 281–283. [Google Scholar] [CrossRef]
- Ying, Q.L.; Rinehart, A.R.; Simon, S.R.; Cheronis, J.C. Inhibition of human leucocyte elastase by ursolic acid. Evidence for a binding site for pentacyclic triterpenes. Biochem. J. 1991, 277, 521–526. [Google Scholar]
- Meyer, E.F.; Clore, G.M.; Gronenborn, A.M.; Hansen, H.A.S. Analysis of an enzyme substrate complex. Biochemistry 1988, 27, 725–730. [Google Scholar]
- Edwards, P.D.; Bernstein, P.R. Synthetic inhibitors of elastase. Med. Res. Rev. 1994, 14, 127–194. [Google Scholar] [CrossRef]
- Navia, M.A.; Springer, J.P.; Lin, T.-Y.; Williams, H.R.; Firestone, R.A.; Pisano, J.M.; Doherty, J.B.; Finke, P.E.; Hoogsteen, K. Crystallographie study of a β-lactam inhibitor complex with elastase at 1.84 Å resolution. Nature 1987, 327, 79–82. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Edwards, J.V.; Caston-Pierre, S. Citrate-Linked Keto- and Aldo-Hexose Monosaccharide Cellulose Conjugates Demonstrate Selective Human Neutrophil Elastase-Lowering Activity in Cotton Dressings. J. Funct. Biomater. 2013, 4, 59-73. https://doi.org/10.3390/jfb4020059
Edwards JV, Caston-Pierre S. Citrate-Linked Keto- and Aldo-Hexose Monosaccharide Cellulose Conjugates Demonstrate Selective Human Neutrophil Elastase-Lowering Activity in Cotton Dressings. Journal of Functional Biomaterials. 2013; 4(2):59-73. https://doi.org/10.3390/jfb4020059
Chicago/Turabian StyleEdwards, Judson V., and Sonya Caston-Pierre. 2013. "Citrate-Linked Keto- and Aldo-Hexose Monosaccharide Cellulose Conjugates Demonstrate Selective Human Neutrophil Elastase-Lowering Activity in Cotton Dressings" Journal of Functional Biomaterials 4, no. 2: 59-73. https://doi.org/10.3390/jfb4020059




