Development of an Interaction Assay between Single-Stranded Nucleic Acids Trapped with Silica Particles and Fluorescent Compounds
Abstract
:1. Introduction
2. Experimental Procedures
2.1. Preparation of SiO2 Particles Modified with Aminoalkyl Chains
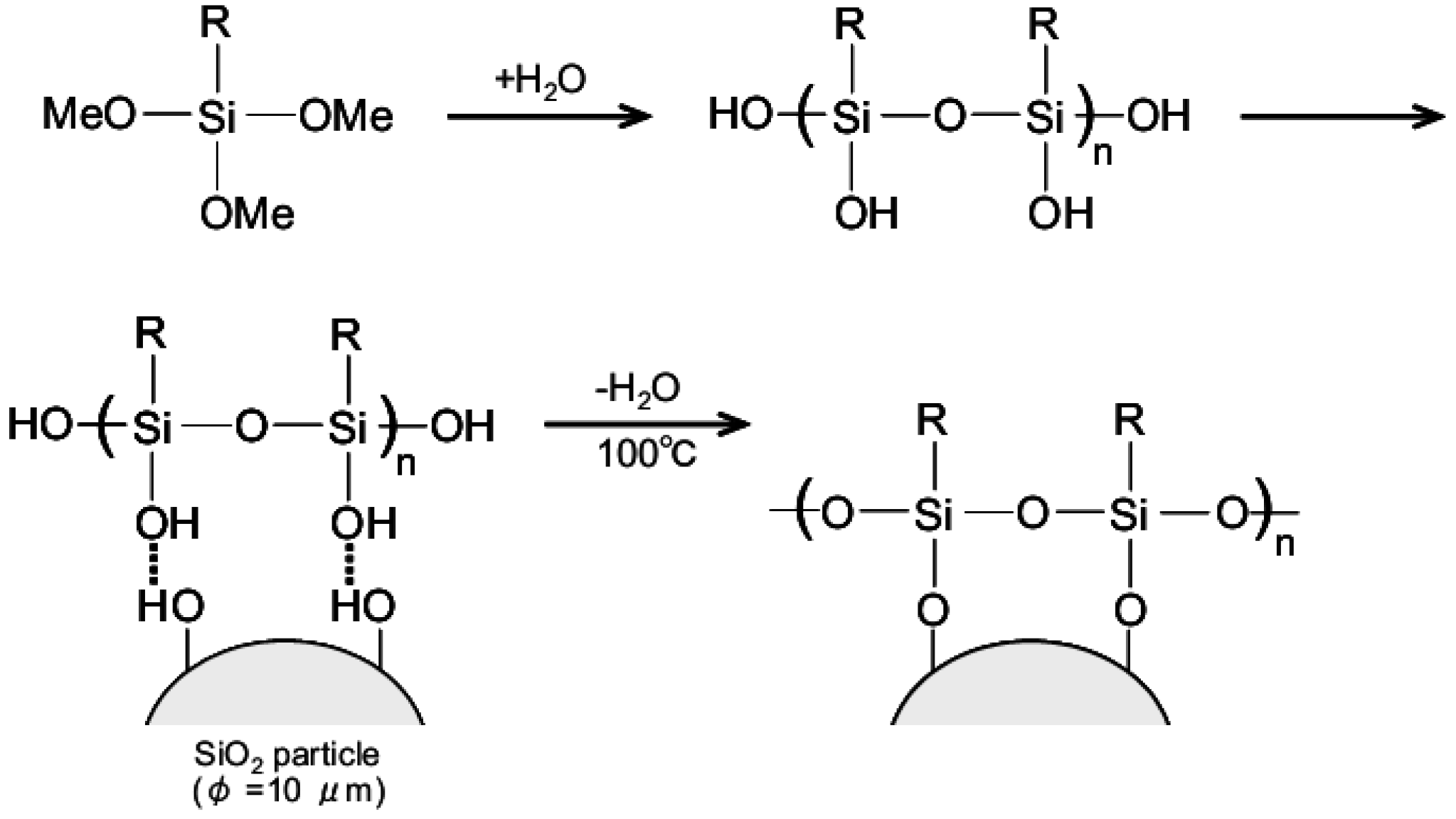
2.2. Analysis of Surface Chemical Structure
2.3. Trapping Procedure of Single-Stranded Nucleic Acids Using Aminoalkylated SiO2 Particles
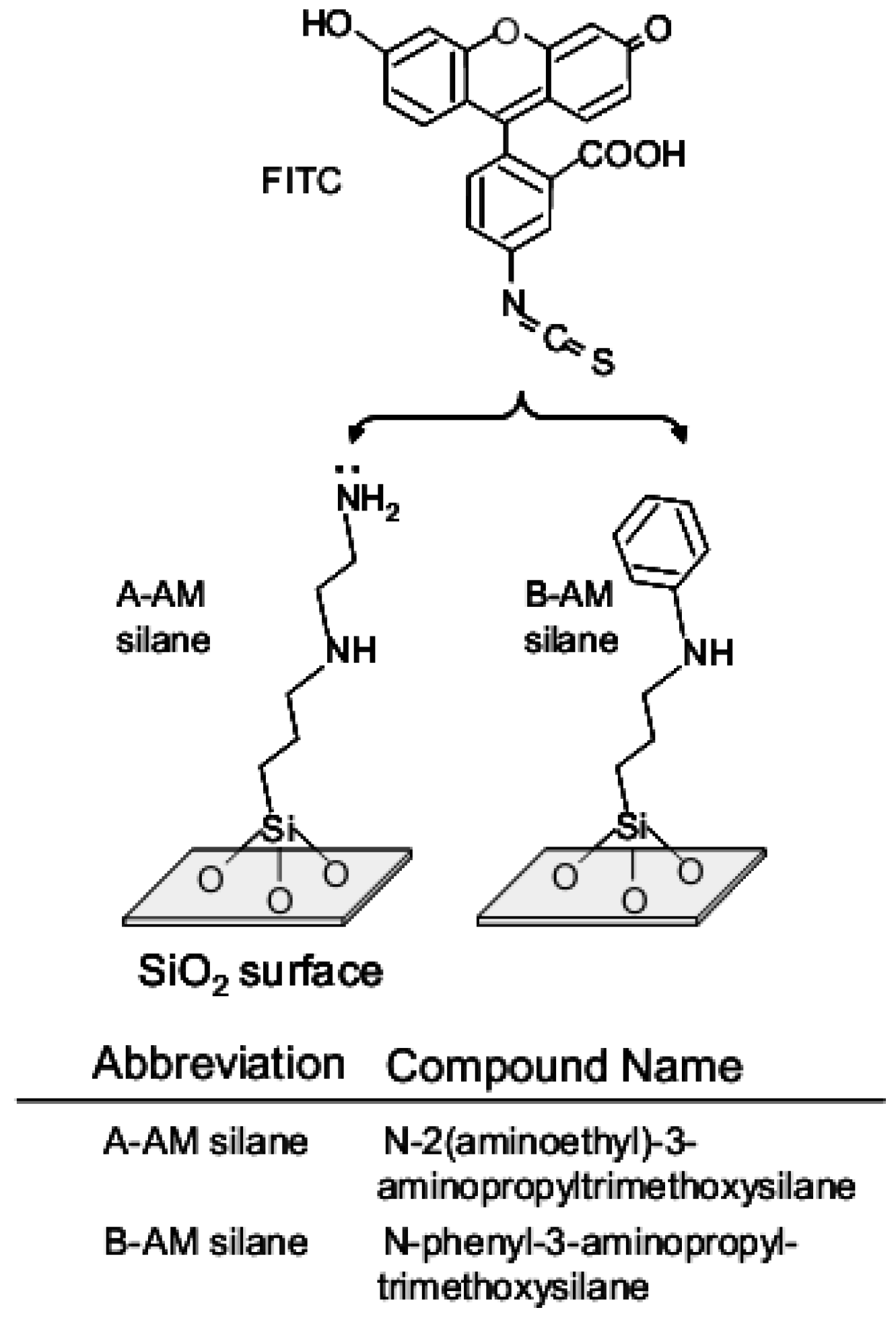
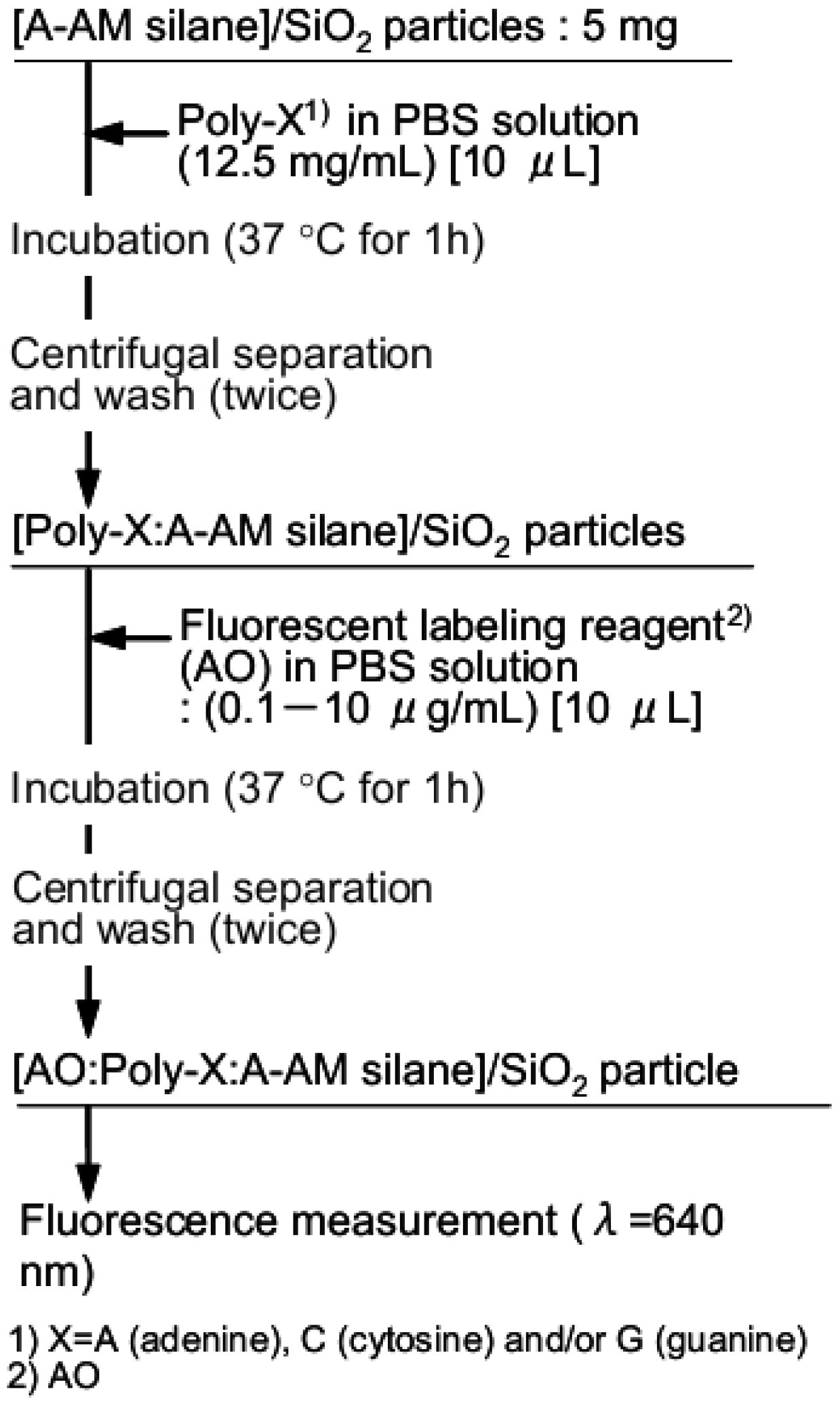
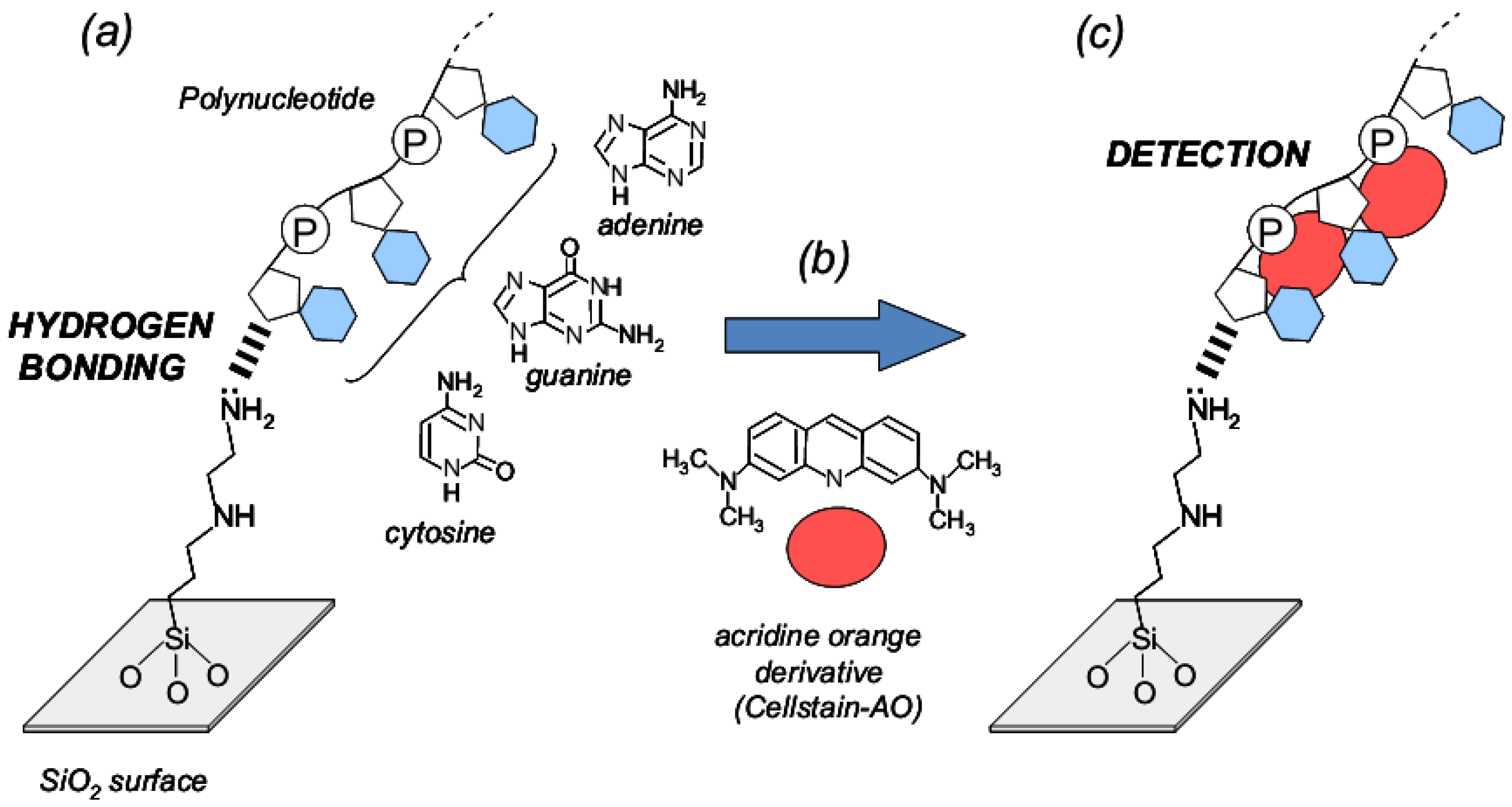
2.4. Molecular Orbital Calculations of Surface Model Structure of Aminoalkylated SiO2, Polynucleotides and Fluorescent Compound
3. Results
3.1. Chemical Structure of Aminoalkylated SiO2 Surface
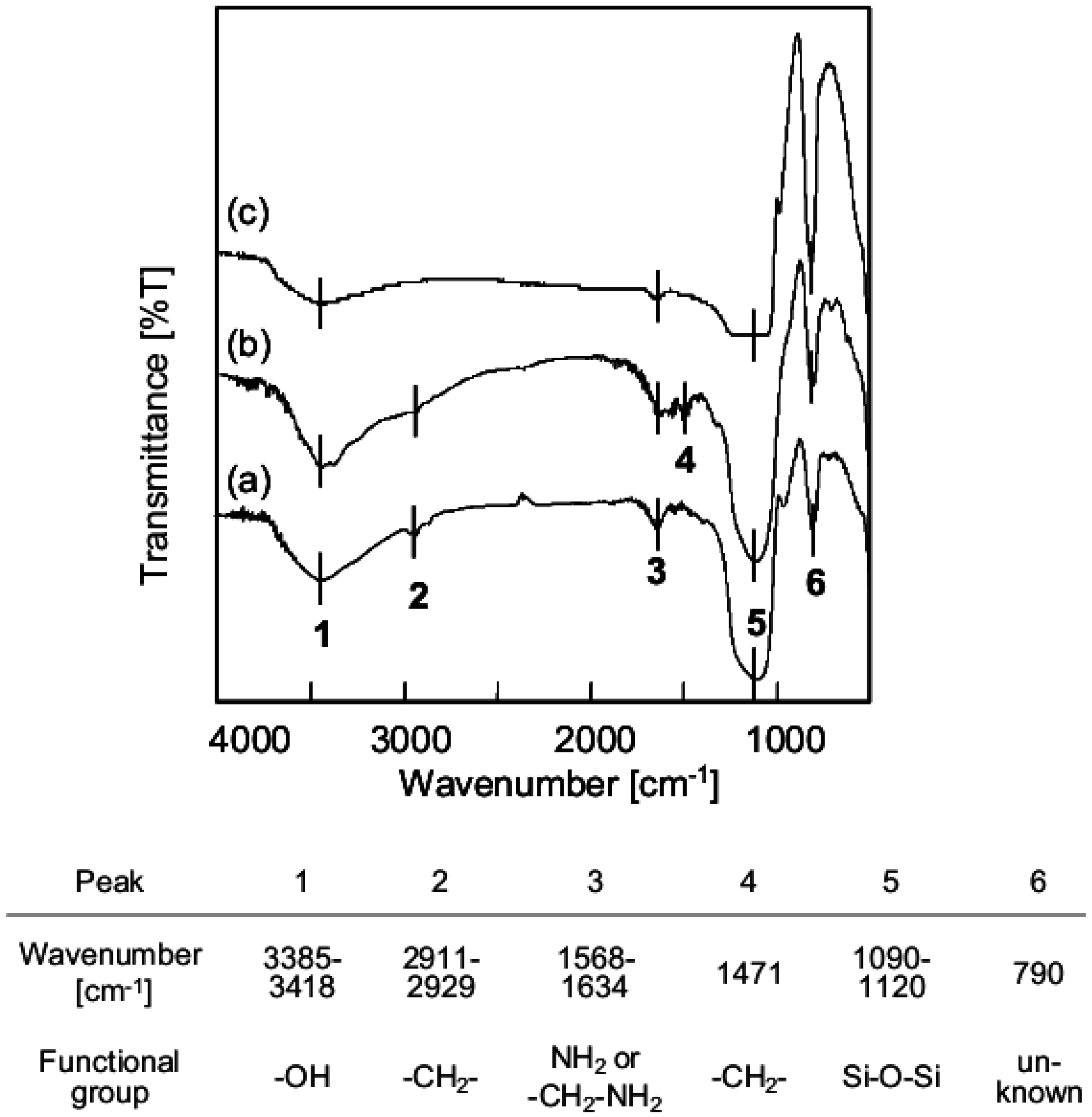
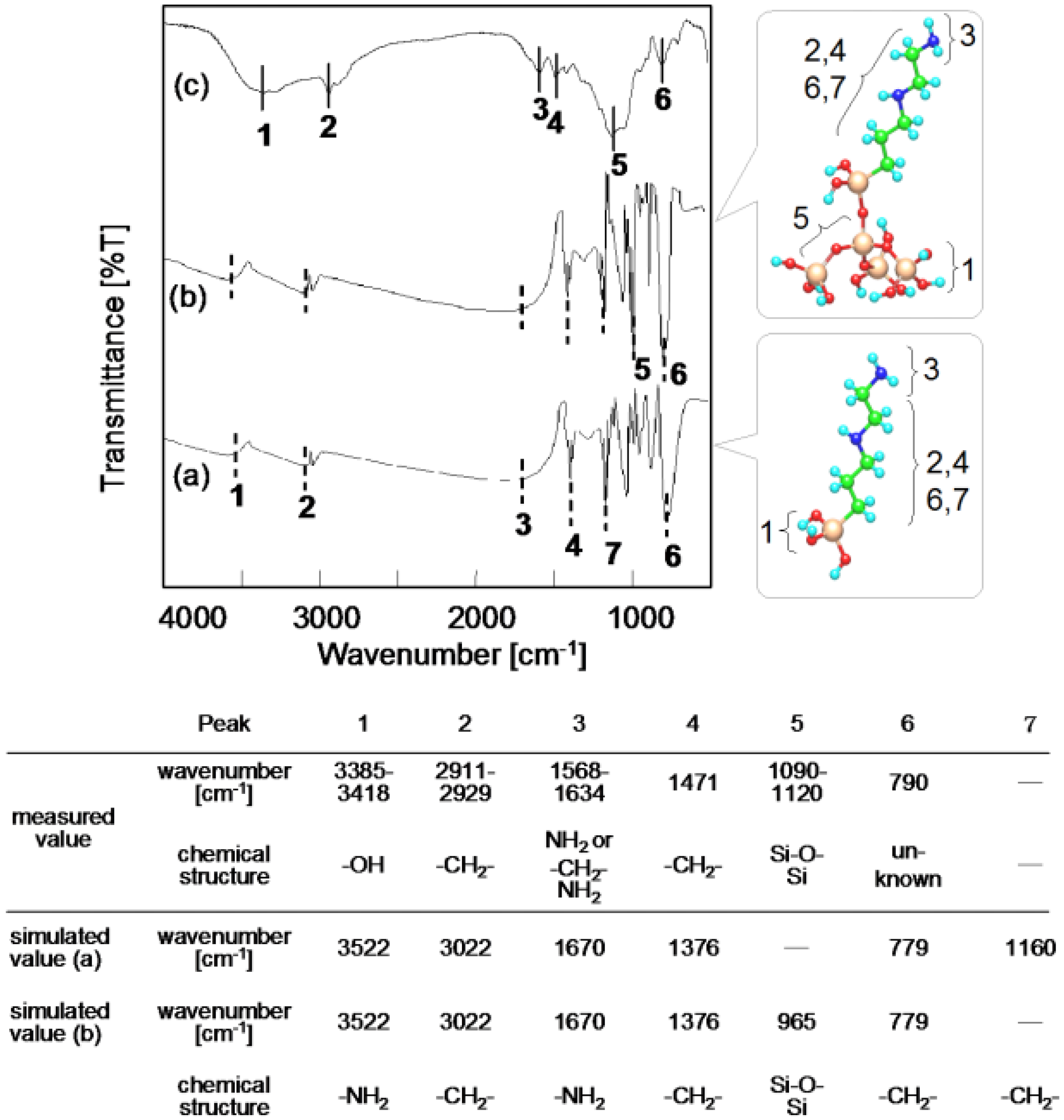
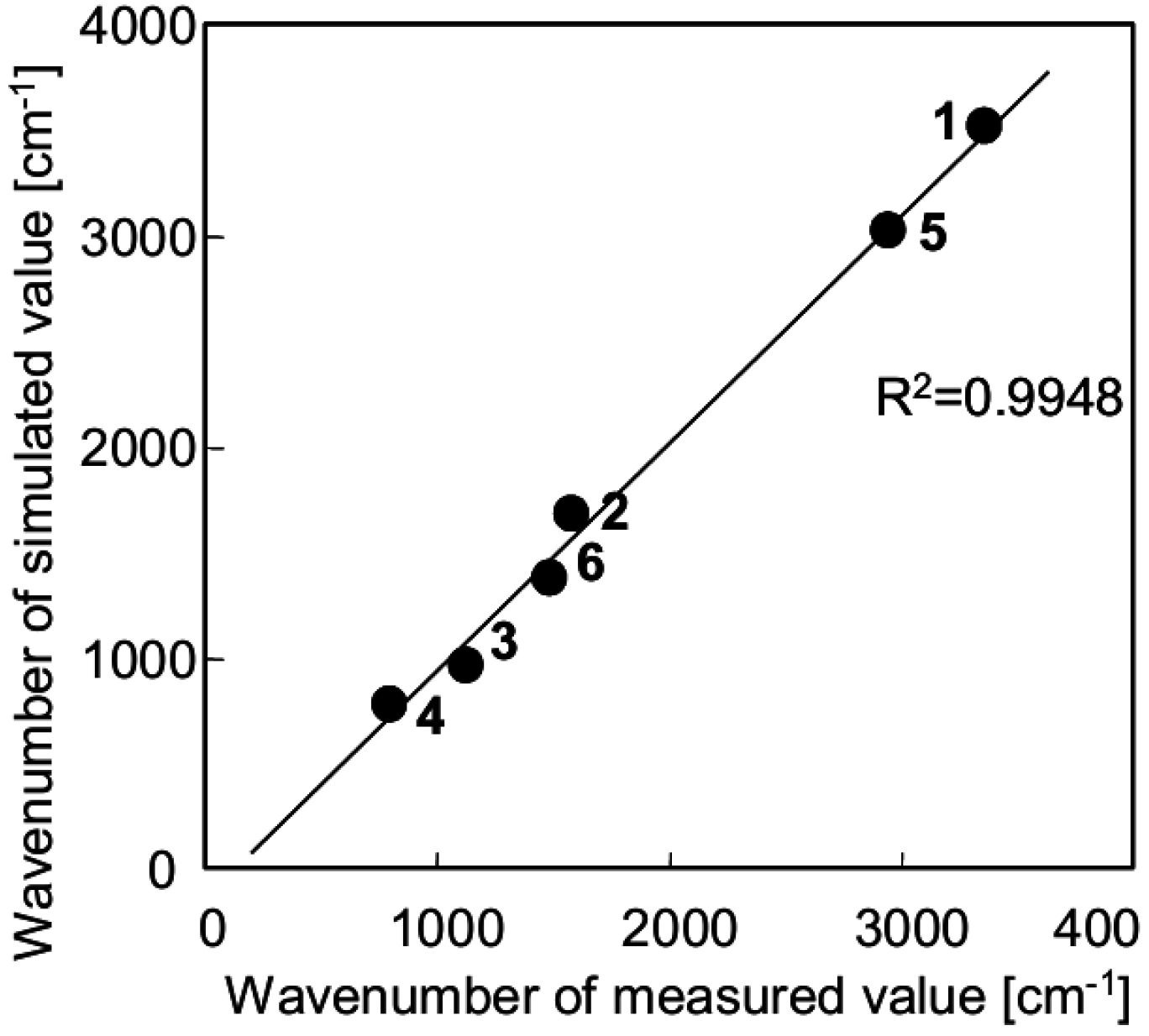
3.2. Orientation of Aminoalkyl Chains on the SiO2 Surface
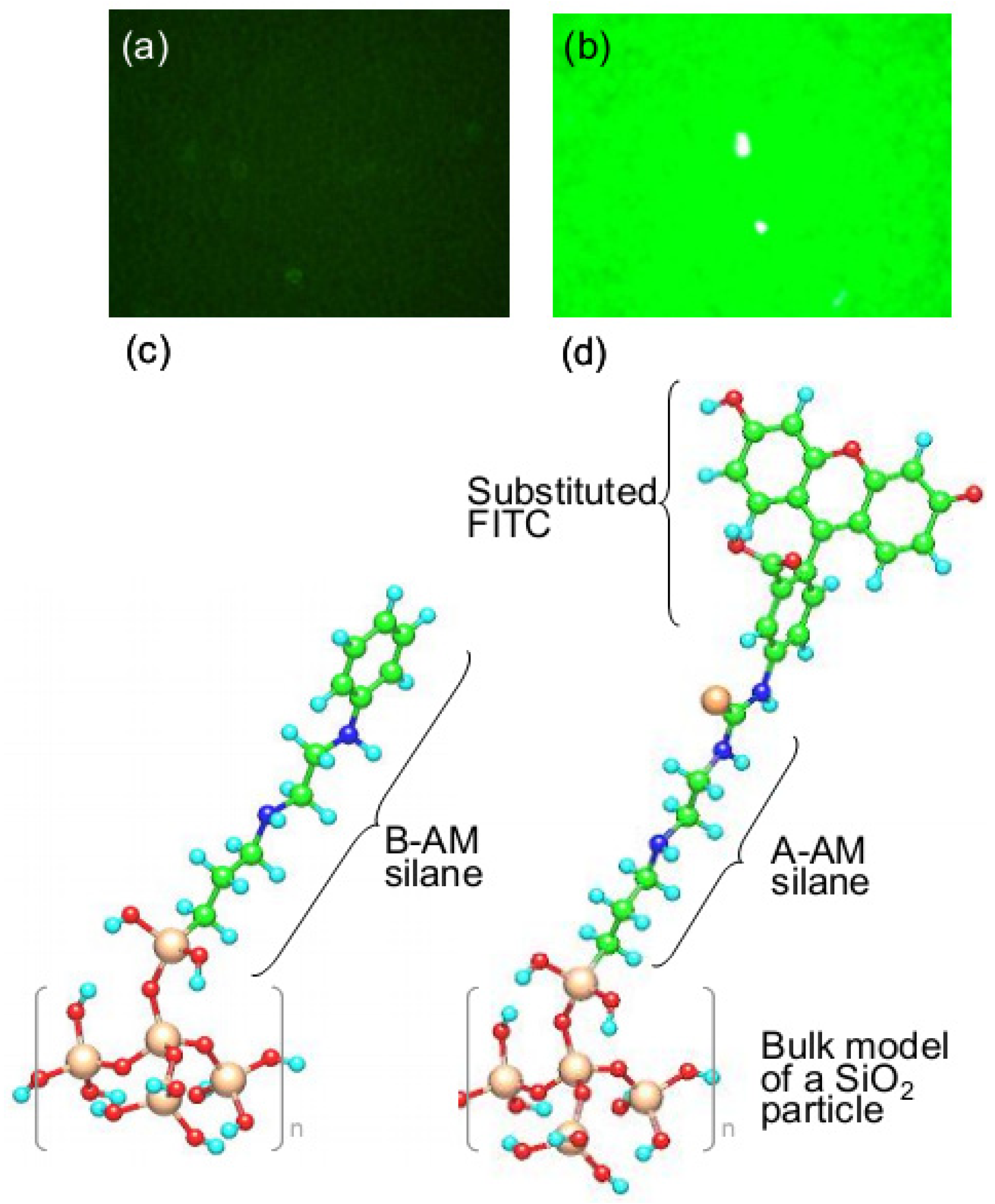
3.3. Evaluation for Interaction of Single-stranded Nucleic Acids and Fluorescence Compound

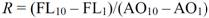
| single-stranded nucleic acid | [AO:Poly-X:A-AM silane]/SiO2 | [AO:Poly-X] | ||||
|---|---|---|---|---|---|---|
| FL10 | FL1 | R(1) | FL10 | FL1 | R(1) | |
| Poly-G | 177 | 45 | 14.7 | 141 | 37 | 11.6 |
| Poly-A | 144 | 41 | 11.4 | 130 | 20 | 12.2 |
| Poly-C | 54 | 27 | 3.0 | 40 | 27 | 1.4 |
3.4. Evaluation of the Interaction Between Nucleic-Acid Base in Nucleotides and Fluorescent Compound
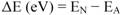
| Calculated MO | AO(1) | Poly-C(2) | Poly-A(3) | Poly-G(4) |
|---|---|---|---|---|
| HOMO [eV] | −7.695 | −9.140 | −8.840 | −8.820 |
| LUMO [eV] | −0.819 | −0.345 | −0.449 | −0.482 |
| ΔE | – | 7.350 | 7.246 | 7.213 |
4. Discussion
5. Conclusions
Abbreviations
| A-AM silane | N-2-aminoethyl-3-aminopropyltrimethoxysilane (KBM-603, Shin-Etsu Chemical Co., Ltd., Tokyo, Japan) |
| B-AM silane | N-phenyl-3-aminopropyltrimethoxysilane (KBM-573, Shin-Etsu Chemical Co., Ltd., Tokyo, Japan) |
| [A-AM silane]/SiO2 | silica particles surface-modified with aminoalkyl chains |
| [X-AM silane]/SiO2 | X = A: amino group and B: phenyl group |
| Poly-X | polynucleotides-X, X = A (adenine), G (guanine) and C (cytosine) |
| AO | derivatives of acridine orange (Cellstain-AO; Dojindo Laboratories, Kumamoto, Japan).) |
| FITC | fluorescein isothiocyanate |
| MO | Molecular orbital |
| HOMO | highest occupied molecular orbital |
| LUMO | lowest unoccupied molecular orbital |
Acknowledgements
References
- Ge, H. UPA, a universal protein array system for quantitative detection of protein-protein, protein-DNA, protein-RNA and protein-ligand interactions. Nucleic Acid Res. 2000, 28, e3. [Google Scholar] [CrossRef]
- Holt, L.J.; Bussow, K.; Walter, G.; Tomlinson, I.M. By-passing selection: Direct screening for antibody-antigen interactions using protein arrays. Nucleic Acid Res. 2000, 28, E72. [Google Scholar] [CrossRef]
- MacBeath, G.; Schreiber, S.L. Printing proteins as microarrays for high-throughput function determination. Science 2000, 289, 1760–1763. [Google Scholar]
- Zhu, H.; Klemic, J.F.; Chang, S.; Bertone, P.; Casamayor, A.; Klemic, K.G.; Smith, D.; Gerstein, M.; Reed, M.A.; Snyder, M. Analysis of yeast protein kinases using protein chips. Nat. Genetics. 2000, 26, 283–289. [Google Scholar] [CrossRef]
- Jung, H.J.; Hwang, I.; Kim, B.J.; Min, H.; Yu, H.; Lee, T.G.; Chung, T.D. Selective and direct immobilization of cysteinyl biomolecules by electrochemical cleavage of azo linkage. Langmuir 2010, 26, 15087–15091. [Google Scholar]
- Byeon, J.-Y.; Limpoco, F.T.; Bailey, R.C. Efficient bioconjugation of protein capture agents to biosensor surfaces using aniline-catalyzed hydrazone ligation. Langmuir 2010, 26, 15430–15435. [Google Scholar]
- Kimura, T.; Koumoto, K.; Mizu, M.; Sakurai, K.; Shinkai, S. Polysaccharaide-polynucleotide Interaction (XI); Novel separation system of RNAs by using schizophyllan appended column. Chem. Lett. 2002, 12, 1240–1241. [Google Scholar]
- Isoda, T.; Takahara, N.; Imanaga, H.; Imamura, R.; Hasegawa, S.; Noguchi, K.; Kimura, T. Measurement of non-electrolyte concentrations in an ion solution using a micro-electrode. Sens. Actuators B 2006, 120, 1–9. [Google Scholar] [CrossRef]
- Zhu, H.; Bilgin, M.; Bangham, R.; Hall, D.; Casamayor, A.; Bertone, P.; Lan, N.; Jansen, R.; Bidlingmaier, S.; Houfek, T.; Mitchell, T.; Miller, P.; Dean, R.A.; Gerstein, M.; Snyder, M. Global analysis of protein activities using proteome chips. Science 2001, 293, 2101–2105. [Google Scholar]
- Murata, M.; Nakayama, M.; Irie, H.; Yakabe, K.; Fukuma, K.; Katayama, Y.; Maeda, M. Novel biosensor for the rapid measurement of estrogen based on a ligand-receptor interaction. Anal. Sci. 2001, 17, 387–390. [Google Scholar] [CrossRef]
- Murata, M.; Yano, K.; Kuroki, S.; Suzutani, T.; Katayama, Y. Protein-immobilized electrode for rapid and convenient sensing of thyroid hormone receptor-ligand interaction. Anal. Sci. 2003, 19, 1569–1573. [Google Scholar] [CrossRef]
- Arkles, B. Hybrid polymers in the marketplace: Mix and match molecular building blocks to create better contact lenses, smoother-sailing ships, slippery surfaces, and more. Chem. Tech. 1999, 29, 7–14. [Google Scholar]
- Miyoshi, M.; Hara, K.; Yokoyama, I.; Tomita, G.; Taylor, I.W.; Milthorpe, B.K. An evaluation of DNA fluorochromes, staining techniques, and analysis for flow cytometr. I. Unperturbed cell populations. J. Histochem. Cytochem. 1980, 28, 1224–1232. [Google Scholar] [CrossRef]
- El-Naggar, A.K.; Batsakis, J.G.; Teague, K.; Garnsey, L.; Barlogie, B. Single- and double-stranded RNA measurements by flow cytometry in solid neoplasms. Cytometry 1991, 12, 330–335. [Google Scholar] [CrossRef]
- Stewart, J.J.P. MOPAC: A semiempirical molecular orbital program. J. Comput. Aided Mol. Des. 1990, 4, 1–103. [Google Scholar] [CrossRef]
- Fukui, K.; Yonezawa, T.; Shingu, H. A molecular orbital theory of reactivity in aromatic hydrocarbons. J. Chem. Phys. 1952, 20, 722–725. [Google Scholar] [CrossRef]
- Levit-Binnun, N.; Lindner, A.B.; Zik, O.; Eshhar, Z.; Moses, E. Quantitative detection of protein arrays. Anal. Chem. 2003, 75, 1436–1441. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Isoda, T.; Maeda, R. Development of an Interaction Assay between Single-Stranded Nucleic Acids Trapped with Silica Particles and Fluorescent Compounds. J. Funct. Biomater. 2012, 3, 601-614. https://doi.org/10.3390/jfb3030601
Isoda T, Maeda R. Development of an Interaction Assay between Single-Stranded Nucleic Acids Trapped with Silica Particles and Fluorescent Compounds. Journal of Functional Biomaterials. 2012; 3(3):601-614. https://doi.org/10.3390/jfb3030601
Chicago/Turabian StyleIsoda, T., and R. Maeda. 2012. "Development of an Interaction Assay between Single-Stranded Nucleic Acids Trapped with Silica Particles and Fluorescent Compounds" Journal of Functional Biomaterials 3, no. 3: 601-614. https://doi.org/10.3390/jfb3030601





