Cell Attachment to Hydrogel-Electrospun Fiber Mat Composite Materials
Abstract
:1. Introduction
2. Results and Discussion
| Material 1 | Topography | Surface Composition |
|---|---|---|
| PEG G | Gel | PEG |
| PEGPCL G | Gel | PEGPCL |
| PCL EFM | Fibers | PCL |
| PCL/PEGPCL 2 EFM | Fibers | PEGPCL |
| PEGPCL G-PCL EFM | Composite | PCL |
| PEGPCL G-PCL/PEGPCL2 EFM | Composite | PEGPCL |
2.1. Characterization of Electrospun Fiber Mats
| EFM | Water contact angle (°) | Average fiber width ω (µm) | Mean pore size (µm) | Porosity ε (%) | Available surface fraction (1 − Φ) |
|---|---|---|---|---|---|
| PCL | 135.2 ± 5.4 | 1.726 ± 0.102 | 1.517 ± 0.203 | 82 ± 2 | ~0.79 |
| PCL/PEGPCL | ~0 | 1.512 ± 0.141 | 1.557 ± 0.229 | 83 ± 2 | ~0.73 |

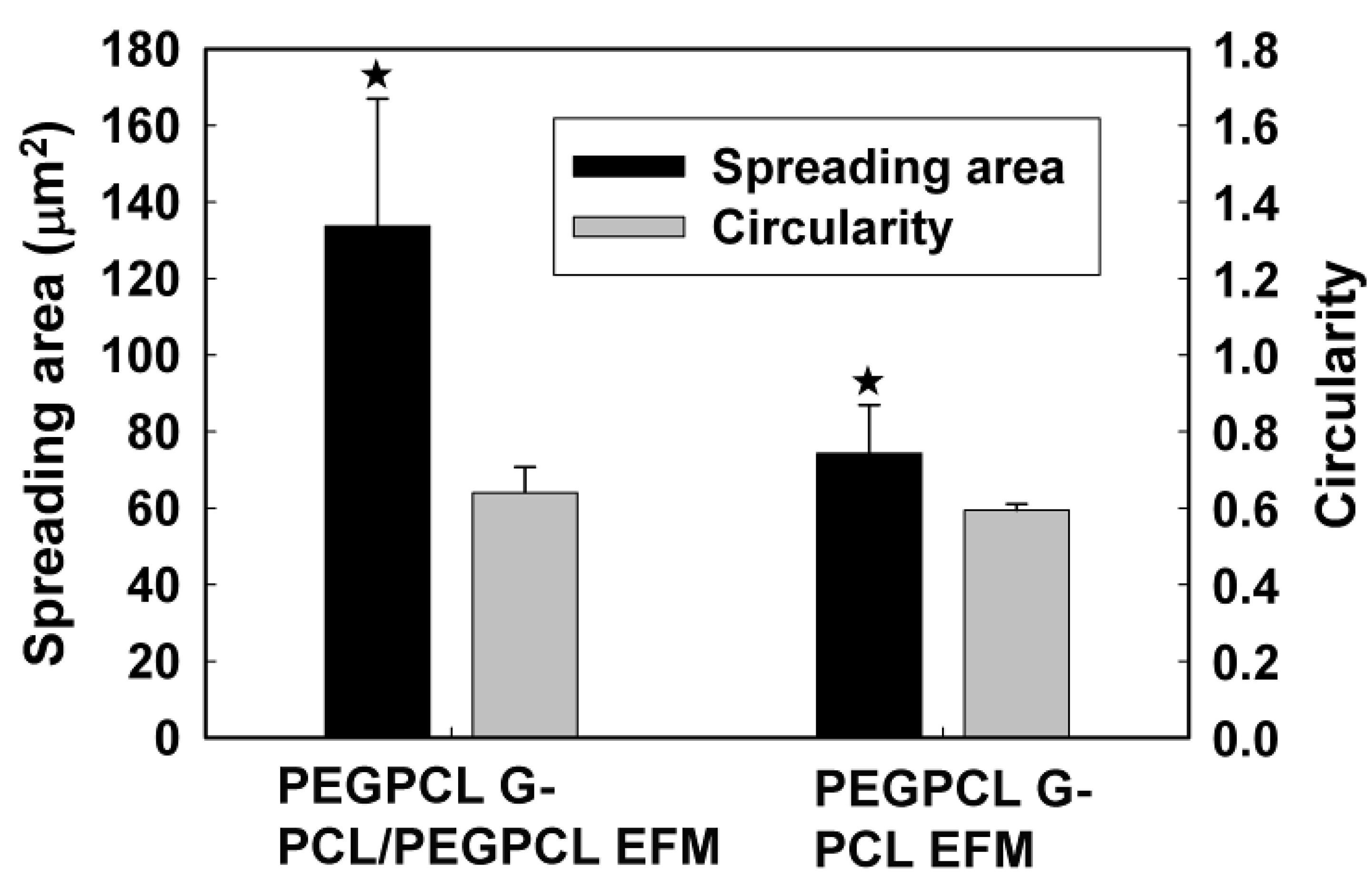
2.2. SK-N-SH Neuroblastoma Cell Attachment and Proliferation

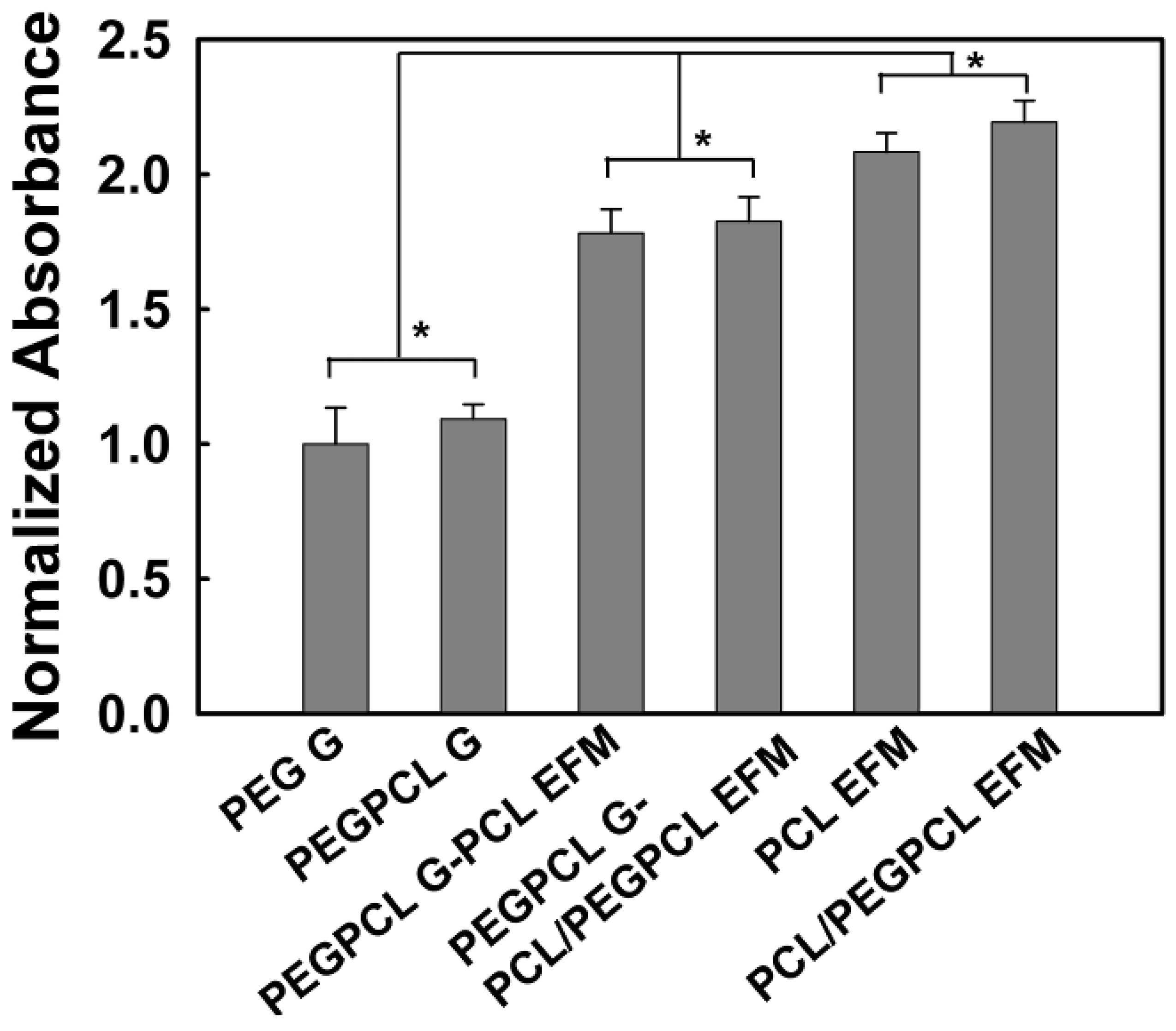


2.3. Rat Cortical Cell Attachment to Composite Materials
3. Experimental Section
3.1. Electrospun Fiber Mat Fabrication
3.1.1. PCL Mat Fabrication

3.1.2. PCL/PEGPCL Core/Shell Mat Fabrication
3.2. Electrospun Fiber Mat Characterization
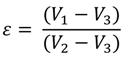
3.3. Synthesis of Diacryl-PEGPCL Copolymer
3.4. Formation of Hydrogels and Hydrogel-Electrospun Fiber Mat Composite Materials
3.4.1. PEG and PEGPCL Hydrogels
3.4.2. Composite Materials
3.5. SK-N-SH Cell Culture
3.6. SK-N-SH Cell Fluorescent Imaging and the MTT Proliferation Assay
3.7. Rat Cortical Neuron Culture
3.8. Cell Fixation and Scaffold SEM Imaging
3.9. Statistical Analysis
4. Conclusions
Acknowledgments
References
- Kashyap, N.; Kumar, N.; Kumar, M.N. Hydrogels for pharmaceutical and biomedical applications. Crit. Rev. Ther. Drug Carrier Syst. 2005, 22, 107–149. [Google Scholar] [CrossRef]
- Sill, T.J.; von Recum, H.A. Electro spinning: Applications in drug delivery and tissue engineering. Biomaterials 2008, 29, 1989–2006. [Google Scholar] [CrossRef]
- Peppas, N.A.; Hilt, J.Z.; Khademhosseini, A.; Langer, R. Hydrogels in biology and medicine: From molecular principles to bionanotechnology. Adv. Mater. 2006, 18, 1345–1360. [Google Scholar]
- Chew, S.Y.; Wen, Y.; Dzenis, Y.; Leong, K.W. The role of electrospinning in the emerging field of nanomedicine. Curr. Pharm. Des. 2006, 12, 4751–4770. [Google Scholar] [CrossRef]
- Murugan, R.; Ramakrishna, S. Nano-featured scaffolds for tissue engineering: A review of spinning methodologies. Tissue Eng. 2006, 12, 435–447. [Google Scholar] [CrossRef]
- Liang, D.; Hsiao, B.S.; Chu, B. Functional electrospun nanofibrous scaffolds for biomedical applications. Adv. Drug Deliv. Rev. 2007, 59, 1392–1412. [Google Scholar] [CrossRef]
- Yoo, H.S.; Kim, T.G.; Park, T.G. Surface-functionalized electrospun nanofibers for tissue engineering and drug delivery. Adv. Drug Deliv. Rev. 2009, 61, 1033–1042. [Google Scholar] [CrossRef]
- Lee, K.Y.; Mooney, D.J. Hydrogels for tissue engineering. Chem. Rev. 2001, 101, 1869–1879. [Google Scholar] [CrossRef]
- Han, N.; Rao, S.S.; Johnson, J.; Parikh, K.S.; Bradley, P.A.; Lannutti, J.J.; Winter, J.O. Hydrogel-electrospun fiber mat composite coatings for neural prostheses. Front. Neuroeng. 2011, 4, 1–8, Article 2.. [Google Scholar]
- Hayami, J.W.; Surrao, D.C.; Waldman, S.D.; Amsden, B.G. Design and characterization of a biodegradable composite scaffold for ligament tissue engineering. J. Biomed. Mater. Res. A 2010, 92, 1407–1420. [Google Scholar]
- Hong, Y.; Huber, A.; Takanari, K.; Amoroso, N.J.; Hashizume, R.; Badylak, S.F.; Wagner, W.R. Mechanical properties and in vivo behavior of a biodegradable synthetic polymer microfiber-extracellular matrix hydrogel biohybrid scaffold. Biomaterials 2011, 32, 3387–3394. [Google Scholar]
- McMahon, R.E.; Qu, X.; Jimenez-Vergara, A.C.; Bashur, C.A.; Guelcher, S.A.; Goldstein, A.S.; Hahn, M.S. Hydrogel-electrospun mesh composites for coronary artery bypass grafts. Tissue Eng. Part C Methods 2011, 17, 451–461. [Google Scholar] [CrossRef]
- Rnjak, J.; Li, Z.; Maitz, P.K.; Wise, S.G.; Weiss, A.S. Primary human dermal fibroblast interactions with open weave three-dimensional scaffolds prepared from synthetic human elastin. Biomaterials 2009, 30, 6469–6477. [Google Scholar]
- Xu, W.; Ma, J.; Jabbari, E. Material properties and osteogenic differentiation of marrow stromal cells on fiber-reinforced laminated hydrogel nanocomposites. Acta Biomater. 2010, 6, 1992–2002. [Google Scholar] [CrossRef]
- Abidian, M.R.; Corey, J.M.; Kipke, D.R.; Martin, D.C. Conducting-polymer nanotubes improve electrical properties, mechanical adhesion, neural attachment, and neurite outgrowth of neural electrodes. Small 2010, 6, 421–429. [Google Scholar]
- Alberts, B.; Bray, D.; Lewis, B.; Raff, M.; Roberts, K.; Watson, J.D. Molecular Biology of the Cell; Garland Publishing, Inc.: New York, NY, USA, 1996. [Google Scholar]
- Anderson, J.M. Inflammation, wound healing, and the foreign-body response. In Biomaterials Science: An Introduction to Materials in Medicine, 2nd; Ratner, B.D., Hoffman, A.S., Schoen, F.J., Lemons, J.E., Eds.; Elsevier: London, UK, 2004; pp. 296–304. [Google Scholar]
- Anderson, J.M. Biological responses to materials. Ann. Rev. Mater. Res. 2001, 31, 81–110. [Google Scholar] [CrossRef]
- Anderson, J.M.; Rodriguez, A.; Chang, D.T. Foreign body reaction to biomaterials. Semin. Immunol. 2008, 20, 86–100. [Google Scholar] [CrossRef]
- LeBaron, R.G.; Athanasiou, K.A. Extracellular matrix cell adhesion peptides: Functional applications in orthopedic materials. Tissue Eng. 2000, 6, 85–103. [Google Scholar] [CrossRef]
- Berthiaume, F.; Moghe, P.V.; Toner, M.; Yarmush, M.L. Effect of extracellular matrix topology on cell structure, function, and physiological responsiveness: Hepatocytes cultured in a sandwich configuration. FASEB J. 1996, 10, 1471–1484. [Google Scholar]
- Lannutti, J.; Reneker, D.; Ma, T.; Tomasko, D.; Farson, D.F. Electrospinning for tissue engineering scaffolds. Mat. Sci. Eng. C-Biomim. Supram. S. 2007, 27, 504–509. [Google Scholar] [CrossRef]
- Eugene, M. Polyethyleneglycols and immunocamouflage of the cells tissues and organs for transplantation. Cell Mol. Biol. (Noisy-le-grand) 2004, 50, 209–215. [Google Scholar]
- Zhu, Y.B.; Gao, C.Y.; Shen, J.C. Surface modification of polycaprolactone with poly(methacrylic acid) and gelatin covalent immobilization for promoting its cytocompatibility. Biomaterials 2002, 23, 4889–4895. [Google Scholar] [CrossRef]
- Koh, H.S.; Yong, T.; Chan, C.K.; Ramakrishna, S. Enhancement of neurite outgrowth using nano-structured scaffolds coupled with laminin. Biomaterials 2008, 29, 3574–3582. [Google Scholar]
- Moxon, K.A.; Kalkhoran, N.M.; Markert, M.; Sambito, M.A.; McKenzie, J.L.; Webster, J.T. Nanostructured surface modification of ceramic-based microelectrodes to enhance biocompatibility for a direct brain-machine interface. IEEE Trans. Biomed. Eng. 2004, 51, 881–889. [Google Scholar] [CrossRef]
- Biedler, J.L.; Helson, L.; Spengler, B.A. Morphology and growth, tumorigenicity, and cytogenetics of human neuroblastoma cells in continuous culture. Cancer Res. 1973, 33, 2643–2652. [Google Scholar]
- Eichhorn, S.J.; Sampson, W.W. Statistical geometry of pores and statistics of porous nanofibrous assemblies. J. R. Soc. Interface 2005, 2, 309–318. [Google Scholar] [CrossRef]
- Davis, K.A.; Burdick, J.A.; Anseth, K.S. Photoinitiated crosslinked degradable copolymer networks for tissue engineering applications. Biomaterials 2003, 24, 2485–2495. [Google Scholar] [CrossRef]
- Nam, J.; Johnson, J.K.; Lannutti, J.J.; Agarwal, S. Modulation of embryonic mesenchymal progenitor cell differentiation via control over pure mechanical modulus in electrospun nanofibers. Acta Biomater. 2011, 7, 1516–1524. [Google Scholar] [CrossRef]
- Wang, J.H.; Thampatty, B.P. An introductory review of cell mechanobiology. Biomech. Model. Mechanobiol. 2006, 5, 1–16. [Google Scholar] [CrossRef]
- Peyton, S.R.; Ghajar, C.M.; Khatiwala, C.B.; Putnam, A.J. The emergence of ECM mechanics and cytoskeletal tension as important regulators of cell function. Cell Biochem. Biophys. 2007, 47, 300–320. [Google Scholar] [CrossRef]
- Rao, S.S.; Han, N.; Winter, J.O. Polylysine-modified PEG-based hydrogels to enhance the neuro-electrode interface. J. Biomater. Sci. Polym. Ed. 2011, 22, 611–625. [Google Scholar] [CrossRef]
- Zhu, X.L.; Cui, W.G.; Li, X.H.; Jin, Y. Electrospun fibrous mats with high porosity as potential scaffolds for skin tissue engineering. Biomacromolecules 2008, 9, 1795–1801. [Google Scholar] [CrossRef]
- Guan, J.; Fujimoto, K.L.; Sacks, M.S.; Wagner, W.R. Preparation and characterization of highly porous, biodegradable polyurethane scaffolds for soft tissue applications. Biomaterials 2005, 26, 3961–3971. [Google Scholar] [CrossRef]
- Sawhney, A.S.; Pathak, C.P.; Hubbell, J.A. Bioerodible hydrogels based on photopolymerized poly(ethylene glycol)-co-poly(alpha-hydroxy acid) diacrylate macromers. Macromolecules 1993, 26, 581–587. [Google Scholar]
- Han, N.; Johnson, J.; Lannutti, J.J.; Winter, J.O. Hydrogel-electrospun fiber composite materials for hydrophilic protein release. J. Control. Release 2012, 158, 165–170. [Google Scholar] [CrossRef]
- Stichel, C.C.; Muller, H.W. Dissociated cell-culture of rat cerebral cortical-neurons in serum-free, conditioned media- gaba-immunopositive neurons. Dev. Brain Res. 1991, 64, 145–154. [Google Scholar] [CrossRef]
- Dichter, M.A. Rat cortical-neurons in cell-culture—culture methods, cell morphology, electrophysiology, and synapse formation. Brain Res. 1978, 149, 279–293. [Google Scholar] [CrossRef]
- Chung, T.W.; Liu, D.Z.; Wang, S.Y.; Wang, S.S. Enhancement of the growth of human endothelial cells by surface roughness at nanometer scale. Biomaterials 2003, 24, 4655–4661. [Google Scholar] [CrossRef]
- Kim, D.H.; Kim, P.; Song, I.; Cha, J.M.; Lee, S.H.; Kim, B.; Suh, K.Y. Guided three-dimensional growth of functional cardiomyocytes on polyethylene glycol nanostructures. Langmuir 2006, 22, 5419–5426. [Google Scholar]
- Huang, Y.; Siewe, M.; Madihally, S.V. Effect of spatial architecture on cellular colonization. Biotechnol. Bioeng. 2006, 93, 64–75. [Google Scholar] [CrossRef]
- Curtis, A.; Wilkinson, C. New depths in cell behaviour: reactions of cells to nanotopography. Biochem. Soc. Symp. 1999, 65, 15–26. [Google Scholar]
- Ma, Z.W.; Kotaki, M.; Inai, R.; Ramakrishna, S. Potential of nanofiber matrix as tissue-engineering scaffolds. Tissue Eng. 2005, 11, 101–109. [Google Scholar] [CrossRef]
- Matsuzawa, M.; Potember, R.S.; Stenger, D.A.; Krauthamer, V. Containment and growth of neuroblastoma-cells on chemically patterned substrates. J. Neurosci. Meth. 1993, 50, 253–260. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Han, N.; Johnson, J.K.; Bradley, P.A.; Parikh, K.S.; Lannutti, J.J.; Winter, J.O. Cell Attachment to Hydrogel-Electrospun Fiber Mat Composite Materials. J. Funct. Biomater. 2012, 3, 497-513. https://doi.org/10.3390/jfb3030497
Han N, Johnson JK, Bradley PA, Parikh KS, Lannutti JJ, Winter JO. Cell Attachment to Hydrogel-Electrospun Fiber Mat Composite Materials. Journal of Functional Biomaterials. 2012; 3(3):497-513. https://doi.org/10.3390/jfb3030497
Chicago/Turabian StyleHan, Ning, Jed K. Johnson, Patrick A. Bradley, Kunal S. Parikh, John J. Lannutti, and Jessica O. Winter. 2012. "Cell Attachment to Hydrogel-Electrospun Fiber Mat Composite Materials" Journal of Functional Biomaterials 3, no. 3: 497-513. https://doi.org/10.3390/jfb3030497
APA StyleHan, N., Johnson, J. K., Bradley, P. A., Parikh, K. S., Lannutti, J. J., & Winter, J. O. (2012). Cell Attachment to Hydrogel-Electrospun Fiber Mat Composite Materials. Journal of Functional Biomaterials, 3(3), 497-513. https://doi.org/10.3390/jfb3030497




