A Review of Gene Delivery and Stem Cell Based Therapies for Regenerating Inner Ear Hair Cells
Abstract
:1. Introduction
2. Hair Cells, Supporting Cells and Their Function
2.1. Hair Cells
| Factor | Inner hair cells | Outer hair cells | Ref. |
|---|---|---|---|
| Arrangement | Arranged in a single row | Arranged in three parallel rows | [31,32,33] |
| Shape | Round and small | Long and slim | [10] |
| Function | Transduce mechanical energy to neural signals | Appear to impact and regulate the sensitivity of the cochlea over a range of 32 dB | [10] |
| Effects | Sensory Neural Hearing loss | Alter properties of cochlear input to the brain | [34] |
| Approximate number | 3,000 to 3,500 | 9,000 to 12,000 | [32,33] |
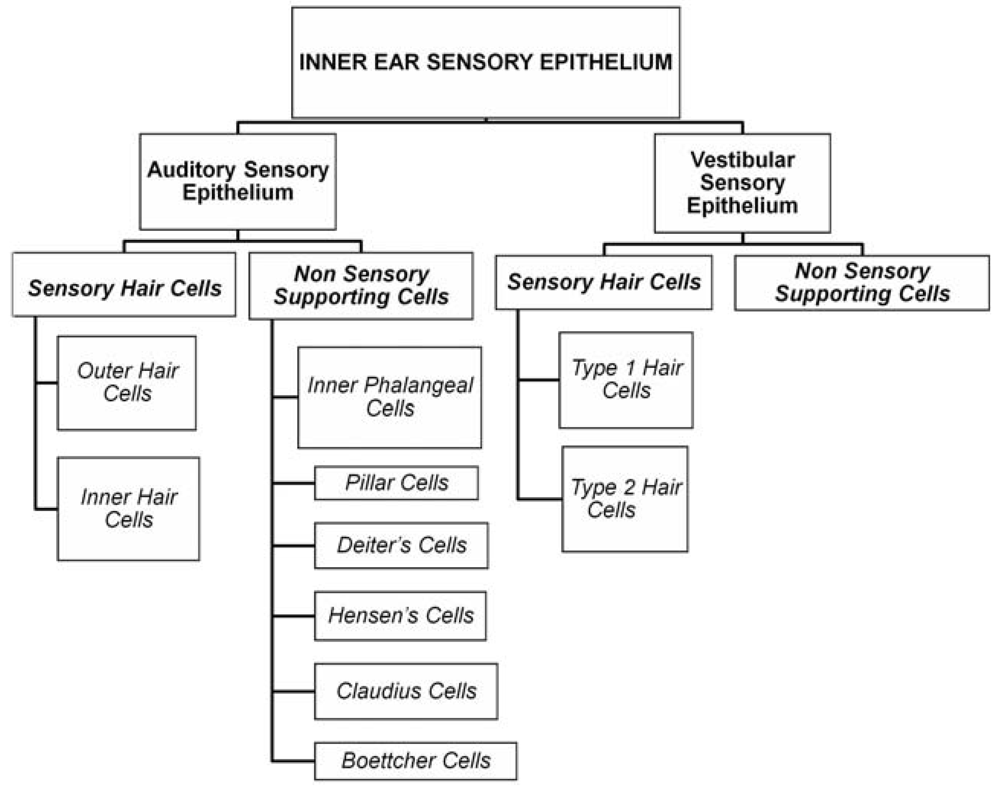
2.1.1. Hair Cell Structure
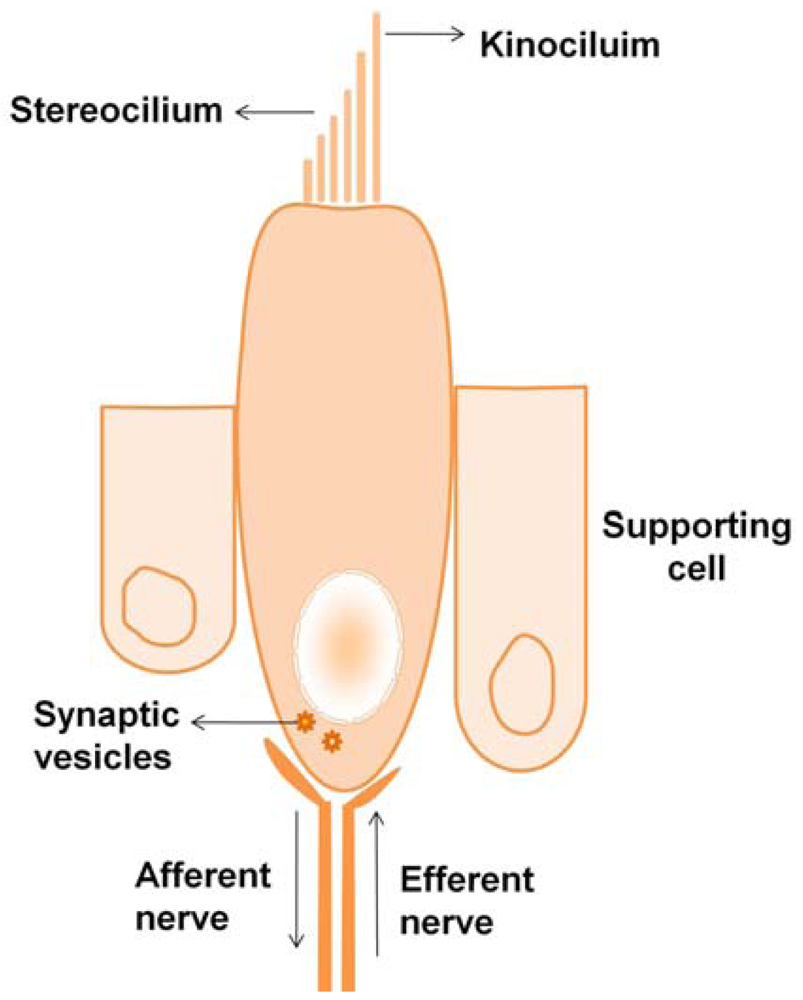
2.1.2. Transduction in Hair Cells
2.2. Supporting Cells
2.3. Summary
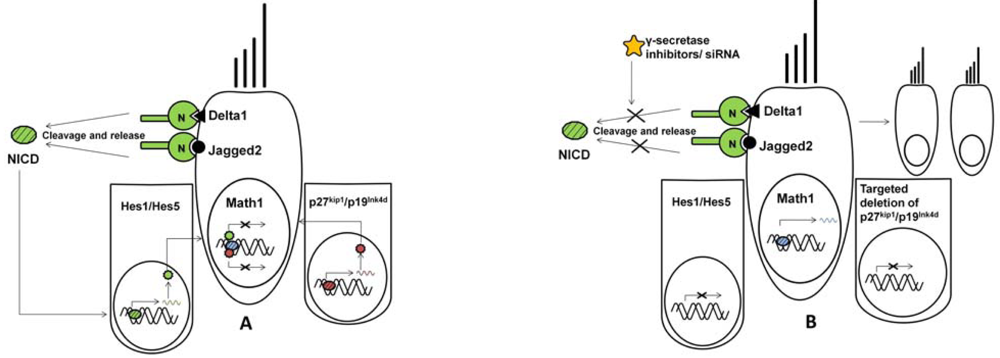
3. Essential Genes in Hair Cell Differentiation
| Gene | Role | Reference |
|---|---|---|
| Math1 | Also known as Atoh1. Primary gene responsible for hair cell differentiation. Other homologues include Hath1, Cath1 and Xath1. | [47,49,53,54,55] |
| Hes1 and Hes5 | Mammalian homologues of Hairy and Enhancer-of-split gene. Expressed in supporting cells and known to be negative regulators of Math1. However, a balance between Hes1/Hes5 is required to control the production of supernumerary hair cells and normal development of inner ear. | [56,57] |
| Sox2 | Responsible for development of inner ear sensory epithelium and is expressed in supporting cells and inner ear progenitors. Acts upstream of Math1 and maintains mitotic and transdifferentiation functions of supporting cells. | [59,60,61] |
| Jag2 | Member of the notch signaling pathway. Expressed in supporting cells of auditory and vestibular system. Required for the normal development of inner ear sensory organs. | [19,61,62] |
| BETA2/Neurod1 | Expressed in neurons and neural precursor cells. Promotes the formation of ganglion neurons in the cochlea. Absence of BETA2/NeuroD can compromise hair cell function. It is known to differentiate outer hair cells to inner ear hair cells and neurons to hair cells. | [58,63] |
| Rb1/Rbl2 | Required for hair cell quiescence and cell-cycle exit of embryonic mammalian hair cells but not for their early differentiation. Deletion of Rb1 from progenitor cells leads to aberrant hair cell and supporting cell. Deletion of Rbl2 results in extra row of hair cells and supporting cells in apical regions of the cochlea. | [64,65,66,67,68] |
| Cdkn1b and Cdkn2d | Cyclin-dependent kinase inhibitor. Expressed in sensory progenitors during the early embryonic development of the cochlea. Regulates cell cycle and inhibits hair cell differentiation. | [69,70,71] |
| MYCN | Member of the Myc family that regulates proliferation. Regulates the growth of the ear as a whole and promotes differentiation of certain sensory and non-sensory components. Absence of MYCN can lead to abnormal development of inner ear organs and disorganized neuronal innervations. | [72] |
4. Gene Therapy and Stem Cell-Based Approaches for Treatment of Sensory Neural Hearing Loss
4.1. Gene Therapy in the Inner Ear
| Vector | Advantages | Disadvantages | Ref. |
|---|---|---|---|
| Adeno virus |
|
| [49,51, 82,83,84,87,88] |
| Adeno-associated Virus |
|
| [76,89,90,91,92] |
| Herpes Simplex Virus |
|
| [84,85,93] |
| Lentivirus |
|
| [86,94] |
4.2. Stem Cell-Based Therapy for Inner Ear Hair Cell Regeneration
4.2.1. Embryonic Stem Cells
4.2.2. Adult Stem Cells
4.2.3. Induced Pluripotent Stem Cells
4.2.4. Summary of Stem Cell-Based Therapies
5. Discussion
References
- Cotanche, D.; Kaiser, C. Hair cell fate decisions in cochlear development and regeneration. Hear. Res. 2010, 226, 18–25. [Google Scholar]
- Oshima, K.; Suchert, S.; Blevins, N.; Heller, S. Curing hearing loss: Patient expectations, health care practitioners, and basic science. J. Commun. Disord. 2010, 43, 311–318. [Google Scholar]
- Bodmer, D. Protection, regeneration and replacement of hair cells in the cochlea: Implications for the future treatment of sensorineural hearing loss. Swiss Med. Wkly. 2008, 138, 708–712. [Google Scholar]
- Suter, A.H.; von Gierke, H.E. Noise and public policy. Ear Hear. 1987, 8, 188–191. [Google Scholar]
- Clark, W.W.; Bohne, B.A. Effects of noise on hearing. J. Am. Med. Assoc. 1999, 281, 1658–1659. [Google Scholar]
- Fligor, B.J.; Cox, L.C. Output levels of commercially available portable compact disc players and the potential risk to hearing. Ear Hear. 2004, 25, 513–527. [Google Scholar]
- Pauley, S.; Kopecky, B.; Beisel, K.; Soukup, G.; Fritzsch, B. Stem cells and molecular strategies to restore hearing. Panminerva Med. 2008, 50, 41–53. [Google Scholar]
- Cheng, A.G.; Cunningham, L.L.; Rubel, E.W. Mechanisms of hair cell death and protection. Curr. Opin. Otolaryngol. Head Neck Surg. 2005, 13, 343–348. [Google Scholar]
- Matsui, J.; Cotanche, D. Sensory hair cell death and regeneration: Two halves of the same equation. Curr. Opin. Otolaryngol. Head Neck Surg. 2004, 12, 418–425. [Google Scholar]
- Kharlamova, A.; Aarts, N. A review of past and present hair cell regeneration techniques. Contemp. Issues Commun. Sci. Disord. 2007, 34, 134–144. [Google Scholar]
- Gillespie, P.; Müller, U. Mechanotransduction by hair cells: Models, molecules, and mechanisms. Cell. 2009, 139, 33–44. [Google Scholar]
- Corwin, J.T.; Cotanche, D.A. Regeneration of sensory hair cells after acoustic trauma. Science 1988, 240, 1772–1774. [Google Scholar]
- Jørgensen, J.; Mathiesen, C. The avian inner ear. Naturwissenschaften 1988, 75, 319–320. [Google Scholar]
- Rubel, E.; Oesterle, E.; Weisleder, P. Hair cell regeneration in the avian inner ear. Ciba Found. Symp. 1991, 160, 77–96. [Google Scholar]
- Weisleder, P.; Rubel, E. Hair cell regeneration in the avian vestibular epithelium. Exp. Neurol. 1992, 115, 2–6. [Google Scholar]
- Huang, Y.; Chi, F.; Han, Z.; Yang, J.; Gao, W.; Li, Y. New ectopic vestibular hair cell-like cells induced by Math1 gene transfer in postnatal rats. Brain Res. 2009, 1276, 31–38. [Google Scholar]
- Dooling, R.; Dent, M. New studies on hair cell regeneration in birds. Acoust. Sci. Technol. 2001, 22, 93–99. [Google Scholar]
- Wang, G.; Chatterjee, I.; Batts, S.; Wong, H.; Gong, T.; Gong, S.; Raphael, Y. Notch signaling and Atoh1 expression during hair cell regeneration in the mouse utricle. Hear. Res. 2010, 267, 61–70. [Google Scholar]
- Zine, A.; van de Water, T.; de Ribaupierre, F. Notch signaling regulates the pattern of auditory hair cell differentiation in mammals. Development 2000, 127, 3373. [Google Scholar]
- Corwin, J. Regeneration in the auditory system. Exp. Neurol. 1992, 115, 7–12. [Google Scholar]
- Stone, J.; Rubel, E. Cellular studies of auditory hair cell regeneration in birds. Proc. Natl. Acad. Sci. USA. 2000, 97, 11714–11721. [Google Scholar]
- Kwan, T.; White, P.; Segil, N. Development and regeneration of the inner ear. Ann. N. Y. Acad. Sci. 2009, 1170, 28–33. [Google Scholar]
- Edge, A.; Chen, Z. Hair cell regeneration. Curr. Opin. Neurobiol. 2008, 18, 377–382. [Google Scholar]
- Matsui, J.; Ryals, B. Hair cell regeneration: An exciting phenomenon… But will restoring hearing and balance be possible? J. Rehabil. Res. Dev. 2005, 42, 187–196. [Google Scholar]
- Bermingham-McDonogh, O.; Rubel, E. Hair cell regeneration: Winging our way towards a sound future. Curr. Opin. Neurobiol. 2003, 13, 119–126. [Google Scholar]
- Brigande, J.; Heller, S. Quo vadis, hair cell regeneration? Nat. Neurosci. 2009, 12, 679–685. [Google Scholar]
- Collado, M.; Burns, J.; Hu, Z.; Corwin, J. Recent advances in hair cell regeneration research. Curr. Opin. Otolaryngol. Head Neck Surg. 2008, 16, 465–471. [Google Scholar]
- Beisel, K.; Hansen, L.; Soukup, G.; Fritzsch, B. Regenerating cochlear hair cells: Quo vadis stem cell. Cell Tissue Res. 2008, 333, 373–379. [Google Scholar]
- Li, H.; Corrales, C.; Edge, A.; Heller, S. Stem cells as therapy for hearing loss. Trends Mol. Med. 2004, 10, 309–315. [Google Scholar]
- Matsui, J.I.; Parker, M.A.; Ryals, B.M.; Cotanche, D.A. Regeneration and replacement in the vertebrate inner ear. Drug Discov. Today 2005, 10, 1307–1312. [Google Scholar]
- Martin, F.; Clark, J. Introduction to Audiology (with CD-ROM); Allyn & Bacon Inc.: Needham Heights, MA, USA, 2005. [Google Scholar]
- Newby, H.; Popelka, G. Audiology, Englewood Cliffs; Prentice-Hall: Upper Saddle River, NJ, USA, 1992. [Google Scholar]
- Sataloff, J. Hearing Loss; Lippincott: Philadephia, PA, USA, 1966. [Google Scholar]
- Willott, J.F. Aging and the Auditory System: Anatomy, Physiology and Psychophysics; Whurr Pub Ltd: London, UK, 1991. [Google Scholar]
- LeMasurier, M.; Gillespie, P. Hair-cell mechanotransduction and cochlear amplification. Neuron 2005, 48, 403–415. [Google Scholar]
- Lindeman, H.; Ades, H.; Bredberg, G.; Engström, H. The sensory hairs and the tectorial membrane in the development of the cat's organ of Corti: A scanning electron microscopic study. Acta Oto-Laryngol. 1971, 72, 229–242. [Google Scholar]
- Roberts, W.; Howard, J.; Hudspeth, A. Hair cells: Transduction, tuning, and transmission in the inner ear. Annu. Rev. Cell Biol. 1988, 4, 63–92. [Google Scholar]
- Axelrod, J.D. Basal bodies, kinocilia and planar cell polarity. Nat. Genet. 2008, 40, 10–11. [Google Scholar]
- Hudspeth, A.; Jacobs, R. Stereocilia mediate transduction in vertebrate hair cells (auditory system/cilium/vestibular system). Proc. Natl. Acad. Sci. USA 1979, 76, 1506–1509. [Google Scholar]
- Moran, D.T.; Rowley, J.C.; Asher, D.L. Calcium-binding sites on sensory processes in vertebrate hair cells. Proc. Natl. Acad. Sci. USA 1981, 78, 3954–3958. [Google Scholar]
- White, P.; Doetzlhofer, A.; Lee, Y.; Groves, A.; Segil, N. Mammalian cochlear supporting cells can divide and trans-differentiate into hair cells. Nature 2006, 441, 984–987. [Google Scholar]
- Stone, J.; Oesterle, E.; Rubel, E. Recent insights into regeneration of auditory and vestibular hair cells. Curr. Opin. Neurol. 1998, 11, 17–24. [Google Scholar]
- Fritzsch, B.; Eberl, D.F.; Beisel, K.W. The role of bHLH genes in ear development and evolution: Revisiting a 10-year-old hypothesis. Cell, Mol. Life Sci. 2010, 67, 3089–3099. [Google Scholar]
- Chen, P.; Johnson, J.; Zoghbi, H.; Segil, H. The role of Math1 in inner ear development: Uncoupling the establishment of the sensory primordium from hair cell fate determination. Development 2002, 129, 2495–2505. [Google Scholar]
- Matei, V.; Pauley, S.; Kaing, S.; Rowitch, D.; Beisel, K.; Morris, K.; Feng, F.; Jones, K.; Lee, J.; Fritzsch, B. Smaller inner ear sensory epithelia in Neurog1 null mice are related to earlier hair cell cycle exit. Dev. Dyn. 2005, 234, 633–650. [Google Scholar]
- Pan, N.; Jahan, I.; Kersigo, J.; Kopecky, B.; Santi, P.; Johnson, S.; Schmitz, H.; Fritzsch, B. Conditional deletion of Atoh1 using Pax2-Cre results in viable mice without differentiated cochlear hair cells that have lost most of the organ of Corti. Hear. Res. 2010, 275, 66–80. [Google Scholar]
- Bermingham, N.; Hassan, B.; Price, S.; Vollrath, M.; Ben-Arie, N.; Eatock, R.; Bellen, H.; Lysakowski, A.; Zoghbi, H. Math1: An essential gene for the generation of inner ear hair cells. Science 1999, 284, 1837–1841. [Google Scholar]
- Izumikawa, M.; Minoda, R.; Kawamoto, K.; Abrashkin, K.; Swiderski, D.; Dolan, D.; Brough, D.; Raphael, Y. Auditory hair cell replacement and hearing improvement by Atoh1 gene therapy in deaf mammals. Nat. Med. 2005, 11, 271–276. [Google Scholar]
- Kawamoto, K.; Ishimoto, S.I.; Minoda, R.; Brough, D.E.; Raphael, Y. Math1 gene transfer generates new cochlear hair cells in mature guinea pigs in vivo. J.Neurosci. 2003, 23, 4395–4440. [Google Scholar]
- Zheng, J.; Gao, W. Overexpression of Math1 induces robust production of extra hair cells in postnatal rat inner ears. Nat. Neurosci. 2000, 3, 580–586. [Google Scholar]
- Baker, K.; Brough, D.; Staecker, H. Repair of the vestibular system via adenovector delivery of Atoh1: A potential treatment for balance disorders. Adv. Otorhinolaryngol. 2009, 66, 52–63. [Google Scholar]
- Staecker, H.; Praetorius, M.; Baker, K.; Brough, D. Vestibular hair cell regeneration and restoration of balance function induced by math1 gene transfer. Otol. Neurotol. 2007, 28, 223–231. [Google Scholar]
- Hong, S.; Huang, A.; Cao, S. The current status and prospects of gene therapy for the inner ear. Hum. Gene Ther. 2011, 22, 1–12. [Google Scholar]
- Kim, P.; Helms, A.W.; Johnson, J.E.; Zimmerman, K. XATH-1, a vertebrate homolog of drosophila atonal, induces neuronal differentiation within ectodermal progenitors* 1. Dev. Biol. 1997, 187, 1–12. [Google Scholar]
- Shou, J.; Zheng, J.; Gao, W. Robust generation of new hair cells in the mature mammalian inner ear by adenoviral expression of Hath1. Mol. Cell. Neurosci. 2003, 23, 169–179. [Google Scholar]
- Zine, A.; Aubert, A.; Qiu, J.; Therianos, S.; Guillemot, F.; Kageyama, R.; de Ribaupierre, F. Hes1 and Hes5 activities are required for the normal development of the hair cells in the mammalian inner ear. J. Neurosci. 2001, 21, 4712–4720. [Google Scholar]
- Zheng, J.; Shou, J.; Guillemot, F.; Kageyama, R.; Gao, W. Hes1 is a negative regulator of inner ear hair cell differentiation. Development 2000, 127, 4551–4560. [Google Scholar]
- Liu, M.; Pereira, F.A.; Price, S.D.; Chu, M.; Shope, C.; Himes, D.; Eatock, R.A.; Brownell, W.E.; Lysakowski, A.; Tsai, M.J. Essential role of BETA2/NeuroD1 in development of the vestibular and auditory systems. Genes Dev. 2000, 14, 2839–2854. [Google Scholar]
- Kiernan, A.E.; Pelling, A.L.; Leung, K.K.H.; Tang, A.S.P.; Bell, D.M.; Tease, C.; Lovell-Badge, R.; Steel, K.P.; Cheah, K.S.E. Sox2 is required for sensory organ development in the mammalian inner ear. Nature 2005, 434, 1031–1035. [Google Scholar]
- Oesterle, E.C.; Campbell, S.; Taylor, R.R.; Forge, A.; Hume, C.R. Sox2 and JAGGED1 expression in normal and drug-damaged adult mouse inner ear. J. Assoc. Res. Otoaryngol. 2008, 9, 65–89. [Google Scholar]
- Kiernan, A.E.; Cordes, R.; Kopan, R.; Gossler, A.; Gridley, T. The Notch ligands DLL1 and JAG2 act synergistically to regulate hair cell development in the mammalian inner ear. Development 2005, 132, 4353–4362. [Google Scholar]
- Kiernan, A.E.; Xu, J.; Gridley, T. The Notch ligand JAG1 is required for sensory progenitor development in the mammalian inner ear. PLoS Genet. 2006. [Google Scholar] [CrossRef]
- Jahan, I.; Pan, N.; Kersigo, J.; Fritzsch, B. Neurod1 suppresses hair cell differentiation in ear ganglia and regulates hair cell subtype development in the cochlea. PLoS One 2010. [Google Scholar] [CrossRef]
- Sage, C.; Huang, M.; Vollrath, M.A.; Brown, M.C.; Hinds, P.W.; Corey, D.P.; Vetter, D.E.; Chen, Z.Y. Essential role of retinoblastoma protein in mammalian hair cell development and hearing. Natl. Acad. Sci. 2006, 139, 7345–7350. [Google Scholar]
- Sage, C.; Huang, M.; Karimi, K.; Gutierrez, G.; Vollrath, M.A.; Zhang, D.S.; García-Añoveros, J.; Hinds, P.W.; Corwin, J.T.; Corey, D.P. Proliferation of functional hair cells in vivo in the absence of the retinoblastoma protein. Science 2005, 307, 1114–1118. [Google Scholar]
- Bodson, M.; Breuskin, I.; Lefebvre, P.; Malgrange, B. Hair cell progenitors: Identification and regulatory genes. Acta Oto-Laryngol. 2010, 130, 312–317. [Google Scholar]
- Rocha-Sanchez, S.M.; Scheetz, L.R.; Contreras, M.; Weston, M.D.; Korte, M.; McGee, J.A.; Walsh, E.J. Mature mice lacking Rbl2/p130 gene have supernumerary inner ear hair cells and supporting cells. J. Neurosci. 2011, 31, 8883–8893. [Google Scholar]
- Mantela, J.; Jiang, Z.; Ylikoski, J.; Fritzsch, B.; Zacksenhaus, E.; Pirvola, U. The retinoblastoma gene pathway regulates the postmitotic state of hair cells of the mouse inner ear. Development 2005, 132, 2377–2388. [Google Scholar]
- Chen, P.; Segil, N. p27 (Kip1) links cell proliferation to morphogenesis in the developing organ of Corti. Development 1999, 126, 1581–1590. [Google Scholar]
- Chen, P.; Zindy, F.; Abdala, C.; Liu, F.; Li, X.; Roussel, M.F.; Segil, N. Progressive hearing loss in mice lacking the cyclin-dependent kinase inhibitor Ink4d. Nat. Cell Biol. 2003, 5, 422–426. [Google Scholar]
- Löwenheim, H.; Furness, D.N.; Kil, J.; Zinn, C.; Gültig, K.; Fero, M.L.; Frost, D.; Gummer, A.W.; Roberts, J.M.; Rubel, E.W. Gene disruption of p27Kip1 allows cell proliferation in the postnatal and adult organ of Corti. Proc. Natl. Acad. Sci. USA 1999, 96, 4084–4088. [Google Scholar]
- Kopecky, B.; Santi, P.; Johnson, S.; Schmitz, H.; Fritzsch, B. Conditional deletion of N Myc disrupts neurosensory and non sensory development of the ear. Dev. Dyn. 2011, 240, 1373–1390. [Google Scholar]
- Okano, T.; Nakagawa, T.; Kita, T.; Endo, T.; Ito, J. Cell-gene delivery of brain-derived neurotrophic factor to the mouse inner ear. Mol. Ther. 2004, 14, 866–871. [Google Scholar]
- Oshima, K.; Shimamura, M.; Mizuno, S.; Tamai, K.; Doi, K.; Morishita, R.; Nakamura, T.; Kubo, T.; Kaneda, Y. Intrathecal injection of HVJ-E containing HGF gene to cerebrospinal fluid can prevent and ameliorate hearing impairment in rats. FASEB J. 2004, 18, 212–214. [Google Scholar]
- Rejali, D.; Lee, V.A.; Abrashkin, K.A.; Humayun, N.; Swiderski, D.L.; Raphael, Y. Cochlear implants and ex vivo BDNF gene therapy protect spiral ganglion neurons. Hear. Res. 2007, 228, 180–187. [Google Scholar]
- Jero, J.; Mhatre, A.N.; Tseng, C.J.; Stern, R.E.; Coling, D.E.; Goldstein, J.A.; Hong, K.; Zheng, W.W.; Hoque, A.T.M.S.; Lalwani, A.K. Cochlear gene delivery through an intact round window membrane in mouse. Hum. Gene Ther. 2001, 12, 539–548. [Google Scholar]
- Praetorius, M.; Brough, D.; Hsu, C.; Plinkert, P.; Pfannenstiel, S.; Staecker, H. Adenoviral vectors for improved gene delivery to the inner ear. Hear. Res. 2009, 248, 31–38. [Google Scholar]
- Campochiaro, P.A.; Nguyen, Q.D.; Shah, S.M.; Klein, M.L.; Holz, E.; Frank, R.N.; Saperstein, D.A.; Gupta, A.; Stout, J.T.; Macko, J. Adenoviral vector-delivered pigment epithelium-derived factor for neovascular age-related macular degeneration: Results of a phase I clinical trial. Hum. Gene Ther. 2006, 17, 167–176. [Google Scholar]
- Alton, E.; Kitson, C. Gene therapy for cystic fibrosis. Expert Opin. Invest. Drugs 2000, 9, 1523–1535. [Google Scholar]
- Praetorius, M.; Hsu, C.; Baker, K.; Brough, D.; Plinkert, P.; Staecker, H. Adenovector-mediated hair cell regeneration is affected by promoter type. Acta Oto-Laryngol. 2010, 130, 215–222. [Google Scholar]
- Kawamoto, K.; Yagi, M.; Stöver, T.; Kanzaki, S.; Raphael, Y. Hearing and hair cells are protected by adenoviral gene therapy with TGF- 1 and GDNF. Mol. Ther. 2003, 7, 484–492. [Google Scholar]
- Pfannenstiel, S.; Praetorius, M.; Plinkert, P.; Brough, D.; Staecker, H. Bcl-2 gene therapy prevents aminoglycoside-induced degeneration of auditory and vestibular hair cells. Audiol. Neurotol. 2009, 14, 254–266. [Google Scholar]
- Luebke, A.E.; Foster, P.K.; Muller, C.D.; Peel, A.L. Cochlear function and transgene expression in the guinea pig cochlea, using adenovirus- and adeno-associated virus-directed gene transfer. Hum. Gene Ther. 2001, 12, 773–781. [Google Scholar]
- Staecker, H.; Li, D.; O Malley, B.; van de Water, T. Gene expression in the mammalian cochlea: A study of multiple vector systems. Acta Oto-Laryngol. 2001, 121, 157–163. [Google Scholar]
- Derby, M.L.; Sena-Esteves, M.; Breakefield, X.O.; Corey, D.P. Gene transfer into the mammalian inner ear using HSV-1 and vaccinia virus vectors. Hear. Res. 1999, 134, 1–8. [Google Scholar]
- Han, J.J.; Mhatre, A.N.; Wareing, M.; Pettis, R.; Gao, W.Q.; Zufferey, R.N.; Trono, D.; Lalwani, A.K. Transgene expression in the guinea pig cochlea mediated by a lentivirus-derived gene transfer vector. Hum. Gene Ther. 1999, 10, 1867–1873. [Google Scholar]
- Holt, J.; Johns, D.; Wang, S.; Chen, Z.; Dunn, R.; Marban, E.; Corey, D. Functional expression of exogenous proteins in mammalian sensory hair cells infected with adenoviral vectors. J. Neurophysiol. 1999, 81, 1881–1888. [Google Scholar]
- Weiss, M.A.; Frisancho, J.C.; Roessler, B.J.; Raphael, Y. Viral-mediated gene transfer in the cochlea. Int. J. Dev. Neurosci. 1997, 15, 577–583. [Google Scholar]
- Peter, A.E.L.C.R.; Poulsenb, G.V.D.D.J. Adenoviral and AAV-mediated gene transfer to the inner ear: Role of serotype, promoter, and viral load on in vivo and in vitro infection efficiencies. Adv. Otorhinolaryngol. 2009, 66, 87–98. [Google Scholar]
- Li Duan, M.; Bordet, T.; Mezzina, M.; Kahn, A.; Ulfendahl, M. Adenoviral and adeno-associated viral vector mediated gene transfer in the guinea pig cochlea. Neuroreport 2002, 13, 1295–1299. [Google Scholar]
- Lalwani, A.; Walsh, B.; Reilly, P.; Muzyczka, N.; Mhatre, A. Development of in vivo gene therapy for hearing disorders: Introduction of adeno-associated virus into the cochlea of the guinea pig. Gene Ther. 1996, 3, 588–592. [Google Scholar]
- Kho, S.T.; Pettis, R.M.; Mhatre, A.N.; Lalwani, A.K. Safety of adeno-associated virus as cochlear gene transfer vector: Analysis of distant spread beyond injected cochleae. Mol. Ther. 2000, 2, 368–373. [Google Scholar]
- Glorioso, J.; DeLuca, N.; Fink, D. Development and application of herpes simplex virus vectors for human gene therapy. Annu. Rev. Microbiol. 1995, 49, 675–710. [Google Scholar]
- Qian, H.; Hang, X.; Xu, W.; Zhu, W.; Cao, H.; Chen, Y.; Xu, X.; Wang, M.; Xie, Y.; Sun, J. Lentivirus-modified human umbilical cord mesenchymal stem cells maintain their pluripotency. Biotechnol. Appl. Biochem. 2010, 55, 53–62. [Google Scholar]
- Odorico, J.S.; Kaufman, D.S.; Thomson, J.A. Multilineage differentiation from human embryonic stem cell lines. Stem cells. 2001, 19, 193–204. [Google Scholar]
- Li, H.; Roblin, G.; Liu, H.; Heller, S. Generation of hair cells by stepwise differentiation of embryonic stem cells. Proc. Natl. Acad. Sci. USA 2003, 100, 13495–13500. [Google Scholar]
- Oshima, K.; Shin, K.; Diensthuber, M.; Peng, A.; Ricci, A.; Heller, S. Mechanosensitive hair cell-like cells from embryonic and induced pluripotent stem cells. Cell 2010, 141, 704–716. [Google Scholar]
- Martinez-Monedero, R.; Edge, A. Stem cells for the replacement of inner ear neurons and hair cells. Int. J. Dev. Biol. 2007, 51, 655–661. [Google Scholar]
- Coleman, B.; Fallon, J.; Pettingill, L.; de Silva, M.; Shepherd, R. Auditory hair cell explant co-cultures promote the differentiation of stem cells into bipolar neurons. Exp. Cell Res. 2007, 313, 232–243. [Google Scholar]
- Corrales, C.E.; Pan, L.; Li, H.; Liberman, M.C.; Heller, S.; Edge, A.S.B. Engraftment and differentiation of embryonic stem cell-derived neural progenitor cells in the cochlear nerve trunk: Growth of processes into the organ of corti. J. Neurobiol. 2006, 66, 1489–1500. [Google Scholar]
- Shi, F.; Corrales, C.E.; Liberman, M.C.; Edge, A.S.B. BMP4 induction of sensory neurons from human embryonic stem cells and reinnervation of sensory epithelium. Eur. J. Neurosci. 2007, 26, 3016–3023. [Google Scholar]
- Chen, W.; Jongkamonwiwat, N.; Jonhson, S. FGF signaling can induce the generation of cochlear progenitors from hESCs with the potential to differentiate into functional hair cell-like cells and neurons UK National Stem Cell Network Oxford. UK Natl. Stem Cell Netw. 2009, 104, 99–109. [Google Scholar]
- Rivolta, M.N.; Li, H.; Heller, S. Generation of inner ear cell types from embryonic stem cells. Methods Mol. Biol. 2006, 330, 71–92. [Google Scholar]
- Jeon, S.; Oshima, K.; Heller, S.; Edge, A. Bone marrow mesenchymal stem cells are progenitors in vitro for inner ear hair cells. Mol. Cell. Neurosci. 2007, 34, 59–68. [Google Scholar]
- Sharif, S.; Nakagawa, T.; Ohno, T.; Matsumoto, M.; Kita, T.; Riazuddin, S.; Ito, J. The potential use of bone marrow stromal cells for cochlear cell therapy. Neuroreport 2007, 18, 351–354. [Google Scholar]
- Matsuoka, A.J.; Kondo, T.; Miyamoto, R.T.; Hashino, E. In vivo and in vitro characterization of bone marrow derived stem cells in the cochlea. Laryngoscope 2006, 116, 1363–1367. [Google Scholar]
- Kondo, T.; Johnson, S.A.; Yoder, M.C.; Romand, R.; Hashino, E. Sonic hedgehog and retinoic acid synergistically promote sensory fate specification from bone marrow-derived pluripotent stem cells. Proc. Natl. Acad. Sci. USA 2005, 102, 4789–4794. [Google Scholar]
- Li, H.; Liu, H.; Heller, S. Pluripotent stem cells from the adult mouse inner ear. Nat. Med. 2003, 9, 1293–1299. [Google Scholar]
- Doyle, K.L.; Kazda, A.; Hort, Y.; McKay, S.M.; Oleskevich, S. Differentiation of adult mouse olfactory precursor cells into hair cells in vitro. Stem Cells 2007, 25, 621–627. [Google Scholar]
- Lang, H.; Ebihara, Y.; Schmiedt, R.; Minamiguchi, H.; Zhou, D.; Smythe, N.; Liu, L.; Ogawa, M.; Schulte, B. Contribution of bone marrow hematopoietic stem cells to adult mouse inner ear: Mesenchymal cells and fibrocytes. J. Comp. Neurol. 2006, 496, 187–201. [Google Scholar]
- Low, C.; Liou, Y.; Tang, B. Neural differentiation and potential use of stem cells from the human umbilical cord for central nervous system transplantation therapy. J. Neurosci. Res. 2008, 86, 1670–1679. [Google Scholar]
- Takahashi, K.; Yamanaka, S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 2006, 126, 663–676. [Google Scholar]
- Kim, J.B.; Greber, B.; Araúzo-Bravo, M.J.; Meyer, J.; Park, K.I.; Zaehres, H.; Schöler, H.R. Direct reprogramming of human neural stem cells by OCT4. Nature 2009, 461, 649–643. [Google Scholar]
- Kawasaki, H.; Mizuseki, K.; Nishikawa, S.; Kaneko, S.; Kuwana, Y.; Nakanishi, S.; Nishikawa, S.I.; Sasai, Y. Induction of midbrain dopaminergic neurons from ES cells by stromal cell-derived inducing activity. Neuron 2000, 28, 31–40. [Google Scholar]
- Nishimura, K.; Nakagawa, T.; Ono, K.; Ogita, H.; Sakamoto, T.; Yamamoto, N.; Okita, K.; Yamanaka, S.; Ito, J. Transplantation of mouse induced pluripotent stem cells into the cochlea. Neuroreport 2009, 20, 1250–1254. [Google Scholar]
- Hildebrand, M.S.; Dahl, H.H.M.; Hardman, J.; Coleman, B.; Shepherd, R.K.; de Silva, M.G. Survival of partially differentiated mouse embryonic stem cells in the scala media of the guinea pig cochlea. J. Assoc. Res. Otoaryngol. 2005, 6, 341–354. [Google Scholar]
- Sekiya, T.; Kojima, K.; Matsumoto, M.; Kim, T.S.; Tamura, T.; Ito, J. Cell transplantation to the auditory nerve and cochlear duct. Exp. Neurol. 2006, 198, 12–24. [Google Scholar]
- Hu, Z.; Ulfendahl, M.; Olivius, N. Central migration of neuronal tissue and embryonic stem cells following transplantation along the adult auditory nerve. Brain Res. 2004, 1026, 68–73. [Google Scholar]
- Jongkamonwiwat, N.; Zine, A.; Rivolta, N. Stem cell based therapy in the inner ear: Appropriate donor cell types and routes for transplantation. Curr. Drug Targets 2010, 11, 888–897. [Google Scholar]
- Crain, B.J.; Tran, S.D.; Mezey, E. Transplanted human bone marrow cells generate new brain cells. J. Neurol. Sci. 2005, 233, 121–123. [Google Scholar]
- Naito, Y.; Nakamura, T.; Nakagawa, T.; Iguchi, F.; Endo, T.; Fujino, K.; Kim, T.S.; Hiratsuka, Y.; Tamura, T.; Kanemaru, S. Transplantation of bone marrow stromal cells into the cochlea of chinchillas. Neuroreport 2004, 15, 1–4. [Google Scholar]
- Vlastarakos, P.; Nikolopoulos, T.; Tavoulari, E.; Papacharalambous, G.; Tzagaroulakis, A.; Dazert, S. Sensory cell regeneration and stem cells: What we have already achieved in the management of deafness. Otol. Neurotol. 2008, 29, 758–768. [Google Scholar]
- Kamiya, K.; Fujinami, Y.; Hoya, N.; Okamoto, Y.; Kouike, H.; Komatsuzaki, R.; Kusano, R.; Nakagawa, S.; Satoh, H.; Fujii, M. Mesenchymal stem cell transplantation accelerates hearing recovery through the repair of injured cochlear fibrocytes. Am. J. Pathol. 2007, 171, 214–226. [Google Scholar]
- Lanza, R. Stem cell breakthrough: Don't forget ethics. Science 2007, 318, 1917–1920. [Google Scholar]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Devarajan, K.; Staecker, H.; Detamore, M.S. A Review of Gene Delivery and Stem Cell Based Therapies for Regenerating Inner Ear Hair Cells. J. Funct. Biomater. 2011, 2, 249-270. https://doi.org/10.3390/jfb2030249
Devarajan K, Staecker H, Detamore MS. A Review of Gene Delivery and Stem Cell Based Therapies for Regenerating Inner Ear Hair Cells. Journal of Functional Biomaterials. 2011; 2(3):249-270. https://doi.org/10.3390/jfb2030249
Chicago/Turabian StyleDevarajan, Keerthana, Hinrich Staecker, and Michael S. Detamore. 2011. "A Review of Gene Delivery and Stem Cell Based Therapies for Regenerating Inner Ear Hair Cells" Journal of Functional Biomaterials 2, no. 3: 249-270. https://doi.org/10.3390/jfb2030249




