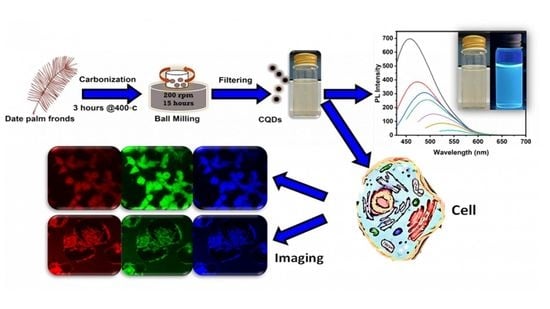
Carbon Nanoparticles Extracted from Date Palm Fronds for Fluorescence Bioimaging: In Vitro Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
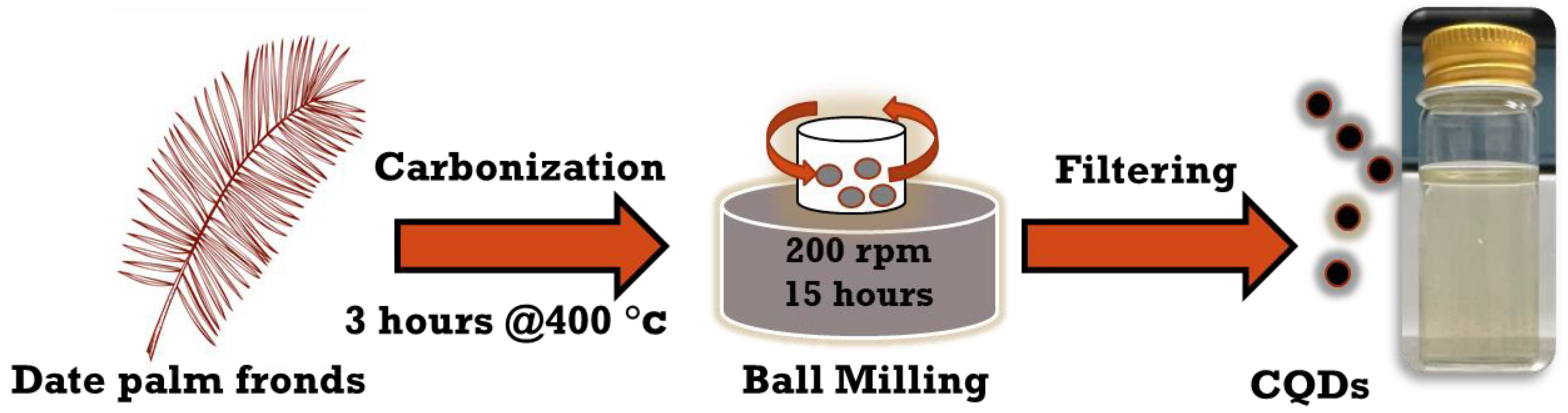
2.2. Synthesis of FCNPs
2.3. Characterization of CNPs
2.4. Cytocompatibility Assessment of CNPs and In Vitro Imaging
2.4.1. Cell Culture
2.4.2. Cell Viability Assay (MTT Assay)
2.4.3. Dose-Response Curve
2.4.4. Laser-Scanning Confocal Imaging
2.5. Statistical Analysis
3. Results and Discussion
3.1. Physiochemical Characterization
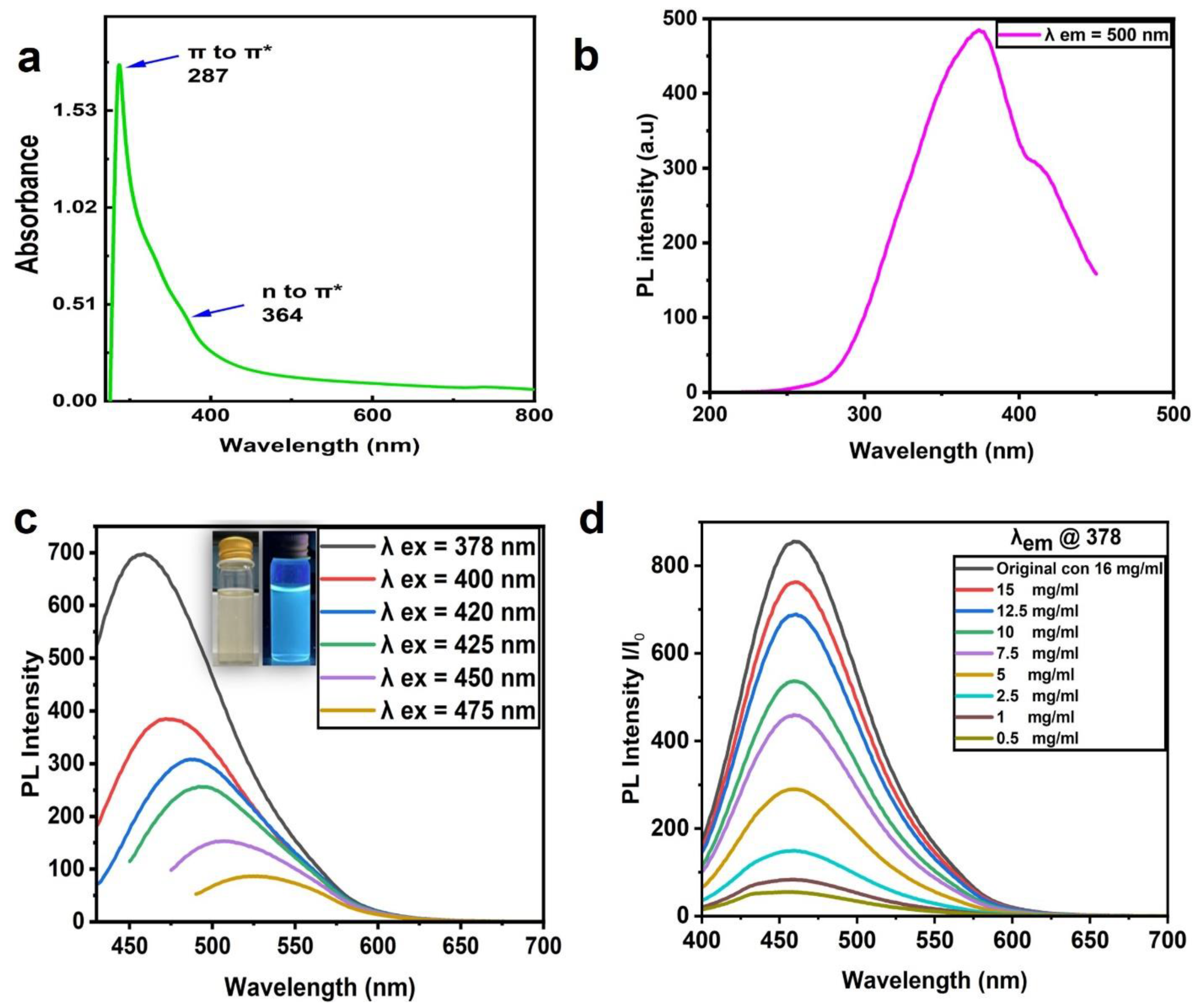
3.2. Optical Characterization
3.3. Cell Viability and Dose-Response Curves
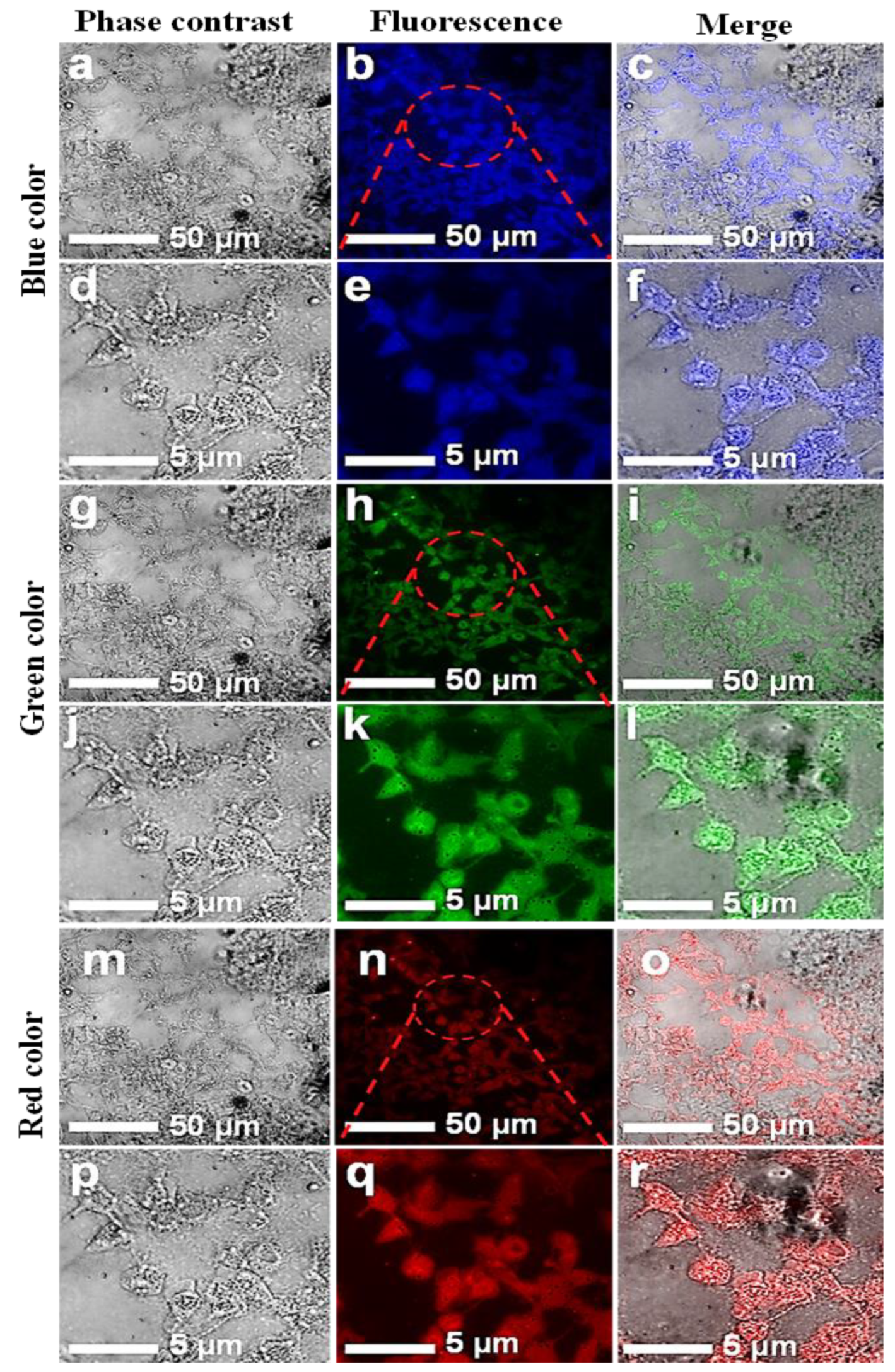
3.4. Cell Imaging by Laser-Scanning Confocal Microscope
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Atabaev, T.S. Doped Carbon Dots for Sensing and Bioimaging Applications: A Minireview. Nanomaterials 2018, 8, 342. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Campuzano, S.; Yáñez-Sedeño, P.; Pingarrón, J.M. Carbon Dots and Graphene Quantum Dots in Electrochemical Biosensing. Nanomaterials 2019, 9, 634. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khan, Z.M.S.H.; Saifi, S.; Shumaila; Aslam, Z.; Khan, S.A.; Zulfequar, M. A Facile One Step Hydrothermal Synthesis of Carbon Quantum Dots for Label -Free Fluorescence Sensing Approach to Detect Picric Acid in Aqueous Solution. J. Photochem. Photobiol. A Chem. 2020, 388, 112201. [Google Scholar] [CrossRef]
- Zhao, W.B.; Liu, K.K.; Song, S.Y.; Zhou, R.; Shan, C.X. Fluorescent Nano-Biomass Dots: Ultrasonic-Assisted Extraction and Their Application as Nanoprobe for FE3+ Detection. Nanoscale Res. Lett. 2019, 14, 130. [Google Scholar] [CrossRef] [Green Version]
- Sharma, A.; Das, J. Small Molecules Derived Carbon Dots: Synthesis and Applications in Sensing, Catalysis, Imaging, and Biomedicine. J. Nanobiotechnol. 2019, 17, 92. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bhunia, S.K.; Saha, A.; Maity, A.R.; Ray, S.C.; Jana, N.R. Carbon Nanoparticle-Based Fluorescent Bioimaging Probes. Sci. Rep. 2013, 3, 1473. [Google Scholar] [CrossRef] [Green Version]
- Kong, B.; Tang, J.; Zhang, Y.; Jiang, T.; Gong, X.; Peng, C.; Wei, J.; Yang, J.; Wang, Y.; Wang, X.; et al. Incorporation of Well-Dispersed Sub-5-Nm Graphitic Pencil Nanodots into Ordered Mesoporous Frameworks. Nat. Chem. 2016, 8, 171–177. [Google Scholar] [CrossRef]
- Russo, A.P.; Hu, G.; Compagnini, W.W.; Duley, N.Y.Z. Femtosecond Laser Large-Scale, Ablation of Highly Oriented Pyrolytic Graphite: A Green Route for Production of Porous Graphene and Graphene Quantum Dots. Nanoscale 2014, 6, 2381–2389. [Google Scholar] [CrossRef]
- Miao, S.; Liang, K.; Kong, B. Förster Resonance Energy Transfer (FRET) Paired Carbon Dot-Based Complex Nanoprobes: Versatile Platforms for Sensing and Imaging Applications. Mater. Chem. Front. 2020, 4, 128–139. [Google Scholar] [CrossRef]
- Soldevilla, B.; Díaz, R.; Silva, J.; Campos-Martín, Y.; Muñoz, C.; García, V.; García, J.M.; Peña, C.; Herrera, M.; Rodriguez, M.; et al. Prognostic Impact of ΔTAp73 Isoform Levels and Their Target Genes in Colon Cancer Patients. Clin. Cancer Res. 2011, 17, 6029–6039. [Google Scholar] [CrossRef]
- Qu, S.; Zhou, D.; Li, D.; Ji, W.; Jing, P.; Han, D.; Liu, L.; Zeng, H.; Shen, D. Toward Efficient Orange Emissive Carbon Nanodots through Conjugated Sp2-Domain Controlling and Surface Charges Engineering. Adv. Mater. 2016, 28, 3516–3521. [Google Scholar] [CrossRef] [PubMed]
- Kavitha, T.; Kumar, S. Turning Date Palm Fronds into Biocompatible Mesoporous Fluorescent Carbon Dots. Sci. Rep. 2018, 8, 16269. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Athinarayanan, J.; Periasamy, V.S.; Alshatwi, A.A. Unveiling the Biocompatible Properties of Date Palm Tree ( Phoenix Dactylifera L. ) Biomass-Derived Lignin Nanoparticles. ACS Omega 2022, 7, 19270–19279. [Google Scholar] [CrossRef]
- Rozila, I.; Abdul Manap, N.M.; Ghazali, L.N.; Kamal, N.; Abdul Hakeem, W.; Mohd Manzor, N.; Chowdhury, S.R.; Abdul Rahman, S. The Antioxidant Properties and Anticancer Effect of Medjool Dates (Phoenix Dactylifera L.) on Human Breast Adenocarcinoma (MCF-7) Cells: In Vitro Study. Front. Pharmacol. 2019, 63, 38. [Google Scholar] [CrossRef]
- Kučikas, V.; Werner, M.P.; Schmitz-Rode, T.; Louradour, F.; van Zandvoort, M.A.M.J. Two-Photon Endoscopy: State of the Art and Perspectives. Mol. Imaging Biol. 2021, 1–15. [Google Scholar] [CrossRef]
- Zhang, H.; Wang, G.; Zhang, Z.; Lei, J.H.; Liu, T.M.; Xing, G.; Deng, C.X.; Tang, Z.; Qu, S. One Step Synthesis of Efficient Red Emissive Carbon Dots and Their Bovine Serum Albumin Composites with Enhanced Multi-Photon Fluorescence for in Vivo Bioimaging. Light Sci. Appl. 2022, 11, 113. [Google Scholar] [CrossRef]
- Bao, X.; Yuan, Y.; Chen, J.; Zhang, B.; Li, D.; Zhou, D.; Jing, P.; Xu, G.; Wang, Y.; Holá, K.; et al. In Vivo Theranostics with Near-Infrared-Emitting Carbon Dots—Highly Efficient Photothermal Therapy Based on Passive Targeting after Intravenous Administration. Light Sci. Appl. 2018, 7, 91. [Google Scholar] [CrossRef] [Green Version]
- Salah, N.A.A.; Salah, Y.N.A.; Alshahrie, A.S.A. Methods of Producing Carbon Foreign Patent Documents Nanoparticles. U. Pat. U 2021, 10, 812–813. [Google Scholar]
- Mohiuddin, S.M.U.G.; Aydarous, A.; Alshahrie, A.; Saeed, A.; Memić, A.; Abdullahi, S.; Salah, N. Structural, Morphological, and Optical Properties of Carbon Nanoparticles Unsheathed from Date Palm Fronds. RSC Adv. 2022, 12, 27411–27420. [Google Scholar] [CrossRef]
- Lei, C.; Cao, Y.; Hosseinpour, S.; Gao, F.; Liu, J.; Fu, J.; Staples, R.; Ivanovski, S.; Xu, C. Hierarchical Dual-Porous Hydroxyapatite Doped Dendritic Mesoporous Silica Nanoparticles Based Scaffolds Promote Osteogenesis in Vitro and in Vivo. Nano Res. 2021, 14, 770–777. [Google Scholar] [CrossRef]
- Scherließ, R. The MTT Assay as Tool to Evaluate and Compare Excipient Toxicity in Vitro on Respiratory Epithelial Cells. Int. J. Pharm. 2011, 411, 98–105. [Google Scholar] [CrossRef] [PubMed]
- Saeed, A.; Razvi, M.A.; Madkhli, A.Y.; Abdullahi, S.; Aljoud, F.; Zughaibi, T.A.; Aboushoushah, S.F.; Alshahrie, A.; Memic, A.; Al-Hazmi, F.E.; et al. Investigation of the Tris(8-Hydroxyquinoline) Aluminum as a Promising Fluorescent Optical Material for in Vitro Bioimaging. Opt. Mater. 2022, 127, 112260. [Google Scholar] [CrossRef]
- Wang, D.H.; Li, W.; Liu, X.F.; Zhang, J.M.; Wang, S.M. Chinese Medicine Formula “Jian-Pi-Zhi-Dong Decoction” Attenuates Tourette Syndrome via Downregulating the Expression of Dopamine Transporter in Mice. Evid.-Based Complement. Altern. Med. 2013, 2013, 385685. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sangam, S.; Gupta, A.; Shakeel, A.; Bhattacharya, R.; Sharma, A.K.; Suhag, D.; Chakrabarti, S.; Garg, S.K.; Chattopadhyay, S.; Basu, B.; et al. Sustainable Synthesis of Single Crystalline Sulphur-Doped Graphene Quantum Dots for Bioimaging and Beyond. Green Chem. 2018, 20, 4245–4259. [Google Scholar] [CrossRef]
- Li, Y.; Zhong, X.; Rider, A.E.; Furman, S.A.; Ostrikov, K. Fast, Energy-Efficient Synthesis of Luminescent Carbon Quantum Dots. Green Chemistry 2014, 16, 2566–2570. [Google Scholar] [CrossRef] [Green Version]
- Larichev, Y.V.; Yeletsky, P.M.; Yakovlev, V.A. Study of Silica Templates in the Rice Husk and the Carbon-Silica Nanocomposites Produced from Rice Husk. J. Phys. Chem. Solids 2015, 87, 58–63. [Google Scholar] [CrossRef]
- Döring, A.; Ushakova, E.; Rogach, A.L. Chiral Carbon Dots: Synthesis, Optical Properties, and Emerging Applications. Light Sci. Appl. 2022, 11, 75. [Google Scholar] [CrossRef]
- Zhu, C.; Zhai, J.; Dong, S. Bifunctional Fluorescent Carbon Nanodots: Green Synthesis via Soy Milk and Application as Metal-Free Electrocatalysts for Oxygen Reduction. Chem. Commun. 2012, 48, 9367–9369. [Google Scholar] [CrossRef]
- Alam, A.M.; Park, B.Y.; Ghouri, Z.K.; Park, M.; Kim, H.Y. Synthesis of Carbon Quantum Dots from Cabbage with Down- and up-Conversion Photoluminescence Properties: Excellent Imaging Agent for Biomedical Applications. Green Chem. 2015, 17, 3791–3797. [Google Scholar] [CrossRef]
- Wang, B.; Song, A.; Feng, L.; Ruan, H.; Li, H.; Dong, S.; Hao, J. Tunable Amphiphilicity and Multifunctional Applications of Ionic-Liquid-Modified Carbon Quantum Dots. ACS Appl. Mater. Interfaces 2015, 7, 6919–6925. [Google Scholar] [CrossRef]
- Qian, Z.; Ma, J.; Shan, X.; Feng, H.; Shao, L.; Chen, J. Highly Luminescent N-Doped Carbon Quantum Dots as an Effective Multifunctional Fluorescence Sensing Platform. Chemistry 2014, 20, 2254–2263. [Google Scholar] [CrossRef] [PubMed]
- Gerakines, P.A.; Schutte, W.A.; Greenberg, J.M.; van Dishoeck, E.F. The Infrared Band Strengths of H2O, CO and CO2 in Laboratory Simulations of Astrophysical Ice Mixtures. Astron. Astrophys. 1995, 296, 810–818. [Google Scholar] [CrossRef]
- Li, Y.; Wang, K.; Zhou, W.; Li, Y.; Vila, R.; Huang, W.; Wang, H.; Chen, G.; Wu, G.H.; Tsao, Y.; et al. Cryo-EM Structures of Atomic Surfaces and Host-Guest Chemistry in Metal-Organic Frameworks. Matter 2019, 1, 428–438. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gómez-Hernández, R.; Panecatl-Bernal, Y.; Méndez-Rojas, M.Á. High Yield and Simple One-Step Production of Carbon Black Nanoparticles from Waste Tires. Heliyon 2019, 5, e02139. [Google Scholar] [CrossRef] [PubMed]
- Qu, D.; Wang, X.; Bao, Y.; Sun, Z. Recent Advance of Carbon Dots in Bio-Related Applications. J. Phys. Mater. 2020, 3, 22003. [Google Scholar] [CrossRef]
- Athinarayanan, J.; Periasamy, V.S.; Alatiah, K.A.; Alshatwi, A.A. Synthesis and Cytocompatibility Analysis of Carbon Nanodots Derived from Palmyra Palm Leaf for Multicolor Imaging Applications. Sustain. Chem. Pharm. 2020, 18, 100334. [Google Scholar] [CrossRef]
- Das, A.; Kundelev, E.V.; Vedernikova, A.A.; Cherevkov, S.A.; Danilov, D.V.; Koroleva, A.V.; Zhizhin, E.V.; Tsypkin, A.N.; Litvin, A.P.; Baranov, A.V.; et al. Revealing the Nature of Optical Activity in Carbon Dots Produced from Different Chiral Precursor Molecules. Light Sci. Appl. 2022, 11, 92. [Google Scholar] [CrossRef]
- Wu, Z.L.; Liu, Z.X.; Yuan, Y.H. Carbon Dots: Materials{,} Synthesis{,} Properties and Approaches to Long-Wavelength and Multicolor Emission. J. Mater. Chem. B 2017, 5, 3794–3809. [Google Scholar] [CrossRef]
- Siddique, A.B.; Pramanick, A.K.; Chatterjee, S.; Ray, M. Amorphous Carbon Dots and Their Remarkable Ability to Detect 2,4,6-Trinitrophenol. Sci. Rep. 2018, 8, 9770. [Google Scholar] [CrossRef] [Green Version]
- Ganesan, K.; Ghosh, S.; Gopala Krishna, N.; Ilango, S.; Kamruddin, M.; Tyagi, A.K. A Comparative Study on Defect Estimation Using XPS and Raman Spectroscopy in Few Layer Nanographitic Structures. Phys. Chem. Chem. Phys. 2016, 18, 22160–22167. [Google Scholar] [CrossRef] [Green Version]
- Tang, X.; Wang, L.; Ye, H.; Zhao, H.; Zhao, L. Biological Matrix-Derived Carbon Quantum Dots: Highly Selective Detection of Tetracyclines. J. Photochem. Photobiol. A Chem. 2022, 424, 113653. [Google Scholar] [CrossRef]
- Chen, G.; Qiu, H.; Prasad, P.N.; Chen, X. Upconversion Nanoparticles: Design, Nanochemistry, and Applications in Theranostics. Chem. Rev. 2014, 114, 5161–5214. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.F.; Xiao, B.; Cheng, J.; Liu, Z.B.; Yang, X.; Li, Q. Theoretical Design of Near-Infrared Fluorescent Sensor for F Anion Detection Based on 10-Hydroxybenzo[h]Quinoline Backbone. ACS Omega 2019, 4, 10516–10523. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arya, N.; Arora, A.; Vasu, K.S.; Sood, A.K.; Katti, D.S. Combination of Single Walled Carbon Nanotubes/Graphene Oxide with Paclitaxel: A Reactive Oxygen Species Mediated Synergism for Treatment of Lung Cancer. Nanoscale 2013, 5, 2818–2829. [Google Scholar] [CrossRef]
- Donahue, N.D.; Acar, H.; Wilhelm, S. Concepts of Nanoparticle Cellular Uptake, Intracellular Trafficking, and Kinetics in Nanomedicine. Adv. Drug Deliv. Rev. 2019, 143, 68–96. [Google Scholar] [CrossRef]
- Meng, W.; Bai, X.; Wang, B.; Liu, Z.; Lu, S.; Yang, B. Biomass-Derived Carbon Dots and Their Applications. Energy Environ. Mater. 2019, 2, 172–192. [Google Scholar] [CrossRef]
- Fan, R.J.; Sun, Q.; Zhang, L.; Zhang, Y.; Lu, A.H. Photoluminescent Carbon Dots Directly Derived from Polyethylene Glycol and Their Application for Cellular Imaging. Carbon 2014, 71, 87–93. [Google Scholar] [CrossRef]
- Chang, S.; Chen, B.B.; Lv, J.; Fodjo, E.K.; Qian, R.C.; Li, D.W. Label-Free Chlorine and Nitrogen-Doped Fluorescent Carbon Dots for Target Imaging of Lysosomes in Living Cells. Microchim. Acta 2020, 187, 435. [Google Scholar] [CrossRef]
- Qin, J.; Gao, X.; Chen, Q.; Liu, H.; Liu, S.; Hou, J.; Sun, T. PH Sensing and Bioimaging Using Green Synthesized Carbon Dots from Black Fungus. RSC Adv. 2021, 11, 31791–31794. [Google Scholar] [CrossRef]
- Kumar, P.; Dua, S.; Kaur, R.; Kumar, M.; Bhatt, G. A Review on Advancements in Carbon Quantum Dots and Their Application in Photovoltaics. RSC Adv. 2022, 12, 4714–4759. [Google Scholar] [CrossRef]
- Al-Abbad, A.; Al-Jamal, M.; Al-Elaiw, Z.; Al-Shreed, F.; Belaifa, H. A Study on the Economic Feasibility of Date Palm Cultivation in the Al-Hassa Oasis of Saudi Arabia. J. Dev. Agric. Econ. 2011, 3, 463–468. [Google Scholar]

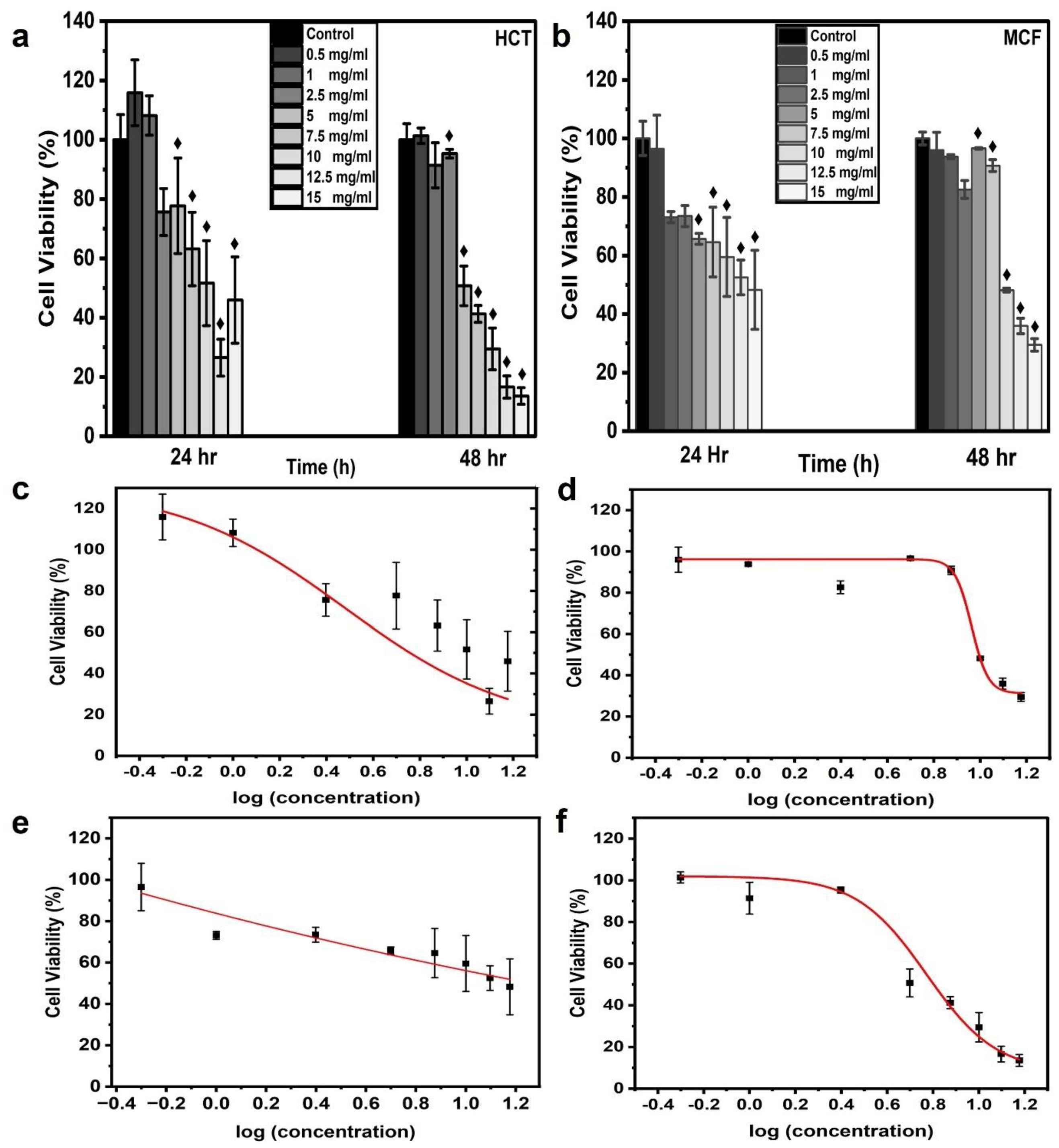

| Parameters | HCT | MCF | ||
|---|---|---|---|---|
| 24 h | 48 h | 24 h | 48 h | |
| EC20 (mg/mL) | 9.57 ± 16.11 | 9.61 ± 1.43 | ̶ | 10.29 ± 0.37 |
| EC50 (mg/mL) | 3.01 ± 1.61 | 5.90 ± 0.55 | ̶ | 9.17 ± 0.34 |
| EC80 (mg/mL) | 0.94 ± 1.07 | 3.63 ± 0.44 | ̶̶ | 8.18 ± 0.61 |
| EC90 (mg/mL) | 0.48 ± 0.90 | 2.73 ± 0.45 | ̶ | 7.64 ± 0.76 |
| R2 | 0.970 | 0.993 | ̶ | 0.993 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mohiuddin, S.M.U.G.; Saeed, A.; Alshahrie, A.; Memić, A.; Aljoud, F.; Abdullahi, S.; Organji, H.A.; Salah, N. Carbon Nanoparticles Extracted from Date Palm Fronds for Fluorescence Bioimaging: In Vitro Study. J. Funct. Biomater. 2022, 13, 218. https://doi.org/10.3390/jfb13040218
Mohiuddin SMUG, Saeed A, Alshahrie A, Memić A, Aljoud F, Abdullahi S, Organji HA, Salah N. Carbon Nanoparticles Extracted from Date Palm Fronds for Fluorescence Bioimaging: In Vitro Study. Journal of Functional Biomaterials. 2022; 13(4):218. https://doi.org/10.3390/jfb13040218
Chicago/Turabian StyleMohiuddin, Shaik Muhammad U. G., Abdu Saeed, Ahmed Alshahrie, Adnan Memić, Fadwa Aljoud, Shittu Abdullahi, Hussam A. Organji, and Numan Salah. 2022. "Carbon Nanoparticles Extracted from Date Palm Fronds for Fluorescence Bioimaging: In Vitro Study" Journal of Functional Biomaterials 13, no. 4: 218. https://doi.org/10.3390/jfb13040218