Studies on Mitigating Lipid Oxidation Reactions in a Value-Added Dairy Product Using a Standardized Cranberry Extract
Abstract
:1. Introduction
2. Results
2.1. Characterization and Standardization of WCE
2.1.1. Phenolic, Anthocyanin, Proanthocyanidin and Organic Acid Composition
| Component | |
|---|---|
| Phenolics (mg/g) | 42.16 ± 4.21 |
| Anthocyanins (mg/g) | 4.09 ± 0.20 |
| Proanthocyanidins (mg/g) | 5.41 ± 0.56 |
| Organic acids | |
| Quinic (mg/g) | 64.30 ±1.61 |
| Malic (mg/g) | 59.11 ± 0.57 |
| Citric (mg/g ) | 86.85 ± 11.94 |
| Ascorbic (mg/g) | 6.17 ± 0.01 |
| Antioxidant activity 1 | |
| H-ORAC (µmol TE/g) | 348.31 ± 33.45 |
| L-ORAC (µmol TE/g) | 11.02 ± 0.85 |
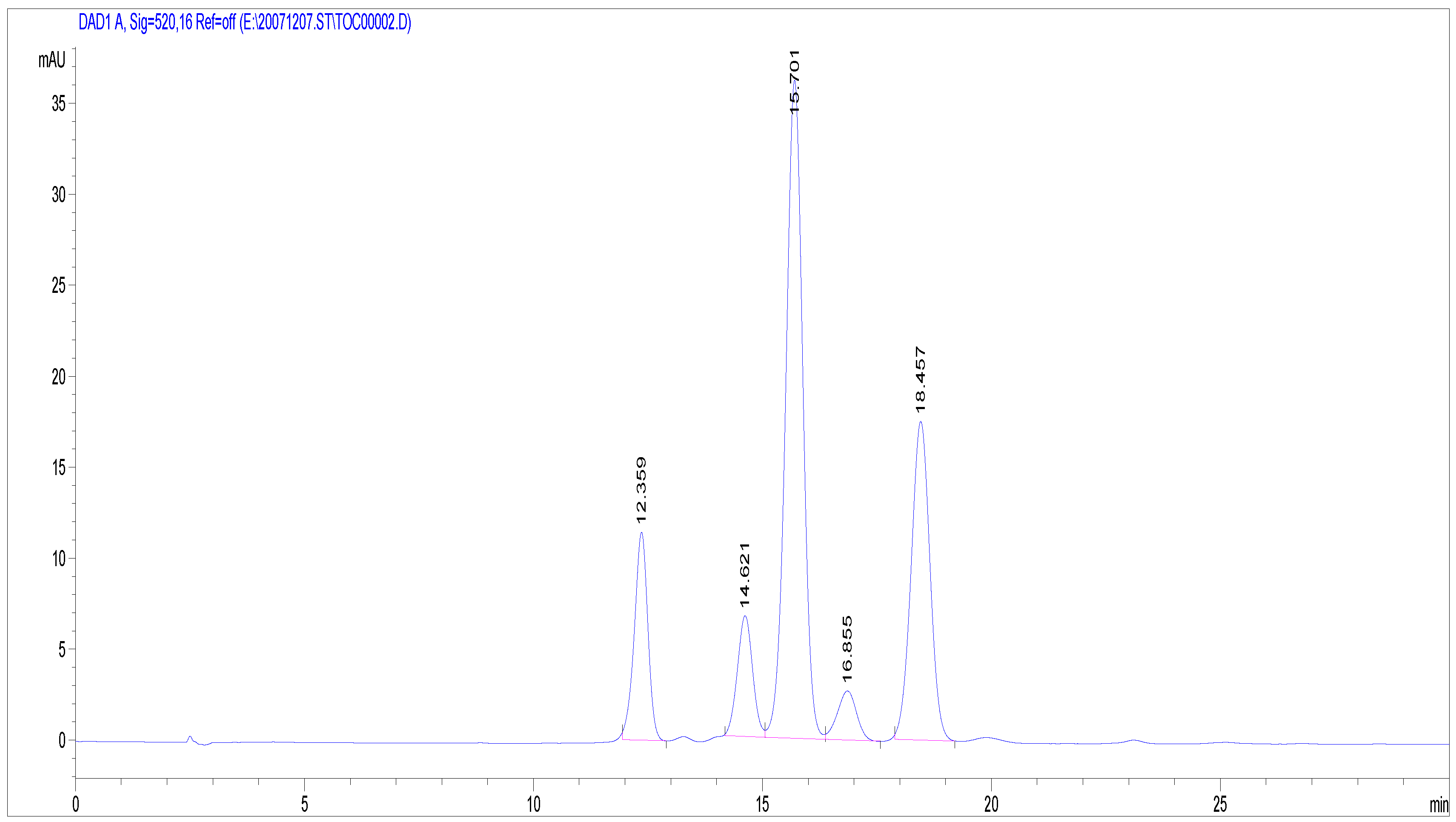
2.1.2. Antioxidant Activity
2.2. Emulsion Model
2.2.1. Emulsion Stability
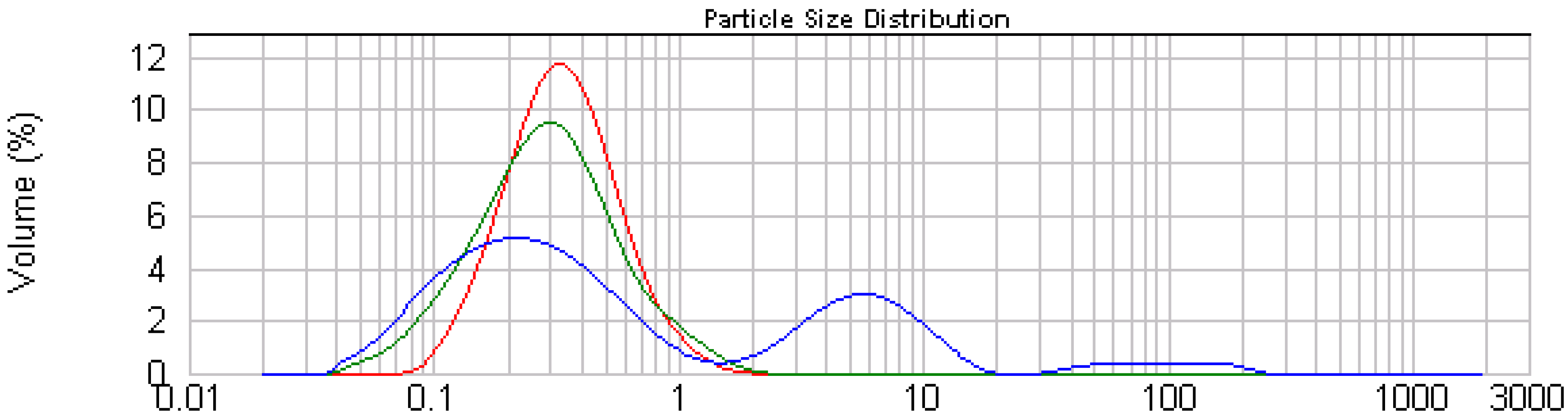
| Emulsion | pH | Paricle size (µm ± SD) |
|---|---|---|
| Control | 6.8 | 0.261 ± 0.024a |
| T1 | 5.6 | 0.284 ± 0.024a |
| T2 | 5.3 | 0.363 ± 0.032b |
2.2.2. Lipid Oxidation in Emulsions
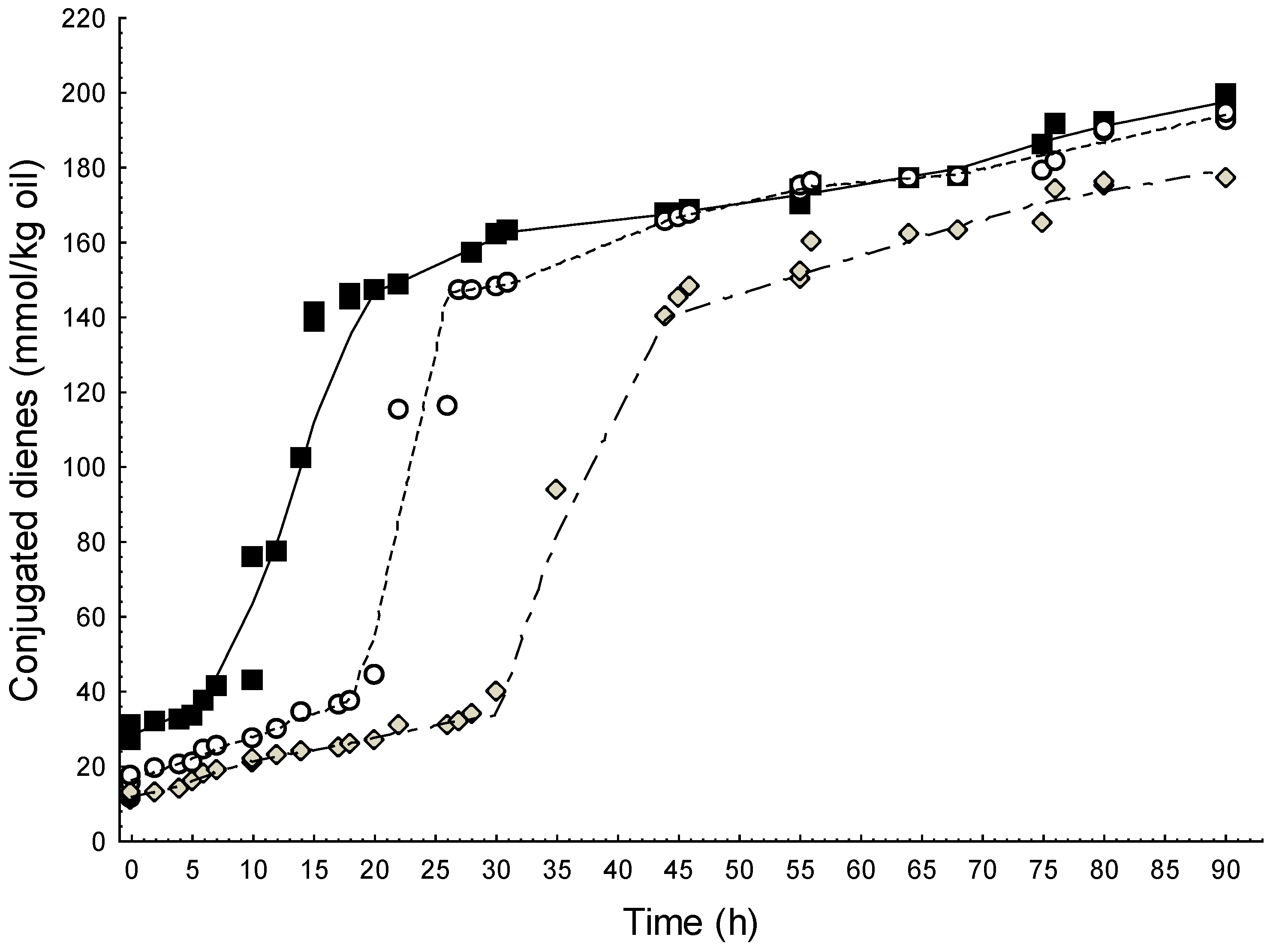
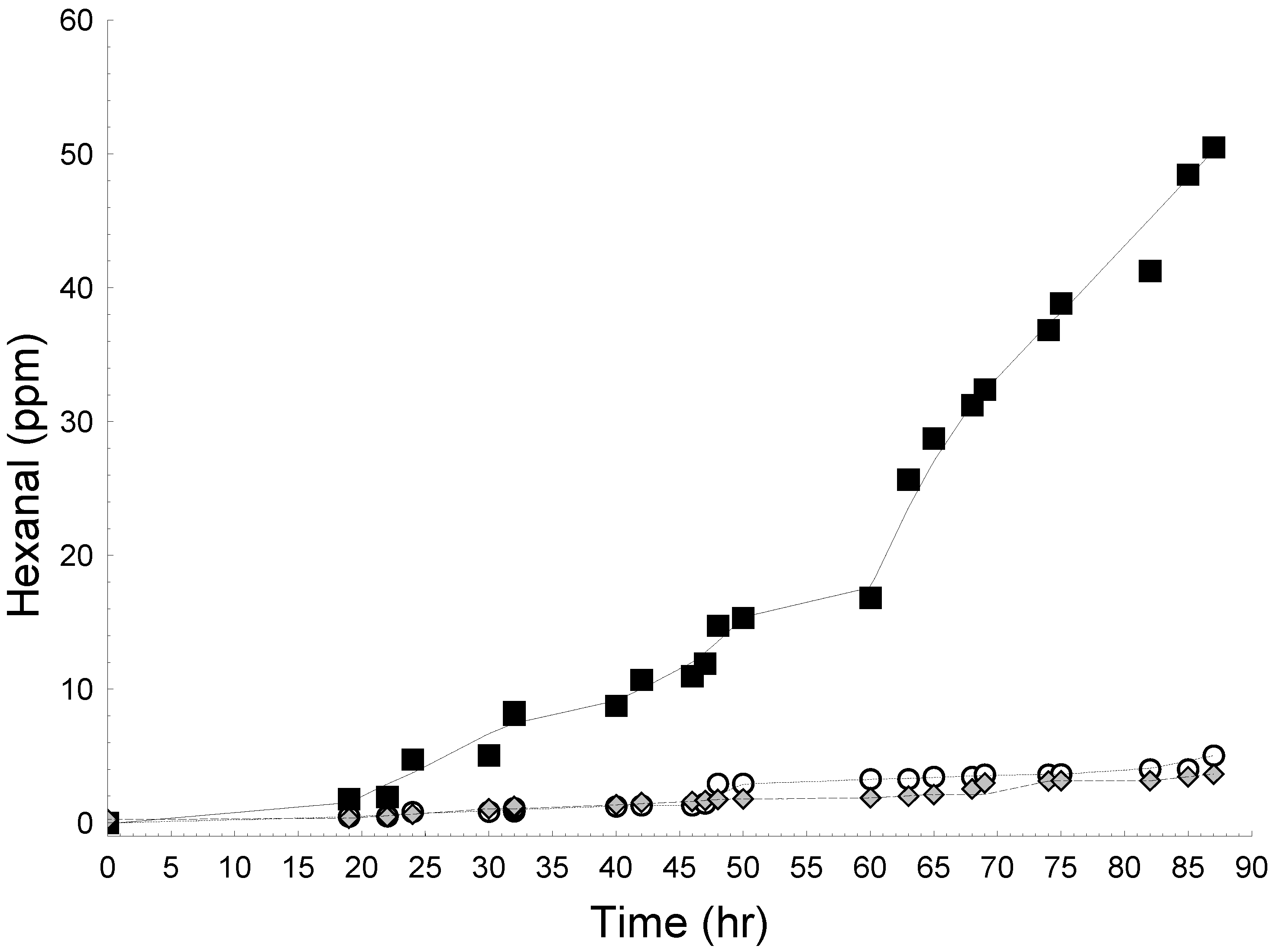
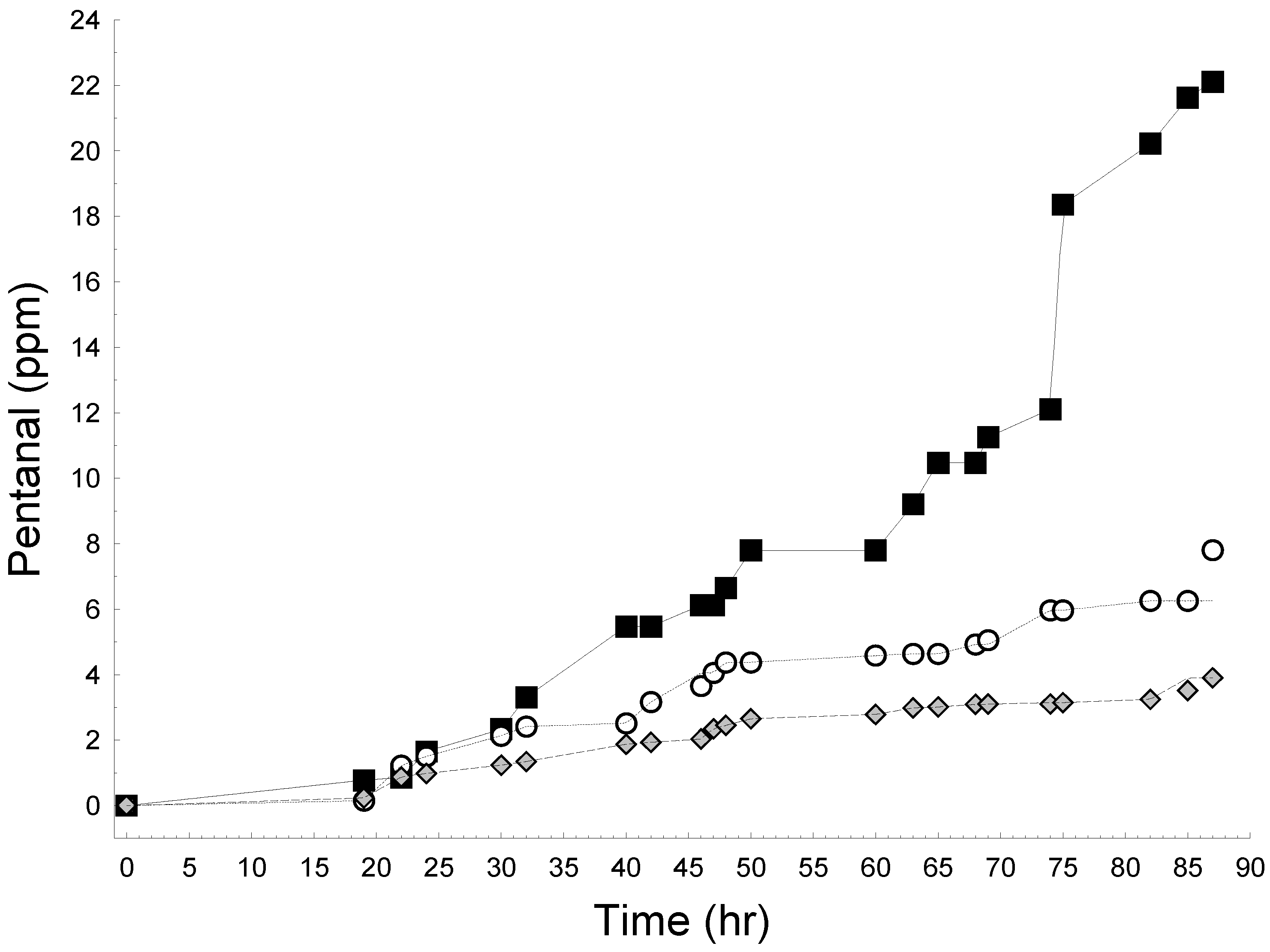
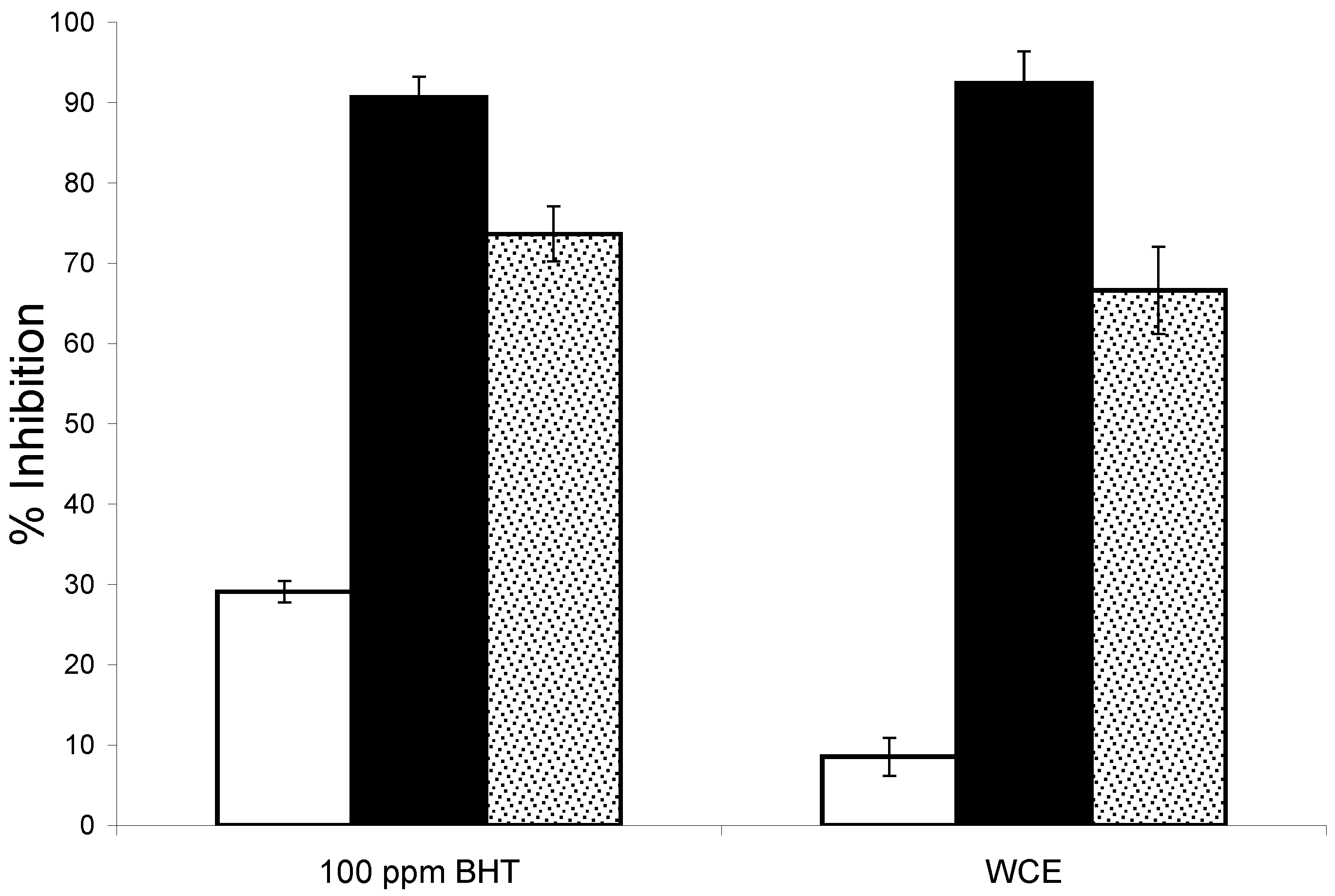
2.2.3. Effect of WCE in Fresh Cream Cheese Product
3. Discussion

4. Materials and Methods
4.1. Materials
4.2. Preparation and Characterization of a Whole Cranberry Extract (WCE)
4.2.1. Preparation of WCE
4.2.2. Phytochemcial Composition of WCE
4.2.3. WCE Antioxidant Activity Capacity
4.3. Lipid Oxidation in Model Emulsion
4.3.1. Model Emulsion Preparation
| Treatment | Na-Cas (% w/w) | Sunflower oil (% w/w) | WCE (% w/w) | pH |
|---|---|---|---|---|
| Control | 2 | 25 | - | 6.80 |
| BHT (100 ppm) | 2 | 25 | - | 6.80 |
| T1 | 2 | 25 | 0.375 | 5.61 |
| T2 | 2 | 25 | 0.75 | 5.33 |
4.3.2. Emulsion Stability Test
4.3.3. Accelerated Lipid Oxidation Test
4.3.4. Measurement of Headspace Hexanal and Pentanal by Static Gas Chromatography
4.3.5. Measurement of Conjugated Dienes (CD)
4.3.6. Measurement of Inhibition of Lipid Oxidation.
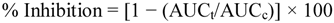
4.4. Lipid Oxidation in a Model Cream Cheese
4.4.1. Formulation of Cream Cheese Product
4.4.2. Determination of Lipid Oxidation in Cream Cheese
4.5. Statistical Analysis
5. Conclusions
Acknowledgments
References
- Moore, R.L.; Duncan, S.E.; Rasor, A.S.; Eigel, W.N.; O’Keefe, S.F. Oxidative stability of an extended shelf-life dairy-based beverage system designed to contribute to heart health. J. Dairy Sci. 2012, 95, 6242–6251. [Google Scholar] [CrossRef]
- Howell, A.B. Bioactive compounds in cranberry and their role in prevention of urinary tract infections. Mol. Nutr. Food Res. 2007, 51, 732–737. [Google Scholar] [CrossRef]
- Fox, P.F.; Guinee, T.P.; Cogan, T.M.; McSweeney, P.L.H. Fundamentals of Cheese Science; Aspen Publishers: Gaithersburg, MD, USA, 2000. [Google Scholar]
- Fedele, E.; Bergamo, P. Protein and lipid oxidative stresses during cheese manufacture. J. Food Sci. 2001, 66, 932–935. [Google Scholar] [CrossRef]
- McClements, D.J.; Decker, E.A. Lipid oxidation in oil-in-water emulsions: Impact of molecular environment on chemical reactions in heterogeneous food systems. J. Food Sci. 2000, 65, 1270–1282. [Google Scholar] [CrossRef]
- Soto-Cantu, C.D.; Graciano-Verdugo, A.Z.; Peralta, E.; Islas-Rubio, A.R.; Gonzalez-Cordova, A.; Gonzalez-Leon, A. Release of butylated hydroxytoluene from an active film packaging to asadero cheese and its effect on oxidation and odor stability. J. Dairy Sci. 2008, 91, 11–19. [Google Scholar] [CrossRef]
- Bandyopadhyay, M.; Chakraborty, R.; Raychaudhuri, U. Antioxidant activity of natural plant sources in dairy dessert (sandesh) under thermal treatment. Food Sci. Tech. 2008, 41, 816–825. [Google Scholar]
- Velasco, J.; Dobarganes, M.C.; Marquez-Ruiz, G. Antioxidant activity of phenolic compounds in sunflower oil-in-water emulsions containing sodium caseinate and lactose. Eur. J. Lipid Sci. Technol. 2004, 106, 4874–4882. [Google Scholar]
- Decker, E.A. Antioxidant mechanisms. In Food Lipids: Chemistry, Nutrition, and Biotechnology; Akoh, C.C., Min, D.B., Eds.; Marcel Dekker: New York, NY, USA, 1998; pp. 397–421. [Google Scholar]
- Rice-Evans, C.; Miller, N.; Paganga, G. Antioxidant properties of phenolic compounds. Trends in Plant Sci. 1997, 2, 152–159. [Google Scholar] [CrossRef]
- Vvedenskaya, I.O.; Vorsa, N. Flavonoid composition over fruit development and maturation in american cranberry, vaccinium macrocarpon ait. Plant Sci. 2004, 167, 1043–1054. [Google Scholar] [CrossRef]
- Hosseini-Beheshti, E.; Lund, S.; Kitts, D.D. Characterization of antioxidant capacity from fruits with distinct anthocyanin biosynthetic pathways. J. Nutr. Food Sci. 2012, 2, 122. [Google Scholar]
- Zheng, W.; Wang, S.Y. Oxygen radical absorbing capacity of phenolics in blueberries, cranberries, chokeberries, and lingonberries. J. Agri. Food Chem. 2003, 51, 502–509. [Google Scholar] [CrossRef]
- Satue-Gracia, M.T.; Heinonen, M.; Frankel, E.N. Anthocyanins as antioxidants on human low-density lipoprotein and lecithin-liposome systems. J. Agri. Food Chem. 1997, 45, 3362–3367. [Google Scholar] [CrossRef]
- Seeram, N.P.; Nair, M.G. Inhibition of lipid peroxidation and structure-activity-related studies of the dietary constituents anthocyanins, anthocyanidins, and catechins. J. Agri. Food Chem. 2002, 50, 5308–5312. [Google Scholar] [CrossRef]
- Vattem, D.A.; Ghaedian, R.; Shetty, K. Enhancing health benefits of berries through phenolic antioxidant enrichment: Focus on cranberry. Asia Pac. J. Clin. Nutr. 2005, 14, 120–130. [Google Scholar]
- Prior, R.L.; Lazarus, S.A.; Cao, G.; Muccitelli, H.; Hammerstone, J.F. Identification of procyanidins and anthocyanins in blueberries and cranberries (Vaccinium spp.) using high-performance liquid chromatography/mass spectrometry. J. Agri. Food Chem. 2001, 49, 1270–1276. [Google Scholar]
- Porter, M.L.; Krueger, C.G.; Wiebe, D.A.; Cunningham, D.G.; Reed, J.D. Cranberry proanthocyanidins associate with low-density lipoprotein and inhibit in vitro Cu2+-induced oxidation. J. Sci. Food Agri. 2001, 81, 1306–1313. [Google Scholar] [CrossRef]
- Škerget, M.; Kotnik, P.; Hadolin, M.; Hraš, A.R.; Simonič, M.; Knez, Ž. Phenols, proanthocyanidins, flavones and flavonols in some plant materials and their antioxidant activities. Food Chem. 2005, 89, 191–198. [Google Scholar] [CrossRef]
- Vvedenskaya, I.O.; Rosen, R.T.; Guido, J.E.; Russell, D.J.; Mills, K.A.; Vorsa, N. Characterization of flavonols in cranberry (Vaccinium macrocarpon) powder. J. Agric. Food Chem. 2004, 52, 188–195. [Google Scholar] [CrossRef]
- Leusink, G.L.; Kitts, D.D.; Yaghmaee, P.; Durance, T.D. Retention of antioxidant capacity of vacuum microwave dried cranberry. J. Food Sci. 2010, 75, 311–316. [Google Scholar]
- Shahidi, F.; Naczk, M.; Griffiths, W. Food Phenolics: Sources, Chemistry, Effects, Applications; Technomic Publishing Company: Lancaster, PA, USA, 1995. [Google Scholar]
- Xagorari, A.; Papapetropoulos, A.; Mauromatis, A.; Economou, M.; Fotsis, T.; Roussos, C. Luteolin inhibits an endotoxin-stimulated phosphorylation cascade and proinflammatory cytokine production in macrophages. J. Pharmacol. Exp. Ther. 2001, 296, 181–187. [Google Scholar]
- Wilson, T.; Porcari, J.P.; Harbin, D. Cranberry extract inhibits low density lipoprotein oxidation. Life Sci. 1998, 62, 381–386. [Google Scholar] [CrossRef]
- Reed, J. Cranberry flavonoids, atherosclerosis and cardiovascular health. Crit. Rev. Food Sci. Nutr. 2002, 42, 301–316. [Google Scholar] [CrossRef]
- Yan, X.; Murphy, B.T.; Hammond, G.B.; Vinson, J.A.; Neto, C.C. Antioxidant activities and antitumor screening of extracts from cranberry fruit (Vaccinium macrocarpon). J. Agri. Food Chem. 2002, 50, 5844–5849. [Google Scholar] [CrossRef]
- Youdim, K.A.; McDonald, J.; Kalt, W.; Joseph, J.A. Potential role of dietary flavonoids in reducing microvascular endothelium vulnerability to oxidative and inflammatory insults. J. Nutr. Biochem. 2002, 13, 282–288. [Google Scholar] [CrossRef]
- Seeram, N.P.; Adams, L.S.; Hardy, M.L.; Heber, D. Total cranberry extract versus its phytochemical constituents: Antiproliferative and synergistic effects against human tumor cell lines. J. Agric. Food Chem. 2004, 52, 2512–2517. [Google Scholar] [CrossRef]
- Girard, K.K.; Sinha, N.K. Cranberry, blueberry, currant, and gooseberry. In Handbook of Fruits and Fruit Processing; Hui, Y.H., Ed.; Blackwell Publishing: New York, NY, USA, 2006; pp. 369–390. [Google Scholar]
- Barrett, D.M.; Somogy, L.P.; Ramaswamy, H.S. Processing Fruits; CRC Press: Boca Raton, FL, USA, 2004. [Google Scholar]
- Kuhnau, J. The flavonoids—A class of semi-essential food components: Their role in human nutrition. World Rev. Nutr. Diet. 1976, 24, 117–119. [Google Scholar]
- Lee, C.H.; Reed, J.D.; Richard, M.P. Ability of various polyphenolic classes from cranberry to inhibit lipid oxidation in mechanically separated turkey and cooked ground pork. J. Muscle Foods 2006, 17, 248–266. [Google Scholar] [CrossRef]
- Huang, S.W.; Frankel, E.N.; German, J.B. Antioxidant activity of. alpha.-and. gamma.-tocopherols in bulk oils and in oil-in-water emulsions. J. Agric. Food Chem. 1994, 42, 2108–2114. [Google Scholar] [CrossRef]
- Kristensen, D.; Hansen, E.; Arndal, A.; Trinderup, R.A.; Skibsted, L.H. Influence of light and temperature on the color and oxidative stability of processed cheese. Int. Dairy J. 2001, 11, 837–843. [Google Scholar] [CrossRef]
- Van Aardt, M.; Duncan, S.E.; Marcy, J.E.; Long, T.E.; O'Keefe, S.F.; Nielsen-Sims, S.R. Aroma analysis of light-exposed milk stored with and without natural and synthetic antioxidants. J. Dairy Sci. 2005, 88, 881–890. [Google Scholar] [CrossRef]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventos, R.M. Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin-Ciocalteu reagent. Method. Enzymoly 1999, 299, 152–177. [Google Scholar] [CrossRef]
- Wrolstad, R.E.; Giusti, M.M. Characterization and measurement of anthocyanins by UV-visible spectroscopy. In Food Analytical Chemistry; Wrolstad, R.E., Ed.; John Wiley & Sons, Inc.: New York, NY, USA, 2001. [Google Scholar]
- Gessner, M.O.; Steiner, D. Acid butanol assay for proanthocyanidins (condensed tannins). In Methods to Study Litter Decomposition: A Practical Guide; Graca, M.A.S., Barlocher, F., Gessner, M.O., Eds.; Springer Verlag GmbH: Heidelberg, Germany, 2005; pp. 107–114. [Google Scholar]
- Kitts, D.D.; Hu, C. Biological and chemical assessment of antioxidant activity of sugar-lysine model maillard reaction products. Ann. N. Y. Acad. Sci. 2005, 1043, 501–512. [Google Scholar] [CrossRef]
- Davalos, A.; Gomez-Cordoves, C.; Bartolome, B. Extending applicability of the oxygen radical absorbance capacity (ORAC-fluorescein) assay. J. Agric. Food Chem. 2004, 52, 48–54. [Google Scholar] [CrossRef]
- AOCS, Method Ti. In Official Methods and Recommended Practices of the American Oil Chemists Society, 5th ed; AOCS Press: Urbana, IL, USA, 1998; pp. 1a–64.
- Cuvelier, M.E.; Lagunes-Galvez, L.; Berset, C. Do antioxidants improve the oxidative stability of oil-in-water emulsions? J. Am. Oil Chem. Soc. 2003, 80, 1101–1105. [Google Scholar] [CrossRef]
- AOAC, Official Methods of Analysis of AOAC International; AOAC International: Arlington, MA, USA, 1995.
- Bot, A.; Kleinherenbrink, F.A.M.; Mellema, M.; Magnani, C.K. Cream cheese as an acidified protein-stabilized emulsion gel. In Handbook of Food Products Manufacturing; Hui, Y.H., Ed.; Wiley-Interscience: New York, NY, USA, 2007; pp. 651–672. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kitts, D.D.; Tomiuk, S. Studies on Mitigating Lipid Oxidation Reactions in a Value-Added Dairy Product Using a Standardized Cranberry Extract. Agriculture 2013, 3, 236-252. https://doi.org/10.3390/agriculture3020236
Kitts DD, Tomiuk S. Studies on Mitigating Lipid Oxidation Reactions in a Value-Added Dairy Product Using a Standardized Cranberry Extract. Agriculture. 2013; 3(2):236-252. https://doi.org/10.3390/agriculture3020236
Chicago/Turabian StyleKitts, David D., and Stephen Tomiuk. 2013. "Studies on Mitigating Lipid Oxidation Reactions in a Value-Added Dairy Product Using a Standardized Cranberry Extract" Agriculture 3, no. 2: 236-252. https://doi.org/10.3390/agriculture3020236
APA StyleKitts, D. D., & Tomiuk, S. (2013). Studies on Mitigating Lipid Oxidation Reactions in a Value-Added Dairy Product Using a Standardized Cranberry Extract. Agriculture, 3(2), 236-252. https://doi.org/10.3390/agriculture3020236




