Elicitors: A Tool for Improving Fruit Phenolic Content
Abstract
:1. The Importance of Plant Phenolic Compounds
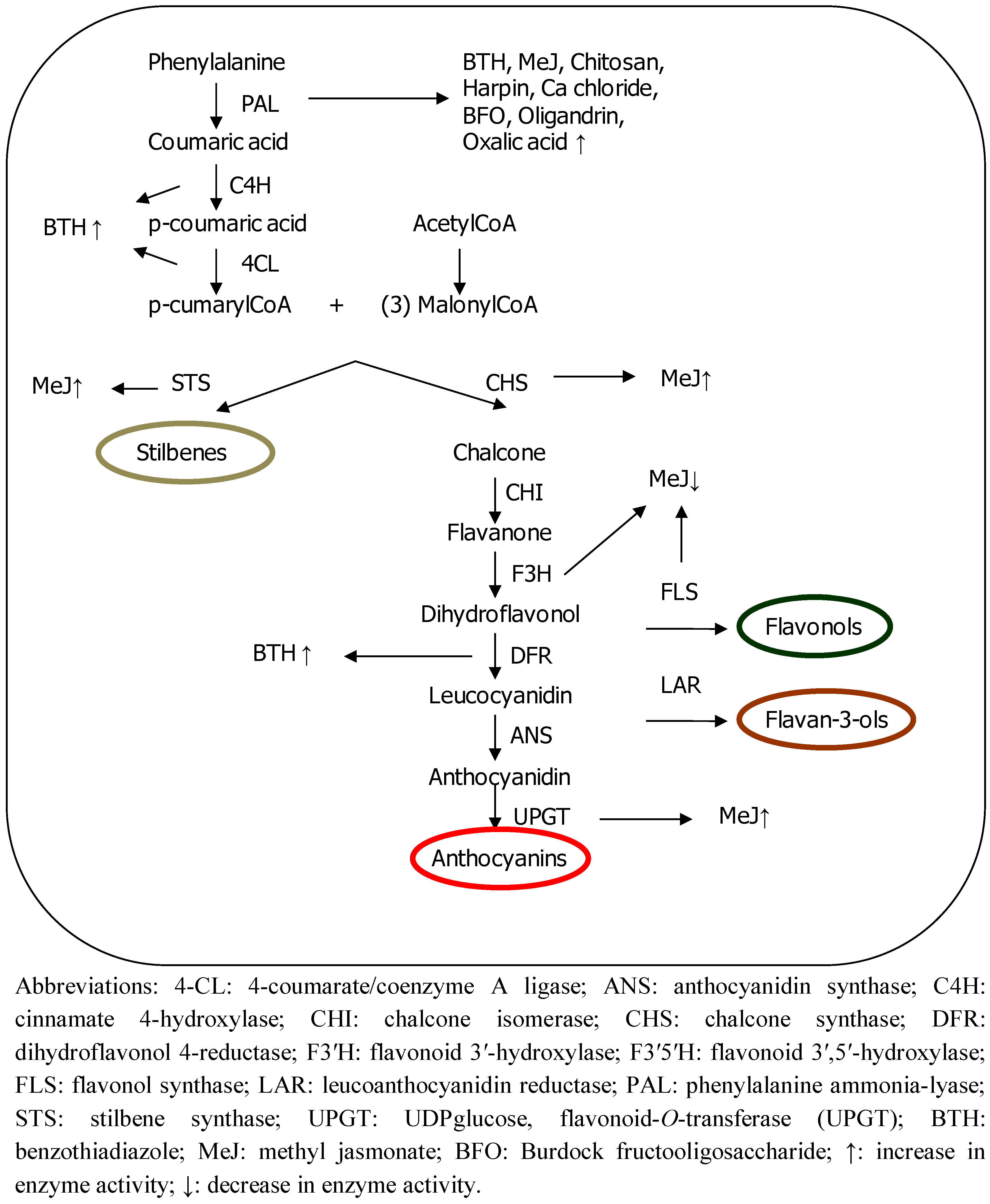
2. Methods for Improving Plant Phenolic Content
3. Phenolic Compounds and Induced Resistance in Plants
4. The Use of Elicitors
4.1. Benzo(1,2,3)thiadiazole-7-Carbothioic Acid S-methyl Ester (BTH)
| Elicitor | Fruit | Preharvest | Postharvest | Activated enzyme and/or increased compound | References |
|---|---|---|---|---|---|
| BTH | Mango; peach | Yes | PAL; total phenol content | [55,60] | |
| Strawberry | Yes | G6PDH, SKDH, TAL, PAL, C4H, DFR, anthocyanins; | [61,71] | ||
| Strawberry | Yes | quercetin and kaempferol; ellagic, p-coumaric, and gallic acids | [65,66,67] | ||
| Banana; mango | Yes | total phenolic content | [62,63] | ||
| Loquat | Yes | POD and PPO | [64] | ||
| Grapevine | Yes | resveratrol, anthocyanins; proanthocyanidins; flavonols | [44,68,69,70] | ||
| MeJ | Lychee | Yes | PAL | [72] | |
| Peach | Yes | PAL; total phenols | [73] | ||
| Apple, plum, table grape, strawberry | Yes | PAL; total phenols | [74] | ||
| Grapevine | Yes | CHS, STS, UPGT, stilbenes and anthocyanins | [75] | ||
| Red raspberry | Yes | myricetin, quercetin and kaempferol | [76] | ||
| Tomato | Yes | total phenols | [77] | ||
| Pomegranate, strawberry | Yes | total phenols and anthocyanins | [78,79] | ||
| Bayberry | Yes | total phenols | [80] | ||
| Berries; apple; grape | Yes | flavonoids | [70,81,82,83] | ||
| Strawberry | Yes | resveratrol | [84] | ||
| Grapevine | Yes | resveratrol and viniferins | [85,86] | ||
| Harpin | Melon | Yes | PAL, total phenol content, flavonoids, and lignins | [87] | |
| Jujube | Yes | PAL | [88] | ||
| Peach | Yes | PAL, total phenols | [89] | ||
| Chitosan | Table grape | Yes | Yes | PAL | [90,91] |
| Banana | Yes | PAL | [92] | ||
| Strawberry | Yes | total phenols | [93] | ||
| Grapevine | Yes | total phenols in grapes and wine | [94] | ||
| Oxalic acid and calcium chloride | Pear | Yes | PAL | [95] | |
| BFO | Tomato | Yes | PAL | [58] | |
| Oligandrin | Tomato | Yes | PAL | [96] | |
| Phosphite and acibenzolar- S-methyl | Apple | Yes | trans-resveratrol | [50] | |
| Potassium silicate | Avocado | Yes | catechin and epicatechin | [97] |
4.2. Methyl Jasmonate (MeJ)
4.3. Harpin
4.4. Chitosan
4.5. Other Elicitors
5. Conclusions
Acknowledgments
References
- Waterhouse, A.L. Wine phenolics. Ann. N. Y. Acad. Sci. 2002, 957, 21–36. [Google Scholar] [CrossRef]
- Cantos, E.; Espín, J.C.; Fernández, M.J.; Oliva, J.; Tomás-Barberán, A. Postharvest UV-C-Irradiated grapes as a potencial source for producing stilbene-enriched red wines. J. Agric. Food Chem. 2003, 51, 1208–1214. [Google Scholar] [CrossRef]
- Harborne, J.B.; Williams, C.A. Advances in flavonoids research since 1992. Phytochemistry 2000, 55, 481–504. [Google Scholar] [CrossRef]
- Noble, A.C. Bitterness in wine. Physiol. Behav. 1994, 56, 1251–1255. [Google Scholar] [CrossRef]
- Halsam, E.; Lilley, T.H. Natural astringency in foodstuffs—A molecular interpretation. Crit. Rev. Food Sci. Nutr. 1988, 27, 1–40. [Google Scholar]
- Zhao, J.; Davis, L.C.; Verpoorte, R. Elicitor signal transduction leading to production of plant secondary metabolites. Biotechnol. Adv. 2005, 23, 283–333. [Google Scholar]
- Leifert, W.R.; Abeywardena, M.Y. Cardioprotective actions of grape polyphenols. Nutr. Res. 2008, 28, 729–737. [Google Scholar] [CrossRef]
- Croizier, A.; Jaganath, I.B.; Clifford, M.N. Dietary phenolics: Chemistry, bioavailability and effects on health. Nat. Prod. Rep. 2009, 26, 1001–1046. [Google Scholar] [CrossRef]
- De Pascual-Teresa, S.; Moreno, D.A.; Garcia-Viguera, C. Flavanols and anthocyanins in cardiovascular health: A review of current evidence. Int. J. Mol. Sci. 2010, 11, 1679–1703. [Google Scholar] [CrossRef]
- Pezzuto, J.M. Grapes and human health: A perspective. J. Agric. Food Chem. 2008, 56, 6777–6784. [Google Scholar] [CrossRef]
- Parr, A.J.; Bolwell, G.P. Phenols in the plant and in man: The potential for possible nutritional enhancement of the diet by modifying the phenols content or profile. J. Sci. Food Agric. 2000, 80, 985–1012. [Google Scholar] [CrossRef]
- Perez-Lamela, C.; García-Falcon, M.S.; Simal-Gándara, J.; Orriols-Fernández, I. Influence of grape variety, vine system and enological treatments on the colour stability of young red wines. Food Chem. 2007, 101, 601–606. [Google Scholar] [CrossRef]
- Singh, S.K.; Singh, S.K.; Sharma, R.R. Effects of pruning intensity on the biochemical status of shoot buds in three mango (Mangifera indica L.) cultivars planted at high density. J. Hortic. Sci. Biotechnol. 2010, 85, 483–490. [Google Scholar]
- González-Neves, G.; Gil, G.; Ferrer, M. Effect of different vineyard treatments on the phenolic contents in tannat (Vitis vinifera L.) grapes and their respective wines. Food Sci. Technol. Int. 2002, 8, 315–321. [Google Scholar]
- Fanzone, M.; Zamora, F.; Jofre, V.; Assof, M.; Peña-Neira, A. Phenolic composition of malbec grape skins and seeds from Valle de Uco (Mendoza, Argentina) during Ripening: Effect of cluster thinning. J. Agric. Food Chem. 2011, 59, 6120–6136. [Google Scholar] [CrossRef]
- Soufleros, E.H.; Stavridou, K.; Dagkli, V. The effect of cluster thinning on phenolic maturity of Vitis vinifera cv. Xinomavro grapes. Int. J. Vine Wine Sci. 2011, 45, 171–179. [Google Scholar]
- Koundouras, S.; Hatzidimitriou, E.; Karamolegkou, M.; Dimopoulou, E.; Kallithraka, S.; Tsialtas, J.T.; Zioziou, E.; Nikolaou, N.; Kotseridis, Y. Irrigation and rootstock effects on the phenolic concentration and aroma potential of Vitis vinifera L. cv. Cabernet Sauvignon grapes. J. Agric. Food Chem. 2009, 57, 7805–7813. [Google Scholar] [CrossRef]
- Basile, B.; Marsal, J.; Mata, M.; Vallverdu, X.; Bellvert, J.; Girona, J. Phenological sensitivity of Cabernet Sauvignon to water stress: Vine physiology and berry composition. Am. J. Enol. Vitic. 2011, 62, 452–461. [Google Scholar] [CrossRef]
- Romero, P.; Fernandez-Fernandez, J.I.; Martinez-Cutillas, A. Physiological thresholds for efficient regulated deficit-irrigation management in winegrapes grown under semiarid conditions. Am. J. Enol. Vitic. 2010, 61, 300–312. [Google Scholar]
- Motilva, M.J.; Tovar, M.J.; Romero, M.P.; Alegre, S.; Girona, J. Influence of regulated deficit irrigation strategies applied to olive trees (Arbequina cultivar) on oil yield and oil composition during the fruit ripening period. J. Sci. Food Agric. 2000, 80, 2037–2043. [Google Scholar] [CrossRef]
- Gómez-Plaza, E.; Gil-Muñoz, R.; Hernández-Jiménez, A.; López-Roca, J.M.; Ortega-Regules, A.; Martínez-Cutillas, A. Studies on the anthocyanin profile of Vitis Vinifera intraspecific hybrids (Monastrell × Cabernet Sauvignon). Eur. Food Res. Technol. 2008, 227, 479–484. [Google Scholar] [CrossRef]
- Hernández-Jiménez, A.; Gómez-Plaza, E.; Martínez-Cutillas, A.; Kennedy, J. Grape skin and seed proanthocyanidins from Monastrell × Syrah grapes. J. Agric. Food Chem. 2009, 57, 10798–10803. [Google Scholar] [CrossRef]
- Tusa, N.; Abbate, L.; Renda, A.; Ruberto, G. Polyphenols distribution in juices from citrus allotetraploid somatic hybrids and their sexual hybrids. J. Agric. Food Chem. 2007, 55, 9089–9094. [Google Scholar] [CrossRef]
- Rapisarda, P.; Bellon, S.E.; Fabroni, S.; Russo, G. Juice quality of two new mandarin-like hybrids (Citrus clementina Hort. ex Tan × Citrus sinensis L. Osbeck) containing antocyanins. J. Agric. Food Chem. 2008, 56, 2074–2078. [Google Scholar] [CrossRef]
- Rapisarda, P.; Fabroni, S.; Peterek, S.; Russo, G.; Mock, H.P. Juice of New citrus hybrids (Citrus clementina Hort. ex Tan × C. sinensis L. Osbeck) as a source of natural antioxidants. Food Chem. 2009, 117, 212–218. [Google Scholar] [CrossRef]
- Pantelidis, G.E.; Vasilakakis, M.; Manganaris, G.A.; Diamantidis, G. Antioxidant capacity, phenol, anthocyanin and ascorbic acid contents in raspberries, blackberries, red currants, gooseberries and cornelian cherries. Food Chem. 2007, 102, 777–783. [Google Scholar] [CrossRef]
- Bugaud, C.; Alter, P.; Daribo, M.O.; Brillouet, J.M. Comparison of the physico-chemical characteristics of a new triploid banana hybrid, FLHORBAN 920, and the Cavendish variety. J. Sci. Food Agric. 2009, 89, 407–413. [Google Scholar] [CrossRef]
- Revilla, E.; Garcia-Beneytez, E.; Cabello, F. Anthocyanin fingerprint of clones of Tempranillo grapes and wines made with them. Aust. J. Grape Wine Res. 2009, 15, 70–78. [Google Scholar] [CrossRef]
- Ferrandino, A.; Guidoni, S. Anthocyanins, flavonols and hydroxycinnamates: An attempt to use them to discriminate Vitis vinifera L. cv “Barbera” clones. Eur. Food Res. Technol. 2010, 230, 417–427. [Google Scholar]
- Gómez-Plaza, E.; Gil-Muñoz, R.; Martínez-Cutillas, A. Multivariate classification of wines from seven clones of Monastrell grapes. J. Sci. Food Agric. 2000, 80, 497–501. [Google Scholar] [CrossRef]
- Burin, V.M.; Costa, L.L.F.; Rosier, J.P.; Bordignon-Luiz, M.T. Cabernet Sauvignon wines from two different clones, characterization and evolution during bottle ageing. Food Sci. Technol. 2011, 44, 1931–1938. [Google Scholar]
- Gonzalez-Molina, E.; Moreno, D.A.; Garcia-Viguera, C. Comparison of Verna lemon juice quality for new ingredients and food products. Sci. Hort. 2009, 120, 353–359. [Google Scholar] [CrossRef]
- Romero, A.; Tous, J.; Diaz, I. Vigin oil characteristics for selected clones from Arbequina variety. Acta Hort. 2008, 791, 713–717. [Google Scholar]
- Lopes, M.M.D.; de Miranda, M.R.A.; Moura, C.F.H.; Eneas, J. Bioactive compounds and total antioxidant capacity of cashew apples (Anacardium occidentale L.) during the ripening of early dwarf cashew clones. Cienc. Agrotec. 2012, 36, 325–332. [Google Scholar] [CrossRef]
- Abreu, C.R.A.; Maia, G.A.; Figueiredo, R.W.; Sousa, P.H.M.; Alves, R.E.; Brito, E.S.; Moura, C.F.H.; Rufino, M.S.M. Bioactive compounds and antioxidant activity of cashew apple (Anacardium occidentale L.) from commercial early dwaf clones. Acta Hort. 2009, 841, 451–454. [Google Scholar]
- Rufino, M.D.M.; Perez-Jimenez, J.; Tabernero, M.; Alves, R.E.; de Brito, E.S.; Saura-Calixto, F. Acerola and cashew apple as sources of antioxidants and dietary fibre. Int. J. Food Sci. Technol. 2010, 45, 2227–2233. [Google Scholar] [CrossRef]
- Sampaio, C.G.; Morais, S.M.; Rufino, M.S.M.; Alves, R.E.; Brito, E.S. Quality, bioactive compound content, and antioxidant activity in fruits of Brazilian Acerola clone. Acta Hort. 2009, 841, 463–466. [Google Scholar]
- Oliveira, L.D.; Moura, C.F.H.; de Brito, E.S.; Mamede, R.V.S.; de Miranda, M.R.A. Antioxidant metabolism during fruit development of different Acerola (Malpighia emarginata D.C) clones. J. Agric. Food Chem. 2012, 60, 7957–7964. [Google Scholar]
- Martens, S.; Knott, J.; Seitz, C.A.; Janvari, L.; Yu, S.N.; Forkmann, G. Impact of biochemical pre-studies on specific metabolic engineering strategies of flavonoid biosynthesis in plant tissues. Biochem. Eng. J. 2003, 14, 227–235. [Google Scholar] [CrossRef]
- Schijlen, E.; de Vos, C.H.R.; Jonker, H.; van den Broeck, H.; Molthoff, J.; van Tunen, A.; Martens, S.; Bovy, A. Pathway engineering for healthy phytochemicals leading to the production of novel flavonoids in tomato fruit. Plant Biotechnol. J. 2006, 4, 433–444. [Google Scholar] [CrossRef]
- D’Introno, A.; Paradiso, A.; Scoditti, E.; D’Amico, L.; de Paolis, A.; Carluccio, M.A.; Nicoletti, I.; DeGara, L.; Santino, A.; Giovinazzo, G. Antioxidant and anti-inflammatory properties of tomato fruits synthesizing different amounts of stilbenes. Plant Biotechnol. J. 2009, 7, 422–429. [Google Scholar] [CrossRef]
- Li, H.H.; Flachowsky, H.; Fischer, T.C.; Hanke, M.V.; Forkmmann, G.; Treutter, D.; Schwab, W.; Hoffmann, T.; Szankowski, I. Maize Lc transcription factor enhances biosynthesis of anthocyanins, distinct proanthocyanidins and phenylpropanoids in apple (Malus domestica Borkh.). Planta 2007, 226, 1243–1254. [Google Scholar] [CrossRef]
- Beckers, G.J.M.; Spoel, S.H. Fine-tuning plant defence signalling: Salicylate versus jasmonate. Plant Biol. 2006, 8, 1–10. [Google Scholar] [CrossRef]
- Iriti, M.; Rossoni, M.; Borgo, M.; Ferrara, L.; Faoro, F. Induction of resistance to gray mold with benzothiadiazole modifies amino acid profile and increases proanthocyanidins in grape: Primary versus secondary metabolism. J. Agric. Food Chem. 2005, 53, 9133–9139. [Google Scholar]
- Sticher, L.; Mauch-Mani, B.; Métraux, J.P. Systemic acquired resistance. Ann. Rev. Phytopathol. 1997, 35, 235–270. [Google Scholar] [CrossRef]
- Gozzo, F. Systemic acquired resistance in crop protection: From nature to a chemical approach. J. Agric. Food Chem. 2003, 51, 4487–4503. [Google Scholar] [CrossRef]
- Ding, C.K.; Wang, C.Y.; Gross, K.C.; Smith, D.L. Jasmonate and salicylate induce the expression of pathogenesis-related-protein genes and increase resistance to chilling injury in tomato fruit. Planta 2002, 214, 895–901. [Google Scholar] [CrossRef]
- Iqbal, Z.; Singh, Z.; Khangura, R.; Ahmad, S. Management of citrus blue and green moulds through application of organic elicitors. Aust. Plant Pathol. 2012, 41, 69–77. [Google Scholar] [CrossRef]
- Wang, Y.Y.; Li, B.Q.; Qin, G.Z.; Li, L.; Tian, S.P. Defense response of tomato fruit at different maturity stages to salicylic acid and ethephon. Sci. Hortic. 2011, 129, 183–188. [Google Scholar]
- Sautter, C.K.; Storck, L.; Rizzatti, M.R.; Mallmann, C.A.; Brackmann, A. Synthesis of trans-resveratrol and rotting control in apples with use of elicitors in post-harvest. Pesq. Agropec. Bras. 2008, 43, 1097–1103. [Google Scholar] [CrossRef]
- Eichholz, I.; Huyskens-Keil, S.; Keller, A.; Ulrich, D.; Kroh, L.W.; Rohn, S. UV-B-induced changes of volatile metabolites and phenolic compounds in blueberries (Vaccinium corymbosum L.). Food Chem. 2011, 126, 60–64. [Google Scholar] [CrossRef]
- Crifo, T.; Puglisi, I.; Petrone, G.; Recupero, G.R.; Lo Piero, A.R. Expression analysis in response to low temperature stress in blood oranges: Implication of the flavonoid biosynthetic pathway. Gene 2011, 476, 1–9. [Google Scholar] [CrossRef]
- Becatti, E.; Chkaiban, L.; Tonutti, P.; Forcato, C.; Bonghi, C.; Ranieri, A.M. Short-term postharvest carbon dioxide treatments induce selective molecular and metabolic changes in grape berries. J. Agric. Food Chem. 2010, 58, 8012–8020. [Google Scholar] [CrossRef]
- Pan, Y.G.; Liu, X.H. Effect of benzo-thiadiazole-7-carbothioic acid S-methyl ester (BTH) treatment on the resistant substance in postharvest mango fruits of different varieties. Afr. J. Biotechnol. 2011, 10, 15521–15528. [Google Scholar]
- Liu, H.X.; Jiang, W.B.; Bi, Y.; Luo, Y.B. Postharvest BTH treatment induces resistance of peach (Prunus persica L. cv. Jiubao) fruit to infection by Penicillium expansum and enhances activity of fruit defense mechanisms. Postharvest Biol. Technol. 2005, 35, 263–269. [Google Scholar] [CrossRef]
- Doke, N.; Miura, Y.; Sanchez, L.M.; Park, H.J.; Noritake, T.; Yoshioka, H.; Kawakita, K. The oxidative burst protects plants against pathogen attack: Mechanism and role as an emergency signal for plant bio-defence—A review. Gene 1996, 179, 45–51. [Google Scholar] [CrossRef]
- Obradovic, A.; Jones, J.B.; Momol, M.T.; Balogh, B.; Olson, S.M. Management of tomato bacterial spot in the field by foliar applications of bacteriophages and SAR inducers. Plant Dis. 2004, 88, 736–740. [Google Scholar] [CrossRef]
- Wang, F.D.; Feng, G.H.; Chen, K.S. Defense responses of harvested tomato fruit to burdock fructooligosaccharide, a novel potential elicitor. Postharvest Biol. Technol. 2009, 52, 110–116. [Google Scholar] [CrossRef]
- Kunz, W.; Schurter, R.; Maetzke, T. The chemistry of benzothiadiazole plant activators. Pestic. Sci. 1997, 50, 275–282. [Google Scholar] [CrossRef]
- Zhu, X.; Cao, J.; Wang, Q.; Jiang, W. Postharvest infiltration of BTH reduces of mango fruits (Mangifera indica L. cv. Tainong) by Colletotrichum gloeosporioides and enhances resistance inducing compounds. J. Phytopathol. 2008, 156, 68–74. [Google Scholar] [CrossRef]
- Cao, S.F.; Hu, Z.C.; Zheng, Y.H.; Lu, B.H. Effect of BTH on anthocyanin content and activities of related enzymes in strawberry after harvest. J. Agric. Food Chem. 2010, 58, 5801–5805. [Google Scholar]
- Zhu, S.J.; Ma, B.C. Benzothiadiazole- or methyl jasmonate-induced resistance to Colletotrichum musae in harvested banana fruit is related to elevated defense enzyme activities. J. Hortic. Sci. Biotechnol. 2007, 82, 500–506. [Google Scholar]
- Lin, J.H.; Gong, D.Q.; Zhu, S.J.; Zhang, L.J.; Zhang, L.B. Expression of PPO and POD genes and contents of polyphenolic compounds in harvested mango fruits in relation to Benzothiadiazole-induced defense against anthracnose. Sci. Hortic. 2011, 130, 85–89. [Google Scholar]
- Zhu, S.J.; Zhang, Z.W.; Xu, J.W.; Ma, L.Y.; Tang, W.L. Effect of BTH treatment on storability and activity of related enzymes of harvested loquat fruit. Acta Hortic. 2007, 750, 445–450. [Google Scholar]
- Anttonen, M.; Hukkanen, A.; Tiilikkala, K.; Karjalainen, R. Benzothiadiazole induces defense responses in berry crops. Acta Hortic. 2003, 567, 177–182. [Google Scholar]
- Hukkanen, A.T.; Kokko, H.I.; Buchala, A.J.; Mcdougall, G.J.; Stewart, D.; Karenlampi, S.O.; Karjalainen, R.O. Benzothiadiazole induces the accumulation of phenolics and improves resistance to powdery mildew in strawberries. J. Agric. Food Chem. 2007, 55, 1862–1870. [Google Scholar]
- Karjalainen, R.; Lehtinen, A.; Keinanen, M.; Julkunen-Tiitto, R.; Hietaniemi, V.; Pihlava, J.M.; Tiilikkala, K.; Jokinen, K. Benzothiadiazole and glycine betaine treatments enhance phenolic compound production in strawberry. Acta Hortic. 2002, 567, 353–356. [Google Scholar]
- Iriti, M.; Rossoni, M.; Borgo, M.; Faoro, F. Benzothiadiazole enhances resveratrol and anthocyanin biosynthesis in grapevine, meanwhile improving resistance to Botrytis cinerea. J. Agric. Food Chem. 2004, 52, 4406–4413. [Google Scholar]
- Fumagalli, F.; Rossoni, M.; Iriti, M.; di Gennaro, A.; Faoro, F.; Borroni, E.; Borgo, M.; Scienza, A.; Sala, A.; Folco, G. From field to health: A simple way to increase the nutraceutical content of grape as shown by NO-dependent vascular relaxation. J. Agric. Food Chem. 2006, 54, 5344–5349. [Google Scholar]
- Ruiz-Garcia, Y.; Romero Cascales, I.; Gil-Muñoz, R.; Fernandez-Fernandez, J.I.; Lopez-Roca, J.M.; Gómez-Plaza, E. Improving grape phenolic content and wine chromatic characteristics through the use of two different elicitors: Methyl Jasmonate versus Benzothiadiazole. J. Agric. Food Chem. 2012, 60, 1283–1290. [Google Scholar]
- Cao, S.F.; Hu, Z.C.; Zheng, Y.H.; Yang, Z.F.; Lu, B.H. Effect of BTH on antioxidant enzymes, radical-scavenging activity and decay in strawberry fruit. Food Chem. 2011, 125, 145–149. [Google Scholar] [CrossRef]
- Yang, S.Y.; Chen, Y.L.; Feng, L.Y.; Yang, E.; Su, X.G.; Jiang, Y.M. Effect of Methyl jasmonate on pericarp browning of postharvest lychees. J. Food Process. Preserv. 2011, 35, 417–422. [Google Scholar] [CrossRef]
- Jin, P.; Zheng, Y.H.; Tang, S.S.; Rui, H.J.; Wang, C.Y. Enhancing disease resistance in peach fruit with methyl jasmonate. J. Sci. Food Agric. 2009, 89, 802–808. [Google Scholar] [CrossRef]
- Heredia, J.B.; Cisneros-Zevallos, L. The effects of exogenous ethylene and methyl jasmonate on the accumulation of phenolic antioxidants in selected whole and wounded fresh produce. Food Chem. 2009, 115, 1500–1508. [Google Scholar] [CrossRef]
- Belhadj, A.; Telef, N.; Saigne, C.; Cluzet, S.; Barrieu, F.; Hamdi, S.; Merillon, J.M. Effect of methyl jasmonate in combination with carbohydrates on gene expression of PR proteins, stilbene and anthocyanin accumulation in grapevine cell cultures. Plant Physiol. Biochem. 2008, 46, 493–499. [Google Scholar] [CrossRef]
- Moreno, F.D.; Blanch, G.P.; del Castillo, M.L.R. Methyl jasmonate-induced bioformation of myricetin, quercetin and kaempferol in red raspberries. J. Agric. Food Chem. 2010, 58, 11639–11644. [Google Scholar] [CrossRef]
- Tzortzakis, N.G.; Economakis, C.D. Maintaining postharvest quality of the tomato fruit by employing methyl jasmonate and ethanol vapor treatment. J. Food Qual. 2007, 30, 567–580. [Google Scholar] [CrossRef]
- Sayyari, M.; Babalar, M.; Kalantari, S.; Martinez-Romero, D.; Guillen, F.; Serrano, M.; Valero, D. Vapour treatments with methyl salicylate or methyl jasmonate alleviated chilling injury and enhanced antioxidant potential during postharvest storage of pomegranates. Food Chem. 2011, 124, 964–970. [Google Scholar] [CrossRef]
- Moreno, F.D.; Monagas, M.; Blanch, G.P.; Bartolome, B.; del Castillo, M.L.R. Enhancement of anthocyanins and selected aroma compounds in strawberry fruits through methyl jasmonate vapor treatment. Eur. Food Res. Technol. 2010, 230, 989–999. [Google Scholar] [CrossRef]
- Wang, K.T.; Jin, P.; Shang, H.T.; Zheng, Y.H. Effect of methyl jasmonate in combination with ethanol treatment on postharvest decay and antioxidant capacity in Chinese bayberries. J. Agric. Food Chem. 2010, 58, 9597–9604. [Google Scholar] [CrossRef]
- Percival, D.; MacKenzie, J.L. Use of plant growth regulators to increase polyphenolic compounds in the wild blueberry. Can. J. Plant Sci. 2007, 87, 333–336. [Google Scholar] [CrossRef]
- Wang, S.Y.; Bowman, L.; Ding, M. Methyl jasmonate enchances antioxidant activity and flavonoid content in blackberries (Rubus sp.) and promotes antiproliferation of human cancer cells. Food Chem. 2008, 107, 1261–1269. [Google Scholar]
- Shafid, M.; Singh, Z.; Khan, A.S. Pre-harvest spray application of methyl jasmonate improves red blush and flavonoid content in Cripps Pink apple. J. Hortic. Sci. Biotechnol. 2011, 86, 422–430. [Google Scholar]
- Wang, S.Y.; Chen, C.T.; Wang, C.Y.; Chen, P. Resveratrol content in strawberry fruit is affected by preharvest conditions. J. Agric. Food Chem. 2007, 55, 8269–8274. [Google Scholar] [CrossRef]
- Esna-Ashari, M.; Pour, A.M. Effect of methyl jasmonate on resveratrol production in organs and cell suspension cultures of two Iranian grapevine (Vitis vinifera L.) cultivars. J. Hortic. Sci. Biotechnol. 2011, 86, 557–562. [Google Scholar]
- Vezzulli, S.; Civardi, S.; Ferrari, F.; Bavaresco, L. Methyl jasmonate treatment as a trigger of resveratrol synthesis in cultivated grapevine. Am. J. Enol. Vitic. 2007, 58, 530–533. [Google Scholar]
- Wang, J.; Yang, B.; Zhang, Z.; Zhang, H.; Ge, Y.H. Reduction of Latent infection and enhancement of disease resistance in muskmelon by preharvest application of harpin. J. Agric. Food Chem. 2011, 59, 12527–12533. [Google Scholar]
- Danner, M.A.; Sasso, S.A.Z.; Medeiros, J.G.S.; Marchese, J.A.; Mazaro, S.M. Induction of resistance to brown-rot on peaches by elicitors use in post-harvest. Pesq. Agropecu. Bras. 2008, 43, 793–799. [Google Scholar] [CrossRef]
- Li, M.; Yu, M.L.; Zhang, Z.Q.; Liu, Z.G.; Pan, Y. Control of black spot disease caused by Alternaria alternata on jujube (Ziziphus jujuba Mill. cv. Dongzao) using HarpinXoo protein. J. Hortic. Sci. Biotechnol. 2012, 87, 250–254. [Google Scholar]
- Yan, J.Q.; Cao, J.K.; Jiang, W.B.; Zhao, Y.M. Effects of preharvest oligochitosan sprays on portharvest fungal disease, storage quality, and defense responses in jujube (Zizyphus jujuba Mill. cv. Dongzao) fruit. Sci. Hortic. 2012, 142, 196–204. [Google Scholar] [CrossRef]
- Dos Santos, N.S.T.; Athayde Aguiar, A.J.A.; de Oliveira, C.E.V.; Verissimo de Sales, C.; de Melo E Silva, S.; Sousa da Silva, R.; Stamford, T.C.M.; de Souza, E.L. Efficacy of the application of a coating composed of chitosan and Origanum vulgare L. essential oil to control Rhizopus stolonifer and Aspergillus niger in grapes (Vitis labrusca L.). Food Microbiol. 2012, 32, 345–353. [Google Scholar] [CrossRef]
- Romanazzi, G.; Nigro, E.; Ippolito, A.; Di Venere, D. Effects of pre- and postharvest chitosan treatments to control storage grey mold of table grapes. J. Food Sci. 2002, 67, 1862–1867. [Google Scholar] [CrossRef]
- Meng, X.H.; Qin, G.Z.; Tian, S.P. Influences of preharvest spraying Cryptococcus laurentii combined with postharvest chitosan coating on postharvest diseases and quality of table grapes in storage. Food Sci. Technol. 2010, 43, 596–601. [Google Scholar]
- Meng, X.C.; Tang, Y.X.; Zhang, A.Y.; Huang, X.M.; Zhang, Z.Q. Effect of oligochitosan on development of Colletotrichum musae in vitro and in situ and its role in protection of banana fruits. Fruits 2012, 67, 147–155. [Google Scholar] [CrossRef]
- Mazaro, S.M.; Deschamps, C.; de Mio, L.L.; Biasi, L.A.; de Gouvea, A.; Sautter, C.K. Post harvest behavior of strawberry fruits after pre harvest treatment with chitosan and acibenzolar-S-methyl. Revista Brasileira de Fruticultura 2012, 30, 185–190. [Google Scholar]
- Iriti, M.; Vitalini, S.; Di Tommaso, G.; D’Amico, S.; Borgo, M.; Faoro, F. New chitosan formulation prevents grapevine powdery mildew infection and improves polyphenol content and free radical scavenging activity of grape and wine. Aust. J. Grape Wine Res. 2011, 17, 263–269. [Google Scholar] [CrossRef]
- Tian, S.P.; Qin, G.Z.; Xu, Y. Induction of defense responses against Alternaria rot by different elicitors in harvested pear fruit. Appl. Microbiol. Biotechnol. 2006, 70, 729–734. [Google Scholar] [CrossRef]
- Wei, Z.M.; Laby, R.J.; Zumoff, C.H.; Bauer, D.W.; He, S.Y.; Collmer, A.; Beer, S.V. Harpin, elicitor of the hypersensitive response produced by the plant pathogen Erwinia amylovora. Science 1992, 257, 85–88. [Google Scholar]
- He, S.Y.; Huang, H.-C.; Collmer, A. Pseudomonas syringae pv. syringae harpinPss: A protein that is secreted via the HRP pathway and elicits the hypersensitive response in plants. Cell 1993, 73, 1255–1266. [Google Scholar] [CrossRef]
- Dong, H.S.; Delaney, T.P.; Bauer, D.W.; Beer, S.V. Harpin induces disease resistance in Arabidopsis through the systemic acquired resistance pathway mediated by salicylic acid and the NIM1 gene. Plant J. 1999, 20, 207–215. [Google Scholar] [CrossRef]
- Dong, H.P.; Peng, J.L.; Bao, Z.L.; Meng, X.D.; Bonasera, J.M.; Chen, G.Y.; Beer, S.V.; Dong, H.S. Downstream divergence of the ethylene signaling pathway for harpin-stimulated Arabidopsis growth and insect defense. Plant Physiol. 2004, 136, 3628–3638. [Google Scholar] [CrossRef]
- Li, W.Q.; Shao, M.; Zhong, W.G.; Yang, J.; Okada, K.; Yamane, H.; Zhang, L.; Wang, G.; Wang, D.; Xiao, S.S.; et al. Ectopic expression of Hrf1 enhances bacterial resistance via regulation of diterpene phytoalexins, silicon and reactive oxygen species burst in rice. PloS One 2012, 7, 1–10. [Google Scholar]
- Lucon, C.M.M.; Guzzo, S.D.; de Jesus, C.O.; Pascholati, S.F. Postharvest harpin or Bacillus thuringiensis treatments suppress citrus black spot in Valencia oranges. Crop Prot. 2010, 29, 766–772. [Google Scholar] [CrossRef]
- Bi, Y.; Li, Y.C.; Ge, Y.H.; Wang, Y. Induced Resistance in melons by elicitors for the control of postharvest diseases. Postharvest Pathol. 2010, 2, 31–41. [Google Scholar]
- De Capdeville, G.; Beer, S.V.; Watkins, C.B.; Wilson, C.L.; Tedeschi, L.O.; Aist, J.R. Pre- and post-harvest harpin treatments of apples induce resistance to blue mold. Plant Dis. 2003, 87, 39–44. [Google Scholar] [CrossRef]
- Wang, J.J.; Wang, Y.; Ge, Y.H.; Bi, Y. Inhibiting effect of postharvest harpin treatment on Alternaria rot and induction to resistance enzymes of Pyrus bretschneideri cv Pingguoli. J. Gansu Agric. Univ. 2006, 41, 114–117. [Google Scholar]
- Boro, M.C.; Beriam, L.O.S.; Guzzo, S.D. Induced resistance against Xanthomonas axonopodis pv. passiflorae in passion fruit plants. Trop. Plant Pathol. 2011, 36, 74–80. [Google Scholar]
- Saour, G.; Ismail, H.; Hashem, A. Impact of kaolin particle film, spirodiclofen acaricide, harpin protein, and an organic biostimulant on pear psylla Cacopsylla pyri (Hemiptera: Psyllidae). Int. J. Pest Manag. 2010, 56, 75–79. [Google Scholar] [CrossRef]
- Bastas, K.K.; Maden, S. Evaluation of host resistance inducers and conventional products for fire blight management in loquat and quince. Phytoprotection 2007, 88, 93–101. [Google Scholar] [CrossRef]
- Bautista-Baños, S.; Hernanadez-Lauzardo, A.N.; Velazquez-del Valle, M.G.; Hernandez-Lopez, M.; Barka, E.A.; Bosquez-Molina, E.; Wilson, C.L. Chitosan as a potential natural compound to control pre and postharvest diseases of horticultural commodities. Crop Prot. 2006, 25, 108–118. [Google Scholar] [CrossRef]
- Zhang, H.; Li, R.; Liu, W. Effects of chitin and its derivative chitosan on postharvest decay of fruits: A review. Int. J. Mol. Sci. 2011, 12, 917–934. [Google Scholar] [CrossRef]
- Badawyl, M.E.; Rabea, E.I. A biopolymer chitosan and its derivatives as promising antimicrobial agents against plant pathogens and their applications in crop protection. Int. J. Carbohyd. Chem. 2011. [Google Scholar] [CrossRef]
- Reddy, M.V.B.; Belkacemi, K.; Corcuff, R.; Castaigne, F.; Arul, J. Effect of pre-harvest chitosan sprays on post-harvest infection by Botrytis cinerea and quality of strawberry fruit. Postharvest Biol. Technol. 2000, 20, 39–51. [Google Scholar] [CrossRef]
- Romanazzi, G.; Nigro, F.; Ippolito, A. Effectiveness of pre and postharvest chitosan treatments on storage decay of strawberries. Revista di Frutticoltura e di Ortofloricoltura 2000, 62, 71–75. [Google Scholar]
- Wang, A.Y.; Lou, B.G.; Xu, T.; Lin, C. Defense responses in tomato fruit induced by oligandrin against Botrytis cinerea. Afr. J. Biotechnol. 2011, 10, 4596–4601. [Google Scholar]
- Tesfay, S.Z.; Bertling, I.; Bower, J.P. Effects of postharvest potassium silicate application on phenolics and other anti-oxidant systems aligned to avocado fruit quality. Postharvest Biol. Technol. 2011, 60, 92–99. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ruiz-García, Y.; Gómez-Plaza, E. Elicitors: A Tool for Improving Fruit Phenolic Content. Agriculture 2013, 3, 33-52. https://doi.org/10.3390/agriculture3010033
Ruiz-García Y, Gómez-Plaza E. Elicitors: A Tool for Improving Fruit Phenolic Content. Agriculture. 2013; 3(1):33-52. https://doi.org/10.3390/agriculture3010033
Chicago/Turabian StyleRuiz-García, Yolanda, and Encarna Gómez-Plaza. 2013. "Elicitors: A Tool for Improving Fruit Phenolic Content" Agriculture 3, no. 1: 33-52. https://doi.org/10.3390/agriculture3010033
APA StyleRuiz-García, Y., & Gómez-Plaza, E. (2013). Elicitors: A Tool for Improving Fruit Phenolic Content. Agriculture, 3(1), 33-52. https://doi.org/10.3390/agriculture3010033





