Fetal Cell Based Prenatal Diagnosis: Perspectives on the Present and Future
Abstract
:1. Introduction

2. Current Status of Fetal Cell Based Non-Invasive Prenatal Diagnosis
3. Revisiting the Pursuit
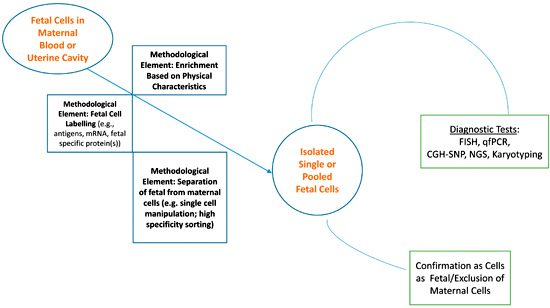
3.1. Fetal Cells in Maternal Blood
- An acknowledged rarity of intact fetal cells in maternal circulation;
- The fragility of target cells that makes delays between blood draw and analysis difficult;
- The relatively low efficiency of enrichment methods leading to loss of fetal samples, sample to sample;
- The possible disintegration of chromosomes before the elimination of the nucleus—specifically from nucleated erythrocytes [19]—thus making FISH potentially unreliable;
- The persistence of white cells from prior pregnancies in maternal circulation for considerable periods of time [20]; and, the difficulty of pinning down markers that are sufficiently distinct or differentially expressed to provide a pointer to a fetal cell time after time.
| Cell Type | Comparison | Comments |
|---|---|---|
| Trophoblasts | Advantages |
|
| Disadvantages |
| |
| nRBC’s | Advantages |
|
| Disadvantages |
| |
| Fetal Leucocytes | Advantages |
|
| Disadvantages |
|
3.2. Fetal Cells from the Uterine Cavity
4. Where to from Here?
| Question | DNA Source | Distinctive Qualities | Common Qualities |
|---|---|---|---|
| What are the instrument needs? | Fetal Cells | Cell sorting Microscopes Cell culture (?) CGH-SNP Platform | PCR related NextGen Sequencing |
| Fetal Cell-Free DNA | DNA isolation Mass spectrometer | ||
| What are the advantageous capabilities? | Fetal Cells | Potential to be a diagnostic test and not limited to screening Direct analysis of single (?) or pooled (?) cells using biological measurements Amenable to FISH and/or qfPCR analyses for rapid analysis of aneuploidy Capacity for single gene analysis, variation screening or sequencing Potential for functional and polygenic analyses Amenable to CNV (copy number variation) determinations by CGH-SNP analysis | Non-invasive Capacity for both aneuploidy and CNV analyses |
| Fetal Cell-Free DNA | Preparation more rapid than cells Minimal problems in transporting blood from clinic to centralized labs Potential for CNV determinations by deep sequencing and analysis | ||
| What are the apparent disadvantages or important challenges to be met? | Fetal Cells | Isolation of cells may be labor intensive; cost effective throughput has not been demonstrated Integrity of DNA in possibly apoptotic cells may dictate consistency and quantity of cells required for reliable evaluations Requires faithful and complete amplification of DNA Likely to be few cells analyzed from a sample and thus less likely to be representative of a mosaic condition Stability and integrity maintenance requirements for transport of cells from phlebotomist to laboratory | Requisite equipment and expertise may limit distribution beyond centralized laboratories |
| Fetal Cell-Free DNA | Becoming validated as an effective diagnostic as well as screening test Accuracy seems to depend on level of proportion of extracted DNA derived from fetus Results are based on (powerful) statistical methods rather than direct biological measurements Unable to determine mosaicism if present Degree of extension of analyses beyond aneuploidy yet to be determined and validated |
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Lapaire, O.; Holzgreve, W.; Oosterwijk, J.C.; Brinkhaus, R.; Bianchi, D.W. Georg Schmorl on trophoblasts in the maternal circulation. Placenta 2005, 28, 1–5. [Google Scholar]
- Krabchi, K.; Gros-Louis, F.; Yan, J.; Bronsard, M.; Massé, J.; Forest, J.C.; Drouin, R. Quantification of all fetal nucleated cells in maternal blood between the 18th and 22nd weeks of pregnancy using molecular cytogenetic techniques. Clin. Genet. 2001, 60, 145–150. [Google Scholar]
- Bianchi, D.W.; Simpson, D.W.; Jackson, L.G.; Elias, S.; Holzgreve, W.; Bischoff, F.Z.; Hahn, S.; Johnson, K.L.; Lewis, D.; Wapner, R.J.; et al. Fetal gender and aneuploidy detection using fetal cells in maternal blood: Analysis of nifty I data. Prenat. Diagn. 2002, 22, 609–615. [Google Scholar] [CrossRef]
- Bayrak-Toydemir, P.; Pergament, E.; Fiddler, M. Are fetal cells in maternal plasma really there? We think they are. J. Hum. Genet. 2003, 48, 665–667. [Google Scholar]
- Bianchi, D.W.; Hanson, J. Sharpening the Tools: A summary of a national institutes of health workshop on new technologies for detection of fetal cells in maternal blood for early prenatal diagnosis. J. Matern. Fetal Neonatal Med. 2006, 19, 199–207. [Google Scholar]
- Christensen, B.; Philip, J.; Kolvraa, S.; Lykke-Hansen, L.; Hromodnikova, I.; Gohel, D.; Lorch, T.; Plesch, A.; Bang, J.; Smidt-Jensen, S.; et al. Fetal cells in maternal blood: A comparison of methods for cell isolation and identification. Fetal Diagn. Ther. 2005, 20, 106–112. [Google Scholar]
- Jackson, L. Fetal cells and DNA in maternal blood. Prenat. Diagn. 2003, 23, 837–846. [Google Scholar]
- Yamanishi, D.T.; Xu, J.; Hujsak, P.G.; Yan, Z.; Wang, X.; Wu, L. Enrichment of rare fetal cells from maternal peripheral blood. Exp. Rev. Mol. Diagn. 2002, 2, 303–311. [Google Scholar]
- Kavanagh, D.M.; Kersaudy-Kerhoas, M.; Dhariwal, R.S.; Desmulliez, Y.M.P. Current and emerging techniques of fetal cell separation from maternal blood. J. Chromatogr. B 2010, 878, 1905–1911. [Google Scholar]
- Comas-Gabriel, C.; Echevarria, M.; Rodriguez, M.A.; Serra, B. Noninvasive prenatal testing for fetal aneuploidy. Donald Sch. J. Ultrasound Obstet. Gynecol. 2013, 7, 443–452. [Google Scholar] [CrossRef]
- Futch, T.; Spinosa, J.; Bhatt, S.; de Feo, E.; Rava, R.P.; Sehnert, A.J. Initial clinical laboratory experience in noninvasive prenatal testing for fetal aneuploidy from maternal plasma DNA samples. Prenat. Diagn. 2013, 33, 569–574. [Google Scholar]
- Morain, S.; Greene, M.F.; Mello, M. A new era in noninvasive prenatal testing. N. Engl. J. Med. 2013, 369, 499–501. [Google Scholar] [CrossRef]
- Ashoor, G.; Syngelaki, A.; Wagner, M.; Birdir, C.; Nicolaides, K.H. Chromosome-selective sequencing of maternal plasma cell-free DNA for first trimester detection of trisomy 21 and trisomy 18. Am. J. Obstet. Gynecol. 2012, 31, 237–243. [Google Scholar]
- Benn, P.; Cuckle, H.; Pergament, E. Non-invasive prenatal testing for aneuploidy: Current status and future prospects. Ultrasound Obstet. Gynecol. 2013, 42, 15–33. [Google Scholar] [CrossRef]
- Benn, P.A.; Hsu, L.Y.F. Prenatal diagnosis of chromosomal abnormalities through amniocentesis. In Genetic Disorders and the Fetus, 5th ed.; Milunsky, A., Ed.; The Johns Hopkins University Press: Baltimore, MD, USA, 2004; pp. 214–296. [Google Scholar]
- FIDD Home—Frequency of Inherited Disorders Database. Available online: http://medic.cardiff.ac.uk/fidd/index.aspx (accessed on 4 January 2014).
- Forabosco, A.; Percesepe, A.; Santucci, S. Incidence of non-age-dependent chromosomal abnormalities: A population-based study on 88965 amniocenteses. Eur. J. Hum. Genet. 2009, 17, 897–903. [Google Scholar] [CrossRef]
- Hook, E.B.; Hamerton, J.L. The frequency of chromosome abnormalities detected in consecutive newborn studies; difference between studies; results by sex and severity of phenotypic involvement. In Population Cytogenetics: Studies in Humans; Hook, E.B., Porter, I.H., Eds.; Academic Press: New York, NY, USA, 1977; pp. 63–79. [Google Scholar]
- Babochkina, T.; Merganthaler, S.; de Napoli, G.; Hristoskova, S.; Tercanli, S.; Holzgreve, W.; Hahn, S. Numerous erthyroblasts in maternal blood are impervious to fluorescent in situ hybridization analysis, a feature related to a dense compact nucleus with apoptotic character. Haematologica 2005, 90, 740–745. [Google Scholar]
- Bianchi, D.W.; Zickwolf, G.K.; Weil, G.J.; Maria, M.A.D. Male fetal progenitor cells persist in maternal blood for as long as 27 years postpartum. Proc. Natl. Acad. Sci. USA 1996, 93, 705–708. [Google Scholar]
- Walknoska, J.; Conte, F.A.; Grumbach, M.M. Practical and theoretical implication of fetal/maternal lymphoctye transfer. Lancet 1969, 1, 1119–1122. [Google Scholar]
- Schindler, A.M.; Graf, E.; Marin-du-Pan, R. Prenatal diagnsois of fetal lymphocytes in the maternal blood. Obstet. Gynecol. 1972, 40, 340–345. [Google Scholar]
- Sargent, I.L.; Choo, Y.S.; Redman, C.W.G. Isolating and analyzing fetal leukocytes in maternal blood. Ann. N. Y. Acad. Sci. 1994, 731, 154–161. [Google Scholar]
- Van Wijk, I.J.; Griffioen, S.; Tjoa, M.L.; Mulders, M.A.; van Vugt, J.M.; Loke, Y.W.; Oudejans, C.B. HLA-G expression in trophoblast cells circulating in maternal peripheral blood during early pregnancy. Am. J. Obstet. Gynecol. 2001, 184, 991–997. [Google Scholar]
- Mouawia, H.; Saker, A.; Jais, J.; Benachi, A.; Bussieres, L.; Lacour, B.; Bonnefont, J.; Frydman, R.; Simpson, J.L.; Paterlini-Brechot, P. Circulating trophoblastic cells provide genetic diagnosis in 63 fetuses at risk for cystic fibrosis or spinal muscular atrophy. Reprod. BioMed. Online 2012, 25, 508–520. [Google Scholar]
- Morgenthaler, S.; Babochkina, T.; Kiefer, V.; Lapaire, O.; Holzgreve, W.; Hahn, S. FISH analysis of all fetal nucleated cells in maternal whole blood: Improved specificity by the use of two Y-chromosome probes. J. Histochem. Cytochem. 2005, 53, 319–322. [Google Scholar] [CrossRef]
- Calabrese, G.; Baldi, M.; Fantasia, D.; Sessa, M.T.; Kalantar, M.; Holzhauer, C.; Alunni-Fabbrni, M.; Palka, G.; Sitar, G. Detection of chromosomal aneuploidies in fetal cells isolated from maternal blood using single-chromosome dual-probe fish analysis. Clin. Genet. 2012, 82, 131–139. [Google Scholar]
- NCBI Blood Group Antigen Gene Mutation Database. dbRBC: I Blood Group System. Available online: http://www.ncbi.nlm.nih.gov/gv/rbc/xslcgi.fcgi?cmd=bgmut/systems_info&system=i (accessed on 2 April 2014).
- Hristoskova, S.; Holzgreve, W.; Hahn, S. More than one-half of the erythroblasts in the fetal circulation and cord blood are TUNEL positive. Clin. Chem. 2001, 47, 1870–1871. [Google Scholar]
- Rodeck, C.; Tutschek, B.; Sherlock, J.; Kingdom, J. Methods for the transcervical collection of fetal cells during the first trimester of pregnancy. Prenat. Diagn. 1995, 15, 933–942. [Google Scholar]
- Schueler, P.A.; Yamanishi, D.T.; Collarini, E.J.; Pearson, J.; Lee, Y.; Cain, C.A.; Edwards, J.; Foltz, L.; Wu, X.; Mahoney, W. Enrichment and identification of fetal trophoblast cells from first trimester maternal cervical lavage and uterine blood specimens. Placenta 2001, 22, 688–701. [Google Scholar] [CrossRef]
- Imudia, A.N.; Kumar, S.; Diamond, M.P.; DeCherney, A.H.; Armant, D.R. Transcervical retrieval of fetal cells in the practice of modern medicine: A review of the current literature and future direction. Fertil. Steril. 2010, 6, 1725–1730. [Google Scholar]
- Adinolfi, M.; Sherlock, J. First trimester prenatal diagnosis using transcervical cells: An evaluation. Hum. Reprod. Update 1997, 3, 383–392. [Google Scholar]
- Cioni, R.; Bussani, C.; Bucciantini, S.; Scarselli, G. Fetal cells in a transcervical cell sample collected at 5 weeks of gestation. J. Matern. Fetal Neonatal Med. 2005, 18, 271–273. [Google Scholar]
- Sinosich, M. Abstracts: Isolation of syncytiotrophoblast cells: Potential for non-invasive prenatal molecular analysis. In Proceedings of 12th World Congress in Fetal Medicine, Marbella, Spain, 23–27 June, 2013.
- Bulmer, J.N.; Rodeck, C.; Adinolfi, M. Immunohistochemical characterization of cells retrieved by transcervical sampling in early pregnancy. Prenat. Diagn. 1995, 15, 1143–1153. [Google Scholar]
- Loke, Y.W.; King, A.; Gardner, L.; Bowen, M.; Hiby, S.; Howlett, S.; Holmes, N.; Jacobs, D. Evaluation of trophoblast HLA-G antigen with a specific monoclonal antibody. Tissue Antigens 1997, 50, 135–146. [Google Scholar]
- Prigodich, A.E.; Randeria, P.S.; Briley, W.E.; Kim, N.J.; Daniel, W.L.; Giljohann, D.A.; Mirkin, C.A. Multiplexed nanoflares: mRNA detection in live cells. Anal. Chem. 2012, 84, 2062–2066. [Google Scholar] [CrossRef]
- Sifakis, S.; Ghatpande, S.; Seppo, A.; Kilpatrick, M.W.; Tafas, T.; Tsipouras, P.; Fejgin, M.; Amiel, A. Prenatal diagnosis of trisomy 21 through detection of trophoblasts in cervical smears. Early Hum. Dev. 2010, 86, 311–313. [Google Scholar] [CrossRef]
- Fejgin, M.D.; Diukman, R.; Cotton, Y.; Weinstein, G.; Amiel, A. Fetal cells in the uterine cervix: A source for early non-invasive prenatal diagnosis. Prenat. Diagn. 2001, 21, 619–621. [Google Scholar]
- Imudia, A.N.; Suzuki, Y.; Kilburn, B.A.; Yelian, F.D.; Diamond, M.P.; Romero, R.; Armant, D.R. Retrieval of trophoblast cells from the cervical canal for prediction of abnormal pregnancy: A pilot study. Hum. Reprod. 2009, 24, 2086–2092. [Google Scholar]
- Biron-Shental, T.; Fejgin, M.D.; Sifakis, S.; Liberman, M.; Anstsaklis, A.; Amiel, A. Endoreduplication in cervical trophoblast cells from normal pregnancies. J. Matern. Fetal Neonatal Med. 2012, 25, 2625–2628. [Google Scholar] [CrossRef]
- Wapner, R.J.; Martin, C.L.; Levy, B.; Baliff, B.C.; Eng, C.M.; Zachary, J.M.; Savage, M.; Platt, L.D.; Saltzman, M.D.; Grobman, W.A.; et al. Chromosomal microarray versus karyotyping for prenatal diagnosis. N. Engl. J. Med. 2012, 367, 2175–2184. [Google Scholar]
- Alix-Panabieres, C.; Pantel, K. Technologies for detection of circulating tumor cells: Facts and vision. Lab Chip 2013, 14, 57–62. [Google Scholar]
- Silicon Biosystems, Inc. Available online: http://www.siliconbiosystems.com/ (accessed on 17 January 2014).
- Zong, C.; Lu, S.; Chapman, A.R.; Xie, X.S. Genome-wide detection of single-nucleotide and copy-number variations of a single human cell. Science 2012, 338, 1622–1626. [Google Scholar]
- Sokolova, M.; El Emam, K.; Chowdhury, S.; Neri, E.; Rose, S.; Jonker, E. Evaluation of rare event detection. In Advances in Artificial Intelligence; Lecture Notes in Computer Science 6085; Farzindar, A., Kešelj, V., Eds.; Springer: Berlin/Heidelberg, Germany, 2010; pp. 379–383. [Google Scholar]
- Michael, J.W.; Jeffrey, D.S.; Kamal, M.A.; Ashok, N.S. Advances in Machine Learning and Data Mining for Astronomy; CRC Press: Boca Raton, FL, USA, 2012. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Fiddler, M. Fetal Cell Based Prenatal Diagnosis: Perspectives on the Present and Future. J. Clin. Med. 2014, 3, 972-985. https://doi.org/10.3390/jcm3030972
Fiddler M. Fetal Cell Based Prenatal Diagnosis: Perspectives on the Present and Future. Journal of Clinical Medicine. 2014; 3(3):972-985. https://doi.org/10.3390/jcm3030972
Chicago/Turabian StyleFiddler, Morris. 2014. "Fetal Cell Based Prenatal Diagnosis: Perspectives on the Present and Future" Journal of Clinical Medicine 3, no. 3: 972-985. https://doi.org/10.3390/jcm3030972