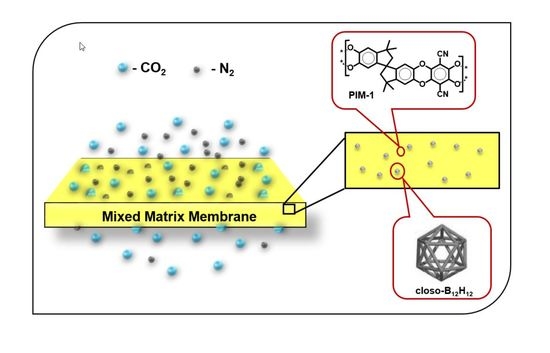
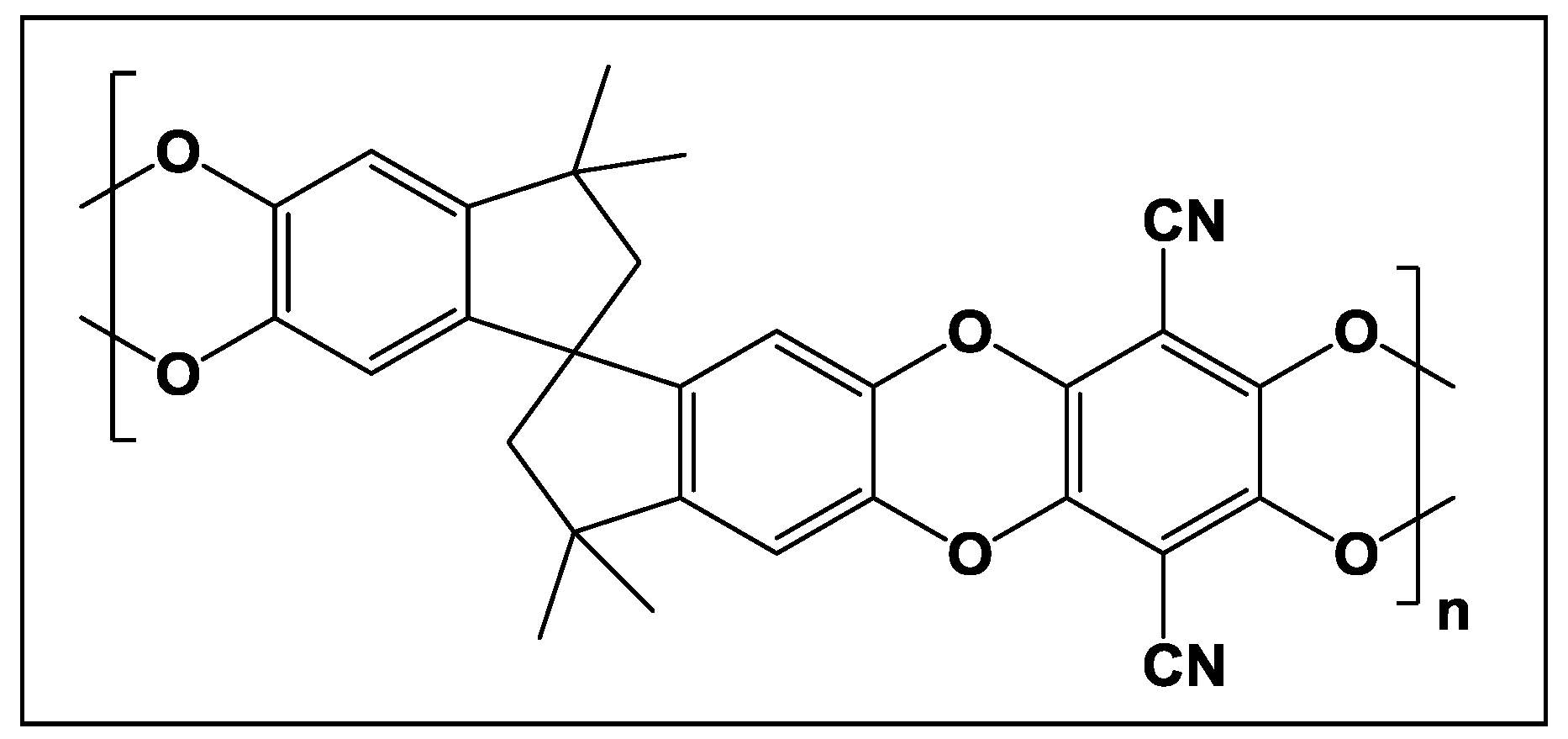
Mixed Matrix Membranes of Boron Icosahedron and Polymers of Intrinsic Microporosity (PIM-1) for Gas Separation
Abstract
:1. Introduction
2. Theory and Background
2.1. Gas Sorption
2.1.1. Static Gas Sorption
2.1.2. Dynamic Gas Sorption
2.2. Gas Permeation
3. Materials
4. Experimental Section
4.1. Pristine PIM-1 Synthesis and Mixed Matrix Membranes Preparation
4.2. Thermal Gravimetric Analysis (TGA)
4.3. Scanning Electron Microscopy (SEM)
4.4. Density Measurements
4.5. Gas Transport Properties
4.6. Gas Sorption
4.6.1. Static Sorption Experiments
4.6.2. Dynamic Sorption Experiments
5. Results and Discussion
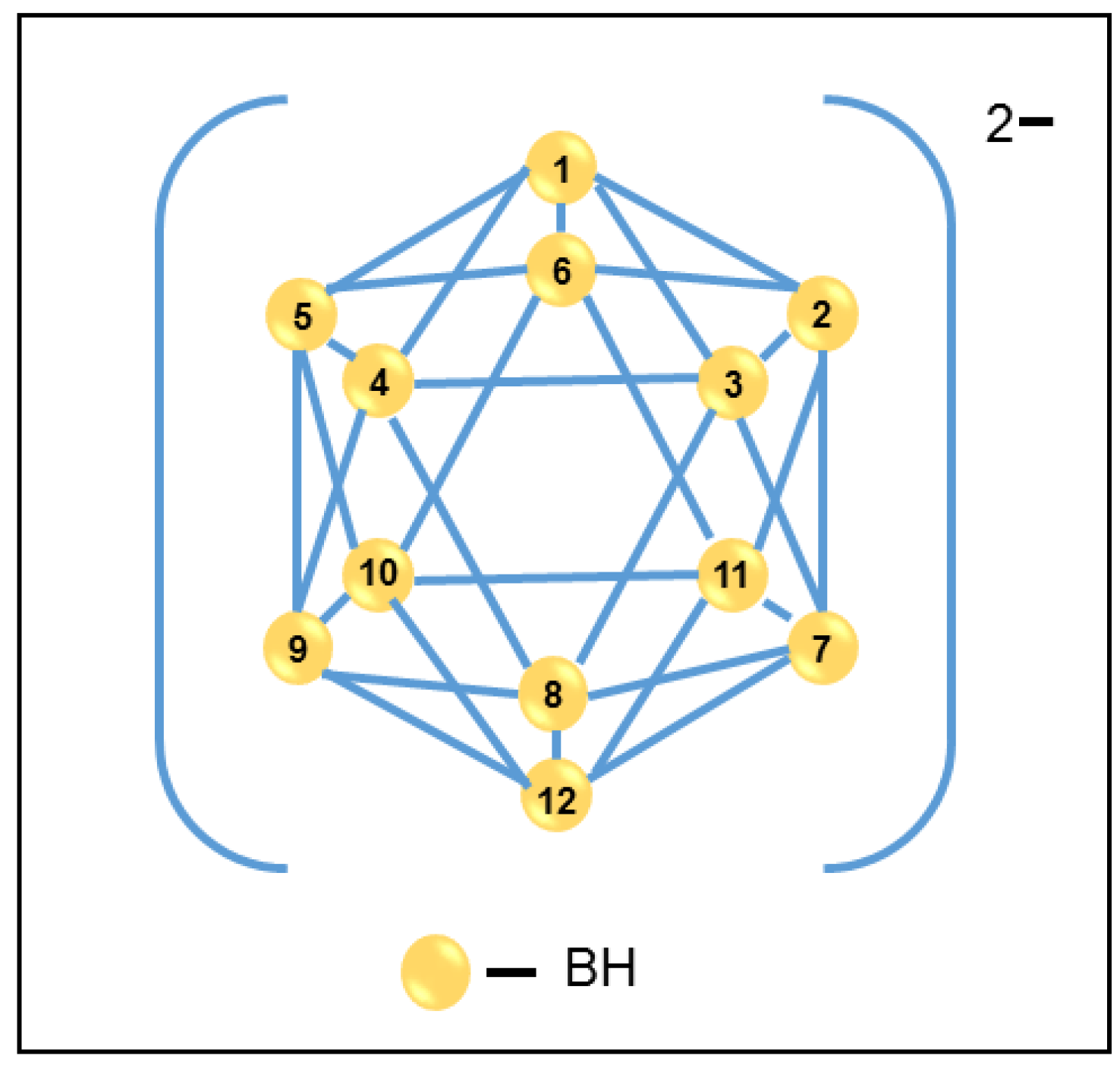
5.1. Inorganic Particle Characterization
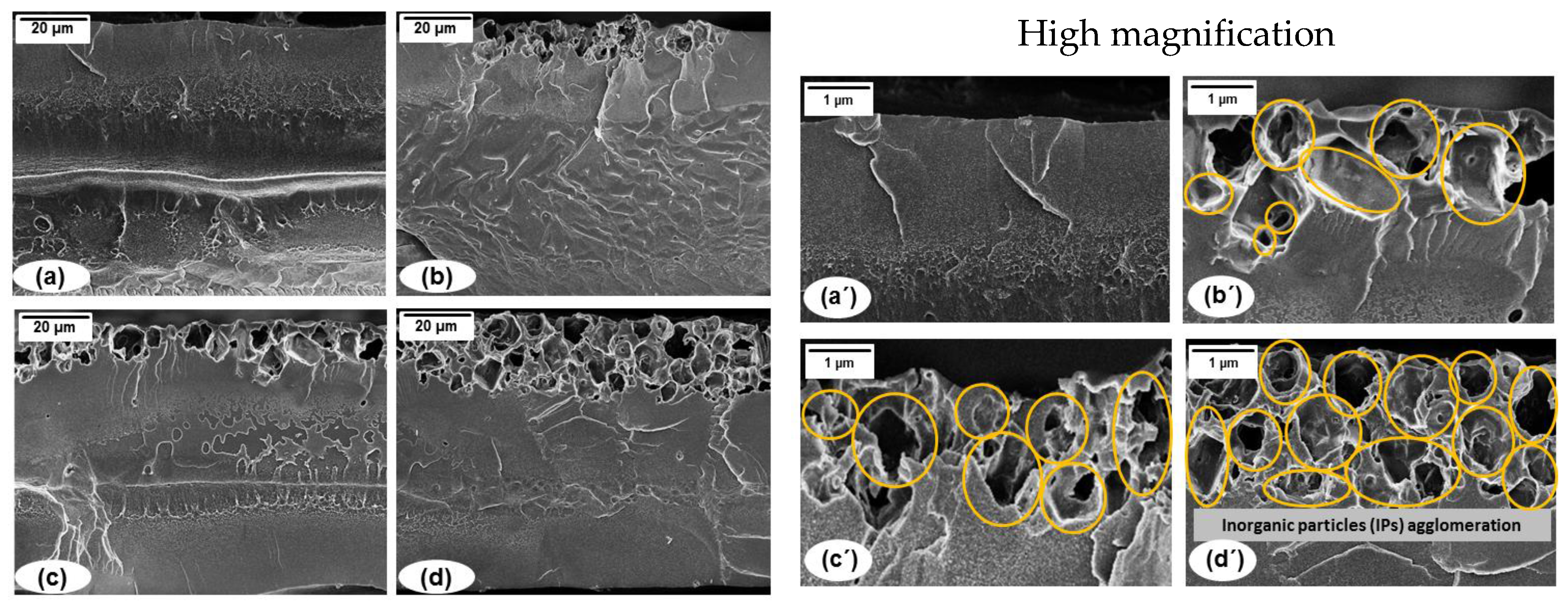
5.2. Mixed Matrix Membranes (MMMs) Characterization
5.3. Gas Permeation Properties
5.3.1. Effects of K2B12H12 Content on PIM1/K2B12H12 MMM Gas Separation Performance
5.3.2. Influence of Temperature on the Gas Separation Performance of PIM1/K2B12H12 MMMs
5.4. Gas Sorption
5.4.1. Static Gas Sorption
5.4.2. Dynamic Gas Sorption
6. Conclusions
Reference
Acknowledgments
Author contribution
Conflicts of interest
References
- Freeman, B.D. Basis of permeability/selectivity tradeoff relations in polymeric gas separation membranes. Macromolecules 1999, 32, 375–380. [Google Scholar] [CrossRef]
- Robeson, L.M. Correlation of separation factor versus permeability for polymeric membranes. J. Membr. Sci. 1991, 62, 165–185. [Google Scholar] [CrossRef]
- Cornelius, C.J.; Marand, E. Hybrid silica-polyimide composite membranes: Gas transport properties. J. Membr. Sci. 2002, 202, 97–118. [Google Scholar] [CrossRef]
- Mahajan, R.; Burns, R.; Schaeffer, M.; Koros, W.J. Challenges in forming successful mixed matrix membranes with rigid polymeric materials. J. Appl. Polym. Sci. 2002, 86, 881–890. [Google Scholar] [CrossRef]
- Bushell, A.F.; Attfield, M.P.; Mason, C.R.; Budd, P.M.; Yampolskii, Y.; Starannikova, L.; Rebrov, A.; Bazzarelli, F.; Bernardo, P.; Carolus Jansen, J.; et al. Gas permeation parameters of mixed matrix membranes based on the polymer of intrinsic microporosity pim-1 and the zeolitic imidazolate framework zif-8. J. Membr. Sci. 2013, 427, 48–62. [Google Scholar] [CrossRef]
- Ismail, A.F.; Rahim, R.A.; Rahman, W.A.W.A. Characterization of polyethersulfone/matrimid® 5218 miscible blend mixed matrix membranes for o2/n2 gas separation. Sep. Purif. Technol. 2008, 63, 200–206. [Google Scholar] [CrossRef]
- Safronov, A.V.; Jalisatgi, S.S.; Lee, H.B.; Hawthorne, M.F. Chemical hydrogen storage using polynuclear borane anion salts. Int. J. Hydrogen Energy 2011, 36, 234–239. [Google Scholar] [CrossRef]
- Stibr, B. Carboranes other than C2B10H12. Chem. Rev. 1992, 92, 225–250. [Google Scholar] [CrossRef]
- Bregadze, V.I.; Sivaev, I.B.; Glazun, S.A. Polyhedral boron compounds as potential diagnostic and therapeutic antitumor agents. Anti-Cancer Agents Med. Chem. Anti-Cancer Agents 2006, 6, 75–109. [Google Scholar] [CrossRef]
- Li, T.; Jalisatgi, S.S.; Bayer, M.J.; Maderna, A.; Khan, S.I.; Hawthorne, M.F. Organic syntheses on an icosahedral borane surface: Closomer structures with twelvefold functionality. J. Am. Chem. Soc. 2005, 127, 17832–17841. [Google Scholar] [CrossRef] [PubMed]
- Plesek, J. Potential applications of the boron cluster compounds. Chem. Rev. 1992, 92, 269–278. [Google Scholar] [CrossRef]
- Sivaev, I.B.; Bregadze, V.V. Polyhedral boranes for medical applications: Current status and perspectives. Eur. J. Inorg. Chem. 2009, 2009, 1433–1450. [Google Scholar] [CrossRef]
- Drissner, D.; Kunze, G.; Callewaert, N.; Gehrig, P.; Tamasloukht, M.B.; Boller, T.; Felix, G.; Amrhein, N.; Bucher, M. Lyso-phosphatidylcholine is a signal in the arbuscular mycorrhizal symbiosis. Science 2007, 318, 265–268. [Google Scholar] [CrossRef] [PubMed]
- Budd, P.M.; Ghanem, B.S.; Makhseed, S.; McKeown, N.B.; Msayib, K.J.; Tattershall, C.E. Polymers of intrinsic microporosity (PIMs): Robust, solution-processable, organic nanoporous materials. Chem. Commu. 2004, 230–231. [Google Scholar] [CrossRef] [PubMed]
- Budd, P.M.; McKeown, N.B.; Ghanem, B.S.; Msayib, K.J.; Fritsch, D.; Starannikova, L.; Belov, N.; Sanfirova, O.; Yampolskii, Y.; Shantarovich, V. Gas permeation parameters and other physicochemical properties of a polymer of intrinsic microporosity: Polybenzodioxane PIM-1. J. Membr. Sci. 2008, 325, 851–860. [Google Scholar] [CrossRef]
- Budd, P.M.; Msayib, K.J.; Tattershall, C.E.; Ghanem, B.S.; Reynolds, K.J.; McKeown, N.B.; Fritsch, D. Gas separation membranes from polymers of intrinsic microporosity. J. Membr. Sci. 2005, 251, 263–269. [Google Scholar] [CrossRef]
- McKeown, N.B.; Budd, P.M. Polymers of intrinsic microporosity (PIMS): Organic materials for membrane separations, heterogeneous catalysis and hydrogen storage. Chem. Soc. Rev. 2006, 35, 675–683. [Google Scholar] [CrossRef] [PubMed]
- Koros, W.J.; Chern, R.T. Handbook of Separation Process Technology; John Wiley & Sons: Hoboken, NJ, USA, 1987. [Google Scholar]
- Story, B.J.; Koros, W.J. Comparison of three models for permeation of CO2/CH4 mixtures in poly(phenylene oxide). J. Polym. Sci. B Polym. Phys. 1989, 27, 1927–1948. [Google Scholar] [CrossRef]
- Crank, J. The Mathematics of Diffusion; Oxford Press: London, UK, 1990. [Google Scholar]
- Wijmans, J.G.; Baker, R.W. The solution-diffusion model: A review. J. Membr. Sci. 1995, 107, 1–21. [Google Scholar] [CrossRef]
- Yampolskii, Y.; Pinnau, I.; Freeman, B.D. Material Science of Membranes; John Wiley & Sons: Chichester, UK, 2007. [Google Scholar]
- Fritsch, D.; Bengtson, G.; Carta, M.; McKeown, N.B. Synthesis and gas permeation properties of spirobischromane-based polymers of intrinsic microporosity. Macromol. Chem. Phys. 2011, 212, 1137–1146. [Google Scholar] [CrossRef]
- Khan, M.M.; Bengtson, G.; Shishatskiy, S.; Gacal, B.N.; Mushfequr Rahman, M.; Neumann, S.; Filiz, V.; Abetz, V. Cross-linking of polymer of intrinsic microporosity (pim-1) via nitrene reaction and its effect on gas transport property. Eur. Polym. J. 2013, 49, 4157–4166. [Google Scholar] [CrossRef]
- Khan, M.; Filiz, V.; Bengtson, G.; Shishatskiy, S.; Rahman, M.; Abetz, V. Functionalized carbon nanotubes mixed matrix membranes of polymers of intrinsic microporosity for gas separation. Nanoscale Res. Lett. 2012, 7, 504. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.; Filiz, V.; Emmler, T.; Abetz, V.; Koschine, T.; Rätzke, K.; Faupel, F.; Egger, W.; Ravelli, L. Free volume and gas permeation in anthracene maleimide-based polymers of intrinsic microporosity. Membranes 2015, 5, 214–227. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.M.; Bengtson, G.; Neumann, S.; Rahman, M.M.; Abetz, V.; Filiz, V. Synthesis, characterization and gas permeation properties of anthracene maleimide-based polymers of intrinsic microporosity. RSC Adv. 2014, 4, 32148–32160. [Google Scholar] [CrossRef]
- Hill, A.J.; Pas, S.J.; Bastow, T.J.; Burgar, M.I.; Nagai, K.; Toy, L.G.; Freeman, B.D. Influence of methanol conditioning and physical aging on carbon spin-lattice relaxation times of poly(1-trimethylsilyl-1-propyne). J. Membr. Sci. 2004, 243, 37–44. [Google Scholar] [CrossRef]
- Nagai, K.; Toy, L.G.; Freeman, B.D.; Teraguchi, M.; Masuda, T.; Pinnau, I. Gas permeability and hydrocarbon solubility of poly[1-phenyl-2-[p-(triisopropylsilyl)phenyl]acetylene]. J. Polym. Sci. B Polym. Phys. 2000, 38, 1474–1484. [Google Scholar] [CrossRef]
- Rahman, M.M.; Filiz, V.; Shishatskiy, S.; Abetz, C.; Neumann, S.; Bolmer, S.; Khan, M.M.; Abetz, V. Pebax® with peg functionalized poss as nanocomposite membranes for CO2 separation. J. Membr. Sci. 2013, 437, 286–297. [Google Scholar] [CrossRef]
- Rahman, M.M.; Filiz, V.; Shishatskiy, S.; Neumann, S.; Khan, M.M.; Abetz, V. Peg functionalized poss incorporated pebax nanocomposite membranes. Procedia Eng. 2012, 44, 1523–1526. [Google Scholar] [CrossRef]
- Shishatskii, A.M.; Yampol’skii, Y.P.; Peinemann, K.V. Effects of film thickness on density and gas permeation parameters of glassy polymers. J. Membr. Sci. 1996, 112, 275–285. [Google Scholar] [CrossRef]
- Macdonald, D.D. The mathematics of diffusion. In Transient Techniques in Electrochemistry; Springer: Boston, MA, USA, 1977; pp. 47–67. [Google Scholar]
- Moore, T.T.; Koros, W.J. Non-ideal effects in organic–inorganic materials for gas separation membranes. J. Mol. Struct. 2005, 739, 87–98. [Google Scholar] [CrossRef]
- Byoyancy Phenomenon in TGA System, Thermal Analysis and Surface Solution Gmbh (Thermo Electron Corporation). Available online: http://www.thass.org/ (accessed on 29 December 2017).
- Rezakazemi, M.; Ebadi Amooghin, A.; Montazer-Rahmati, M.M.; Ismail, A.F.; Matsuura, T. State-of-the-art membrane based CO2 separation using mixed matrix membranes (MMMs): An overview on current status and future directions. Prog. Polym. Sci. 2014, 39, 817–861. [Google Scholar] [CrossRef]
- Robeson, L.M. The upper bound revisited. J. Membr. Sci. 2008, 320, 390–400. [Google Scholar] [CrossRef]
- Khan, M.M.; Filiz, V.; Bengtson, G.; Shishatskiy, S.; Rahman, M.M.; Lillepaerg, J.; Abetz, V. Enhanced gas permeability by fabricating mixed matrix membranes of functionalized multiwalled carbon nanotubes and polymers of intrinsic microporosity (PIM). J. Membr. Sci. 2013, 436, 109–120. [Google Scholar] [CrossRef]
- Pinnau, I.; Toy, L.G. Gas and vapor transport properties of amorphous perfluorinated copolymer membranes based on 2,2-bistrifluoromethyl-4,5-difluoro-1,3-dioxole/tetrafluoroethylene. J. Membr. Sci. 1996, 109, 125–133. [Google Scholar] [CrossRef]
- Koros, W.J.; Chan, A.H.; Paul, D.R. Sorption and transport of various gases in polycarbonate. J. Membr. Sci. 1977, 2, 165–190. [Google Scholar] [CrossRef]
- Wang, R.; Cao, C.; Chung, T.-S. A critical review on diffusivity and the characterization of diffusivity of 6FDA–6FPDA polyimide membranes for gas separation. J. Membr. Sci. 2002, 198, 259–271. [Google Scholar] [CrossRef]
- Paul, D.R. Gas sorption and transport in glassy polymers. Ber. Bunsenges. Phys. Chem. 1979, 83, 294–302. [Google Scholar] [CrossRef]
- Merkel, T.C.; He, Z.; Pinnau, I.; Freeman, B.D.; Meakin, P.; Hill, A.J. Sorption and transport in poly(2,2-bis(trifluoromethyl)-4,5-difluoro-1,3-dioxole-co-tetrafluoroethylene) containing nanoscale fumed silica. Macromolecules 2003, 36, 8406–8414. [Google Scholar] [CrossRef]
- Lagorsse, S.; Magalhães, F.D.; Mendes, A. Carbon molecular sieve membranes: Sorption, kinetic and structural characterization. J. Membr. Sci. 2004, 241, 275–287. [Google Scholar] [CrossRef]





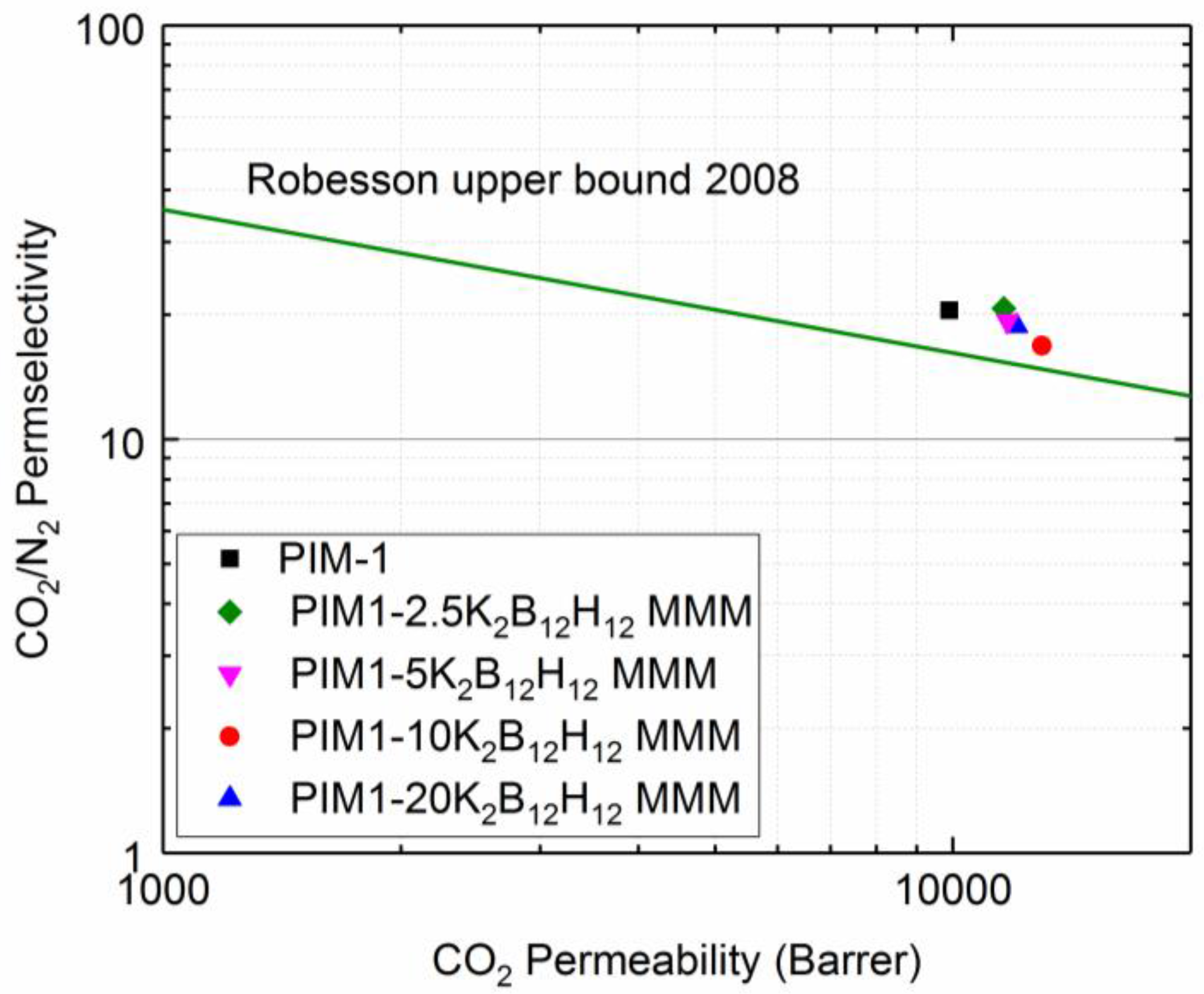
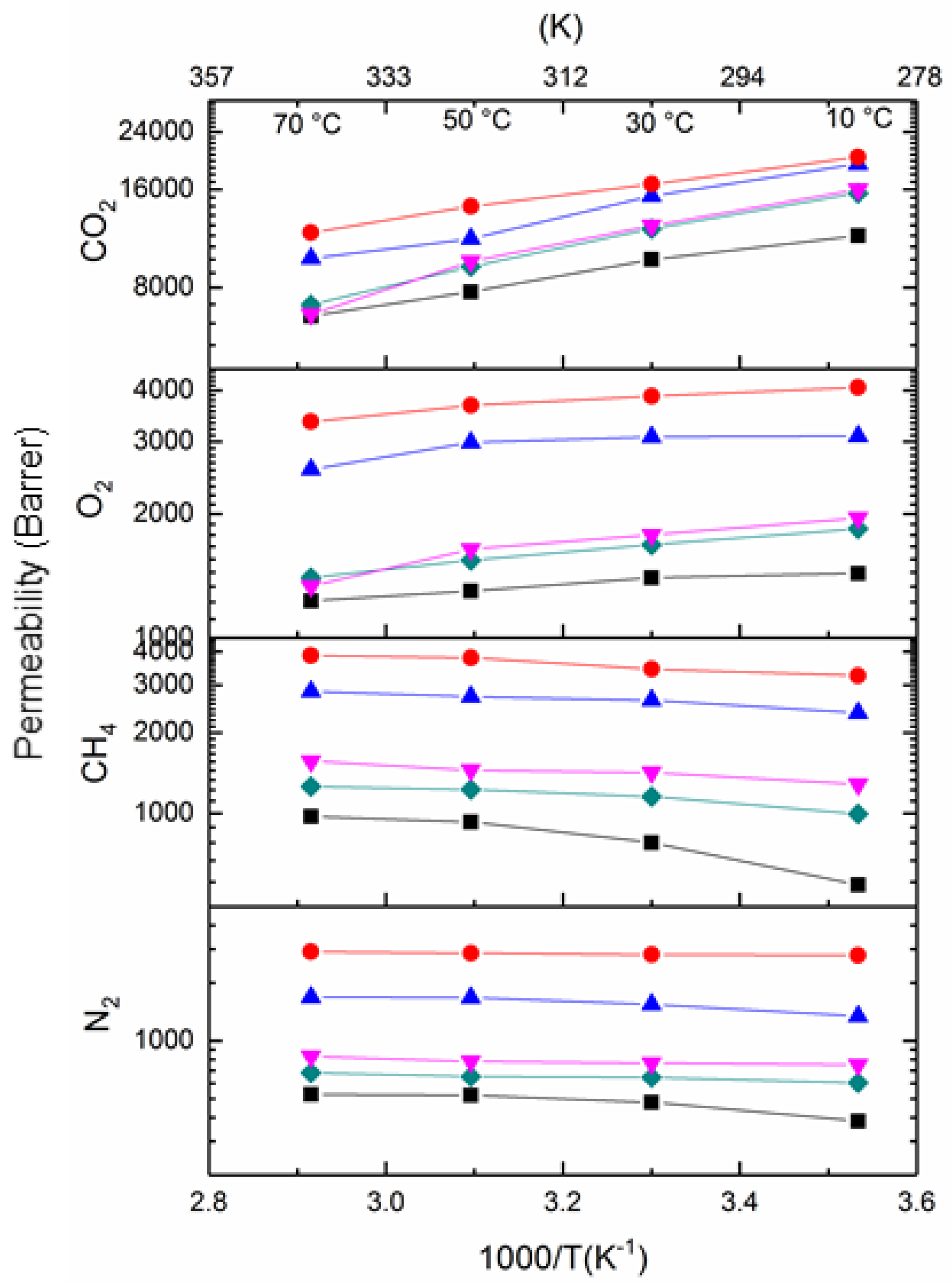
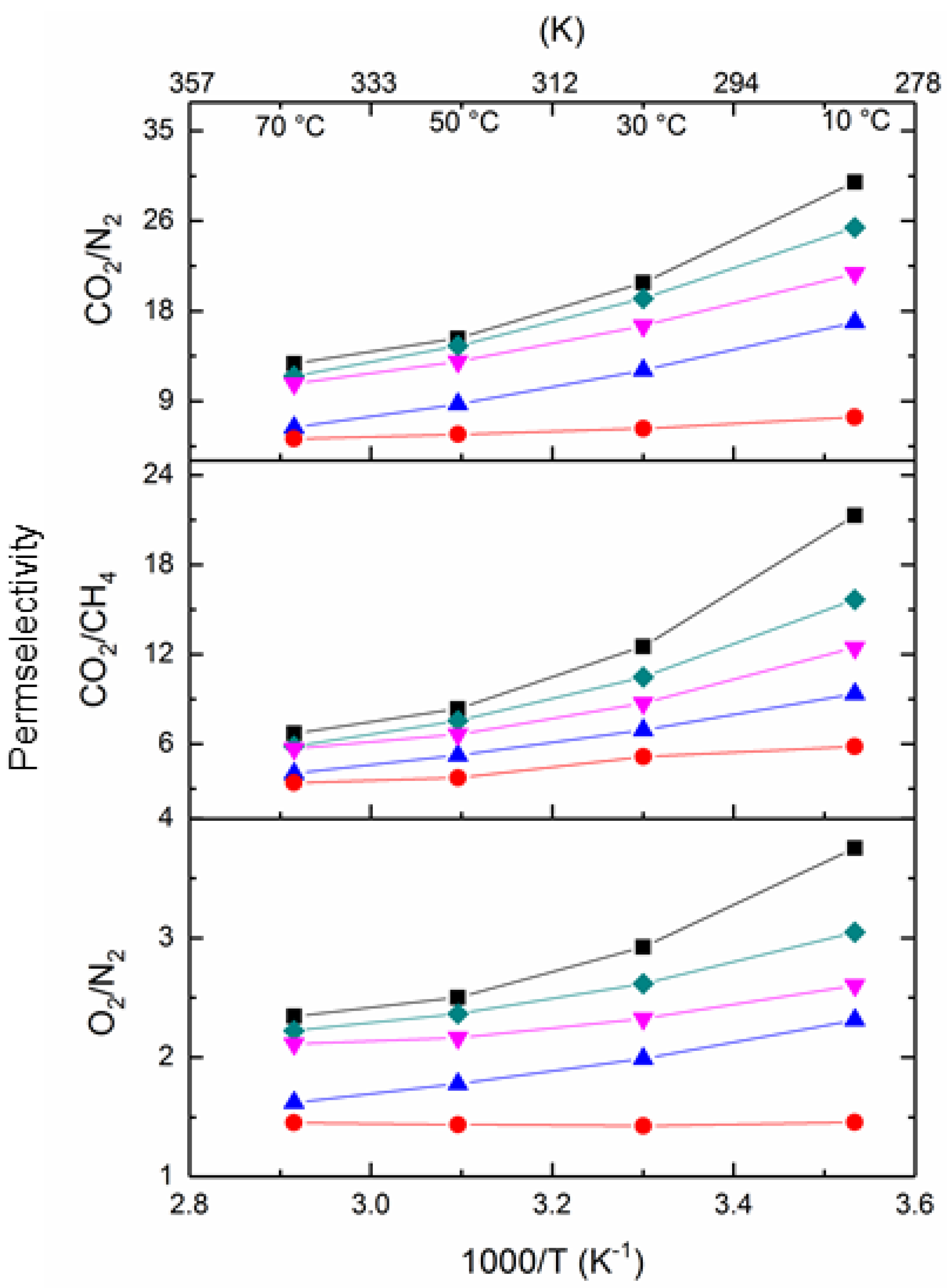
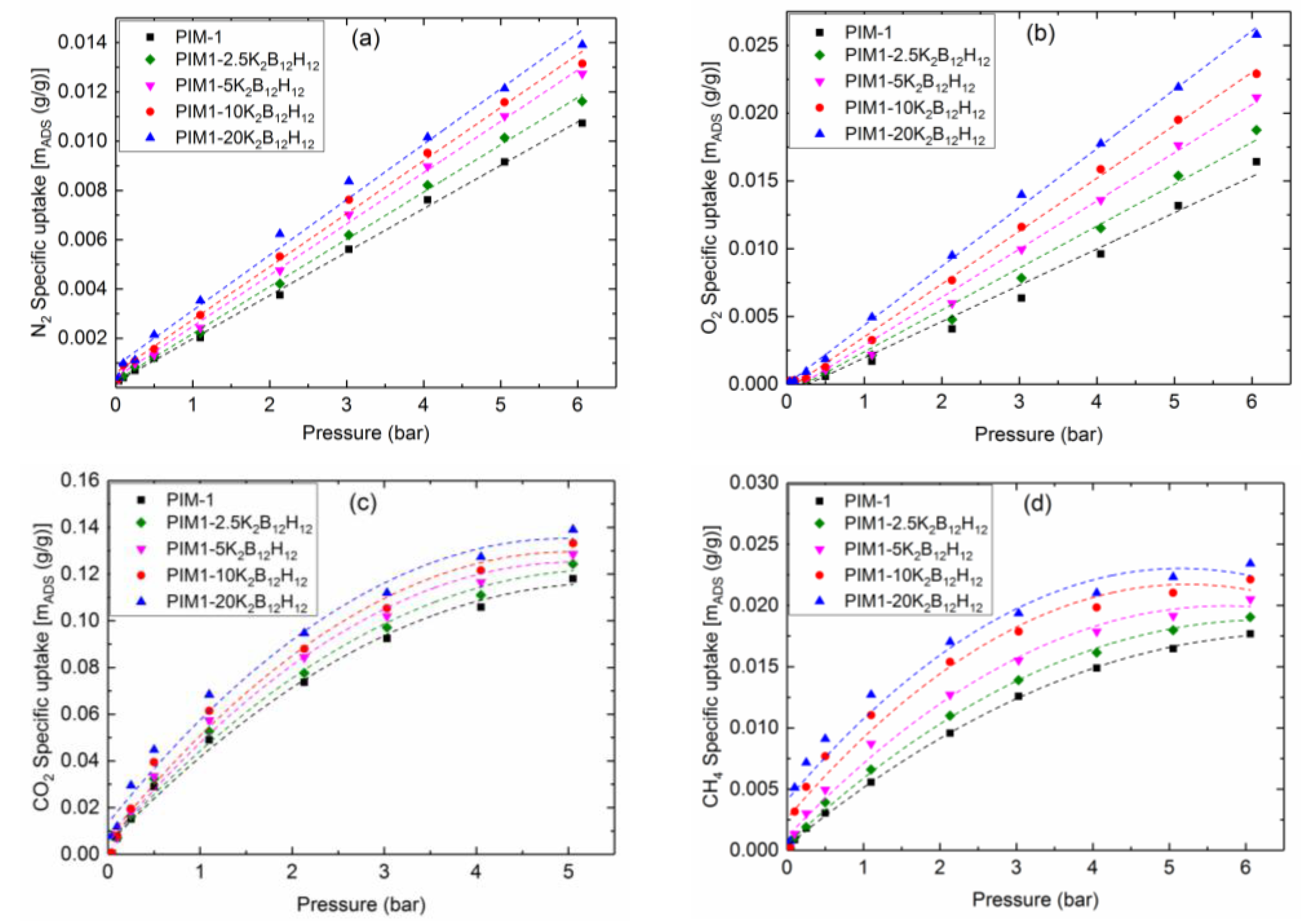


| Membrane | Volume Fraction φIP (%) | K2B12H12 Loading (%) | w700 (%) | ρ (g/cm3) |
|---|---|---|---|---|
| PIM-1 | 0 | 0 | 32.17 | 1.066 |
| PIM-2.5 K2B12H12 | 2.58 | 2.5 | 32.46 | 1.078 |
| PIM-5 K2B12H12 | 5.16 | 5 | 33.10 | 1.077 |
| PIM-10 K2B12H12 | 10.21 | 10 | 33.20 | 1.072 |
| PIM-20 K2B12H12 | 20.53 | 20 | 34.43 | 1.067 |
| K2B12H12 | - | - | 0.6 | 1.031 * |
| Membrane | Permeability (Barrer) | ||||
|---|---|---|---|---|---|
| H2 | N2 | O2 | CO2 | CH4 | |
| PIM-1 | 3274 ± 5 | 483 ± 10 | 1396 ± 13 | 9896 ± 28 | 789 ± 15 |
| PIM1-2.5 K2B12H12 MMM | 3347 ± 8 (3%) | 562 ± 11 (16%) | 1539 ± 12 (10%) | 11598 ± 20 (17%) | 974 ± 18 (23%) |
| PIM1-5 K2B12H12 MMM | 3707 ± 9 (13%) | 641 ± 10 (33%) | 1675 ± 11 (20%) | 12036 ± 21 (22%) | 1148 ± 16 (45%) |
| PIM1-10 K2B12H12 MMM | 4025 ± 8 (22%) | 772 ± 9 (60%) | 1831 ± 14 (31%) | 12954 ± 23 (31%) | 1436 ± 16 (82%) |
| PIM1-20 K2B12H12 MMM | 3436 ± 7 (5%) | 607 ± 12 (25%) | 1600 ± 14 (14%) | 11729 ± 23 (18%) | 1123 ± 14 (42%) |
| Membrane | Permselectivity | |||||
|---|---|---|---|---|---|---|
| H2/N2 | H2/CH4 | CH4/N2 | O2/N2 | CO2/N2 | CO2/CH4 | |
| PIM-1 | 6.8 | 4.2 | 1.6 | 2.9 | 20.5 | 12.5 |
| PIM1-2.5 K2B12H12 MMM | 6.0 | 3.4 | 1.7 | 2.7 | 20.7 | 11.9 |
| PIM1-5 K2B12H12 MMM | 5.8 | 3.2 | 1.8 | 2.6 | 18.8 | 10.5 |
| PIM1-10 K2B12H12 MMM | 5.2 | 2.8 | 1.9 | 2.4 | 16.8 | 9.0 |
| PIM1-20 K2B12H12 MMM | 5.6 | 3.0 | 1.8 | 2.6 | 19.3 | 10.4 |
| Membrane | EP (kJ/mol) | |
|---|---|---|
| N2 | CO2 | |
| PIM-1 | 18.5 | −3.3 |
| PIM1-2.5 K2B12H12 MMM | 13.7 | −4.0 |
| PIM1-5 K2B12H12 MMM | 6.4 | −4.6 |
| PIM1-10 K2B12H12 MMM | 5.5 | −5.0 |
| PIM1-20 K2B12H12 MMM | 2.4 | −3.1 |
| Feed Gas | K2B12H12 Loading (wt %) | Dual Mode Sorption Model Parameter | ||
|---|---|---|---|---|
| kD | C′H | b | ||
| CO2 | 0 | 2.330 | 104.630 | 0.415 |
| 2.5 | 2.440 | 105.830 | 0.422 | |
| 5 | 2.530 | 113.750 | 0.440 | |
| 10 | 2.600 | 120.105 | 0.491 | |
| 20 | 2.58 | 115.81 | 0.444 | |
| CH4 | 0 | 0.581 | 62.097 | 0.135 |
| 2.5 | 0.59 | 63.957 | 0.141 | |
| 5 | 0.604 | 65.042 | 0.154 | |
| 10 | 0.627 | 66.741 | 0.167 | |
| 20 | 0.611 | 65.412 | 0.151 | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khan, M.M.; Shishatskiy, S.; Filiz, V. Mixed Matrix Membranes of Boron Icosahedron and Polymers of Intrinsic Microporosity (PIM-1) for Gas Separation. Membranes 2018, 8, 1. https://doi.org/10.3390/membranes8010001
Khan MM, Shishatskiy S, Filiz V. Mixed Matrix Membranes of Boron Icosahedron and Polymers of Intrinsic Microporosity (PIM-1) for Gas Separation. Membranes. 2018; 8(1):1. https://doi.org/10.3390/membranes8010001
Chicago/Turabian StyleKhan, Muntazim Munir, Sergey Shishatskiy, and Volkan Filiz. 2018. "Mixed Matrix Membranes of Boron Icosahedron and Polymers of Intrinsic Microporosity (PIM-1) for Gas Separation" Membranes 8, no. 1: 1. https://doi.org/10.3390/membranes8010001