Polymeric Pseudo-Liquid Membranes from Poly(N-oleylacrylamide)
Abstract
:1. Introduction
2. Results and Discussion
2.1. Preparation of PC18AAm

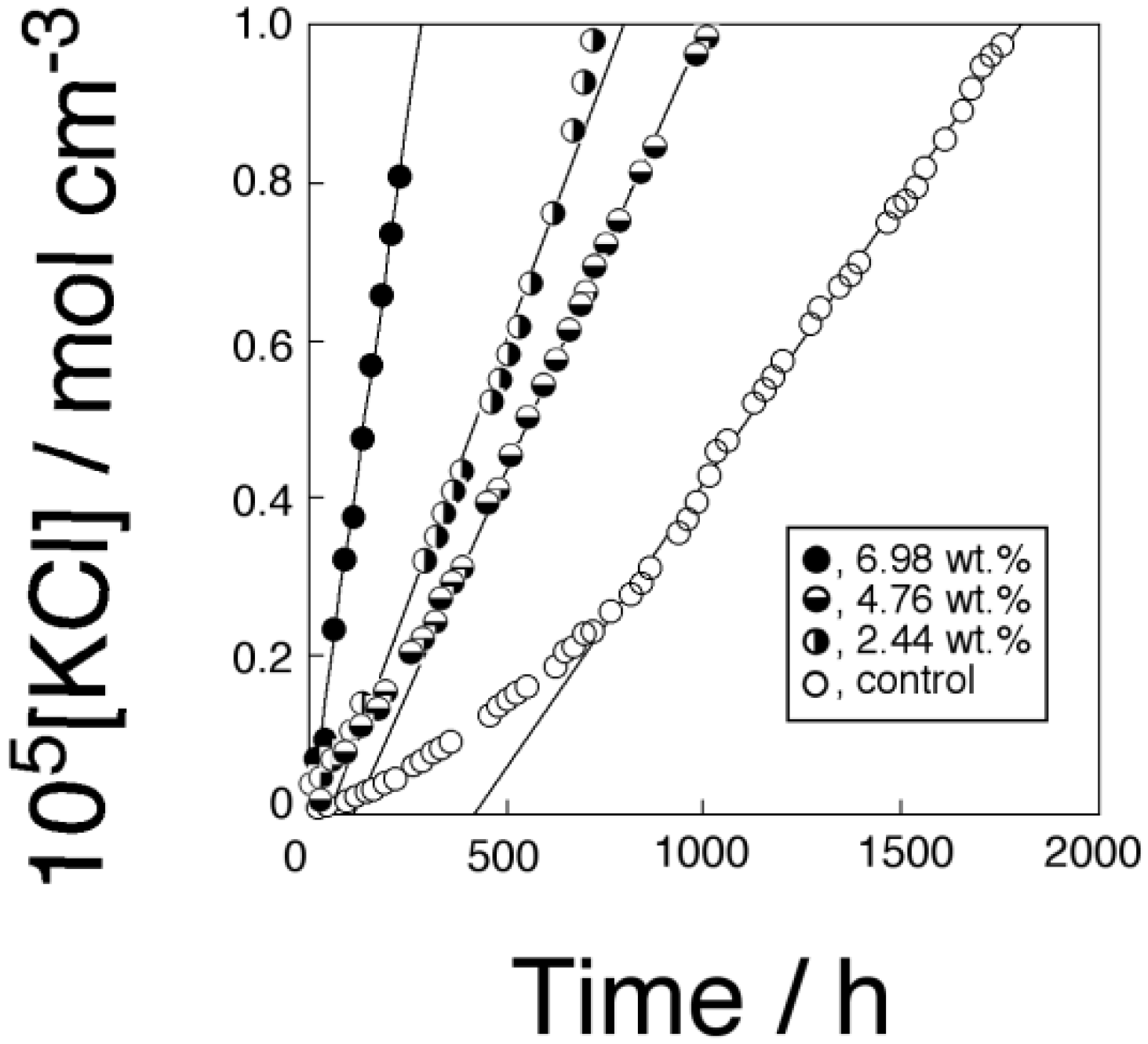
2.2. Transport of KCl through the Membranes
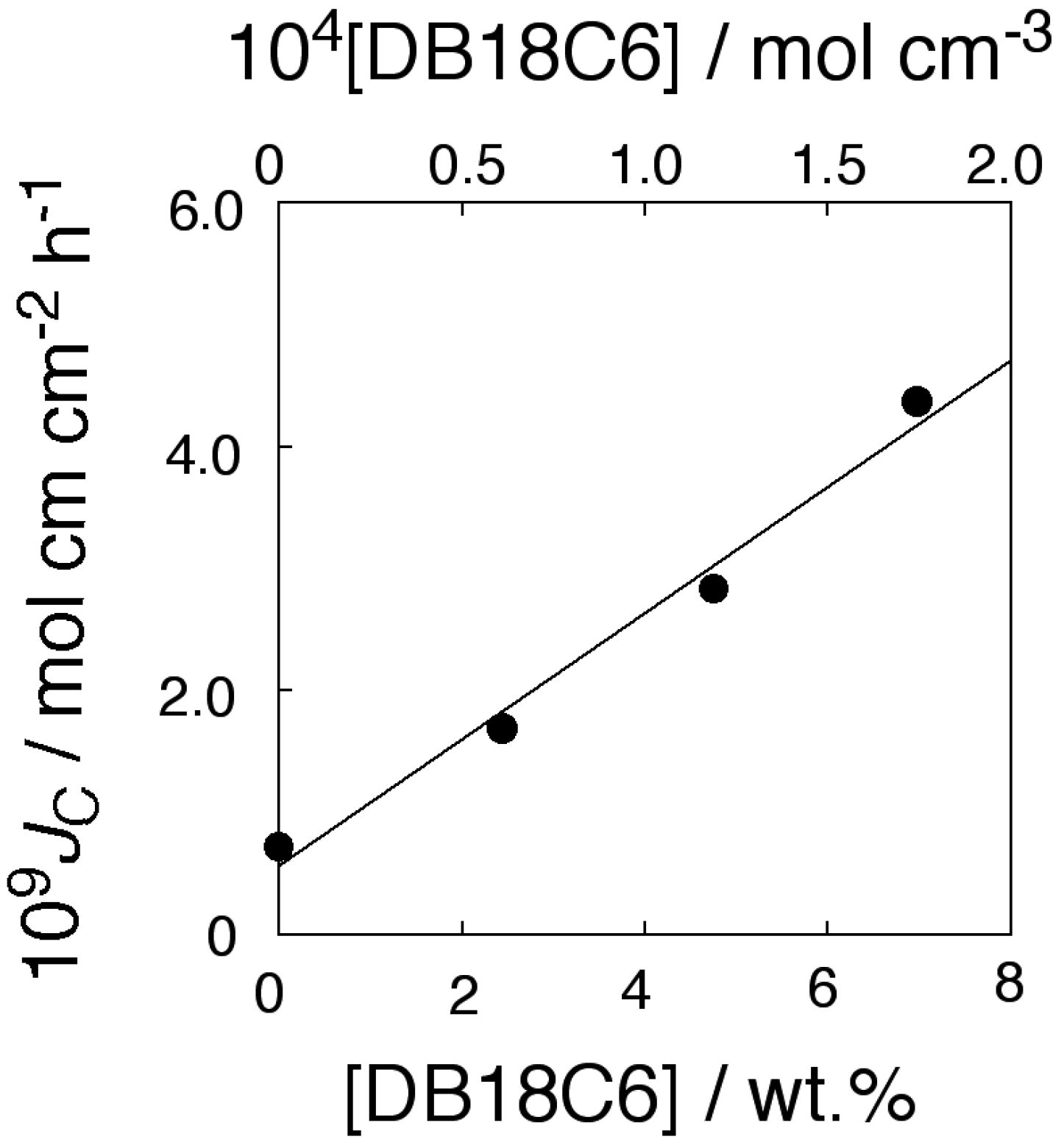

2.3. Dependence of Membrane Transport on Operating Temperature


2.4. Comparison of the Present PPLM with Others
| Liquid membrane | J (normalized flux of K+) | Flux ratio a | Operating temperature (°C) |
|---|---|---|---|
| (mol·cm·cm−2·h−1) | |||
| (mol·cm−3)(mol·cm−3)2 | |||
| PC18AAm/DB18C6 b | 6.75 × 103 | 40 | 70 |
| PC18AAm/DB18C6 b | 6.43 × 103 | 38 | 60 |
| PC18AAm/DB18C6 b | 4.86 × 103 | 29 | 50 |
| PC18MA/DB18C6 c | 6.89 × 103 | 41 | 60 |
| PC12MA/DB18C6 d | 5.79 × 104 | 350 | 40 |
| P2EHA/DB18C6 e | 5.88 × 103 | 35 | 40 |
| P2EHMA/DB18C6 f | 6.20 × 103 | 37 | 40 |
| CHCl3/DB18C6 g | 1.67 × 102 | 1 | 25 |
| PIM/DC18C6 h | 2.37 × 102 | 1.4 | 25 |
2.5. Transport of CsCl through the Membrane
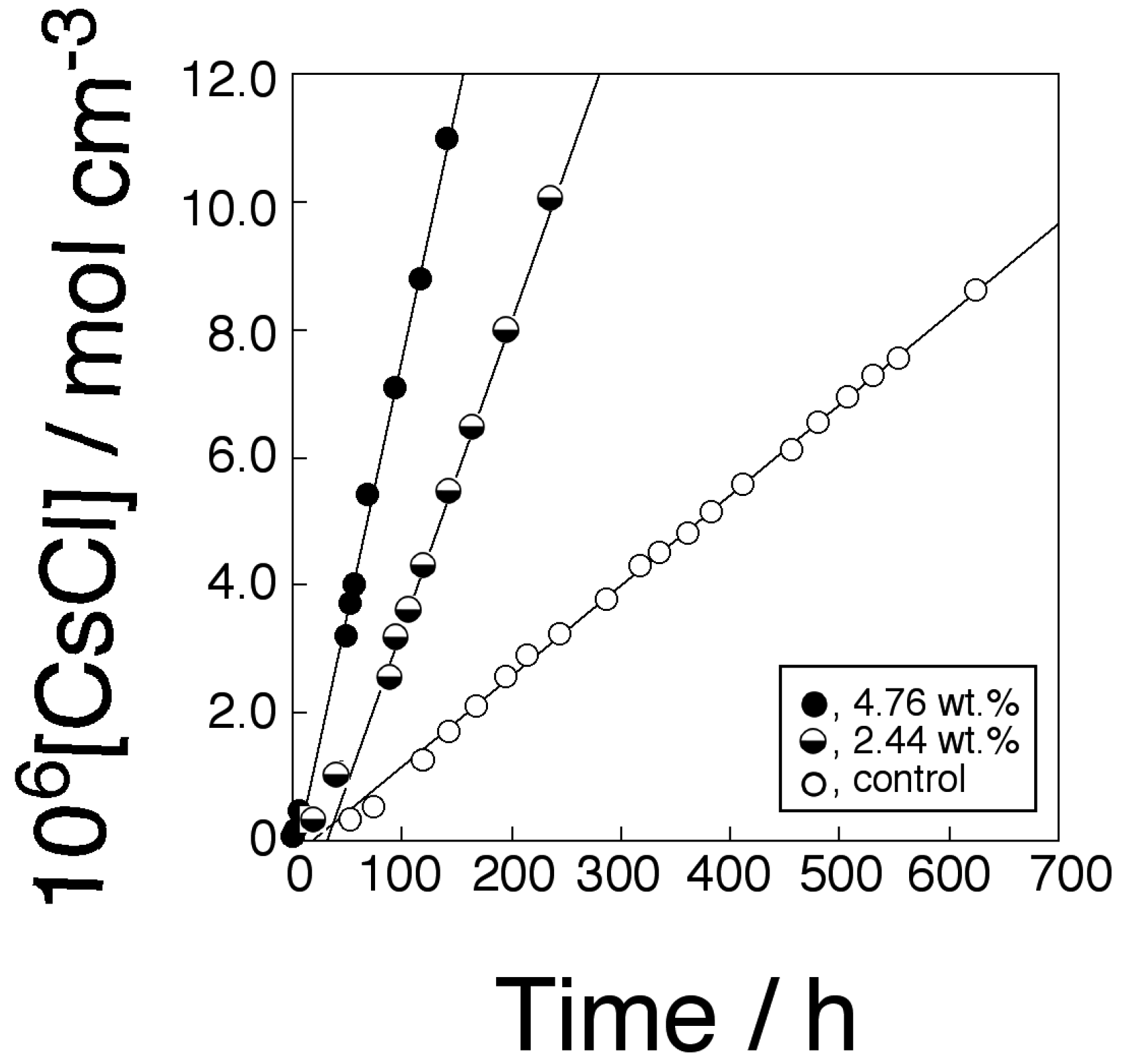
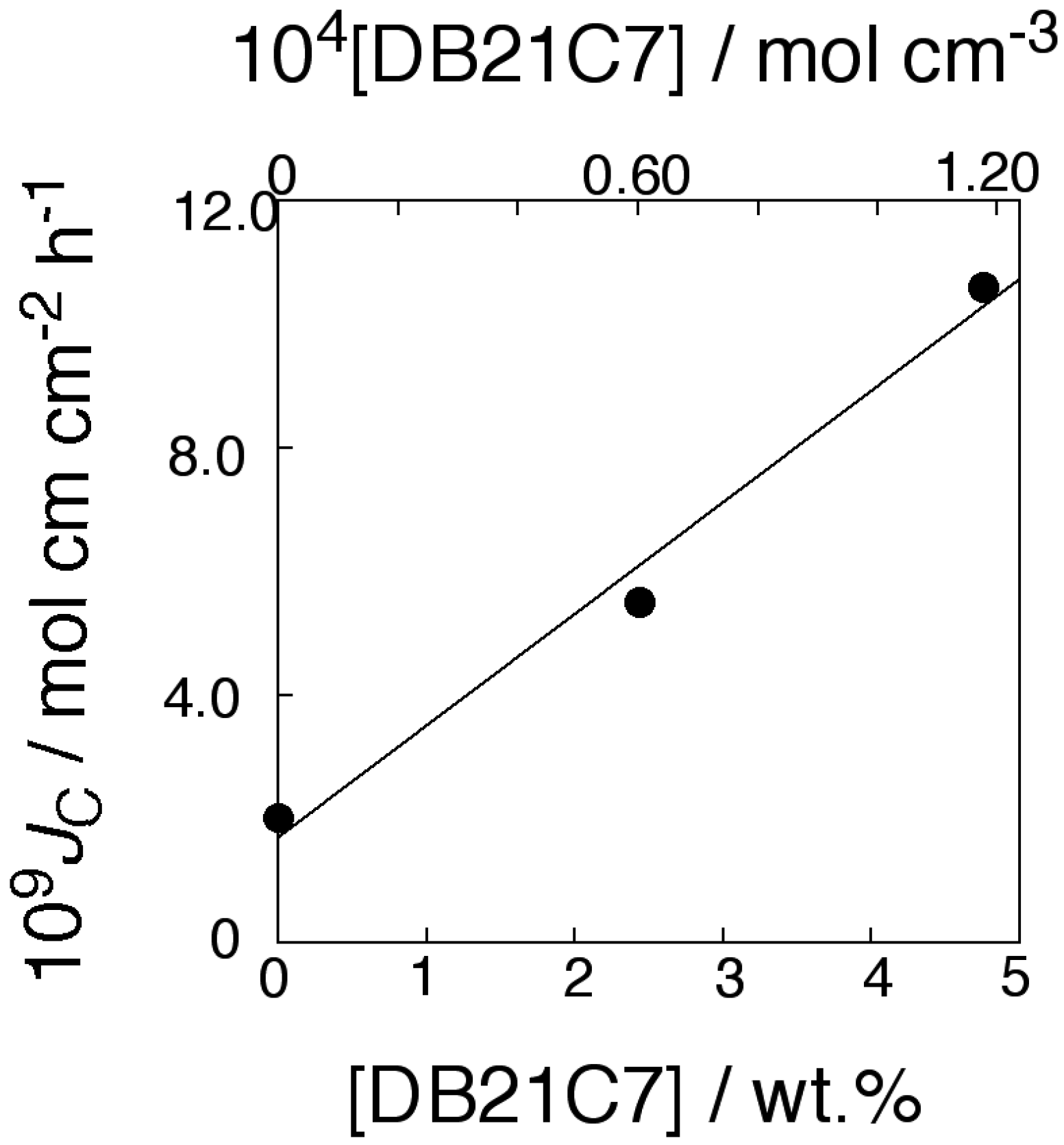
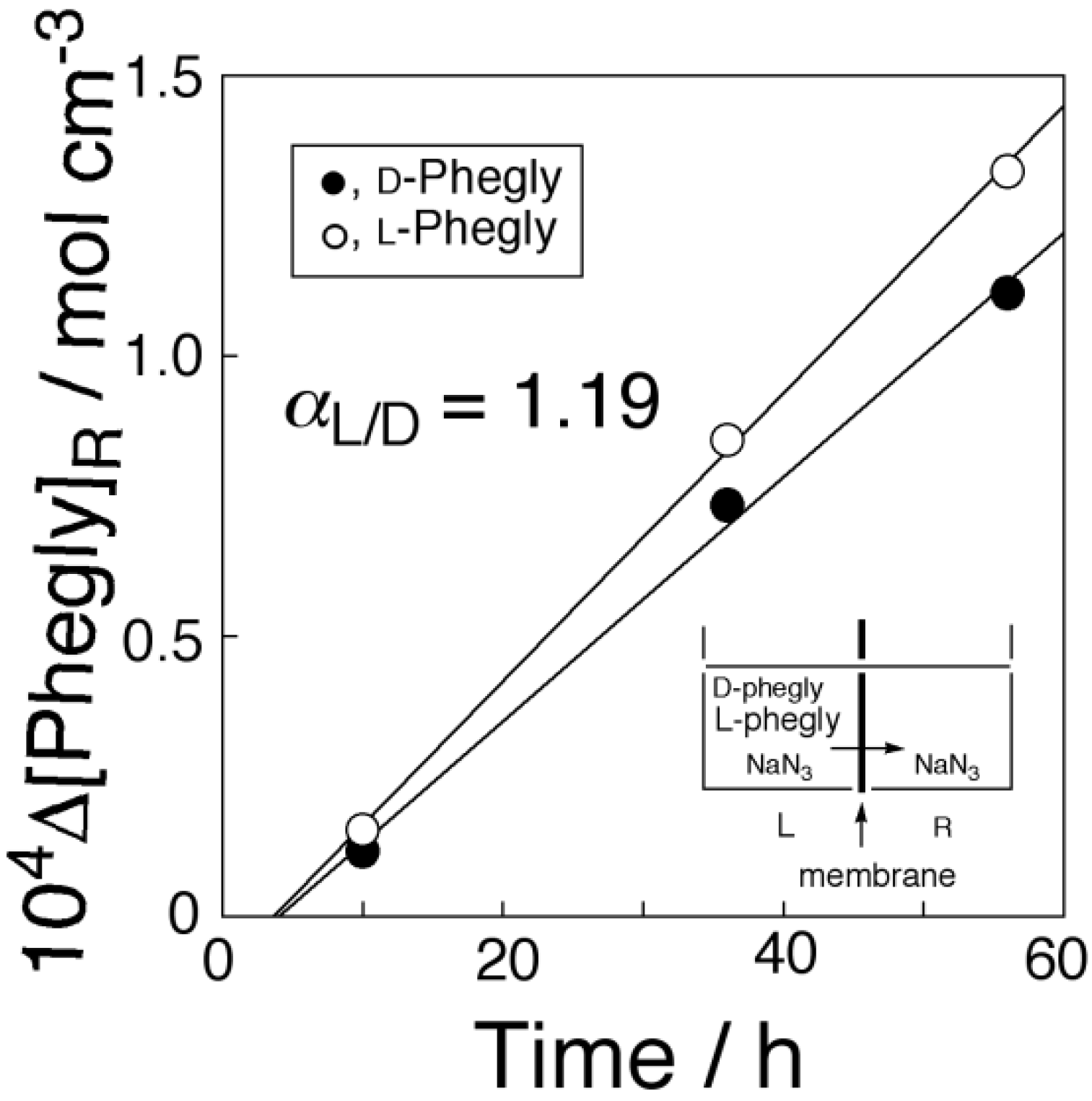
2.6. Chiral Separation of Racemic Mixture of Phenylglycine (Phegly)
3. Experimental Section
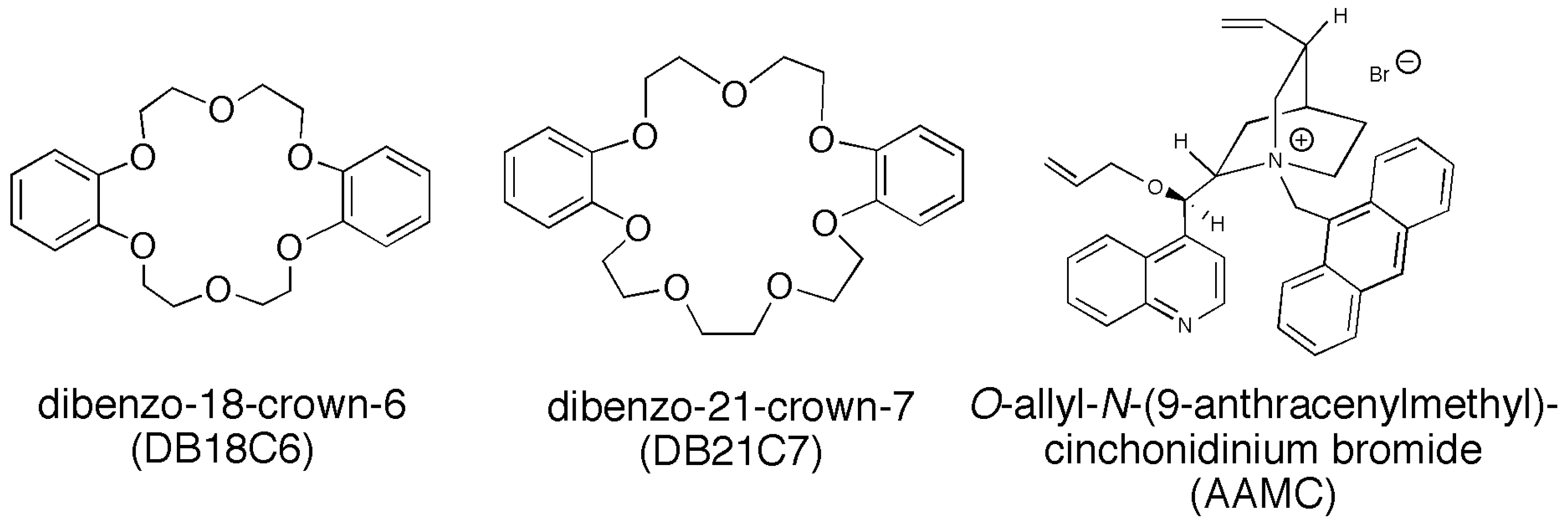
3.1. Materials
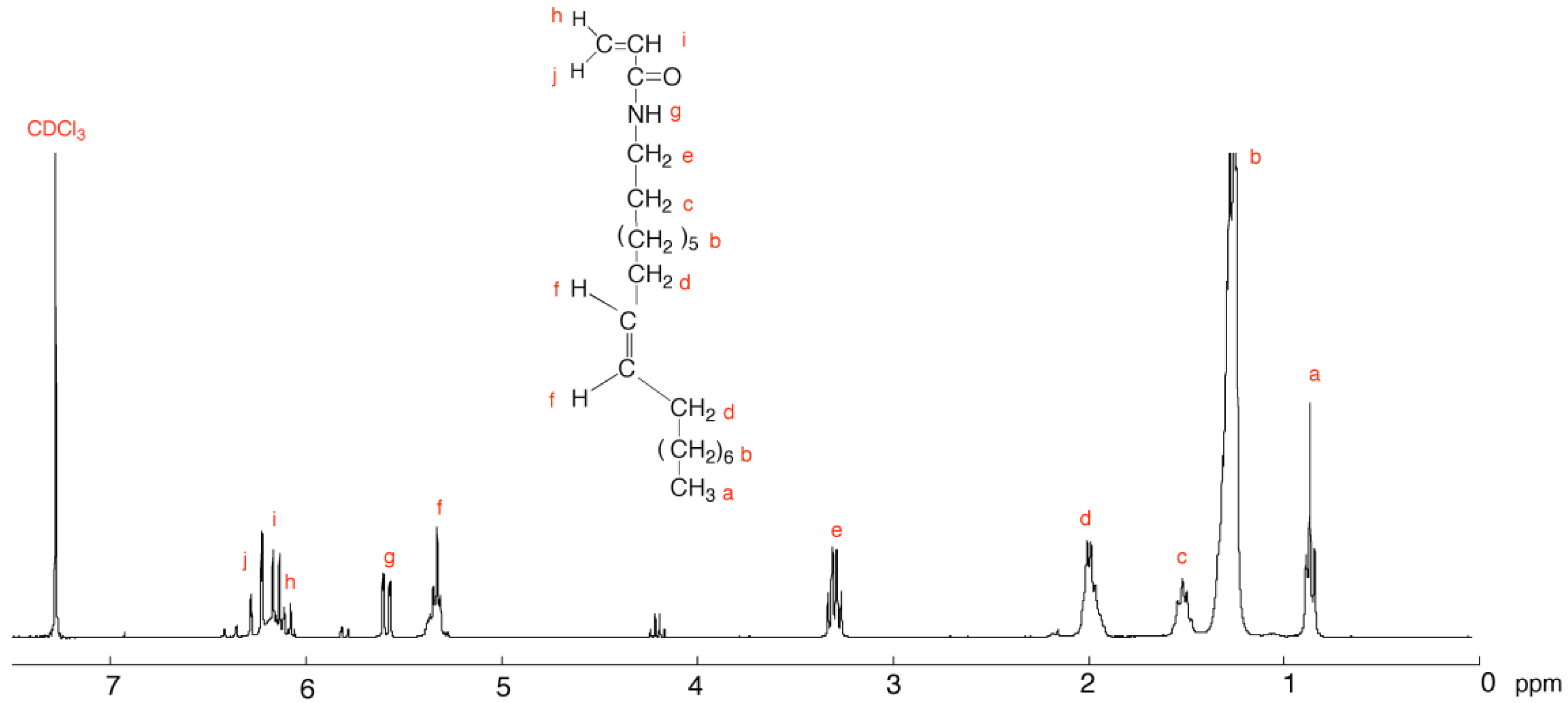
3.2. Characterization of C18AAm
3.3. Preparation of poly(N-oleylacrylamide) (PC18AAm)

3.4. Characterization of PC18AAm
3.5. Preparation of Polymeric Pseudo-Liquid Membranes
3.6. Transport of Alkali Metal Salts
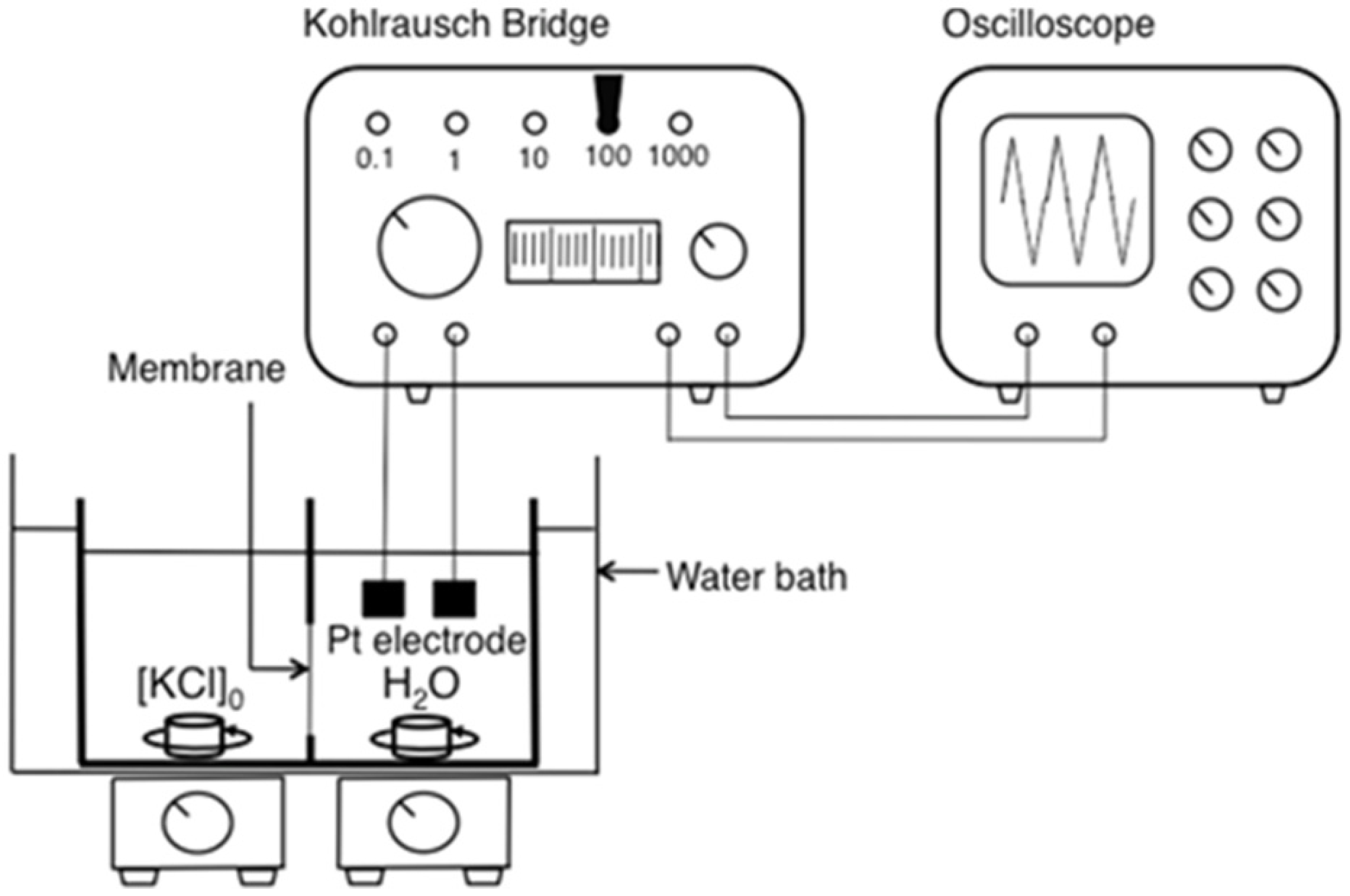
3.7. Transport of Racemic Mixture of Phegly
4. Conclusions
Acknowledgments
Author Contributions
Nomenclature
| DCA | diffusion coefficient of the free solute (cm2·h−1) |
| DCLA | diffusion coefficient of the complexed solute (cm2·h−1) |
| JC | total flux of the diffusing solute, K+, across the membrane per unit membrane thickness (JC = δx JC,obsd) (mol·cm·cm−2·h−1) |
| JC,obsd | observed total flux of the diffusing solute, K+, across the membrane (mol·cm−2·h−1) |
| k | partition coefficient of the solute between water and the organic membrane |
| K | equilibrium constant for the association (mol−1·cm3) |
| δ | membrane thickness (cm) |
| [DB18C6] | total concentration of complexed and uncomplexed transporter, DB18C6, in the membrane (mol·cm−3) |
| [K+] | concentration of the diffusing solutes, K+, in the source phase (mol·cm−3) |
Conflicts of Interest
References
- Ho, W.S.W.; Sirkar, K.K. Membrane Handbook; Chapman & Hall: New York, NY, USA, 1992. [Google Scholar]
- Mulder, M. Basic Principles of Membrane Technology, 2nd ed.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2007. [Google Scholar]
- Baker, R.W. Membrane Technology and Applications, 2nd ed.; John Wiley & Sons: West Sussex, UK, 2004. [Google Scholar]
- Danesi, P.R.; Reichley-Yinger, L.; Rickert, P.G. Lifetime of supported liquid membranes: The influence of interfacial properties, chemical composition and water transport on the long-term stability of the membranes. J. Membr. Sci. 1987, 31, 117–145. [Google Scholar] [CrossRef]
- Takeuchi, H.; Takahashi, K.; Goto, W. Some observations on the stability of supported liquid membranes. J. Membr. Sci. 1987, 34, 19–31. [Google Scholar] [CrossRef]
- Zha, F.F.; Fane, A.G.; Fell, C.J.D.; Schofield, R.W. Critical displacement of a supported liquid membrane. J. Membr. Sci. 1992, 75, 69–80. [Google Scholar] [CrossRef]
- Zha, F.F.; Fane, A.G.; Fell, C.J.D. Instability of supported liquid membranes in phenol transport process. J. Membr. Sci. 1995, 107, 59–74. [Google Scholar] [CrossRef]
- Zha, F.F.; Fabe, A.G.; Fell, C.J.D. Effect of tension gradients on stability of supported liquid membranes. J. Membr. Sci. 1995, 107, 75–86. [Google Scholar] [CrossRef]
- Yang, X.J.; Fane, A.G. Performance and stability of supported liquid membranes using LIX 984N for copper transport. J. Membr. Sci. 1999, 156, 251–263. [Google Scholar] [CrossRef]
- Shinkai, S.; Nakamura, S.; Tachiki, S.; Manabe, O.; Kajiyama, T. Thermocontrol of ion permeation through ternary composite membranes composed of polymer/liquid crystal/amphiphilic crown ether. J. Am. Chem. Soc. 1985, 107, 3363–3365. [Google Scholar] [CrossRef]
- Shinkai, S.; Torigoe, K.; Manabe, O.; Kajiyama, T. Complete’ thermocontrol of ion permeation through ternary composite membranes composed of polymer-liquid crystal-fluorocarbon-containing crown ethers. Chem. Commun. 1986, 12, 933–935. [Google Scholar]
- Sugiura, M. Coupled-ion transport through a solvent polymeric membrane. J. Colloid Interface Sci. 1981, 81, 385–389. [Google Scholar] [CrossRef]
- Sugiura, M.; Kikkawa, M. Effect of plasticizer on carrier-mediated transport of zinc ion through cellulose triacetate membranes. Sep. Sci. Technol. 1987, 22, 2263–2268. [Google Scholar] [CrossRef]
- Schow, A.J.; Peterson, R.T.; Lamb, J.D. Polymer inclusion membranes containing macrocyclic carriers for use in cation separations. J. Membr. Sci. 1996, 111, 291–295. [Google Scholar] [CrossRef]
- Riggs, J.A.; Smith, B.D. Facilitated transport of small carbohydrates through plasticized cellulose triacetate membranes. Evidence of fixed-site jumping transport mechanism. J. Am. Chem. Soc. 1997, 119, 2765–2766. [Google Scholar] [CrossRef]
- Kusumocahyo, S.P.; Kanamori, T.; Sumaru, K.; Aomatsu, S.; Matsuyama, H.; Teramoto, M.; Shinbo, T. Development of polymer inclusion membranes based on cellulose triacetate: Carrier-mediated transport of cerium (III). J. Membr. Sci. 2004, 244, 251–257. [Google Scholar] [CrossRef]
- Nghiem, L.D.H.; Mornane, P.; Potter, I.D.; Perera, J.M.; Cattrall, R.W.; Kolev, S.D. Extraction and transport of metal ions and small organic compounds using polymer inclusion membranes (PIMs). J. Membr. Sci. 2006, 281, 7–41. [Google Scholar] [CrossRef]
- Sodaye, S.; Suresh, G.; Pandey, A.K.; Goswami, A. Determination and theoretical evaluation of selectivity coefficients of monovalent anions in anion-exchange polymer inclusion membrane. J. Membr. Sci. 2007, 295, 108–113. [Google Scholar] [CrossRef]
- Sgarlata, C.; Arena, G.; Longo, E.; Zhang, D.; Yang, Y.; Bartsch, R.A. Heavy metal separation with polymer inclusion membranes. J. Membr. Sci. 2008, 323, 444–451. [Google Scholar] [CrossRef]
- Inês, M.; Cattrall, R.; Kolev, S.D. Recent trends in extraction and transport of metal ions using polymer inclusion membranes (PIMs). J. Membr. Sci. 2012, 415–416, 9–23. [Google Scholar] [CrossRef]
- Kolev, S.D.; St John, A.M.; Cattrall, R.W. Mathematical modeling of the extraction od uranium (VI) into a polymer inclusion membrane composed of PVC and di-(2-ethylhexyl)phosphoric acid. J. Membr. Sci. 2013, 425–426, 169–175. [Google Scholar] [CrossRef]
- St John, A.M.; Cattrall, R.W.; Kolev, S.D. Determination of the initial flux of polymer inclusion membranes. Sep. Purif. Technol. 2013, 116, 41–45. [Google Scholar] [CrossRef]
- Neplenbroek, A.M.; Bargeman, D.; Smolders, C.A. Supported liquid membranes: Stabilization by gelatiopn. J. Membr. Sci. 1992, 67, 149–165. [Google Scholar] [CrossRef]
- Miyako, E.; Maruyama, T.; Kamiya, N.; Goto, M. Selective separation of organic acids through a lipase-facilitated organogel membrane. Membrane 2004, 29, 236–243. [Google Scholar] [CrossRef]
- Kemperman, A.J.B.; Rolevink, H.H.M.; Bargeman, D.; van den Boomgaard, Th.; Dtrathmann, H. Stabilization of supported liquid membranes by interfacial polymerization top layers. J. Membr. Sci. 1998, 138, 43–55. [Google Scholar] [CrossRef]
- Wang, Y.; Thio, Y.S.; Doyle, F.M. Formation of semi-permeable polyamide skin layers on the surface of supported liquid membranes. J. Membr. Sci. 1998, 147, 109–116. [Google Scholar] [CrossRef]
- Fortunato, R.; Afonso, C.A.M.; Reis, M.A.M.; Crespo, J.G. Supported liquid membranes using ionic liquids: Study of stability and transport mechanisms. J. Membr. Sci. 2004, 242, 197–209. [Google Scholar] [CrossRef]
- De los Rios, A.P.; Hemández-Fernández, F.J.; Tomás-Alonso, F.; Palacios, H.M.; Gómez, D.; Rubio, M.; Villora, G. A SEM-EDX study of highly stable supported liquid membranes based on ionic liquids. J. Membr. Sci. 2007, 300, 88–94. [Google Scholar] [CrossRef]
- Pirkle, W.H.; Doherty, E.M. Enantiosleective transport through a silicone-supported liquid membrane. J. Am. Chem. Soc. 1989, 111, 4113–4114. [Google Scholar] [CrossRef]
- Meier, W.; Nardin, C.; Winterhalter, M. Reconstitution of channel proteins in (polymerized) ABA triblock cpopolymer membranes. Angew. Chem. Int. Ed. 2000, 39, 4599–4602. [Google Scholar] [CrossRef]
- Sakiyama, T.; Aoki, T.; Yoshikawa, M. ABA triblock copolymer POE-block-PDMS-block-POE as a component for liquid membranes. Membrane 2004, 29, 377–383. [Google Scholar] [CrossRef]
- Naito, D.; Yoshikawa, M.; Maeda, S.; Okushita, H. Polymeric pseudo-liquid membranes from poly(2-ethylhexyl methacrylate). Polym. J. 2009, 41, 1005–1010. [Google Scholar] [CrossRef]
- Murai, Y.; Asaoka, S.; Yoshikawa, M. Polymeric pseudo-liquid membrane as a stable liquid membrane—Evidence for carrier-mediated mechanism. J. Membr. Sci. 2011, 380, 216–222. [Google Scholar] [CrossRef]
- Murai, Y.; Yoshikawa, M. Polymeric pseudo-liquid membranes from poly(dodecyl methacrylate): KCl transport and optical resolution. Polym. J. 2013, 45, 1058–1063. [Google Scholar] [CrossRef]
- Tsujimoto, H.; Yoshikawa, M. Polymeric pseudo-liquid membranes from poly(octadecyl methacrylate). J. Membr. Sci. 2013, 445, 8–14. [Google Scholar] [CrossRef]
- Jordan, E.F., Jr.; Riser, G.R.; Artymyshyn, B.; Parker, W.E.; Pensabene, J.W. Mechanical properties and transition temperatures of copolymers on N-n-alkylacrylamides and vinylidene chloride. J. Appl. Polym. Sci. 1969, 13, 1777–1794. [Google Scholar] [CrossRef]
- Reusch, C.F.; Cussler, E.L. Selective membrane transport. AIChE J. 1973, 19, 736–741. [Google Scholar] [CrossRef]
- Lamb, J.D.; Christensen, J.J.; Izatt, S.R.; Bedke, K.; Astin, M.S.; Izatt, R.M. Effects of salt concentration and anion on the rate of carrier-facilitated transport of metal cations through bulk liquid membranes containing crown ethers. J. Am. Chem. Soc. 1980, 102, 3399–3403. [Google Scholar] [CrossRef]
- Cussler, E.L.; Aris, R.; Bhown, A. On the limit of facilitated diffusion. J. Membr. Sci. 1989, 43, 149–164. [Google Scholar] [CrossRef]
- Noble, R.D. Analysis of ion transport with fixed site carrier membranes. J. Membr. Sci. 1991, 56, 229–234. [Google Scholar] [CrossRef]
- Igawa, M.; Tanaka, M.; Izumi, S.; Kaneko, Y.; Yamabe, T. Separation of potassium and sodium by liquid membrane. Nippon Kagaku Kaishi 1980, 135–140. [Google Scholar]
- Moyer, B.A.; Deng, Y.; Sun, Y.; Sachleben, A. Extraction of cesium nitrate from concentrated sodium nitrate solutions with 21-crown-7 ethers: Selectivity and equilibrium modeling. Solvent Extr. Ion Exch. 1997, 15, 791–810. [Google Scholar] [CrossRef]
- Šísrková, N.; Kolářová, M.; Lučaníková, M.; Bělohradský, M.; Rais, J. Liquid-liquid extrtaction of microamounts of 137Cs and 85Sr with chlorinated dicarbollide and crown ethers into mixed dodecanenitrile/n-dedecane nonpolar diluent. Sep. Sci. Technol. 2011, 46, 2174–2182. [Google Scholar] [CrossRef]
- The National Academic Press. Radiation Source Use and Replacement; The National Academic Press: Washington, DC, USA, 2008. [Google Scholar]
- Kessler, G. Sustainable and Safe Nuclear Fission Energy Technology and Safety of Fast and Thermal Nuclear Reactors; Springer-Verlag: Berlin, Germany, 2012. [Google Scholar]
- Maier, N.M.; Lindner, W. Chiral recognition applications of molecularly imprinted polymers: A critical review. Anal. Bioanal. Chem. 2007, 389, 377–397. [Google Scholar] [CrossRef]
- Xie, R.; Chu, L.-Y.; Deng, J.-G. Membranes and membrane processes for chiral resolution. Chem. Soc. Rev. 2008, 37, 1243–1263. [Google Scholar] [CrossRef]
- Higuchi, A.; Tamao, M.; Ko, Y.-A.; Tagawa, Y.; Wu, Y.-H.; Freeman, B.D.; Le Bing, J.-T.; Chang, Y.; Ling, Q.-D. Polymeric membranes for chiral separation of pharmaceuticals and chemicals. Polym. Rev. 2010, 50, 113–143. [Google Scholar] [CrossRef]
- Schuur, B.; Verkuijl, B.J.V.; Minnaard, A.J.; de Vries, J.G.; Heeres, H.J.; Feringa, B.L. Chiral separation by enantioselective liquid-liquid extraction. Org. Biomol. Chem. 2011, 9, 36–51. [Google Scholar] [CrossRef]
- Yoshikawa, M.; Higuchi, A. Enantioselective Membranes. In Encyclopedia of Membrane Science and Technology; Hoek, E.M.V., Tarabana, V., Eds.; Wiley: New York, NY, USA, 2013. [Google Scholar]
- Rosini, C.; Altemura, P.; Pini, D.; Bertucci, C.; Zullino, G.; Salvadori, P. Cinchona alkaloids for preparing new, easily accessible chira: Stationary phases II Resolution of binaphthol derivatives on silica-supported quinine. J. Chromatogr. 1985, 348, 79–87. [Google Scholar] [CrossRef]
- Mandl, A.; Nicoletti, L.; Lämmerhofer, M.; Lindner, W. Quinine vs. carbamoylated quinine-baased chiral anion exchangers a comparison tregarding enantioselectivity for N-protected amino acids and other chiral acids. J. Chromatogr. A 1999, 858, 1–11. [Google Scholar] [CrossRef]
- Franco, P.; Blanc, J.; Oberleitner, W.R.; Maier, N.M.; Lindner, W.; Minguillón, C. Enantiomer separation by countercurrent chromatography using chinchona alkaloid derivatives as chiral selectors. Anal. Chem. 2002, 74, 4175–4183. [Google Scholar] [CrossRef]
- Czerwenka, C.; Lämmerhofer, M.; Maierm, N.M.; Rissanen, K.; Lindner, W. Direct high-perfromance liquid chromatographic separation of peptide enantiomers: Study on chiral recognition by systematic evaluation of the influence of structural features of the chiral selectors on enantioselectivity. Anal. Chem. 2002, 74, 5658–5666. [Google Scholar] [CrossRef]
- Canepari, S.; Girelli, A.M.; Mattei, E.; Tosti, D. Enantioselective transport of d,l-Phenylalanine and d,l-Phenylglycine through a bulk liquid membrane containing chinchona alkaloid derivatives as chiral selectors. J. Braz. Chem. Soc. 2009, 20, 429–436. [Google Scholar] [CrossRef]
- Jordan, E.F., Jr.; Roser, G.R.; Parker, W.E.; Wrigley, A.N. Mechanical propertiers and transition temperatures for copolymers of N-n-alkylacrylamides and acrylonitrile. J. Polym. Sci. 1966, 4, 975–996. [Google Scholar] [CrossRef]
- Kice, J.L. Inhibition polymerization. I. Methyl methacrylate. J. Am. Chem. Soc. 1954, 76, 6274–6280. [Google Scholar] [CrossRef]
- Riddick, J.A.; Bunger, W.B.; Sakano, T.K. Organic Solvents, 4th ed.; Wiley: New York, NY, USA, 1986. [Google Scholar]
- Silverstein, R.M.; Webster, F.X. Spectroscopic Identification of Organic Compounds, 6th ed.; Wiley: New York, NY, USA, 1998. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Shiono, H.; Yoshikawa, M. Polymeric Pseudo-Liquid Membranes from Poly(N-oleylacrylamide). Membranes 2014, 4, 210-226. https://doi.org/10.3390/membranes4020210
Shiono H, Yoshikawa M. Polymeric Pseudo-Liquid Membranes from Poly(N-oleylacrylamide). Membranes. 2014; 4(2):210-226. https://doi.org/10.3390/membranes4020210
Chicago/Turabian StyleShiono, Hiroko, and Masakazu Yoshikawa. 2014. "Polymeric Pseudo-Liquid Membranes from Poly(N-oleylacrylamide)" Membranes 4, no. 2: 210-226. https://doi.org/10.3390/membranes4020210