Can Biochemistry Usefully Guide the Search for Better Polymer Electrolytes?
Abstract
:1. Introduction
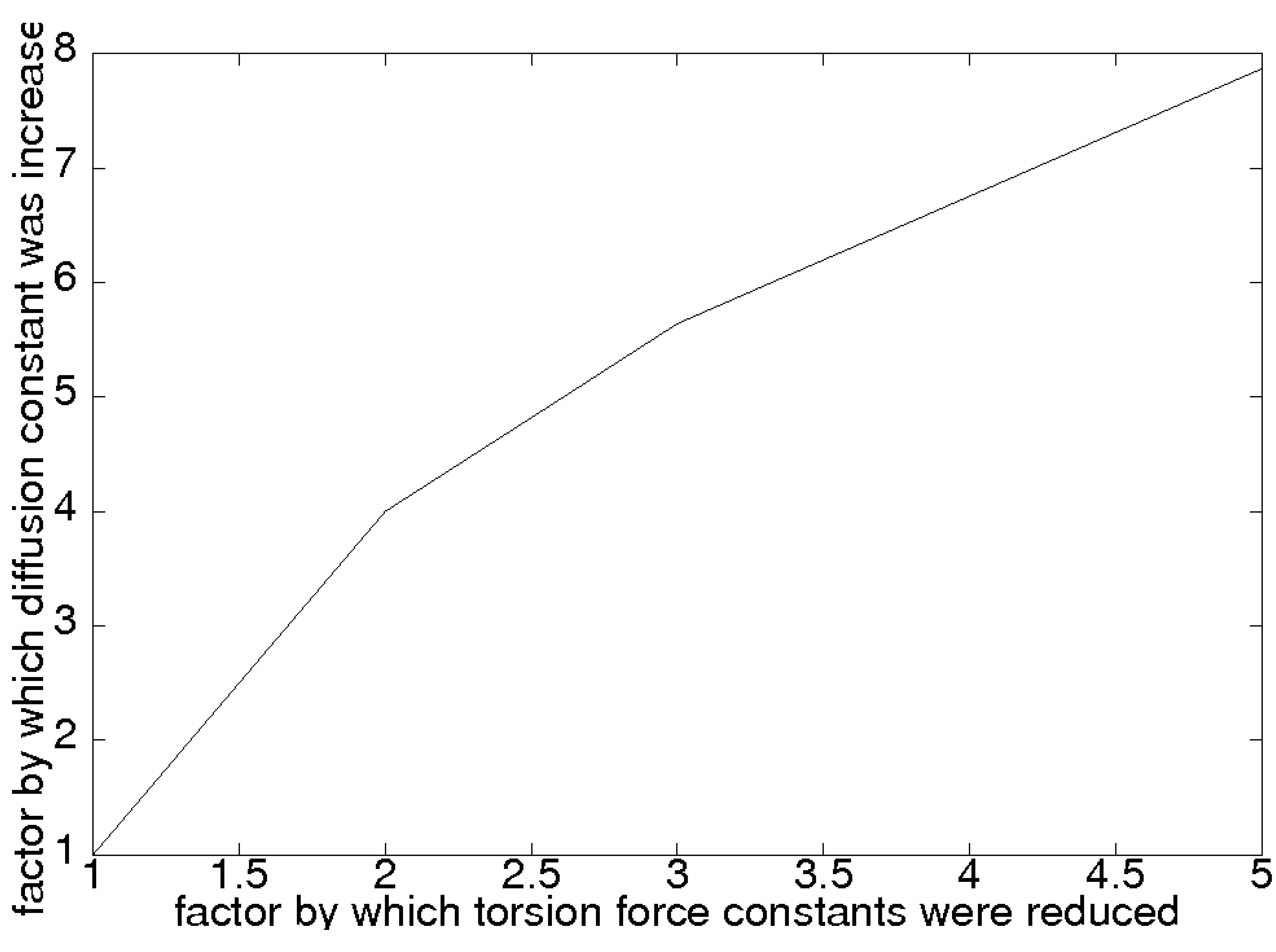
2. Protein PEO Mixtures
3. Membrane Proteins
4. Keeping out the Water
5. Role for Simulation
6. Conclusions
Acknowledgments
Conflicts of Interest
References
- Boden, N.; Leng, S.A.; Ward, I.M. Ionic conductivity and diffusivity in polyethylene oxide/electrolyte solutions as models for polymer electrolytes. Solid State Ionics 1991, 45, 261–270. [Google Scholar]
- Ratner, M. Polymer Electrolyte Reviews; MacCallum, J.R., Vincent, C.A., Eds.; Elsevier: London, UK, 1987; Volume 1, p. 173. [Google Scholar]
- Gray, F.M. Solid Polymer Electrolytes; VCH Pub.: New York, NY, USA, 1991. [Google Scholar]
- Muller-Plathe, F. Permeation of polymers—A computational approach. Acta Polym. 1994, 45, 259–293. [Google Scholar] [CrossRef]
- Mckechnie, J.I.; Brown, D.; Clarke, J.H.R. Methods of generating dense relaxed amorphous polymer samples for use in dynamic simulations. Macromolecules 1992, 25, 1562–1567. [Google Scholar] [CrossRef]
- Brown, D.; Clarke, J.H.R.; Okuda, M.; Yamazaki, T. Preparation of polymer melt samples for computer simulation studies. J. Chem. Phys. 1994, 100, 6011–6018. [Google Scholar] [CrossRef]
- Neyertz, S.; Brown, D. Computer simulation study of the chain configurations in poly(ethylene oxide)-homolog melts. J. Chem. Phys. 1995, 102, 9725–9735. [Google Scholar] [CrossRef]
- Catlow, C.R.A.; Mills, G.E. Computer simulation of ionically conducting polymers. Electrochim. Acta 1995, 40, 2057–2059, 2061–2062. [Google Scholar] [CrossRef]
- Neyertz, S.; Brown, D. Local structure and mobility of ions in polymer electrolytes: A molecular dynamics simulation study of the amorphous PEOxNal system. J. Chem. Phys. 1996, 104, 3797–3809. [Google Scholar] [CrossRef]
- Annis, B.B.K.; Kim, M.-H.; Wignall, G.D.; Borodin, O.; Smith, G.D. Study of the influence of LiI on the chain conformations of poly(ethyleneoxide) in the melt by small-angle neutron scattering and molecular dynamics simulations. Macromolecules 2000, 33, 7544–7548. [Google Scholar] [CrossRef]
- Borodin, O.; Smith, G.D. Molecular dynamics simulations of poly(ethylene) oxide LiI melts 2. Dynamic properties. Macromolecules 2000, 33, 2273–2283. [Google Scholar]
- Ennari, J.; Neelov, I.; Sundholm, F. Molecular dynamics simulation of the structure of PEO based solid polymer electrolytes. Polym. Guildf. 2000, 41, 4057–4063. [Google Scholar]
- Ennari, J.; Neelov, I.; Sundholm, F. Simulation of a PEO based solid polyelectrolyte, comparison of the CMM and the Ewald summation method. Polym. Guildf. 2000, 41, 2149–2155. [Google Scholar]
- Borodin, O.; Smith, G.D. Molecular dynamics simulations of poly(ethylene oxide)/LiI melts. 1. Structural and conformational properties. Macromolecules 1998, 31, 8396–8406. [Google Scholar] [CrossRef]
- Lin, B.; Boinske, P.T.; Halley, J.W. Molecular dynamics model of the amorphous regions of polyethylene oxide. J. Chem. Phys. 1996, 105, 1668–1681. [Google Scholar] [CrossRef]
- Halley, J.W.; Duan, Y.; Nielsen, B.; Redfern, P.C.; Curtiss, L.A. Simulation of polyethylene oxide: Improved structure using better models for hydrogen and flexible walls. J. Chem. Phys. 2001, 115, 3957–3966. [Google Scholar] [CrossRef]
- Johnson, J.; Saboungi, M.-L.; Price, D.L.; Ansell, S.; Russell, T.P.; Halley, J.W.; Nielsen, B. Atomic structure of solid and liquid polyethylene oxide. J. Chem. Phys. 1998, 109, 7005–7010. [Google Scholar] [CrossRef]
- Halley, J.W.; Duan, Y.; Curtiss, L.A.; Baboul, A.G. Lithium perchlorate ion pairing in a model of amorphous polyethylene oxide. J. Chem. Phys. 1999, 111, 3302–3308. [Google Scholar] [CrossRef]
- Duan, Y.; Halley, J.W.; Curtiss, L.; Redfern, P. Mechanisms of lithium transport in amorphous polyethylene oxide. J. Chem. Phys. 2005, 122, 054702:1–054702:8. [Google Scholar]
- Kato, M.; Warshel, A. Through the channel and around the channel: Validating and comparing microscopic approaches for the evaluation of free energy profiles for ion pentration through ion channels. J. Phys. Chem. B 2005, 109, 19516–19522. [Google Scholar] [CrossRef]
- Shin, J.H.; Henderson, W.A.; Passerini, S. PEO-based polymer electrolytes with ionic liquids and their use in lithium metal-polymer electrolyte batteries. J. Electrochem. Soc. 2005, 152, A978–A983. [Google Scholar] [CrossRef]
- Henderson, W.A.; Passerini, S. Phase behavior of ionic liquid–LiX mixtures: Pyrrolidinium cations and TFSI− anions. Chem. Mater. 2004, 16, 2881–2885. [Google Scholar]
- Castriota, M.; Caruso, T.; Agostino, R.G.; Cazzanelli, E.; Henderson, W.; Passerini, S. Raman investigation of the ionic liquid N-methyl-N-propylpyrrolidinium bis(trifluoromethanesulfonyl) imide and its mixture with LiN(SO2CF3)2. J. Phys. Chem. A 2005, 109, 92–96. [Google Scholar] [CrossRef]
- Shin, J.H.; Henderson, W.A.; Passerini, S. An elegant fix for polymer electrolytes. Electrochem. Solid State Lett. 2005, 8, A125–A127. [Google Scholar] [CrossRef]
- Wang, Y.; Li, B.; Ji, J.; Eyler, A.; Zhong, W.-H. A gum-like electrolyte: Safety of a solid, performance of a liquid. Adv. Energy Mater. 2013. [Google Scholar] [CrossRef]
- Ji, J.; Li, B.; Zhong, W.-H. An ultraelastic poly(ethylene oxide)/soy protein film with fully amorphous structure. Macromolecules 2012, 45, 602–606. [Google Scholar] [CrossRef]
- Gokel, G.; Daschbach, M.M. Coordination and transport of metal cations through phospholipid bilayer membranes by hydraphile channels. Coord. Chem. Rev. 2008, 252, 886–902. [Google Scholar] [CrossRef]
- Hauser, H.; Shipley, G.G. Crystallization of Phospatidylserine Bilayers Induced by Lithium. J. Biol. Chem. 1981, 256, 11377–11380. [Google Scholar]
- Perriman, A.W.; Brogan, A.P.S.; Colfen, H.; Tsoureas, N.; Owen, G.R.; Mann, S. Reversible dioxygen binding in solvent-free liquid myoglobin. Nat. Chem. 2010, 2, 622–626. [Google Scholar]
- Morais-Cabral, J.H.; Zhou, Y.; MacKinnon, R. Energetic optimization of ion conduction rate by the K+ selectivity filter. Nature 2001, 414, 37–42. [Google Scholar] [CrossRef]
- Eriksson, U.K.; Fischer, G.; Friemann, R.; Enkavi, G.; Tajkhorshid, E.; Neutze1, R. Subangstrom resolution X-ray structure details aquaporin-water interactions. Science 2013, 340, 1346–1349. [Google Scholar]
- Freites, J.A.; Tobias, D.J.; von Heijne, G.; White, S.H. Interface connections of a transmembrane voltage sensor. Proc. Natl. Acad. Sci. USA 2005, 102, 15059–15064. [Google Scholar]
- Jaud, S.; Tobias, D.J.; Falke, J.J.; White, S.H. Self-induced docking site of a deeply embedded peripheral membrane protein. Biophys. J. 2007 92, 517–524.
- Bondar, A.N.; del Val, C.; White, S.H. Rhomboid protease dynamics and lipid interactions. Structure 2009, 17, 395–405. [Google Scholar] [CrossRef]
- Krepkiy, D.; Mihailescu, M.; Freites, J.A.; Schow, E.V.; Worcester, D.L.; Gawrisch, K.; Tobias, D.J.; White, S.H.; Swartz, K.J. Structure and hydration of membranes embedded with voltage-sensing domains. Nature 2009, 462, 473–479. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Halley, J.W. Can Biochemistry Usefully Guide the Search for Better Polymer Electrolytes? Membranes 2013, 3, 242-248. https://doi.org/10.3390/membranes3030242
Halley JW. Can Biochemistry Usefully Guide the Search for Better Polymer Electrolytes? Membranes. 2013; 3(3):242-248. https://doi.org/10.3390/membranes3030242
Chicago/Turabian StyleHalley, J. Woods. 2013. "Can Biochemistry Usefully Guide the Search for Better Polymer Electrolytes?" Membranes 3, no. 3: 242-248. https://doi.org/10.3390/membranes3030242



