Stimuli Responsive Ionogels for Sensing Applications—An Overview
Abstract
:1. Introduction
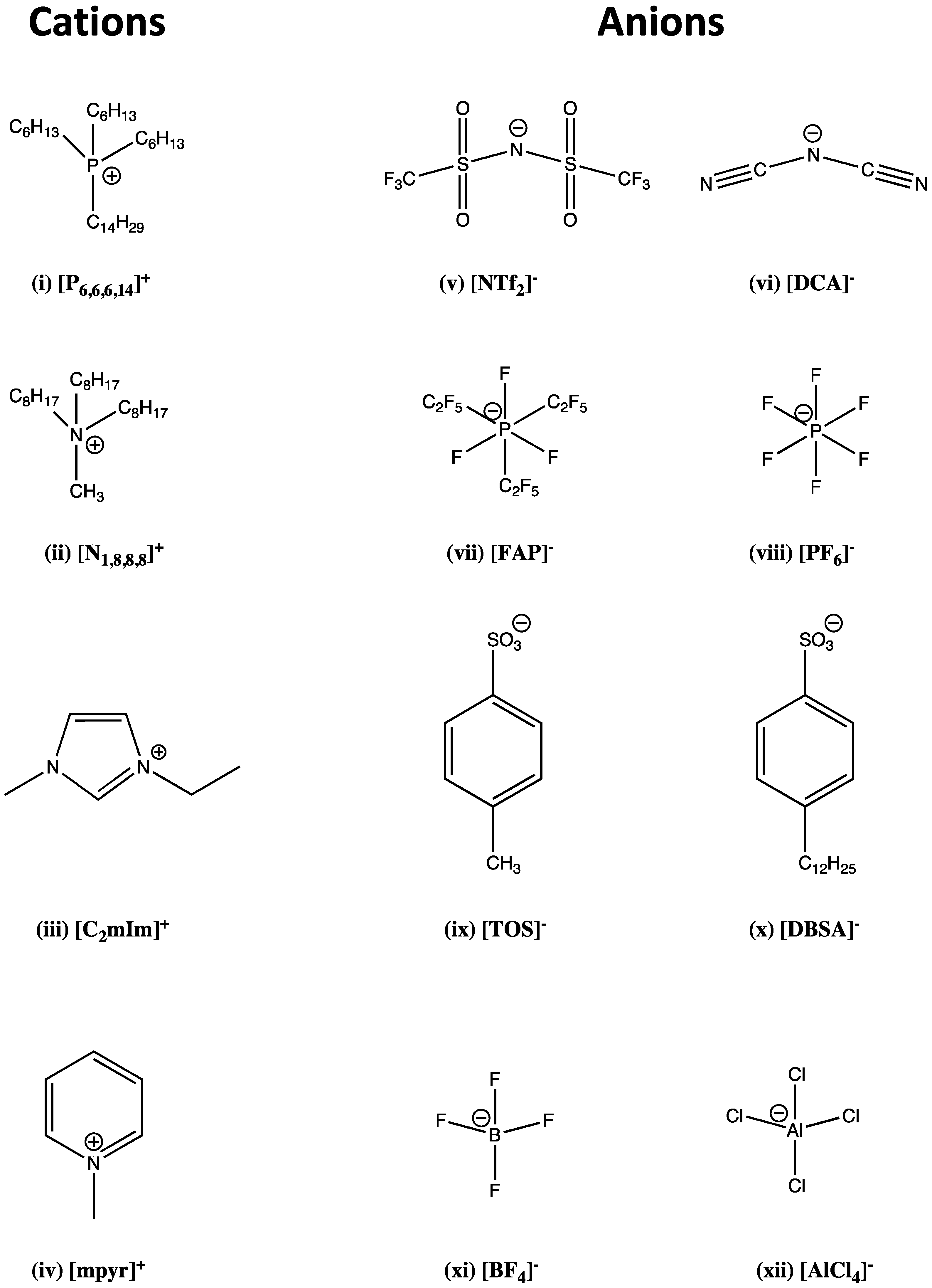
2. Ionic Liquids
Synthesis of ILs
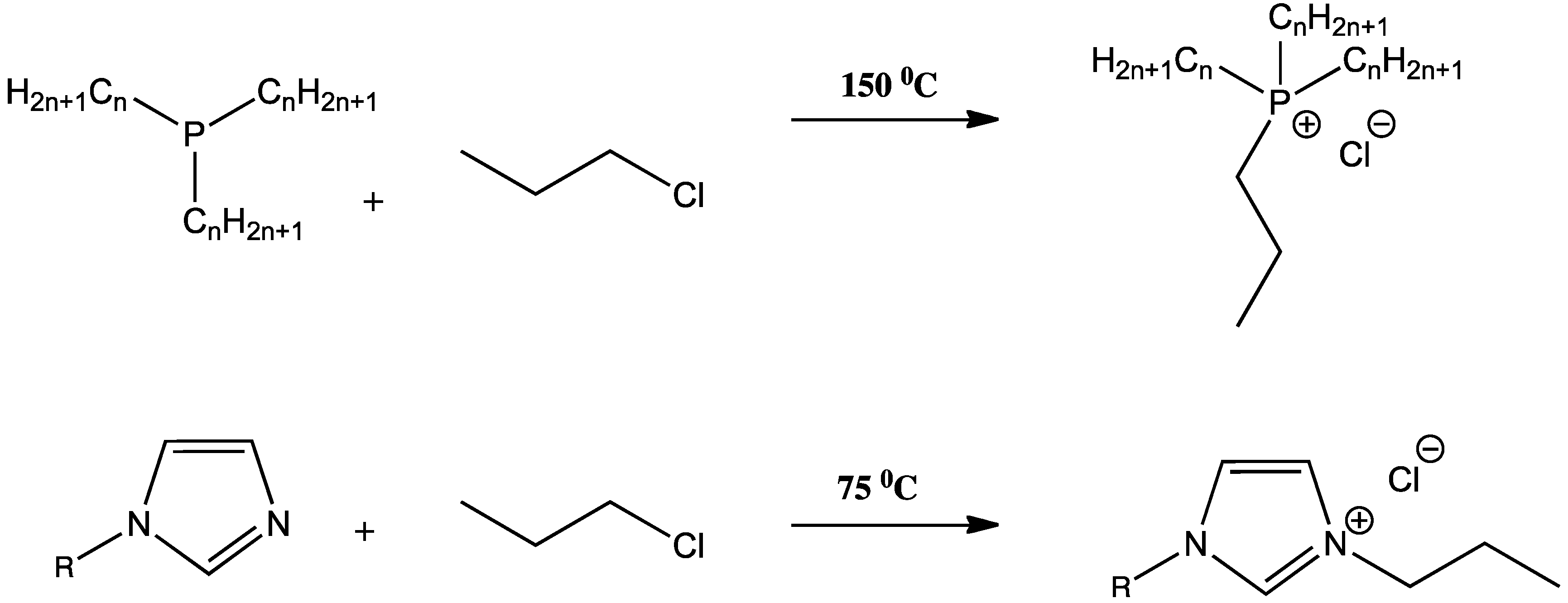
3. Stimuli Responsive Materials
Photo-Responsive Materials
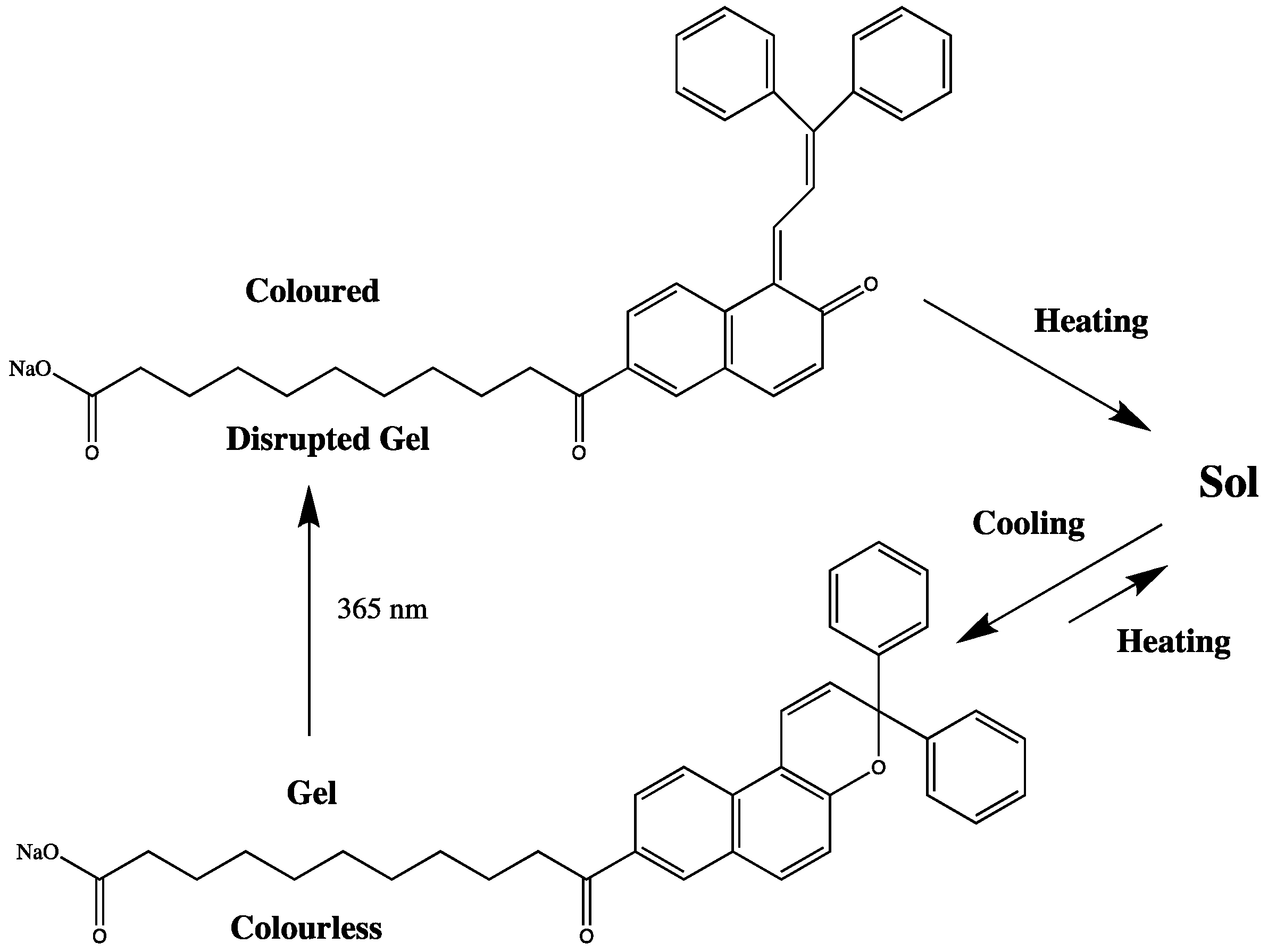
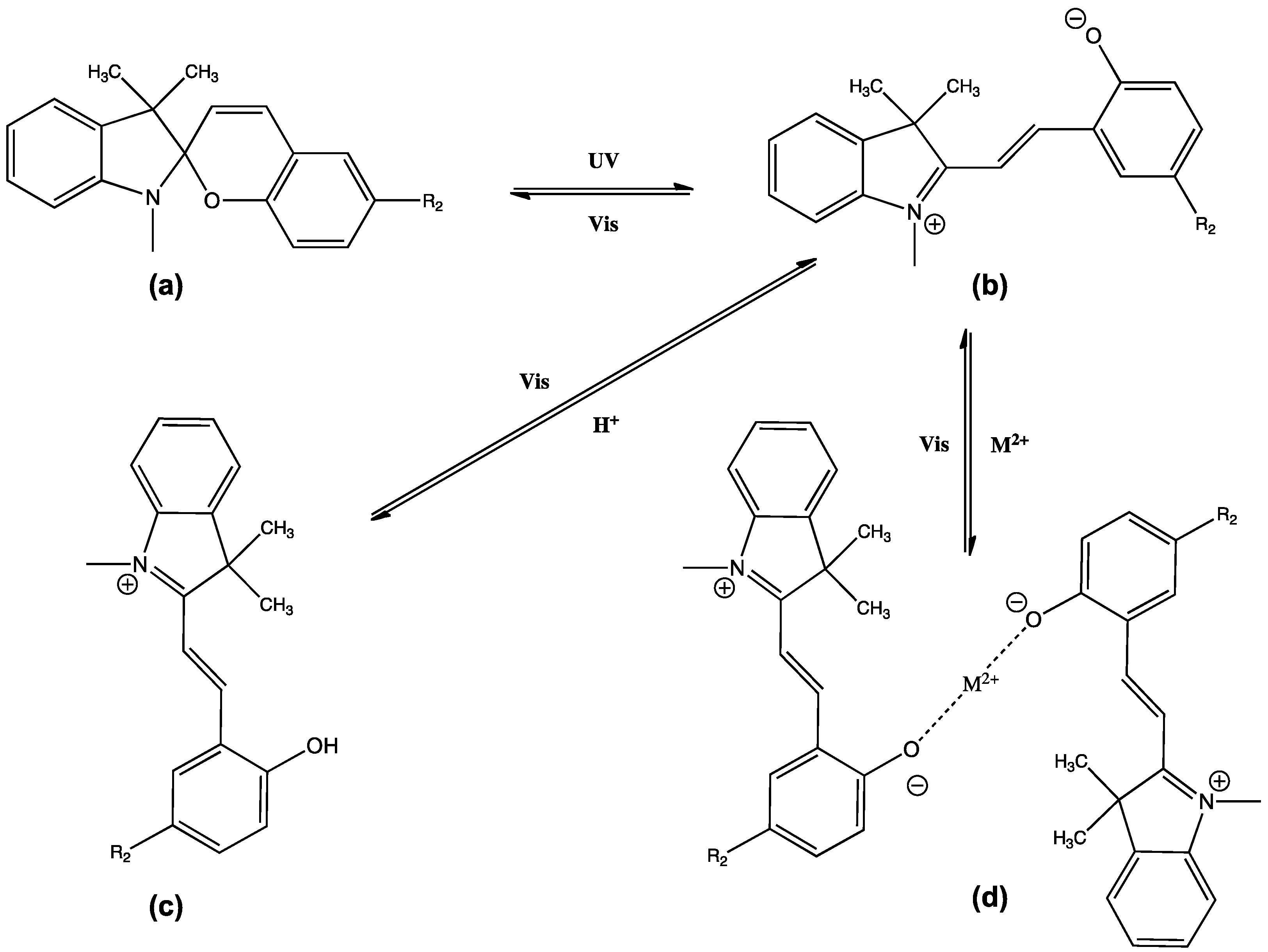
4. Stimuli Responsive Ionogels
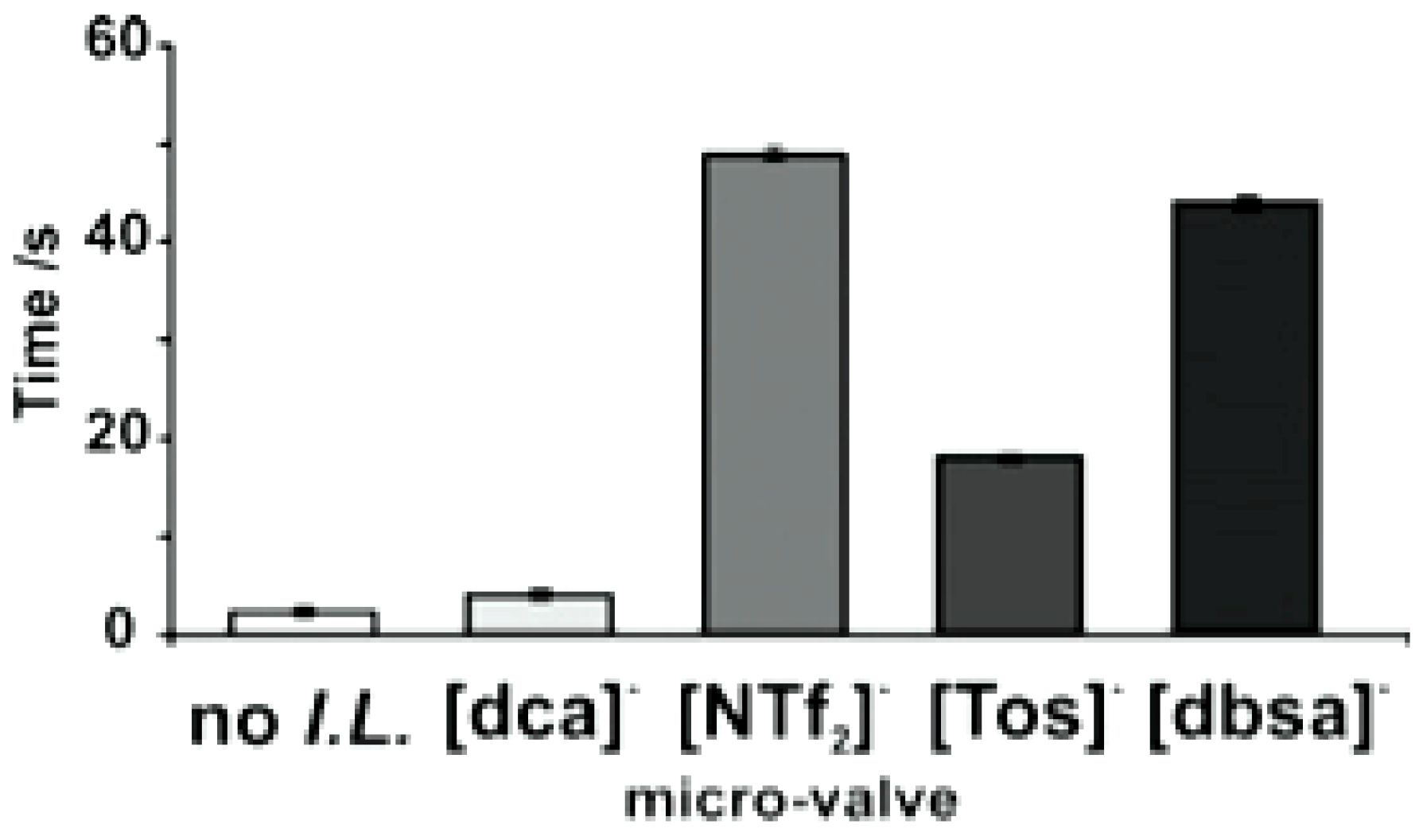
4.1. Photo Responsive Ionogels for Direct Fluid Control in Microfluidic Devices

4.2. Electro Responsive Ionogels for Sensing Applications
4.3. Electro Responsive Ionogels for Electrochromic Applications
4.4. Optically Responsive Ionogels for Sensing Applications

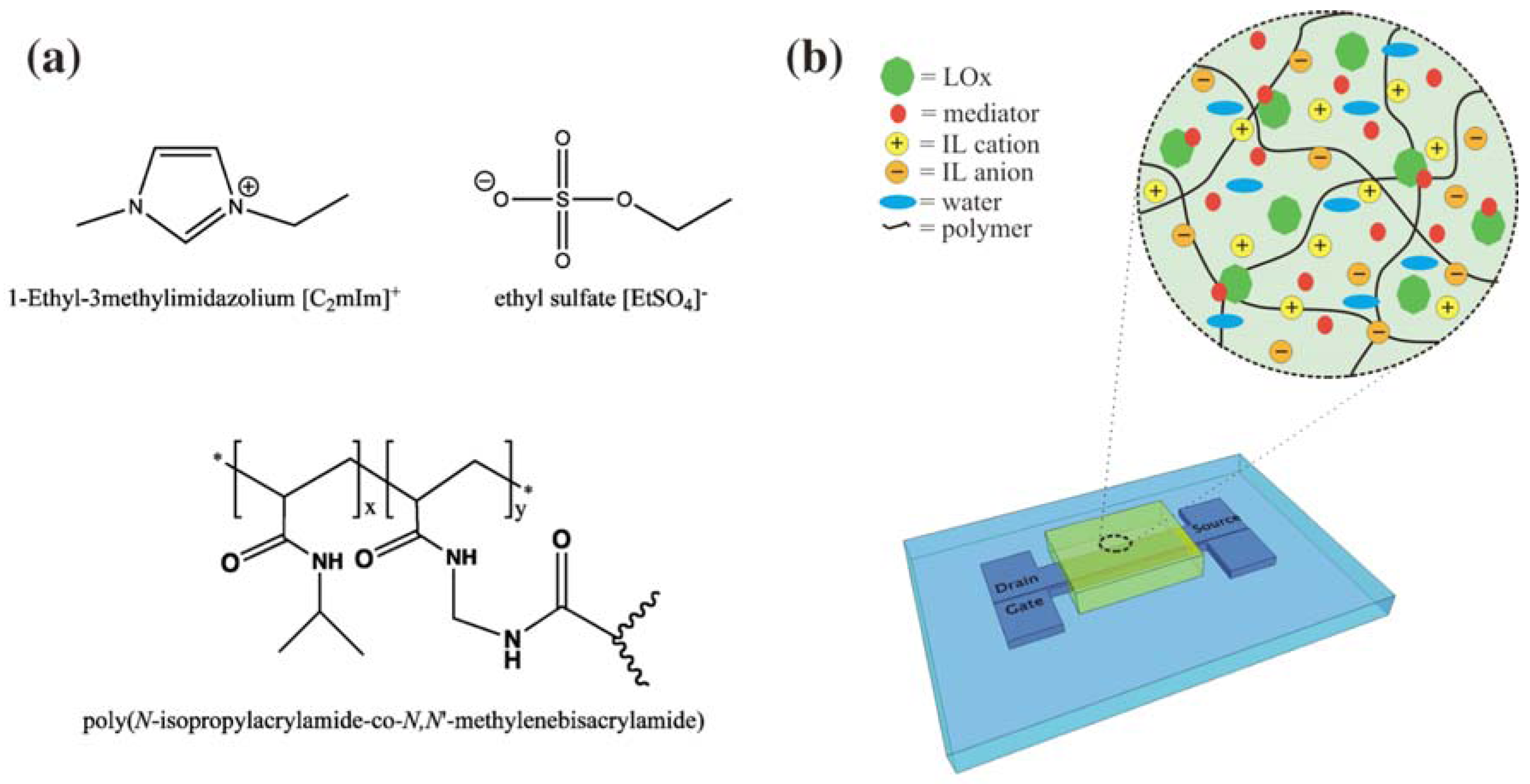
4.5. Ionogels as Bio-Sensing Components
4.5.1. The Need for Wearable Sensors
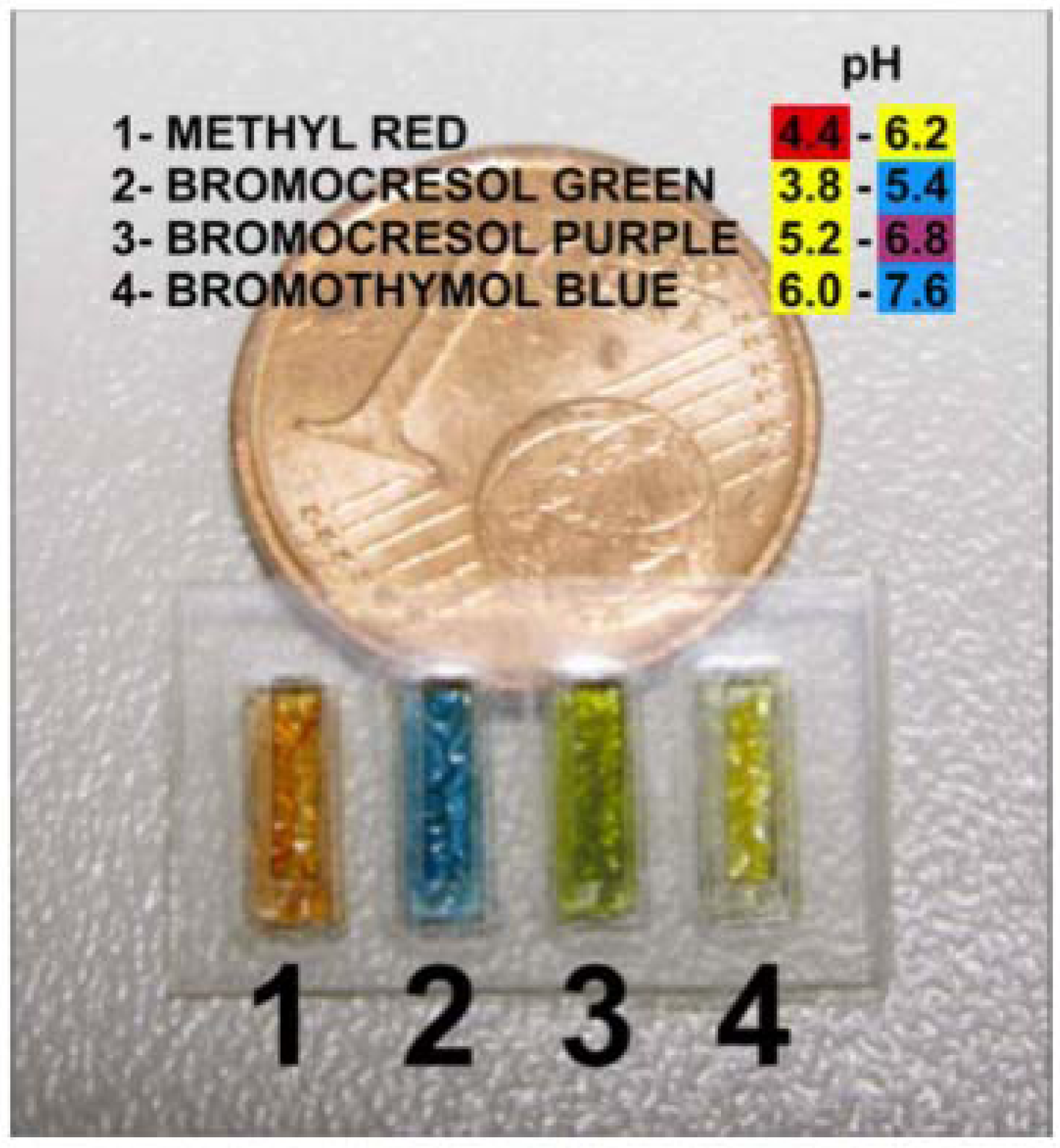

5. What Does the Future of Stimuli Responsive ILs Hold?
5.1. PH, Photo-Responsive ILs

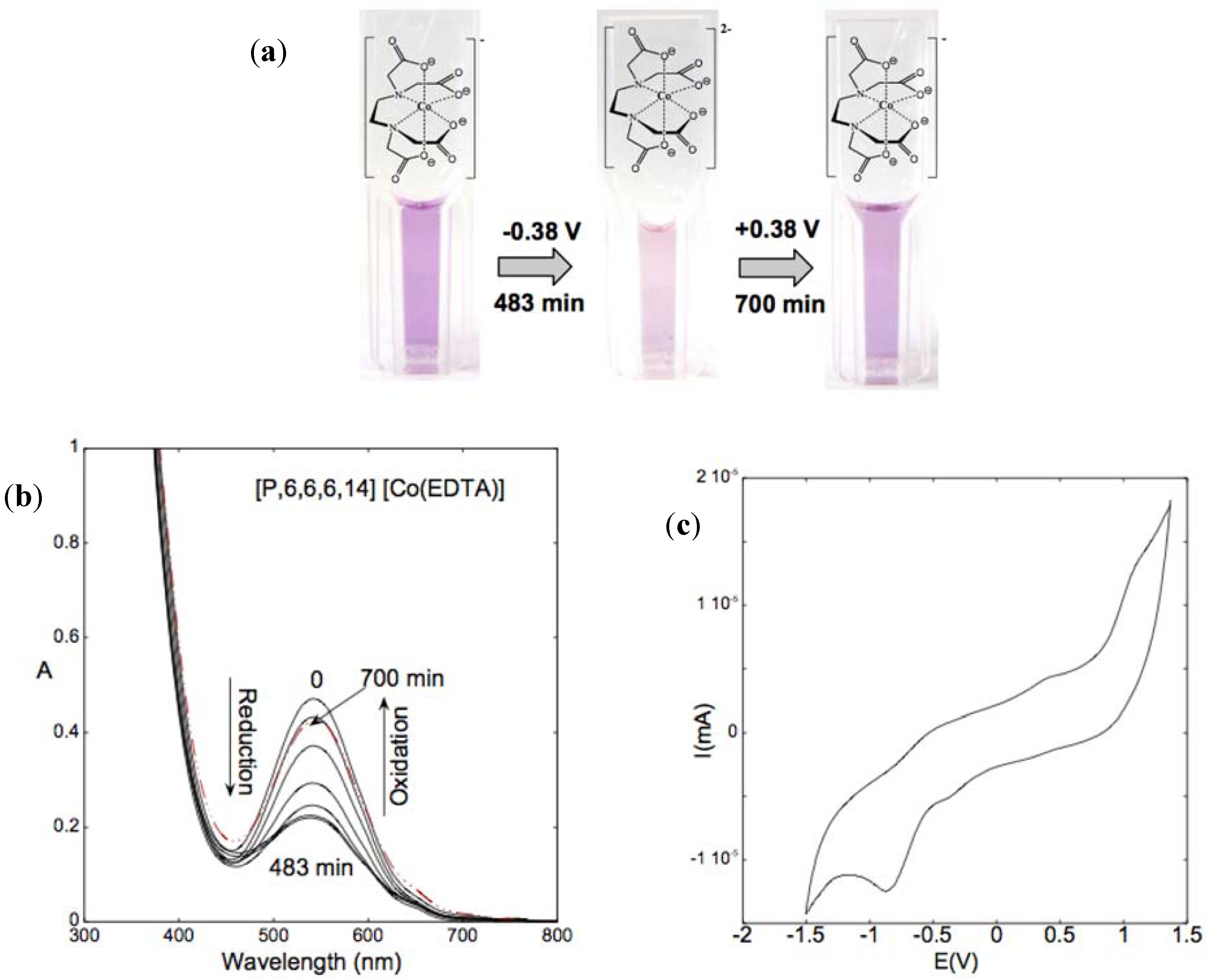
5.2. Electrochromic ILs
6. Conclusions
Acknowledgments
References and Notes
- Diamond, D.; Coyle, S.; Scarmagnani, S.; Hayes, J. Wireless sensor networks and chemo-/biosensing. Chem. Rev. 2008, 108, 652–679. [Google Scholar] [CrossRef]
- Bobacka, J.; Ivaska, A.; Lewenstam, A. Potentiometric ion sensors based on conducting polymers. Electroanal 2003, 15, 366–374. [Google Scholar] [CrossRef]
- Singh, N.; Mulrooney, R.C.; Kaur, N.; Callan, J.F. A nanoparticle based chromogenic chemosensor for the simultaneous detection of multiple analytes. Chem. Commun. 2008, 4900–4902. [Google Scholar]
- Schmittel, M.; Lin, H.W. Quadruple-channel sensing: A molecular sensor with a single type of receptor site for selective and quantitative multi-ion analysis. Angew. Chem.-Inter. Ed. 2007, 46, 893–896. [Google Scholar] [CrossRef]
- Bakker, E.; Diamond, D.; Lewenstam, A.; Pretsch, E. Ion sensors: Current limits and new trends. Anal. Chim. Acta 1999, 393, 11–18. [Google Scholar] [CrossRef]
- Diamond, D. Internet-scale sensing. Anal. Chem. 2004, 76, 278–286. [Google Scholar] [CrossRef]
- Park, C.O.; Fergus, J.W.; Miura, N.; Park, J.; Choi, A. Solid-state electrochemical gas sensors. Ionics 2009, 15, 261–284. [Google Scholar] [CrossRef]
- Anastasova-Ivanova, S.; Mattinen, U.; Radu, A.; Bobacka, J.; Lewenstam, A.; Migdalski, J.; Danielewskic, M.; Diamond, D. Development of miniature all-solid-state potentiometric sensing system. Sensor. Actuator. B-Chem. 2010, 146, 199–205. [Google Scholar] [CrossRef]
- Ahn, S.-K.; Kasi, R.M.; Kim, S.-C.; Sharma, N.; Zhou, Y. Stimuli-responsive polymer gels. Soft Matter 2008, 4, 1151–1157. [Google Scholar] [CrossRef]
- Osada, Y.; Gong, J.P. Soft and wet materials: Polymer gels. Adv. Mater 1998, 10, 827–837. [Google Scholar] [CrossRef]
- Neouze, M.A.; Le Bideau, J.; Gaveau, P.; Bellayer, S.; Vioux, A. Ionogels, new materials arising from the confinement of ionic liquids within silica-derived networks. Chem. Mater 2006, 18, 3931–3936. [Google Scholar] [CrossRef]
- Susan, M.A.; Kaneko, T.; Noda, A.; Watanabe, M. Ion gels prepared by in situ radical polymerization of vinyl monomers in an ionic liquid and their characterization as polymer electrolytes. J. Am. Chem. Soc. 2005, 127, 4976–4983. [Google Scholar]
- Le Bideau, J.; Viau, L.; Vioux, A. Ionogels, ionic liquid based hybrid materials. Chem. Soc. Rev. 2011, 40, 907–925. [Google Scholar] [CrossRef]
- Ueki, T.; Watanabe, M. Macromolecules in ionic liquids: Progress, challenges, and opportunities. Macromolecules 2008, 41, 3739–3749. [Google Scholar] [CrossRef]
- Kavanagh, A.; Byrne, R.; Diamond, D.; Radu, A. A two-component polymeric optode membrane based on a multifunctional ionic liquid. Analyst 2011, 136, 348–353. [Google Scholar] [CrossRef]
- Kavanagh, A.; Hilder, M.; Clark, N.; Radu, A.; Diamond, D. Wireless radio frequency detection of greatly simplified polymeric membranes based on a multifunctional ionic liquid. Electrochim. Acta 2011, 56, 8947–8953. [Google Scholar] [CrossRef]
- Kavanagh, A.; Copperwhite, R.; Oubaha, M.; Owens, J.; McDonagh, C.; Diamond, D.; Byrne, R. Photo-patternable hybrid ionogels for electrochromic applications. J. Mater Chem. 2011, 21, 8687–8693. [Google Scholar]
- Benito-Lopez, F.; Byrne, R.; Raduta, A.M.; Vrana, N.E.; McGuinness, G.; Diamond, D. Ionogel-based light-actuated valves for controlling liquid flow in micro-fluidic manifolds. Lab Chip 2010, 10, 195–201. [Google Scholar] [CrossRef]
- Cicmil, D.; Anastasova, S.; Kavanagh, A.; Diamond, D.; Mattinen, U.; Bobacka, J.; Lewenstam, A.; Radu, A. Ionic liquid-based, liquid-junction-free reference electrode. Electroanalysis 2011, 23, 1881–1890. [Google Scholar] [CrossRef]
- Khodagholy, D.; Curto, V.F.; Fraser, K.J.; Gurfinkel, M.; Byrne, R.; Diamond, D.; Benito-Lopez, F.; Owens, R.M. Organic electrochemical transistor incorporating an ionogel as solid state electolyte for lactate sensing. J. Mater. Chem. 2011. [Google Scholar] [CrossRef]
- Welton, T. Room-temperature ionic liquids. Solvents for synthesis and catalysis. Chem. Rev. 1999, 99, 2071–2083. [Google Scholar] [CrossRef]
- Fraser, K.J.; MacFarlane, D.R. Phosphonium-based ionic liquids: An overview. Aust. J. Chem. 2009, 62, 309–321. [Google Scholar] [CrossRef]
- Zhao, C.; Burrell, G.; Torriero, A.A.J.; Separovic, F.; Dunlop, N.F.; MacFarlane, D.R.; Bond, A.M. Electrochemistry of room temperature protic ionic liquids. J. Phys. Chem. B 2008, 112, 6923–6936. [Google Scholar]
- Plechkova, N.V.; Seddon, K.R. Applications of ionic liquids in the chemical industry. Chem. Soc. Rev. 2008, 37, 123–150. [Google Scholar] [CrossRef]
- Xue, H.; Gao, Y.; Twamley, B.; Shreeve, J.M. New energetic salts based on nitrogen-containing heterocycles. Chem. Mater. 2005, 17, 191–198. [Google Scholar] [CrossRef]
- MacFarlane, D.R.; Forsyth, M.; Izgorodina, E.I.; Abbott, A.P.; Annat, G.; Fraser, K. On the concept of ionicity in ionic liquids. Pccp. Phys. Chem. Chem. Phys. 2009, 11, 4962–4967. [Google Scholar] [CrossRef]
- Bradaric, C.J.; Downard, A.; Kennedy, C.; Robertson, A.J.; Zhou, Y.H. Industrial preparation of phosphonium ionic liquids. Green Chem. 2003, 5, 143–152. [Google Scholar] [CrossRef]
- Ermolaev, V.; Miluykov, V.; Rizvanov, I.; Krivolapov, D.; Zvereva, E.; Katsyuba, S.; Sinyashin, O.; Schmutzler, R. Phosphonium ionic liquids based on bulky phosphines: Synthesis, structure and properties. Dalton Trans. 2010, 39, 5564–5571. [Google Scholar]
- Busi, S.; Lahtinen, M.; Mansikkamaki, H.; Valkonen, J.; Rissanen, K. Synthesis, characterization and thermal properties of small R2R’2N+X- type quaternary ammonium halides. J. Solid State Chem. 2005, 178, 1722–1737. [Google Scholar] [CrossRef]
- MacFarlane, D.R.; Golding, J.; Forsyth, S.; Forsyth, M.; Deacon, G.B. Low viscosity ionic liquids based on organic salts of the dicyanamide anion. Chem. Commun. 2001, 1430–1431. [Google Scholar]
- MacFarlane, D.R.; Pringle, J.M.; Johansson, K.M.; Forsyth, S.A.; Forsyth, M. Lewis base ionic liquids. Chem. Commun. 2006, 1905–1917. [Google Scholar]
- Ercole, F.; Davis, T.P.; Evans, R.A. Photo-responsive systems and biomaterials: photochromic polymers, light-triggered self-assembly, surface modification, fluorescence modulation and beyond. Polym. Chem. 2010, 1, 37–54. [Google Scholar] [CrossRef]
- Mortimer, R.J. Electrochromic materials. In Annual Review of Materials Research; Clarke, D.R.F.P., Ed.; Annual Reviews: Palo Alto, CA, USA, 2011; Volume 41, pp. 241–268. [Google Scholar]
- Zhang, H.X.; Meng, X.; Li, P. Light and thermal-stimuli responsive materials. Prog. Chem. 2008, 20, 657–672. [Google Scholar]
- Einaga, Y. Photo-switching magnetic materials. J. Photochem. Photobiol. C-Photo 2006, 7, 69–88. [Google Scholar] [CrossRef]
- Roy, D.; Cambre, J.N.; Sumerlin, B.S. Future perspectives and recent advances in stimuli-responsive materials. Prog. Polym. Sci. 2010, 35, 278–301. [Google Scholar] [CrossRef]
- Scarmagnani, S.; Walsh, Z.; Slater, C.; Alhashimy, N.; Paull, B.; Macka, M.; Diamond, D. Polystyrene bead-based system for optical sensing using spiropyran photoswitches. J. Mater Chem. 2008, 18, 5063–5071. [Google Scholar] [CrossRef]
- Stitzel, S.; Byrne, R.; Diamond, D. LED switching of spiropyran-doped polymer films. J. Mater Sci. 2006, 41, 5841–5844. [Google Scholar] [CrossRef]
- Radu, A.; Byrne, R.; Alhashimy, N.; Fusaro, M.; Scarmagnani, S.; Diamond, D. Spiropyran-based reversible, light-modulated sensing with reduced photofatigue. J. Photochem. Photobiol. A-Chem. 2009, 206, 109–115. [Google Scholar] [CrossRef]
- Earle, M.J.; Gordon, C.M.; Plechkova, N.V.; Seddon, K.R.; Welton, T. Decolorization of ionic liquids for spectroscopy. Anal. Chem. 2007, 79, 758. [Google Scholar] [CrossRef]
- Miljanic, S.; Frkanec, L.; Meic, Z.; Zinic, M. Photoinduced gelation by stilbene oxalyl amide compounds. Langmuir 2005, 21, 2754–2760. [Google Scholar] [CrossRef]
- Sanchez, A.M.; Barra, M.; de Rossi, R.H. On the mechanism of the acid/base-catalyzed thermal cis-trans isomerization of methyl orange. J. Org. Chem. 1999, 64, 1604–1609. [Google Scholar] [CrossRef]
- Bazarnik, M.; Henzl, J.; Czajka, R.; Morgenstern, K. Light driven reactions of single physisorbed azobenzenes. Chem. Commun. 2011, 47, 7764–7766. [Google Scholar] [CrossRef]
- Ichimura, K.; Oh, S.K.; Nakagawa, M. Light-driven motion of liquids on a photoresponsive surface. Science 2000, 288, 1624–1626. [Google Scholar] [CrossRef]
- Hanabusa, K.; Hiratsuka, K.; Kimura, M.; Shirai, H. Easy preparation and useful character of organogel electrolytes based on low molecular weight gelator. Chem. Mater. 1999, 11, 649–655. [Google Scholar] [CrossRef]
- Terech, P.; Weiss, R.G. Low molecular mass gelators of organic liquids and the properties of their gels. Chem. Rev. 1997, 97, 3133–3159. [Google Scholar] [CrossRef]
- Murata, K.; Aoki, M.; Suzuki, T.; Harada, T.; Kawabata, H.; Komori, T.; Ohseto, F.; Ueda, K.; Shinkai, S. Thermal and light control of the sol-gel phase transition in cholesterol-based organic gels. Novel helical aggregation modes as detected by circular dichroism and electron microscopic observation. J. Am. Chem. Soc. 1994, 116, 6664–6676. [Google Scholar]
- Ahmed, S.A.; Sallenave, X.; Fages, F.; Mieden-Gundert, G.; Mueller, W.M.; Mueller, U.; Voegtle, F.; Pozzo, J.-L. Multiaddressable self-assembling organogelators based on 2H-chromene and N-Acyl-1,w-amino acid units. Langmuir 2002, 18, 7096–7101. [Google Scholar] [CrossRef]
- Minkin, V.I. Photo-, thermo-, solvato-, and electrochromic spiroheterocyclic compounds. Chem. Rev. 2004, 104, 2751–2776. [Google Scholar] [CrossRef]
- Rosario, R.; Gust, D.; Garcia, A.A.; Hayes, M.; Taraci, J.L.; Clement, T.; Dailey, J.W.; Picraux, S.T. Lotus effect amplifies light-induced contact angle switching. J. Phys. Chem. B 2004, 108, 12640–12642. [Google Scholar]
- Athanassiou, A.; Lygeraki, M.I.; Pisignano, D.; Lakiotaki, K.; Varda, M.; Mele, E.; Fotakis, C.; Cingolani, R.; Anastasiadis, S.H. Photocontrolled variations in the wetting capability of photochromic polymers enhanced by surface nanostructuring. Langmuir 2006, 22, 2329–2333. [Google Scholar]
- Rosario, R.; Gust, D.; Hayes, M.; Jahnke, F.; Springer, J.; Garcia, A.A. Photon-modulated wettability changes on spiropyran-coated surfaces. Langmuir 2002, 18, 8062–8069. [Google Scholar] [CrossRef]
- Radu, A.; Scarmagnani, S.; Byrne, R.; Slater, C.; Lau, K.T.; Diamond, D. Photonic modulation of surface properties: A novel concept in chemical sensing. J. Phys. D-Appl. Phys. 2007, 40, 7238–7244. [Google Scholar] [CrossRef]
- Coleman, S.; Byrne, R.; Minkovska, S.; Diamond, D. Thermal reversion of spirooxazine in ionic liquids containing the [NTf2](-) anion. Pccp. Phys. Chem. Chem. Phys. 2009, 11, 5608–5614. [Google Scholar] [CrossRef]
- Byrne, R.; Coleman, S.; Fraser, K.J.; Raduta, A.; MacFarlane, D.R.; Diamond, D. Photochromism of nitrobenzospiropyran in phosphonium based ionic liquids. Pccp. Phys. Chem. Chem. Phys. 2009, 11, 7286–7291. [Google Scholar] [CrossRef]
- Wagner, K.; Byrne, R.; Zanoni, M.; Gambhir, S.; Dennany, L.; Breukers, R.; Higgins, M.; Wagner, P.; Diamond, D.; Wallace, G.G.; et al. A multiswitchable poly(terthiophene) bearing a spiropyran functionality: Understanding photo- and electrochemical control. J. Am. Chem. Soc. 2011, 133, 5453–5462. [Google Scholar]
- Szilagyi, A.; Sumaru, K.; Sugiura, S.; Takagi, T.; Shinbo, T.; Zrinyi, M.; Kanamori, T. Rewritable microrelief formation on photoresponsive hydrogel layers. Chem. Mater 2007, 19, 2730–2732. [Google Scholar] [CrossRef]
- Byrne, R.; Ventura, C.; Lopez, F.B.; Walther, A.; Heise, A.; Diamond, D. Characterisation and analytical potential of a photo-responsive polymeric material based on spiropyran. Biosens. Bioelectron. 2010, 26, 1392–1398. [Google Scholar] [CrossRef]
- Brazel, C.S.; Peppas, N.A. Synthesis and characterization of thermomechanically and chemomechanically responsive poly(N-isopropylacrylamide-co-methacrylic acid) hydrogels. Macromolecules 1995, 28, 8016–8020. [Google Scholar] [CrossRef]
- Schild, H.G.; Tirrell, D.A. Interaction of poly(N-isopropylacrylamide) with sodium normal-alkyl sulfates in aqueous-solution. Langmuir 1991, 7, 665–671. [Google Scholar] [CrossRef]
- Bakker, E.; Buehlmann, P.; Pretsch, E. Carrier-based ion-selective electrodes and bulk optodes. 1. General Characteristics. Chem. Rev. 1997, 97, 3083–3132. [Google Scholar] [CrossRef]
- Bakker, E.; Xu, A.P.; Pretsch, E. Optimum composition of neutral carrier based Ph electrodes. Anal. Chim. Acta 1994, 295, 253–262. [Google Scholar] [CrossRef]
- Malon, A.; Radu, A.; Qin, W.; Qin, Y.; Ceresa, A.; Maj-Zurawska, M.; Bakker, E.; Pretsch, E. Improving the detection limit of anion-selective electrodes: An iodide-selective membrane with a nanomolar detection limit. Anal. Chem. 2003, 75, 3865–3871. [Google Scholar]
- Sutter, J.; Radu, A.; Peper, S.; Bakker, E.; Pretsch, E. Solid-contact polymeric membrane electrodes with detection limits in the subnanomolar range. Anal. Chim. Acta 2004, 523, 53–59. [Google Scholar] [CrossRef]
- Ceresa, A.; Radu, A.; Peper, S.; Bakker, E.; Pretsch, E. Rational design of potentiometric trace level ion sensors. A Ag+-selective electrode with a 100 ppt detection limit. Anal. Chem. 2002, 74, 4027–4036. [Google Scholar] [CrossRef]
- Del Sesto, R.E.; Corley, C.; Robertson, A.; Wilkes, J.S. Tetraalkylphosphonium-based ionic liquids. J. Organomet. Chem. 2005, 690, 2536–2542. [Google Scholar] [CrossRef]
- Peng, B.; Zhu, J.W.; Liu, X.J.; Qin, Y. Potentiometric response of ion-selective membranes with ionic liquids as ion-exchanger and plasticizer. Sensor. Actuator. B-Chem. 2008, 133, 308–314. [Google Scholar] [CrossRef]
- Shvedene, N.V.; Chernyshov, D.V.; Khrenova, M.G.; Formanovsky, A.A.; Baulin, V.E.; Pletnev, I.V. Ionic liquids plasticize and bring ion-sensing ability to polymer membranes of selective electrodes. Electroanalysis 2006, 18, 1416–1421. [Google Scholar] [CrossRef]
- Buhlmann, P.; Pretsch, E.; Bakker, E. Carrier-based ion-selective electrodes and bulk optodes. 2. Ionophores for potentiometric and optical sensors. Chem. Rev. 1998, 98, 1593–1687. [Google Scholar] [CrossRef]
- Chernyshov, D.V.; Shuedene, N.V.; Antipova, E.R.; Pletnev, I.V. Ionic liquid-based miniature electrochemical sensors for the voltammetric determination of catecholamines. Anal. Chim. Acta 2008, 621, 178–184. [Google Scholar] [CrossRef]
- Nadherna, M.; Opekar, F.; Reiter, J. Ionic liquid-polymer electrolyte for amperometric solid-state NO(2) sensor. Electrochim. Acta 2011, 56, 5650–5655. [Google Scholar] [CrossRef]
- Bird, C.L.; Kuhn, A.T. Electrochemistry of the viologens. Chem. Soc. Rev. 1981, 10, 49–82. [Google Scholar]
- Mortimer, R.J.; Dyer, A.L.; Reynolds, J.R. Electrochromic organic and polymeric materials for display applications. Displays 2006, 27, 2–18. [Google Scholar] [CrossRef]
- Rosseinsky, D.R.; Mortimer, R.J. Electrochromic systems and the prospects for devices. Adv. Mater 2001, 13, 783–793. [Google Scholar] [CrossRef]
- Wishart, J.F. Energy applications of ionic liquids. Energy Environ. Sci. 2009, 2, 956–961. [Google Scholar] [CrossRef]
- Ahmad, S.; Deepa, M. Ionogels encompassing ionic liquid with liquid like performance preferable for fast solid state electrochromic devices. Electrochem. Commun. 2007, 9, 1635–1638. [Google Scholar] [CrossRef]
- Kavanagh, A.; Fraser, K.; Byrne, R.; Diamond, D. An electrochromic ionic liquid: Design, characterisation and incorporation into a solid-state devices. Chem. Mater 2011. Submitted. [Google Scholar]
- Deepa, M.; Ahmad, S.; Sood, K.N.; Alam, J.; Ahmad, S.; Srivastava, A.K. Electrochromic properties of polyaniline thin film nanostructures derived from solutions of ionic liquid/polyethylene glycol. Electrochim. Acta 2007, 52, 7453–7463. [Google Scholar] [CrossRef]
- Ahmad, S.; Singh, S. Electrochromic device based on carbon nanotubes functionalized poly(methyl pyrrole) synthesized in hydrophobic ionic liquid medium. Electrochem. Commun. 2008, 10, 895–898. [Google Scholar] [CrossRef]
- Deepa, M.; Awadhia, A.; Bhandari, S. Electrochemistry of poly(3,4-ethylenedioxythiophene)-polyaniline/Prussian blue electrochromic devices containing an ionic liquid based gel electrolyte film. Pccp. Phys. Chem. Chem. Phys. 2009, 11, 5674–5685. [Google Scholar] [CrossRef]
- Oehme, I.; Wolfbeis, O.S. Optical sensors for determination of heavy metal ions. Mikrochim. Acta 1997, 126, 177–192. [Google Scholar] [CrossRef]
- Zhu, J.W.; Zhai, J.Y.; Li, X.; Qin, Y. Applications of hydrophobic room temperature ionic liquids in ion-selective optodes. Sensor. Actuator. B-Chem. 2011, 159, 256–260. [Google Scholar] [CrossRef]
- Topal, S.Z.; Ertekin, K.; Gurek, A.G.; Yenigul, B.; Ahsen, V. Tuning pH sensitivities of zinc phthalocyanines in ionic liquid modified matrices. Sensor. Actuator. B-Chem. 2011, 156, 236–244. [Google Scholar] [CrossRef]
- Lunstroot, K.; Driesen, K.; Nockemann, P.; Gorller-Walrand, C.; Binnemans, K.; Bellayer, S.; Bideau, J.L.; Vioux, A. Luminescent ionogels based on europium-doped ionic liquids confined within silica-derived networks. Chem. Mater 2006, 18, 5711–5715. [Google Scholar] [CrossRef]
- Lunstroot, K.; Driesen, K.; Nockemann, P.; Van Hecke, K.; Van Meervelt, L.; Goerller-Walrand, C.; Binnemans, K.; Bellayer, S.; Viau, L.; Le Bideau, J.; et al. Lanthanide-doped luminescent ionogels. Dalton. Trans. 2009, 298–306. [Google Scholar]
- Cheminet, N.; Jarrosson, T.; Lere-Porte, J.-P.; Serein-Spirau, F.; Cury, L.; Moreau, J.; Viau, L.; Vioux, A. One pot synthesis of fluorescent pi-conjugated materials: Immobilization of phenylene-ethynylene polyelectrolytes in silica confined ionogels. J. Mater. Chem. 2011, 21, 13588–13593. [Google Scholar]
- Wassercheid, P.; Welton, T. Ionic Liquids in Synthesis; Wiley-VCH: Weinheim, Germany, 2003. [Google Scholar]
- Liu, Y.; Wang, M.; Li, J.; Li, Z.; He, P.; Liu, H.; Li, J. Highly active horseradish peroxidase immobilized in 1-butyl-3-methylimidazolium tetrafluoroborate room-temperature ionic liquid based sol-gel host materials. Chem. Commun. 2005, 1778–1780. [Google Scholar]
- Ohno, H.; Suzuki, C.; Fukumoto, K.; Yoshizawa, M.; Fujita, K. Electron transfer process of poly(ethylene oxide)-modified Cytochrome C in imidazolium type ionic liquid. Chem. Lett. 2003, 32, 450–451. [Google Scholar] [CrossRef]
- Baker, S.N.; McCleskey, T.M.; Pandey, S.; Baker, G.A. Fluorescence studies of protein thermostability in ionic liquids. Chem. Commun. 2004, 940–941. [Google Scholar]
- Pernak, A.; Iwanik, K.; Majewski, P.; Grzymislawski, M.; Pernak, J. Ionic liquids as an alternative to formalin in histopathological diagnosis. Acta Histochem. 2005, 107, 149–156. [Google Scholar] [CrossRef]
- Fujita, K.; MacFarlane, D.R.; Forsyth, M. Protein solubilising and stabilising ionic liquids. Chem. Commun. 2005, 4804–4806. [Google Scholar]
- Laszlo, J.A.; Compton, D.L. Comparison of peroxidase activities of hemin, cytochrome c and microperoxidase-11 in molecular solvents and imidazolium-based ionic liquids. J. Mol. Catal. B Enzym. 2002, 18, 109–120. [Google Scholar] [CrossRef]
- Yang, Z.; Pan, W. Ionic liquids: Green solvents for nonaqueous biocatalysis. Enzyme Microb. Technol. 2005, 37, 19–28. [Google Scholar] [CrossRef]
- Zhao, H. Methods for stabilizing and activating enzymes in ionic liquids—A review. J. Chem. Tech. Biotech. 2010, 85, 891–907. [Google Scholar] [CrossRef]
- Abe, Y.; Yoshiyama, K.; Yagi, Y.; Hayase, S.; Kawatsura, M.; Itoh, T. A rational design of phosphonium salt type ionic liquids for ionic liquid coated-lipase catalyzed reaction. Green Chem. 2010, 12, 1976–1980. [Google Scholar] [CrossRef]
- Zhang, Y.; Zheng, J. Direct electrochemistry and electrocatalysis of myoglobin immobilized in hyaluronic acid and room temperature ionic liquids composite film. Electrochem. Commun. 2008, 10, 1400–1403. [Google Scholar] [CrossRef]
- Zhang, J.; Lei, J.; Liu, Y.; Zhao, J.; Ju, H. Highly sensitive amperometric biosensors for phenols based on polyaniline-ionic liquid-carbon nanofiber composite. Biosens. Bioelectron. 2009, 24, 1858–1863. [Google Scholar] [CrossRef]
- Torimoto, T.; Tsuda, T.; Okazaki, K.-I.; Kuwabata, S. New frontiers in materials science opened by ionic liquids. Adv. Mater. 2010, 22, 1196–1221. [Google Scholar] [CrossRef]
- Ohno, K.-I.; Tachikawa, K.; Manz, A. Microfluidics: Applications for analytical purposes in chemistry and biochemistry. Electrophoresis 2008, 29, 4443–4453. [Google Scholar] [CrossRef]
- Brady, S.; Dunne, L.E.; Lynch, A.; Smyth, B.; Diamond, D. Personalised Health Management Systems: The Integration of Innovative Sensing, Textile, Information and Communication Technologies; IOS Press: Amsterdam, The Netherlands, 2005; Volume 117. [Google Scholar]
- Bhandari, P.; Narahari, T.; Dendukuri, D. Fab-chips’: A versatile, fabric-based platform for low-cost, rapid and multiplexed diagnostics. Lab Chip 2011, 11, 2493–2499. [Google Scholar] [CrossRef]
- Schmid-Wendtner, M.H.; Korting, H.C. The pH of the skin surface and its impact on the barrier function. Skin Pharmacol. Physiol. 2006, 19, 296–302. [Google Scholar] [CrossRef]
- Patterson, M.J.; Galloway, S.D.R.; Nimmo, M.A. Effect of induced metabolic alkalosis on sweat composition in men. Acta Physiol. Scand. 2002, 174, 41–46. [Google Scholar] [CrossRef]
- Granger, D.; Marsolais, M.; Burry, J.; Laprade, R. Na+/H+ exchangers in the human eccrine sweat duct. Amer. J. Physiol.-Cell Physiol. 2003, 285, C1047–C1058. [Google Scholar]
- Patterson, M.J.; Galloway, S.D.R.; Nimmo, M.A. Variations in regional sweat composition in normal human males. Exp. Physiol. 2000, 85, 869–875. [Google Scholar] [CrossRef]
- Morgan, R.M.; Patterson, M.J.; Nimmo, M.A. Acute effects of dehydration on sweat composition in men during prolonged exercise in the heat. Acta Physiol. Scand. 2004, 182, 37–43. [Google Scholar] [CrossRef]
- O’Toole, M.; Shepherd, R.; Wallace, G.G.; Diamond, D. Inkjet printed LED based pH chemical sensor for gas sensing. Anal. Chim. Acta 2009, 652, 308–314. [Google Scholar] [CrossRef]
- Smyth, C.N.; Lau, K.T.; Shepherd, R.L.; Diamond, D.; Wu, Y.; Spinks, G.M.; Wallace, G.G. Self-maintained colorimetric acid/base sensor using polypyrrole actuator. Sensor. Actuator. B Chem. 2008, 129, 518–524. [Google Scholar] [CrossRef]
- Benito-Lopez, F.; Coyle, S.; Byrne, R.; O’Toole, C.; Barry, C.; Diamond, D. Simple barcode system based on inonogels for real time pH-sweat monitoring. Body Sensor Networks (BSN) 2010, 291–296. [Google Scholar]
- Coyle, S.; Benito-Lopez, F.; Radu, T.; Lau, K.T.; Diamond, D. Fibers and fabrics for chemical and biological sensing. J. Text. App. 2010, 14, 64. [Google Scholar]
- Nilsson, D.; Kugler, T.; Svensson, P.-O.; Berggren, M. An all-organic sensor-transistor based on a novel electrochemical transducer concept printed electrochemical sensors on paper. Sensor. Actuator. B Chem. 2002, 86, 193–197. [Google Scholar] [CrossRef]
- Chaubey, A.; Pande, K.K.; Singh, V.S.; Malhotra, B.D. Co-immobilization of lactate oxidase and lactate dehydrogenase on conducting polyaniline films. Anal. Chim. Acta 2000, 407, 97–103. [Google Scholar] [CrossRef]
- Weber, J.; Kumar, A.; Kumar, A.; Bhansali, S. Novel lactate and pH biosensor for skin and sweat analysis based on single walled carbon nanotubes. Sensor. Actuator. B Chem. 2006, 117, 308–313. [Google Scholar] [CrossRef]
- Yashina, E.I.; Borisova, A.V.; Karyakina, E.E.; Shchegolikhina, O.I.; Vagin, M.Y.; Sakharov, D.A.; Tonevitsky, A.G.; Karyakin, A.A. Sol-gel immobilization of lactate oxidase from organic solvent: Toward the advanced lactate biosensor. Anal. Chem. 2010, 82, 1601–1604. [Google Scholar]
- Cai, X.; Yan, J.; Chu, H.; Wu, M.; Tu, Y. An exercise degree monitoring biosensor based on electrochemiluminescent detection of lactate in sweat. Sensor. Actuator. B Chem. 2010, 143, 655–659. [Google Scholar] [CrossRef]
- Wang, Y.; Xu, H.; Zhang, J.M.; Li, G. Electrochemical sensors for clinic analysis. Sensors 2008, 8, 2043–2081. [Google Scholar] [CrossRef]
- Weil, M.H.; Afifi, A.A. Experimental and clinical studies on lactate and pyruvate as indicators of the severity of acute circulatory failure (shock). Circulation 1970, 41, 989–1001. [Google Scholar] [CrossRef]
- Green, J.M.; Pritchett, R.C.; Crews, T.R.; McLester, J.R.; Tucker, D.C. Sweat lactate response between males with high and low aerobic fitness. Eur. J. Appl. Physiol. 2004, 91, 1–6. [Google Scholar] [CrossRef]
- Billat, L.V. Use of blood lactate measurements for prediction of exercise performance and for control of training: Recommendations for long-distance running. Sports Med. 1996, 22, 157–175. [Google Scholar] [CrossRef]
- Branco, L.C.; Pina, F. Intrinsically photochromic ionic liquids. Chem. Commun. 2009, 6204–6206. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhang, S.; Liu, S.; Ma, X.; Lu, L.; Deng, Y. Ionic liquid-modified dyes and their sensing performance toward acids in aqueous and non-aqueous solutions. Analyst 2011, 136, 1302–1304. [Google Scholar] [CrossRef]
- Branco, A.; Branco, L.C.; Pina, F. Electrochromic and magnetic ionic liquids. Chem. Commun. 2011, 47, 2300–2302. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kavanagh, A.; Byrne, R.; Diamond, D.; Fraser, K.J. Stimuli Responsive Ionogels for Sensing Applications—An Overview. Membranes 2012, 2, 16-39. https://doi.org/10.3390/membranes2010016
Kavanagh A, Byrne R, Diamond D, Fraser KJ. Stimuli Responsive Ionogels for Sensing Applications—An Overview. Membranes. 2012; 2(1):16-39. https://doi.org/10.3390/membranes2010016
Chicago/Turabian StyleKavanagh, Andrew, Robert Byrne, Dermot Diamond, and Kevin J. Fraser. 2012. "Stimuli Responsive Ionogels for Sensing Applications—An Overview" Membranes 2, no. 1: 16-39. https://doi.org/10.3390/membranes2010016
APA StyleKavanagh, A., Byrne, R., Diamond, D., & Fraser, K. J. (2012). Stimuli Responsive Ionogels for Sensing Applications—An Overview. Membranes, 2(1), 16-39. https://doi.org/10.3390/membranes2010016





