Functionality in Electrospun Nanofibrous Membranes Based on Fiber’s Size, Surface Area, and Molecular Orientation
Abstract
:1. Introduction
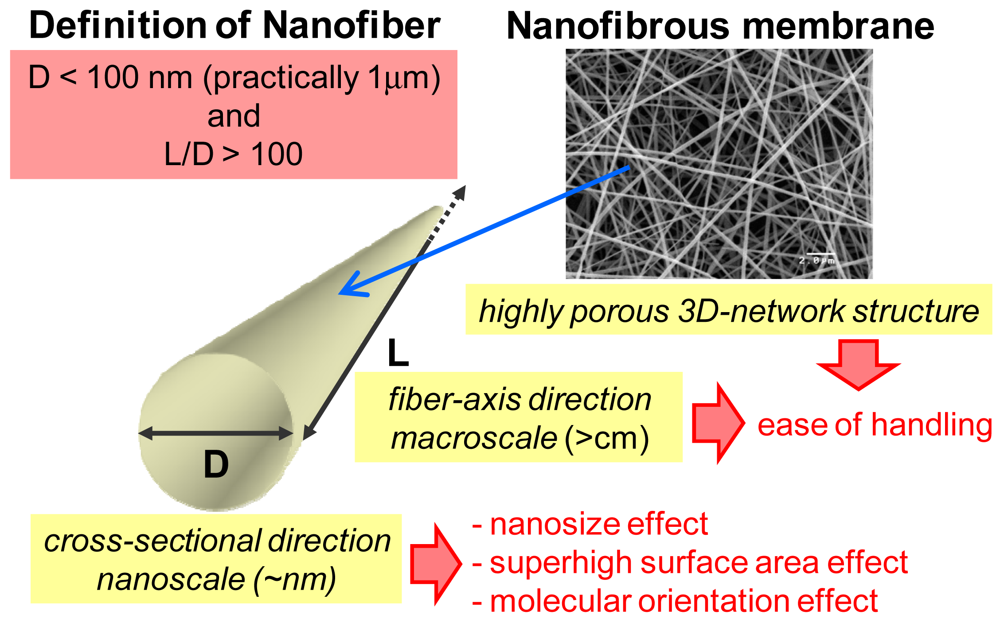
2. Control of Structure of Nanofibers and Nanofibrous Membranes
2.1. Control of Size, Internal Structure, and Surface of Nanofiber
| Solution properties |
| Viscosity (molecular weight of polymer and concentration) |
| Electric conductivity |
| Solvent properties (surface tension, boiling point, polarity, and permittivity) |
| Operating conditions |
| Applied voltage (typically from several kV to several 10 kV) |
| Distance between spinneret and collector (typically from several cm to 50 cm) |
| Feeding rate of polymer solution |
| Spinneret (inner diameter, shape, and material) |
| Surrounding conditions |
| Temperature |
| Humidity |
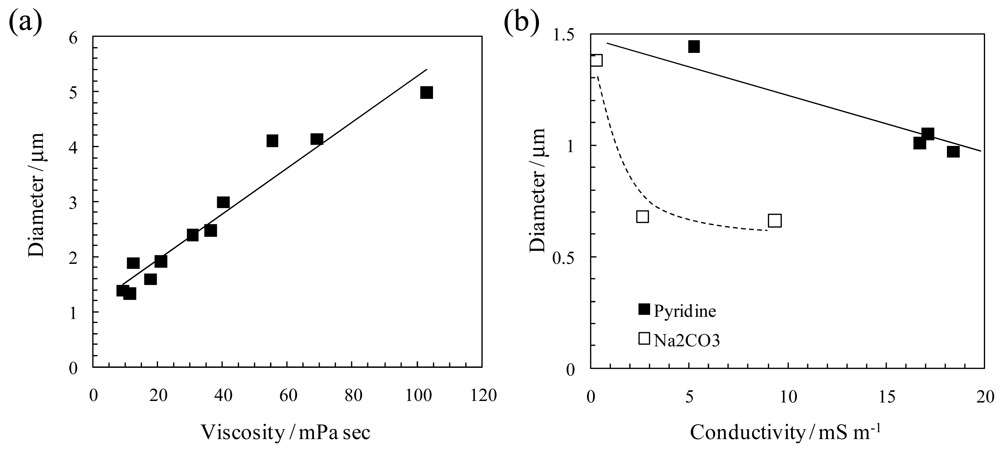

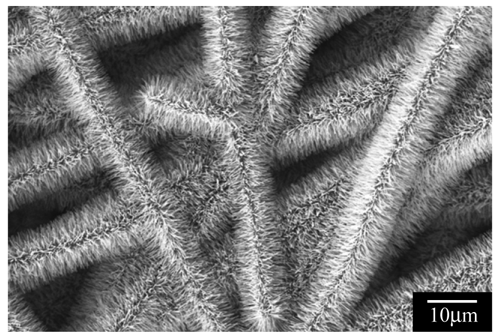
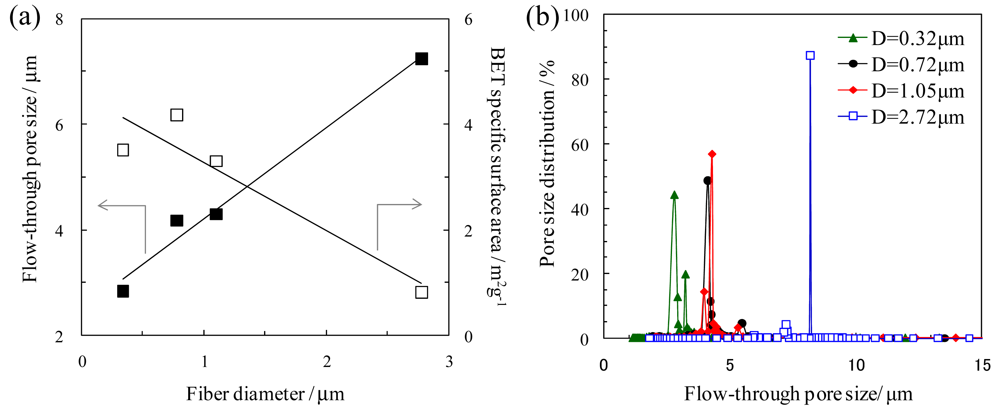
2.2. Pore structure of Nanofibrous Membranes
3. Ion-Exchangers
3.1. Ion-Exchange Nanofibrous Membranes by Electrospinning
| Material | Chitosan | Sulfonated PS | Quaternized P4VP |
|---|---|---|---|
| Type (ion-exchange group) | anion-exchange (amino group) | cation-exchange (sulfonic acid group) | anion-exchange (tertiary pyridyl and quaternary pyridinium groups) |
| Ion-exchange capacity *1 [mmol/g-dry membrane] | 5.4 | 1.3 | 0.8 |
| Average pore size *2 [μm] | 0.5 | 1.9 | 3.5 |
| Membrane porosity *2 [%] | 92 | 75 | 80 |
| Through-pore specific surface area *2 [m2/g] | 26 | 13 | 14 |
| BET specific surface area *3 [m2/g] | 2 | 600 | |
| Thickness [μm] | 59 | 52 | 40 |

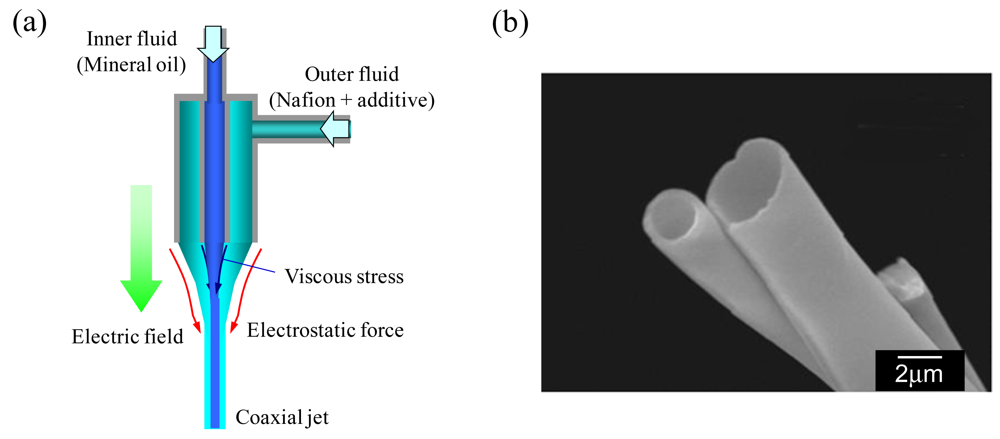
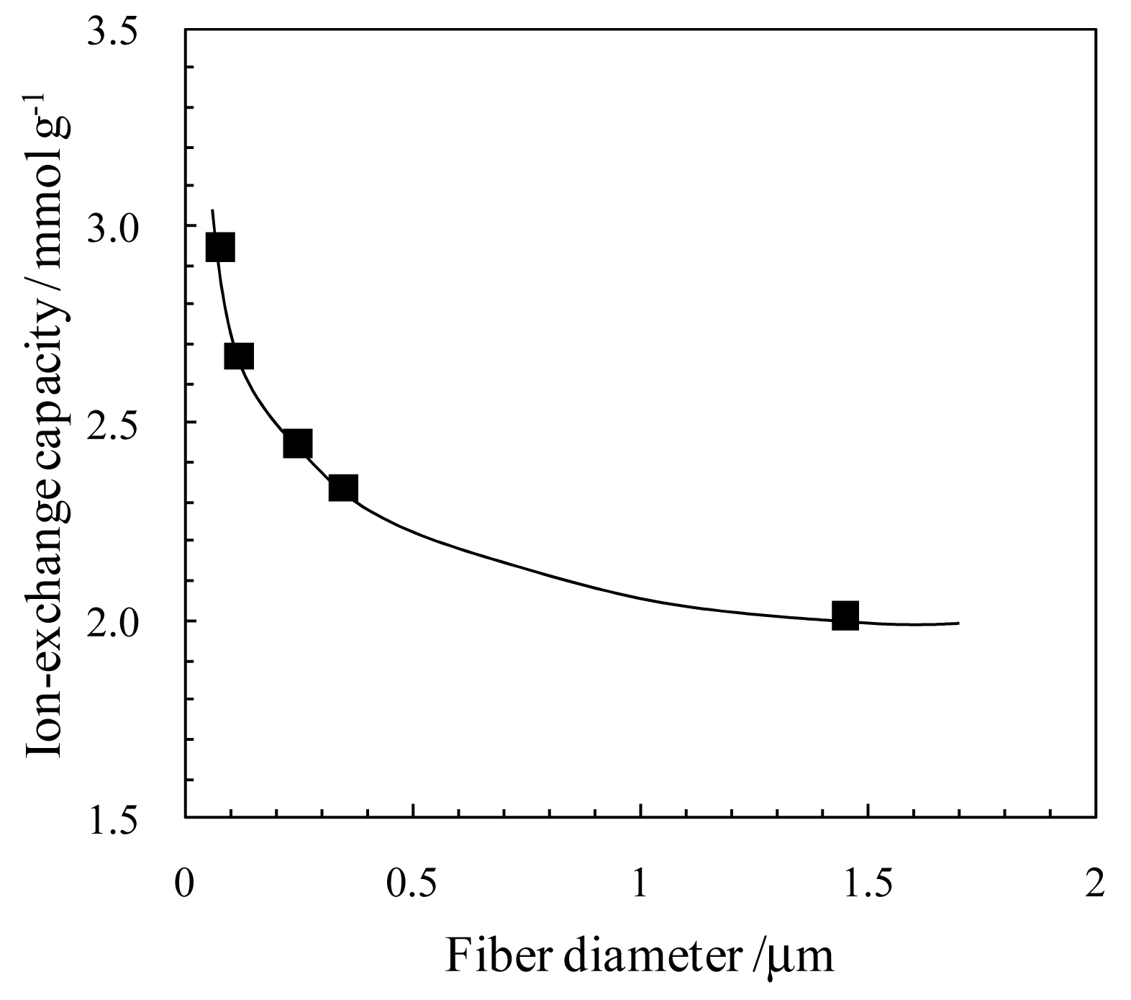
3.2. Catalytic Effect of Ion-Exchange Nanofiber on Water Splitting in Bipolar Membrane Electrodialysis System
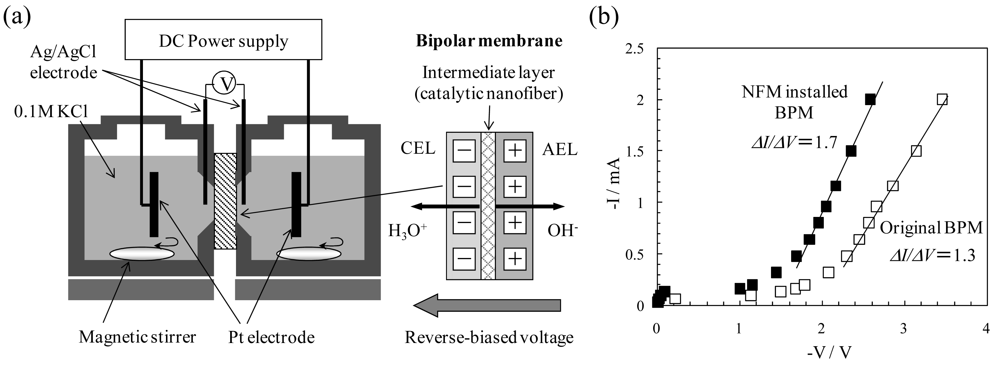
4. Air Filters


5. Antimicrobial Materials
| Bacterial count (cells/vial) *1 | ||||
|---|---|---|---|---|
| Before incubation | After incubation | |||
| Chitosan nanofibrous membrane | 5.4 × 105 | 0 | ||
| Chitosan microfibrous membrane *2 | 5.4 × 105 | 4.2 × 103 | ||
| Polyethylene terephtalate (PET) microfiber membrane *2 | 5.4 × 105 | 2.4 × 105 | ||
| Polymer | Chemical structure | Fiber diameter (nm) | Bacteriostatic activity *1 | Sterilization activity *2 |
|---|---|---|---|---|
| Poly(lactic acid) (PLA) | 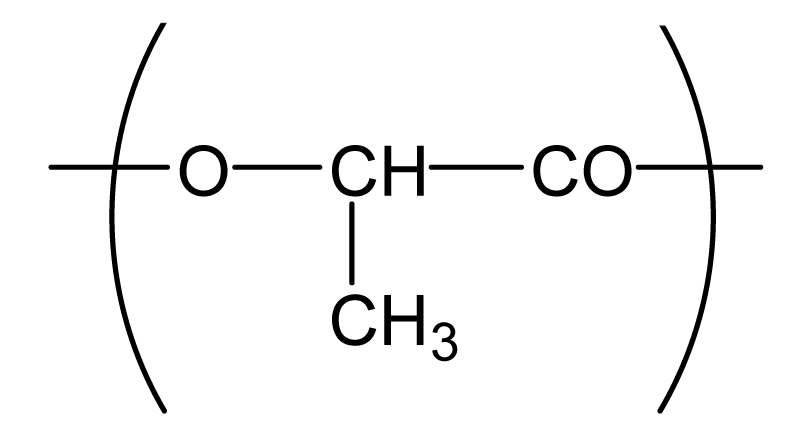 | 80,000 *3 | 0.2 | −2.3 |
| 500 | 4.1 | 1.5 | ||
| Polyamide66 (PA66) | 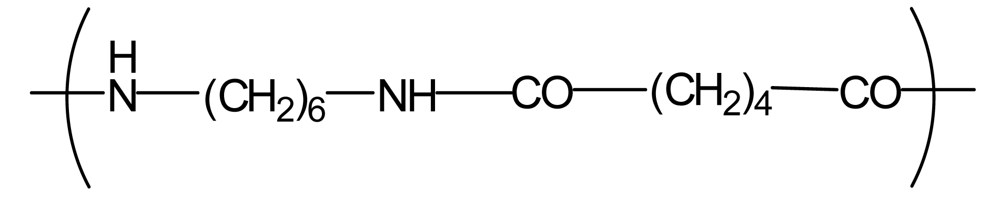 | 1,300 | −0.2 | −1.9 |
| 250 | 4.2 | 1.6 | ||
| Polyamide6 (PA6) | 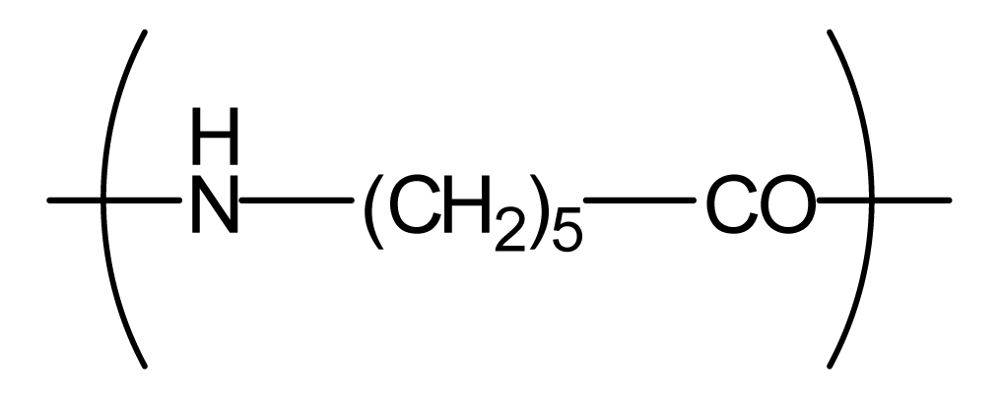 | 1,300 | −0.3 | −2.1 |
| 300 | 4.1 | 1.5 | ||
| Polyamide (PA) | 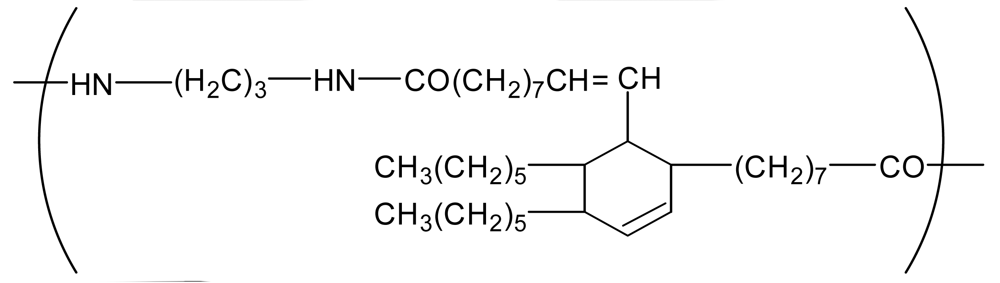 | 800 | 0.7 | −2 |
| Polyurethane (PU(ester)) | 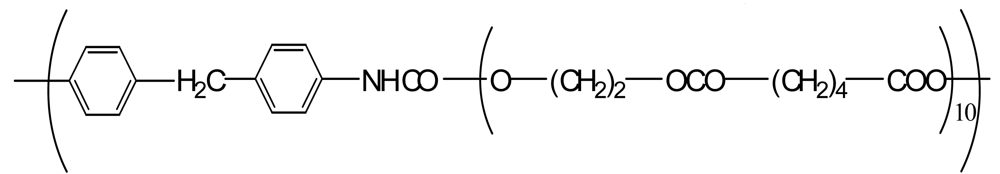 | 40,000 *3 | −0.1 | −1.9 |
| 500 | 3.2 | 0.6 | ||
| Polyurethane (PU(ether)) | 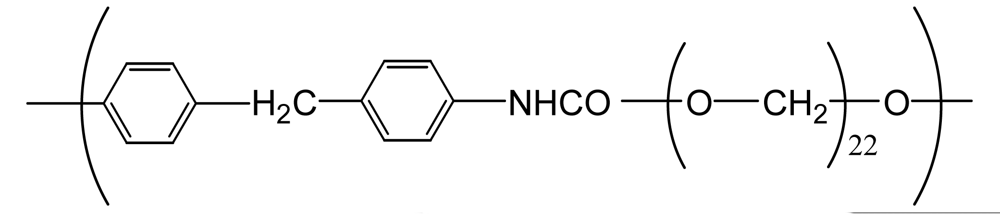 | 500 | 1.3 | −1.4 |
| Polyacrylonitrile (PAN) | 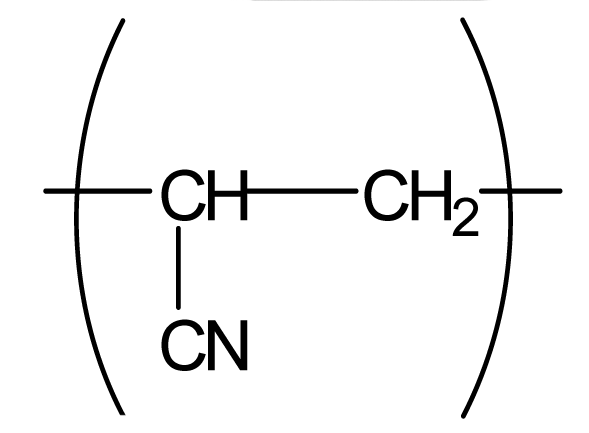 | 100 | 4.1 | 1.5 |
| Polyvinylidene-chloride (PVDC) | 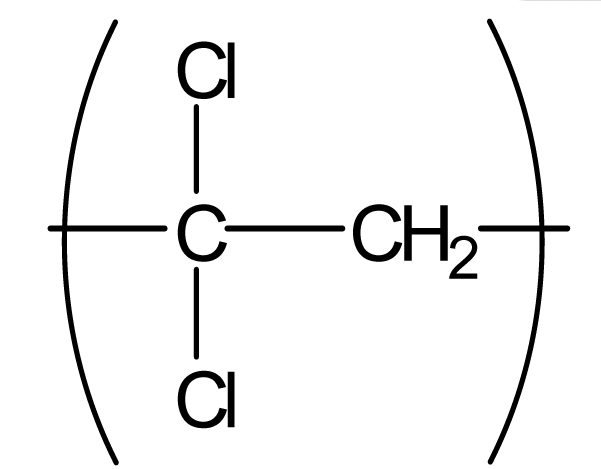 | 800 | 4.1 | 1.5 |
| Polystyrene (PS) |  | 800 | 4.0 | 1.5 |

6. Summary and Future Directions
Acknowledgments
References
- Doshi, J.; Reneker, D.H. Electrospinning process and applications of electrospun fibers. J. Electrost. 1995, 35, 151–160. [Google Scholar]
- Li, D.; Xia, Y. Electrospinning of nanofibers: Reinventing the wheel? Adv. Mater. 2004, 16, 1151–1170. [Google Scholar]
- Greiner, A.; Wendorff, J.H. Electrospinning: A fascinating method for the preparation of ultrathin fibers. Angew. Chem. Int. Ed. 2007, 46, 5670–5703. [Google Scholar]
- Yoon, K.; Hsiao, B.S.; Chu, B. Functional nanofibers for environmental applications. J. Mater. Chem. 2008, 18, 5326–5334. [Google Scholar]
- Matsumoto, H. Nanofibrous membranes—Preparation and application of electrospun membranes. Maku 2010, 35, 113–118. [Google Scholar]
- Son, W.-K.; Cho, D.; Park, W.-H. Direct electrospinning of ultrafine titania fibers in the absence of polymer additives and formation of pure anatase titania fibers at low temperature. Nanotechnology 2006, 17, 439–443. [Google Scholar]
- Zhou, F.-L.; Gong, R.-H.; Porat, I. Mass production of nanofibre assemblies by electrostatic spinning. Polym. Int. 2009, 58, 331–342. [Google Scholar]
- Thompson, C.J.; Chase, G.G.; Yarin, A.L.; Reneker, D.H. Effects of parameters on nanofiber diameter determined from electrospinning model. Polymer 2007, 48, 6913–6922. [Google Scholar]
- Imaizumi, S.; Matsumoto, H.; Suzuki, K.; Minagawa, M.; Kimura, M.; Tanioka, A. Phenolic resin-based carbon nanofibers prepared by electrospinning: Additive effects of poly(vinyl butyral) and electrolytes. Polym. J. 2009, 41, 1124–1128. [Google Scholar]
- Minato, K.; Ohkawa, K.; Yamamoto, H. Chain conformations of poly(γ-benzyl-L-glutamate) pre and post an electrospinning process. Macromol. Biosci. 2006, 6, 487–495. [Google Scholar]
- Kakade, M.V.; Givens, S.; Gardner, K.; Lee, K.H.; Chase, D.B.; Rabolt, J.F. Electric field induced orientation of polymer chains in macroscopically aligned electospun polymer nanofibers. J. Am. Chem. Soc. 2007, 129, 2777–2782. [Google Scholar]
- Kongkhlang, T.; Tashiro, K.; Kotaki, M.; Chirachanchai, S. Electrospinning as a new technique to control the crystal morphology and molecular orientation of polyoxymethylene nanofibers. J. Am. Chem. Soc. 2008, 130, 15460–15466. [Google Scholar]
- Danno, T.; Matsumoto, H.; Nasir, M.; Shimizu, S.; Minagawa, M.; Kawaguchi, J.; Horibe, H.; Tanioka, A. Fine structure of PVDF nanofiber fabricated by electrospray deposition. J. Polym. Sci. B Polym. Phys. 2008, 46, 558–563. [Google Scholar]
- Nakashima, K.; Tsuboi, K.; Matsumoto, H.; Ishige, R.; Tokita, M.; Watanabe, J.; Tanioka, A. Control over internal structure of liquid crystal polymer nanofibers by electrospinning. Macromol. Rapid Commun. 2010, 31, 1641–1645. [Google Scholar]
- Imaizumi, S.; Matsumoto, H.; Konosu, Y.; Tsuboi, K.; Minagawa, M.; Tanioka, A.; Koziol, K.; Windle, A. Top-down process based on electrospinning, twisting, and heating for producing one-dimensional carbon nanotube assembly. ACS Appl. Mater. Interfaces 2011, 3, 469–475. [Google Scholar]
- Suzuki, K.; Matsumoto, H.; Minagawa, M.; Tanioka, A.; Hayashi, Y.; Fukuzono, K.; Gehan, A.J.; Amaratunga, G.A.J. Carbon nanotubes on carbon fabrics for flexible field emitter arrays. Appl. Phys. Lett. 2008, 93, 053107:1–053107:3. [Google Scholar]
- Unalan, H.E.; Wei, D.; Suzuki, K.; Dalal, S.; Hiralal, P.; Matsumoto, H.; Imaizumi, S.; Minagawa, M.; Tanioka, A.; Flewitt, A.J.; et al. Photoelectrochemical cell using dye sensitized zinc oxide nanowires grown on carbon fibers. Appl. Phys. Lett. 2008, 93, 133116:1–133116:3. [Google Scholar]
- Xie, Y. Preparation and Characterization of Silica-Based Nanofibers by Electrospinning. M.Sc. Thesis, Tokyo Institute of Technology, Tokyo, Japan, 2008. [Google Scholar]
- Streat, M. Boom time for ion exchange. Chem. Ind. (London) 2004, 13, 20–21. [Google Scholar]
- Seo, H.; Matsumoto, H.; Hara, S.; Yako, H.; Minagawa, M.; Tanioka, A.; Yamagata, Y.; Inoue, K. Preparation of polysaccharide nanofiber fabrics by electrospray deposition: Additive effects of poly(ethylene oxide). Polym. J. 2005, 37, 391–398. [Google Scholar]
- Matsumoto, H.; Wakamatsu, Y.; Minagawa, M.; Tanioka, A. Preparation of ion-exchange fiber fabrics by electrospray deposition. J. Colloid Interface Sci. 2006, 293, 143–150. [Google Scholar]
- Imaizumi, S.; Matsumoto, H.; Ashizawa, M.; Tsuboi, K.; Minagawa, M.; Tanioka, A. Preparation of ion-exchange carbon nanofibers by electrospinning: Effect of fiber diameter on their adsorption behaviors. Fiber Prepr. Japan 2011, 66, 112. (in Japanese). [Google Scholar]
- Matsumoto, H.; Yako, H.; Minagawa, M.; Tanioka, A. Characterization of chitosan nanofiber fabric by electrospray deposition: Electrokinetic and adsorption behavior. J. Colloid Interface Sci. 2007, 310, 678–681. [Google Scholar]
- Matsumoto, H.; Nagata, T.; Minagawa, M.; Tanioka, A. Preparation of polyelectrolyte nanofiber by electrospray deposition. Polym. Prepr. Japan 2006, 55, 3438. (in Japanese). [Google Scholar]
- Yarin, A.L.; Zussman, E.; Wendorff, J.H.; Greiner, A. Material encapsulation and transport in core–shell micro/nanofibers, polymer and carbon nanotubes and micro/nanochannels. J. Mater. Chem. 2007, 17, 2585–2599. [Google Scholar]
- Dong, B.; Gwee, L.; Salas-de la Cruz, D.; Winey, K.I.; Elabd, Y.A. Super proton conductive high-purity Nafion nanofibers. Nano Lett. 2010, 10, 3785–3790. [Google Scholar]
- Kemperman, A.J.B. Handbook on Bipolar Membrane Technology; Twente University Press: Enschede, The Netherlands, 2000. [Google Scholar]
- Wakamatsu, Y.; Matsumoto, H.; Minagawa, M.; Tanioka, A. Effect of ion-exchange nanofiber fabric on water splitting in bipolar membrane. J. Colloid Interface Sci. 2006, 300, 442–445. [Google Scholar]
- Brown, R.C. Air Filtration: An Integrated Approach to the Theory and Applications of Fibrous Filters; Pergamon Press: Oxford, UK, 1993. [Google Scholar]
- Yamaguchi, T.; Shima, T.; Matsumoto, H.; Minagawa, M.; Tanioka, A. Study on non-continuum air flow through electrospun nanofiber nonwovens. Fiber Prepr. Japan 2010, 65, 222. (in Japanese). [Google Scholar]
- Hinds, W.C. Aerosol Technology, 2nd ed.; Wiley-Intersciences: New York, NY, USA, 1998. [Google Scholar]
- Pich, J. Pressure characteristics of fibrous aerosol filters. J. Colloid Interface Sci. 1971, 37, 912–917. [Google Scholar]
- Nasir, M.; Matsumoto, H.; Minagawa, M.; Tanioka, A.; Danno, T.; Horibe, H. Preparation of porous PVDF nanofiber from PVDF/PVP blend by electrospray deposition. Polym. J. 2007, 39, 1060–1064. [Google Scholar]
- Tan, K.; Obendorf, S.K. Fabrication and evaluation of electrospun nanofibrous antimicrobial nylon 6 membranes. J. Membr. Sci. 2007, 305, 287–298. [Google Scholar]
- Matsumoto, H.; Minagawa, M.; Tanioka, A.; Yako, H. Biological Ion-Exchange Nanofibre Fabrics by Electrospinning. Proceedings of the IEX 2008 Recent Advances in Ion Exchange Theory & Practice Conference, IEX2008, Cambridge, UK, 9–11 July 2008; pp. 495–499.
- Ogushi, Y.; Sasaki, N.; Imashiro, Y.; Minagawa, M.; Matsumoto, H.; Tanioka, A. Antimicrobial activity of ultra-fine fiber nonwoven fabrics produced by electrospinning. Seikei Kakou 2009, 21, 287–290. [Google Scholar]
- Graham, K.; Ouyang, M.; Raether, T.; Grafe, T. Polymeric nanofibers in air filtration applications. Proceedings of the Fifteenth Annual Technical Conference and Expo of American Filtration and Separation Society, Galveston, TX, USA, 9–12 April 2002.
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Matsumoto, H.; Tanioka, A. Functionality in Electrospun Nanofibrous Membranes Based on Fiber’s Size, Surface Area, and Molecular Orientation. Membranes 2011, 1, 249-264. https://doi.org/10.3390/membranes1030249
Matsumoto H, Tanioka A. Functionality in Electrospun Nanofibrous Membranes Based on Fiber’s Size, Surface Area, and Molecular Orientation. Membranes. 2011; 1(3):249-264. https://doi.org/10.3390/membranes1030249
Chicago/Turabian StyleMatsumoto, Hidetoshi, and Akihiko Tanioka. 2011. "Functionality in Electrospun Nanofibrous Membranes Based on Fiber’s Size, Surface Area, and Molecular Orientation" Membranes 1, no. 3: 249-264. https://doi.org/10.3390/membranes1030249





