Change in Hepatitis A Seroprevalence among U.S. Children and Adolescents: Results from the National Health and Nutrition Examination Survey 2003–2006 and 2007–2010
Abstract
:1. Introduction
2. Experimental Section
2.1. Data Source and Sample Design
2.2. Laboratory Testing
2.3. Definitions the FIPR
2.4. Statistical Analysis
3. Results
3.1. HAV Testing Response Rates
3.2. Change in Seropositivity over Time (Survey Cycle) by Birth Cohort, Geographic Region, and Race/Ethnicity
3.3. Predictors of Seropositivity Using Logistic Models for 2007–2010
| NHANES 2003–2006 | NHANES 2007–2010 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| n | Percent | Lower | Upper | n | Percent | Lower | Upper | Increase | p-value | |
| Prevalence | 95% CI | 95% CI | Prevalence | 95% CI | 95% CI | Over time | ||||
| All | 4,185 | 24.4 | 16.6 | 33.9 | 3,804 | 37.6 | 32.6 | 42.7 | 13.1 | p < 0.05 |
| Region of the U.S. * | ||||||||||
| Early region (ref) | 1,759 | 47.4 | 34.8 | 60.3 | 1,600 | 66.0 | 52.2 | 78.0 | 18.5 | NS |
| Later region | 2,426 | 10.1 a | 7.3 | 13.5 | 2,204 | 21.6a | 16.5 | 27.4 | 11.5 | p < 0.01 |
| Birth Cohort ^ | ||||||||||
| Pre-vaccine (ref) | 3,682 | 24.4 | 16.4 | 34.1 | 2,023 | 33.8 | 28.8 | 39.0 | 9.3 | NS |
| Post vaccine | 503 | 24.5 | 16.1 | 34.8 | 1,781 | 43.4 a | 38.0 | 48.9 | 18.9 | p < 0.01 |
| Race/ethnicity | ||||||||||
| White, non-Hispanic (ref) | 1,162 | 20.1 | 10.4 | 33.2 | 1,258 | 25.5 | 20.9 | 30.7 | 5.4 | NS |
| Black, non-Hispanic | 1,508 | 20.0 | 13.9 | 27.3 | 939 | 39.1 b | 30.8 | 47.9 | 19.1 | p < 0.01 |
| Mexican American | 1,190 | 53.2 a | 47.2 | 59.1 | 971 | 76.5 a | 67.6 | 83.9 | 23.3 | p < 0.001 |
| White, non-Hispanic (ref) | 1,162 | 20.1 | 10.4 | 33.2 | 1,258 | 25.5 | 20.9 | 30.7 | 5.4 | NS |
| All others + | 3,023 | 32.0 c | 26.7 | 37.6 | 2,546 | 56.0 a | 49.0 | 62.8 | 24.0 | p < 0.001 |
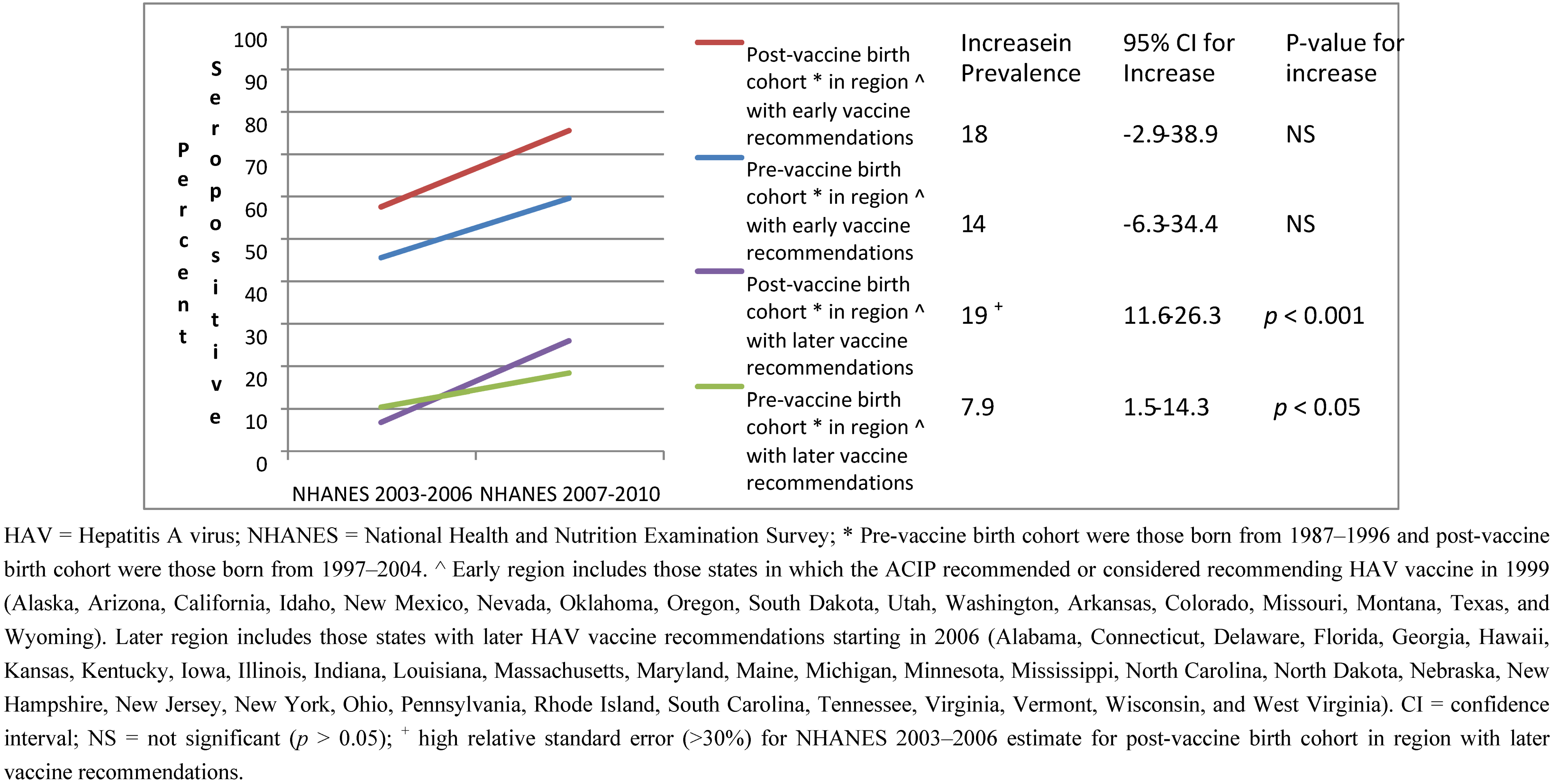

| Pre-vaccine | Post-vaccine | |||||
|---|---|---|---|---|---|---|
| Birth cohort in | Birth cohort in | |||||
| All | Region with Early Vaccine | Region with Later Vaccine | Region with Later Vaccine | Region with Later Vaccine | ||
| Recommendations a | Recommendations b | Recommendations | Recommendations | |||
| Region of the U.S. | ||||||
| Early a | 6.7 (3.5–13.0) c | NA | NA | NA | NA | |
| Later b | ref | |||||
| Birth Cohort * | ||||||
| Pre-vaccine | ref | ref | ref | NA | NA | |
| Post-vaccine | 1.6 (1.4–2.0) c | 1.9 (1.2–2.8)e | 1.5 (1.3–1.7) c | |||
| Race/ethnicity | ||||||
| Non-Hispanic white | ref | ref | ref | ref | ref | |
| All others ^ | 3.0 (2.0–4.6) c | 5.1 (2.2–11.6)c | 2.2 (1.5–3.2) c,f | 1.5 (1.0–2.4) | 3.2 (2.2–4.8) c | |
| Gender | ||||||
| Male | ref | ref | ref | ref | ref | |
| Female | 1.2 (1.0–1.5) d | 1.0 (0.7–1.4) | 1.3 (1.1–1.6)e | 1.3 (0.9–1.9) | 1.4 (1.1–1.8) d | |
| Poverty Index | ||||||
| <1.0 | 1.0 (0.8–1.2) | 1.0 (0.7–1.5) | 1.0 (0.7–1.2) | 1.0 (0.7–1.4) | 0.9 (0.6–1.3) | |
| ≥1.0 | ref | ref | ref | ref | ref | |
| Education of head of household | ||||||
| <High school | 1.1 (0.8–1.6) | 1.0 (0.6–1.8) | 1.1 (0.8–1.6) | 1.0 (0.6–1.6) | 1.3 (0.8–2.2) | |
| High school graduate | 0.8 (0.5–1.1) | 0.9 (0.5–1.6) | 0.7 (0.5–1.1)f | 0.5 (0.3–0.9) d | 1.0 (0.6–1.8) | |
| >High school | ref | ref | ref | ref | ref | |
| Household size | ||||||
| ≤4 persons | ref | ref | ref | ref | ref | |
| 5 or more persons | 1.3 (1.0–1.6) | 1.3 (0.9–1.9) | 1.2 (0.8–1.8) | 1.2 (0.7–2.2) | 1.1 (0.8–1.6) | |
| Health Insurance | ||||||
| Any | 1.7 (1.0–2.8) | 1.6 (0.7–3.8) | 2.0 (1.2–3.3) d | 1.9 (1.0–3.7) d,r | 1.9 (0.7–4.8) | |
| None | ref | ref | ref | ref | ref | |
4. Discussion
5. Conclusions
Conflict of Interest
References
- Wasley, A.; Samandari, T.; Bell, B.P. Incidence of hepatitis a in the United States in the era of vaccination. JAMA 2005, 294, 19–201. [Google Scholar]
- Recommendations of the Advisory Committee on Immunization Practices (ACIP). Prevention of hepatitis a through active or passive immunization: Recommendations of the Advisory Committee onIimmunization Practices (ACIP). MMWR Recomm. Rep. 1996, 45, 1–30.
- Recommendations of the Advisory Committee on Immunization Practices (ACIP). Prevention of hepatitis a through active or passive immunization: Recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm. Rep. 1999, 48, 1–37.
- Fiore, A.E.; Wasley, A.; Bell, B.P. Prevention of hepatitis a through active or passive immunization: Recommendations of the advisory committee on immunization practices (ACIP). MMWR Recomm. Rep. 2006, 55, 1–23. [Google Scholar]
- Daniels, D.; Grytdal, S.; Wasley, A.; Centers for Disease Control and Prevention. Surveillance for acute viral hepatitis—United States, 2007. MMWR Surveill. Summ. 2009, 58, 1–27. [Google Scholar]
- Centers for Disease Control and Prevention. Viral Hepatitis Surveillance—United States, 2009. Available online: http://www.cdc.gov/hepatitis/Statistics/2009Surveillance/Commentary.htm/ (accessed on 18 January 2012).
- Centers for Disease Control and Prevention, U.S.D.H.H.S. Available online: http://www.healthypeople.gov/2020/topicsobjectives2020/objectiveslist.aspx?topicId=23/ (accessed on 17 May 2012).
- Begg, N.; Miller, E. Role of epidemiology in vaccine policy. Vaccine 1990, 8, 180–189. [Google Scholar]
- Bell, B.P.; Kruszon-Moran, D.; Shapiro, C.N.; Lambert, S.B.; McQuillan, G.M.; Margolis, H.S. Hepatitis a virus infection in the United States: Serologic results from the Third National Health and Nutrition Examination Survey. Vaccine 2005, 23, 5798–5806. [Google Scholar]
- Klevens, R.M.; Kruszon-Moran, D.; Wasley, A.; Gallagher, K.; McQuillan, G.M.; Kuhnert, W.; Teshale, E.H.; Drobeniuc, J.; Bell, B.P. Seroprevalence of hepatitis a virus antibodies in the U.S.: Results from the National Health and Nutrition Examination Survey. Public Health Rep. 2011, 126, 522–532. [Google Scholar]
- Centers for Disease Control and Prevention. Hepatitis a vaccination coverage among children aged 24–35 months—United States, 2006 and 2007. MMWR Morb. Mortal. Wkly. Rep. 2009, 58, 689–694.
- Centers for Disease Control and Prevention. NHANES file documentation. Available online: http://www.cdc.gov/nchs/nhanes/about_nhanes.htm/ (accessed on 16 March 2012).
- US Department of Health and Human Services. Poverty Guidelines, Research, and Measurement. US Department of Health and Human Services website. Available online: http://aspe.hhs.gov/POVERTY/index.shtml/ (accessed 16 March 2012).
- National Center for Health Statistics, CDC. National Health and Nutrition Examination Surveys Analytic Guidelines. Available online: http://www.cdc.gov/nchs/nhanes/nhanes2003-2004/analytical_guidelines.htm/ (accessed on 16 March 2012).
- Korn, L. Confidence intervals for proportions with small expected number of positive counts estimated from survey data. Surv. Methodol. 1998, 24, 193–201. [Google Scholar]
- Klevens, R.M.; Miller, J.T.; Iqbal, K.; Thomas, A.; Rizzo, E.M.; Hanson, H.; Sweet, K.; Phan, Q.; Cronquist, A.; Khudyakov, Y.; et al. The evolving epidemiology of hepatitis a in the United States: Incidence and molecular epidemiology from population-based surveillance, 2005–2007. Arch. Intern. Med. 2010, 170, 1811–1818. [Google Scholar]
- Centers for Disease Control and Prevention. National, State, and Local area vaccination coverage among children aged 19–35 months—United States, 2011. MMWR Morb. Mortal. Wkly. Rep. 2012, 61, 689–696.
- Centers for Disease Control and Prevention. Vaccine for Childrens Prgram. Available online: http://www.cdc.gov/vaccines/programs/vfc/default.htm/ (accessed on 5 September 2012).
- Dorell, C.G.; Yankey, D.; Byrd, K.K.; Murphy, T.V. Hepatitis a vaccination coverage among adolescents in the United States. Pediatrics 2012, 129, 213–221. [Google Scholar]
- Centers for Disease Control and Prevention. Adult vaccination coverage—United States, 2010. MMWR Morb. Mortal. Wkly. Rep. 2012, 61, 66–72.
- Chodick, G.; Heymann, A.D.; Ashkenazi, S.; Kokia, E.; Shalev, V. Long-term trends in hepatitis a incidence following the inclusion of hepatitis a vaccine in the routine nationwide immunization program. J. Viral Hepat. 2008, 15, 62–65. [Google Scholar]
- Vacchino, M.N. Incidence of hepatitis a in Argentina after vaccination. J. Viral Hepat. 2008, 15, 47–50. [Google Scholar]
- Chironna, M.; Prato, R.; Sallustio, A.; Martinelli, D.; Tafuri, S.; Quarto, M.; Germinario, C. Hepatitis a in Puglia (South Italy) after 10 years of universal vaccination: Need for strict monitoring and catch-up vaccination. BMC Infect. Dis. 2012, 12, 271. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kruszon-Moran, D.; Klevens, R.M.; McQuillan, G.M. Change in Hepatitis A Seroprevalence among U.S. Children and Adolescents: Results from the National Health and Nutrition Examination Survey 2003–2006 and 2007–2010. Vaccines 2013, 1, 105-119. https://doi.org/10.3390/vaccines1020105
Kruszon-Moran D, Klevens RM, McQuillan GM. Change in Hepatitis A Seroprevalence among U.S. Children and Adolescents: Results from the National Health and Nutrition Examination Survey 2003–2006 and 2007–2010. Vaccines. 2013; 1(2):105-119. https://doi.org/10.3390/vaccines1020105
Chicago/Turabian StyleKruszon-Moran, Deanna, R. Monina Klevens, and Geraldine M. McQuillan. 2013. "Change in Hepatitis A Seroprevalence among U.S. Children and Adolescents: Results from the National Health and Nutrition Examination Survey 2003–2006 and 2007–2010" Vaccines 1, no. 2: 105-119. https://doi.org/10.3390/vaccines1020105




