Testing the Effects of DL-Alpha-Tocopherol Supplementation on Oxidative Damage, Total Antioxidant Protection and the Sex-Specific Responses of Reproductive Effort and Lifespan to Dietary Manipulation in Australian Field Crickets (Teleogryllus commodus)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experimental Animals and Experimental Design
2.2. Construction of Artificial Diets
2.3. Feeding Protocol
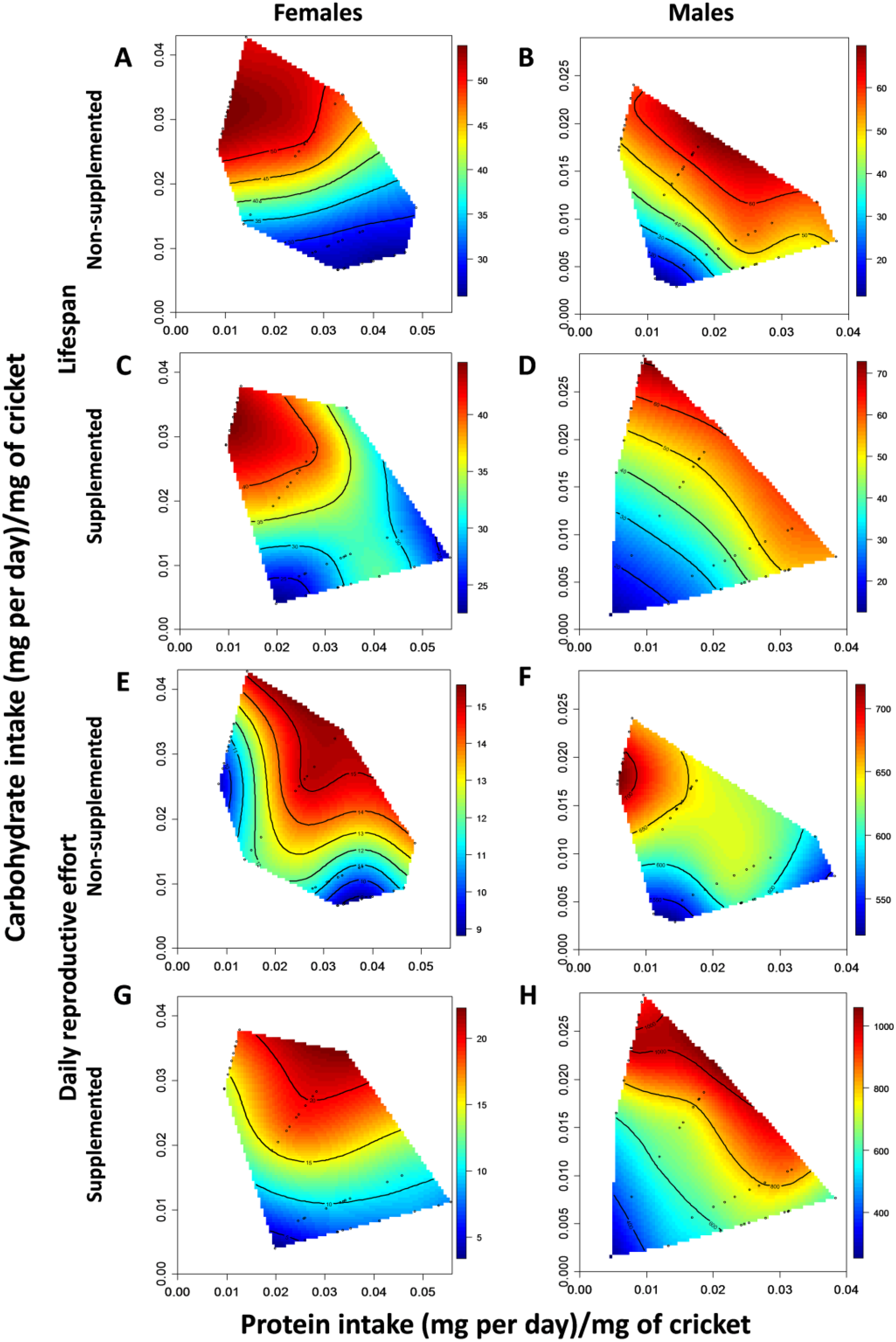
2.4. Experiment 1: The Effect of Diet and dl-Alpha-Tocopherol Supplementation on Lifespan and Reproductive Effort
2.5. Experiment 2: The Effect of Diet and dl-Alpha-Tocopherol Supplementation on Oxidative Damage and Antioxidant Protection
2.6. Statistical Analyses
3. Results
3.1. The Effect of Diet and dl-Alpha-Tocopherol Supplementation on Lifespan and Reproductive Effort
| Non-Supplemented | Supplemented | |||||
|---|---|---|---|---|---|---|
| Coefficient * ± SE | Prop | p Value | Coefficient ± SE | Prop | p Value | |
| (A): Females | ||||||
| LS | ||||||
| P | −0.159 ± 0.171 | 0.181 | 0.362 | −0.095 ± 0.183 | 0.695 | 0.610 |
| C | 0.486 ± 0.159 | 0.998 | 0.004 | 0.271 ± 0.126 | 0.019 | 0.038 |
| P × P | −0.258 ± 0.198 | 0.102 | 0.203 | −0.350 ± 0.201 | 0.953 | 0.094 |
| C × C | −0.374 ± 0.176 | 0.021 | 0.042 | −0.307 ± 0.144 | 0.018 | 0.039 |
| P × C | −0.283 ± 0.266 | 0.853 | 0.295 | −0.661 ± 0.276 | 0.012 | 0.023 |
| DRE | ||||||
| P | 0.192 ± 0.165 | 0.125 | 0.250 | 0.139 ± 0.196 | 0.758 | 0.484 |
| C | 0.280 ± 0.133 | 0.022 | 0.042 | 0.678 ± 0.168 | 0.001 | 0.002 |
| P × P | −0.187 ± 0.083 | 0.016 | 0.031 | −0.416 ± 0.193 | 0.018 | 0.037 |
| C × C | −0.145 ± 0.126 | 0.129 | 0.259 | −0.204 ± 0.194 | 0.851 | 0.298 |
| P × C | 0.179 ± 0.262 | 0.751 | 0.498 | −0.260 ± 0.291 | 0.810 | 0.379 |
| (B): Males | ||||||
| LS | ||||||
| P | 0.521 ± 0.202 | 0.994 | 0.013 | 0.511 ± 0.193 | 0.995 | 0.010 |
| C | 0.961 ± 0.302 | 0.002 | 0.003 | 0.767 ± 0.227 | 0.001 | 0.002 |
| P × P | −0.422 ± 0.290 | 0.912 | 0.176 | −0.137 ± 0.277 | 0.689 | 0.622 |
| C × C | −0.635 ± 0.480 | 0.905 | 0.191 | −0.224 ± 0.322 | 0.245 | 0.490 |
| P × C | −0.379 ± 0.468 | 0.785 | 0.430 | −0.292 ± 0.453 | 0.737 | 0.526 |
| DRE | ||||||
| P | −0.023 ± 0.257 | 0.536 | 0.928 | 0.170 ± 0.276 | 0.729 | 0.543 |
| C | 0.086 ± 0.317 | 0.605 | 0.790 | 0.354 ± 0.149 | 0.021 | 0.043 |
| P × P | −0.126 ± 0.308 | 0.659 | 0.683 | −0.201 ± 0.334 | 0.274 | 0.549 |
| C × C | −0.935 ± 0.421 | 0.017 | 0.033 | −0.064 ± 0.388 | 0.565 | 0.870 |
| P × C | −0.858 ± 0.676 | 0.106 | 0.212 | −0.060 ± 0.547 | 0.543 | 0.914 |
3.2. The Effect of Diet and dl-Alpha-Tocopherol Supplementation on Oxidative Damage and Antioxidant Protection
| Non-Supplemented | Supplemented | |||||
|---|---|---|---|---|---|---|
| Coefficient ± SE | Prop | p Value | Coefficient ± SE | Prop | p Value | |
| (A): Females | ||||||
| PC | ||||||
| P | −0.347 ± 0.162 | 0.981 | 0.039 | −0.300 ± 0.143 | 0.022 | 0.043 |
| C | 0.099 ± 0.157 | 0.266 | 0.532 | 0.026 ± 0.188 | 0.445 | 0.890 |
| P × P | 0.272 ± 0.217 | 0.891 | 0.218 | 0.084 ± 0.227 | 0.643 | 0.714 |
| C × C | 0.160 ± 0.147 | 0.858 | 0.284 | −0.092 ± 0.210 | 0.105 | 0.211 |
| P × C | 0.307 ± 0.330 | 0.179 | 0.358 | −0.074 ± 0.426 | 0.568 | 0.864 |
| TAC | ||||||
| P | −0.249 ± 0.119 | 0.978 | 0.043 | −0.217 ± 0.091 | 0.011 | 0.022 |
| C | −0.076 ± 0.170 | 0.329 | 0.657 | −0.042 ± 0.196 | 0.415 | 0.830 |
| P × P | 0.347 ± 0.231 | 0.928 | 0.144 | 0.199 ± 0.234 | 0.799 | 0.401 |
| C × C | 0.062 ± 0.156 | 0.347 | 0.694 | 0.138 ± 0.217 | 0.735 | 0.530 |
| P × C | 0.228 ± 0.352 | 0.739 | 0.521 | 0.057 ± 0.439 | 0.449 | 0.898 |
| (B): Males | ||||||
| PC | ||||||
| P | −0.355 ± 0.169 | 0.022 | 0.044 | 0.439 ± 0.212 | 0.023 | 0.046 |
| C | 0.065 ± 0.299 | 0.586 | 0.827 | 0.584 ± 0.279 | 0.022 | 0.043 |
| P × P | 0.969 ± 0.280 | 0.001 | 0.002 | 0.012 ± 0.379 | 0.512 | 0.976 |
| C × C | 0.057 ± 0.364 | 0.562 | 0.876 | 0.258 ± 0.322 | 0.215 | 0.429 |
| P × C | 0.600 ± 0.573 | 0.151 | 0.303 | 0.016 ± 0.709 | 0.509 | 0.981 |
| TAC | ||||||
| P | −0.406 ± 0.197 | 0.977 | 0.047 | 0.300 ± 0.265 | 0.868 | 0.265 |
| C | −0.424 ± 0.304 | 0.087 | 0.173 | 0.032 ± 0.287 | 0.456 | 0.912 |
| P × P | 0.478 ± 0.230 | 0.023 | 0.046 | 0.092 ± 0.355 | 0.399 | 0.797 |
| C × C | 0.023 ± 0.428 | 0.521 | 0.958 | 0.392 ± 0.145 | 0.005 | 0.011 |
| P × C | −0.161 ± 0.674 | 0.594 | 0.811 | −0.778 ± 0.664 | 0.875 | 0.250 |

3.3. Do Differences in Oxidative Damage or Protection Mediate Life-History Trade-Offs?
4. Discussion
| Hypothesis | Prediction | Observation | Conclusion | |
|---|---|---|---|---|
| Trade-off between lifespan and reproduction. | Traits peak in different regions of nutrient landscape. | Females—prediction met. Males—prediction met in non-supplemented males but not in supplemented males. | Sex-specific trade-off, pronounced in females, weak in males. | |
| Accumulation of oxidative damage causes aging. | Damage highest in short lived animals. | Generally, high damage, long lifespan. | Oxidative damage to proteins does not appear to mediate variation in lifespan within each sex, following dietary manipulation. | |
| Antioxidant supplementation improves lifespan. | No effect of antioxidants on survival. | |||
| Reproductive effort elevates oxidative stress. | High reproductive effort reduces antioxidant defences. | Mixed support: results depend on sex and supplementation status. | Association between oxidative damage and reproductive effort varies enormously across the sexes. | |
| High reproductive effort increases oxidative damage. | ||||
| Antioxidant supplementation improves reproductive effort. | Weak positive effect but not significant. | |||
4.1. The Effect of Nutrient Intake on Life-History Traits
4.2. Testing the Oxidative Stress Theory of Aging
4.3. Testing a Role for Oxidative Damage in Life-History Trade-Offs
5. Conclusions
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Partridge, L. The new biology of ageing. Philos. Trans. R. Soc. Lond. Ser. B Biol. Sci. 2010, 365, 147–154. [Google Scholar] [CrossRef] [PubMed]
- Harman, D. A theory based on free radical and radiation chemistry. J. Gerontol. 1956, 11, 298–300. [Google Scholar] [CrossRef] [PubMed]
- Barja, G. Mitochondrial oxygen consumption and reactive oxygen species production are independently modulated: Implications for aging studies. Rejuv. Res. 2007, 10, 215–224. [Google Scholar] [CrossRef] [PubMed]
- D’Autreaux, B.; Toledano, M.B. ROS as signalling molecules: Mechanisms that generate specificity in ROS homeostasis. Nat. Rev. Mol. Cell Biol. 2007, 8, 813–824. [Google Scholar] [CrossRef] [PubMed]
- Dalle-Donne, I.; Rossi, R.; Giustarini, D.; Milzani, A.; Colombo, R. Protein carbonyl groups as biomarkers of oxidative stress. Clin. Chim. Acta 2003, 329, 23–38. [Google Scholar] [CrossRef]
- Finkel, T.; Holbrook, N.J. Oxidants, oxidative stress and the biology of ageing. Nature 2000, 408, 239–247. [Google Scholar] [CrossRef] [PubMed]
- Alper, S.; Bronikowski, A.M.; Harper, J.M. Comparative cellular biogerontology: Where do we stand? Exp. Gerontol. 2015, 71, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Robert, K.A.; Bronikowski, A.M. Evolution of senescence in nature: Physiological evolution in populations of garter snake with divergent life histories. Am. Nat. 2010, 175, 147–159. [Google Scholar] [CrossRef] [PubMed]
- Andziak, B.; O’Connor, T.P.; Qi, W.; de Waal, E.M.; Pierce, A.; Chaudhuri, A.R.; van Remmen, H.; Buffenstein, R. High oxidative damage levels in the longest-living rodent, the naked mole-rat. Aging Cell 2006, 5, 463–471. [Google Scholar] [CrossRef] [PubMed]
- Head, E.; Liu, J.; Hagen, T.M.; Muggenburg, B.A.; Milgram, N.W.; Ames, B.N.; Cotman, C.W. Oxidative damage increases with age in a canine model of human brain aging. J. Neurochem. 2002, 82, 375–381. [Google Scholar] [CrossRef] [PubMed]
- Archer, C.R.; Sakaluk, S.K.; Selman, C.; Royle, N.J.; Hunt, J. Oxidative stress and the evolution of sex differences in life span and ageing in the decorated cricket, Gryllodes sigillatus. Evolution 2013, 67, 620–634. [Google Scholar] [CrossRef] [PubMed]
- Costantini, D.; Casagrande, S.; de Filippis, S.; Brambilla, G.; Fanfani, A.; Tagliavini, J.; Dell’Omo, G. Correlates of oxidative stress in wild kestrel nestlings (Falco tinnunculus). J. Comp. Physiol. B 2006, 176, 329–337. [Google Scholar] [CrossRef] [PubMed]
- Jang, Y.C.; Remmen, H.V. The mitochondrial theory of aging: Insight from transgenic and knockout mouse models. Exp. Gerontol. 2009, 44, 256–260. [Google Scholar] [CrossRef] [PubMed]
- Speakman, J.R.; Selman, C. The free-radical damage theory: Accumulating evidence against a simple link of oxidative stress to ageing and lifespan. BioEssays 2011, 33, 255–259. [Google Scholar] [CrossRef] [PubMed]
- Dowling, D.K.; Simmons, L.W. Reactive oxygen species as universal constraints in life-history evolution. Proc. R. Soc. B Biol. Sci. 2009, 276, 1737–1745. [Google Scholar] [CrossRef] [PubMed]
- Monaghan, P.; Metcalfe, N.B.; Torres, R. Oxidative stress as a mediator of life history trade-offs: Mechanisms, measurements and interpretation. Ecol. Lett. 2009, 12, 75–92. [Google Scholar] [CrossRef] [PubMed]
- Metcalfe, N.B.; Monaghan, P. Does reproduction cause oxidative stress? An open question. Trends Ecol. Evol. 2013, 28, 347–350. [Google Scholar] [CrossRef] [PubMed]
- Blount, J.D.; Vitikainen, E.I.; Stott, I.; Cant, M.A. Oxidative shielding and the cost of reproduction. Biol. Rev. 2015. [Google Scholar] [CrossRef] [PubMed]
- Hunt, J.; Brooks, R.; Jennions, M.D.; Smith, M.J.; Bentsen, C.L.; Bussiere, L.F. High-quality male field crickets invest heavily in sexual display but die young. Nature 2004, 432, 1024–1027. [Google Scholar] [CrossRef] [PubMed]
- Harshman, L.G.; Zera, A.J. The cost of reproduction: The devil in the details. Trends Ecol. Evol. 2007, 22, 80–86. [Google Scholar] [CrossRef] [PubMed]
- Kavanagh, M.W. The efficiency of sound production in two cricket species, Gryllotalpa australis and Teleogryllus commodus (Orthoptera: Grylloidea). J. Exp. Biol. 1987, 130, 107–119. [Google Scholar]
- Hulbert, A.J.; Pamplona, R.; Buffenstein, R.; Buttemer, W.A. Life and death: Metabolic rate, membrane composition, and life span of animals. Physiol. Rev. 2007, 87, 1175–1213. [Google Scholar] [CrossRef] [PubMed]
- Blount, J.D.; Metcalfe, N.B.; Birkhead, T.R.; Surai, P.F. Carotenoid modulation of immune function and sexual attractiveness in zebra finches. Science 2003, 300, 125–127. [Google Scholar] [CrossRef] [PubMed]
- Rey, B.; Pélisson, P.-F.; Bel-Venner, M.-C.; Voituron, Y.; Venner, S. Revisiting the link between breeding effort and oxidative balance through field evaluation of two sympatric sibling insect species. Evolution 2015, 69, 815–822. [Google Scholar] [CrossRef] [PubMed]
- Beaulieu, M.; Geiger, R.E.; Reim, E.; Zielke, L.; Fischer, K. Reproduction alters oxidative status when it is traded-off against longevity. Evolution 2015, 69, 1786–1796. [Google Scholar] [CrossRef] [PubMed]
- Speakman, J.R.; Garratt, M. Oxidative stress as a cost of reproduction: Beyond the simplistic trade-off model. Bioessays 2014, 36, 93–106. [Google Scholar] [CrossRef] [PubMed]
- Bonduriansky, R.; Maklakov, A.; Zajitschek, F.; Brooks, R. Sexual selection, sexual conflict and the evolution of ageing and life span. Funct. Ecol. 2008, 22, 443–453. [Google Scholar] [CrossRef]
- Trivers, R. Parental investment and sexual selection. In Sexual Selection & the Descent of Man; Aldine de Gruyter: New York, NY, USA, 1972; pp. 136–179. [Google Scholar]
- Mysterud, A.; Langvatn, R.; Stenseth, N.C. Patterns of reproductive effort in male ungulates. J. Zool. 2004, 264, 209–215. [Google Scholar] [CrossRef]
- Blank, R.H.; Olson, M.H.; Cox, N.R.; Bell, D.S. Black field cricket (Teleogryllus commodus) food preferences between sixteen pasture species. N. Zeal. J. Agric. Res. 1980, 23, 409–415. [Google Scholar] [CrossRef]
- Archer, C.R.; Hunt, J. Understanding the link between sexual selection, sexual conflict and aging using crickets as a model. Exp. Gerontol. 2015, 71, 4–13. [Google Scholar] [CrossRef] [PubMed]
- Bentsen, C.L.; Hunt, J.; Jennions, M.D.; Brooks, R. Complex multivariate sexual selection on male acoustic signalling in a wild population of Teleogryllus commodus. Am. Nat. 2006, 167, E102–E116. [Google Scholar] [CrossRef] [PubMed]
- Maklakov, A.A.; Simpson, S.J.; Zajitschek, F.; Hall, M.D.; Dessmann, J.; Clissold, F.; Raubenheimer, D.; Bonduriansky, R.; Brooks, R.C. Sex-specific fitness effects of nutrient intake on reproduction and lifespan. Curr. Biol. 2008, 18, 1062–1066. [Google Scholar] [CrossRef] [PubMed]
- Archer, C.R.; University of Exeter, UK. Unpublished work. 2008.
- Head, M.L.; Hunt, J.; Jennions, M.D.; Brooks, R. The indirect benefits of mating with attractive males outweigh the direct costs. PLoS Biol. 2005, 3, e33. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Simpson, S.J.; Raubenheimer, D. The Nature of Nutrition: A Unifying Framework from Animal Adaptation to Human Obesity; Princeton University Press: Princeton, NJ, USA, 2012. [Google Scholar]
- Simpson, S.; Abisgold, J. Compensation by locusts for changes in dietary nutrients: Behavioural mechanisms. Physiol. Entomol. 1985, 10, 443–452. [Google Scholar] [CrossRef]
- Jedlička, P.; Cvačka, J.; Sláma, K. Juvenile hormone-stimulated synthesis of acyl-glycerols and vitamin E in female accessory sexual glands of the fire bug, Pyrrhocoris apterus L. Arch. Insect Biochem. Physiol. 2009, 72, 48–59. [Google Scholar] [CrossRef] [PubMed]
- Hunt, J.; Jennions, M.D.; Spyrou, N.; Brooks, R. Artificial selection on male longevity influences age-dependent reproductive effort in the black field cricket Teleogryllus commodus. Am. Nat. 2006, 168, E72–E86. [Google Scholar] [CrossRef] [PubMed]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Lande, R.; Arnold, S.J. The measurement of selection on correlated characters. Evolution 1983, 1210–1226. [Google Scholar] [CrossRef]
- Fields Development Team. Fields: Tools for Spatial Data; National Center for Atmospheric Research: Boulder, CO, USA, 2006. [Google Scholar]
- Green, P.J.; Silverman, B.W. Nonparametric Regression and Generalized Linear Models: A Roughness Penalty Approach; CRC Press: Boca Raton, FL, USA, 1993. [Google Scholar]
- Manly, B.F.J. Randomization, Bootstrap and Monte Carlo Methods in Biology; Chapman and Hall: London, UK, 1997. [Google Scholar]
- Draper, N.R.; John, J.A. Response-surface designs for quantitative and qualitative variables. Technometrics 1988, 30, 423–428. [Google Scholar] [CrossRef]
- Bowerman, B.L.; O’Connell, R.T. Linear Statistical Models: An Applied Approach; PWS-Kent Publishing: Boston, MA, USA, 1990. [Google Scholar]
- Chenoweth, S.F.; Blows, M.W. Contrasting mutual sexual selection on homologous signal traits in Drosophila serrata. Am. Nat. 2005, 165, 281–289. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Howes, R.M. The Free Radical Fantasy. Ann. N. Y. Acad. Sci. 2006, 1067, 22–26. [Google Scholar] [CrossRef] [PubMed]
- Isaksson, C.; Sheldon, B.C.; Uller, T. The challenges of integrating oxidative stress into life-history biology. BioScience 2011, 61, 194–202. [Google Scholar] [CrossRef]
- Bunning, H.; Rapkin, J.; Belcher, L.; Archer, C.R.; Jensen, K.; Hunt, J. Protein and carbohydrate intake influence sperm number and fertility in male cockroaches, but not sperm viability. Proc. R. Soc. Lond. B Biol. Sci. 2015, 282. [Google Scholar] [CrossRef] [PubMed]
- Povey, S.; Cotter, S.C.; Simpson, S.J.; Wilson, K. Dynamics of macronutrient self-medication and illness-induced anorexia in virally infected insects. J. Anim. Ecol. 2014, 83, 245–255. [Google Scholar] [CrossRef] [PubMed]
- Poiani, A.; Wilks, C. Sexually transmitted diseases: A possible cost of promiscuity in birds? Auk 2000, 117, 1061–1065. [Google Scholar] [CrossRef]
- Simpson, S.J.; Raubenheimer, D. Macronutrient balance and lifespan. Aging 2009, 1, 875–880. [Google Scholar] [PubMed]
- Sanz, A.; Caro, P.; Barja, G. Protein restriction without strong caloric restriction decreases mitochondrial oxygen radical production and oxidative DNA damage in rat liver. J. Bioenerg. Biomembr. 2004, 36, 545–552. [Google Scholar] [CrossRef] [PubMed]
- Ayala, V.; Naudí, A.; Sanz, A.; Caro, P.; Portero-Otin, M.; Barja, G.; Pamplona, R. Dietary protein restriction decreases oxidative protein damage, peroxidizability index, and mitochondrial complex I content in rat liver. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2007, 62, 352–360. [Google Scholar] [CrossRef]
- Petzke, K.J.; Elsner, A.; Proll, J.; Thielecke, F.; Metges, C.C. Long-term high protein intake does not increase oxidative stress in rats. J. Nutr. 2000, 130, 2889–2896. [Google Scholar] [PubMed]
- Hands, S.L.; Proud, C.G.; Wyttenbach, A. mTOR’s role in ageing: Protein synthesis or autophagy? Aging 2009, 1, 586–597. [Google Scholar] [PubMed]
- Canavoso, L.E.; Jouni, Z.E.; Karnas, K.J.; Pennington, J.E.; Wells, M.A. Fat metabolism in insects. Annu. Rev. Nutr. 2001, 21, 23–46. [Google Scholar] [CrossRef] [PubMed]
- House, H. Effects of vitamins E and A on growth and development, and the necessity of vitamin E for reproduction in the parasitoid Agria affinis (Fallen) (Diptera, Sarcophagidae). J. Insect Physiol. 1966, 12, 409–417. [Google Scholar] [CrossRef]
- Niki, E. Assessment of antioxidant capacity in vitro and in vivo. Free Radic. Biol. Med. 2010, 49, 503–515. [Google Scholar] [CrossRef] [PubMed]
- Archer, C.R.; Köhler, A.; Pirk, C.W.; Oosthuizen, V.; Apostolides, Z.; Nicolson, S.W. Antioxidant supplementation can reduce the survival costs of excess amino acid intake in honeybees. J. Insect Physiol. 2014, 71, 78–86. [Google Scholar] [CrossRef] [PubMed]
- Vagni, S.; Saccone, F.; Pinotti, L.; Baldi, A. Vitamin E bioavailability: Past and present insights. Food Nutr. Sci. 2011. [Google Scholar] [CrossRef]
- Salin, K.; Auer, S.K.; Rey, B.; Selman, C.; Metcalfe, N.B. Variation in the link between oxygen consumption and ATP production, and its relevance for animal performance. Proc. R. Soc. B Biol. Sci. 2015, 282. [Google Scholar] [CrossRef] [PubMed]
- Okada, K.; Pitchers, W.R.; Sharma, M.D.; Hunt, J.; Hosken, D.J. Longevity, calling effort, and metabolic rate in two populations of cricket. Behav. Ecol. Sociobiol. 2011, 65, 1773–1778. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Archer, C.R.; Hempenstall, S.; Royle, N.J.; Selman, C.; Willis, S.; Rapkin, J.; Blount, J.D.; Hunt, J. Testing the Effects of DL-Alpha-Tocopherol Supplementation on Oxidative Damage, Total Antioxidant Protection and the Sex-Specific Responses of Reproductive Effort and Lifespan to Dietary Manipulation in Australian Field Crickets (Teleogryllus commodus). Antioxidants 2015, 4, 768-792. https://doi.org/10.3390/antiox4040768
Archer CR, Hempenstall S, Royle NJ, Selman C, Willis S, Rapkin J, Blount JD, Hunt J. Testing the Effects of DL-Alpha-Tocopherol Supplementation on Oxidative Damage, Total Antioxidant Protection and the Sex-Specific Responses of Reproductive Effort and Lifespan to Dietary Manipulation in Australian Field Crickets (Teleogryllus commodus). Antioxidants. 2015; 4(4):768-792. https://doi.org/10.3390/antiox4040768
Chicago/Turabian StyleArcher, C. Ruth, Sarah Hempenstall, Nick J. Royle, Colin Selman, Sheridan Willis, James Rapkin, Jon D. Blount, and John Hunt. 2015. "Testing the Effects of DL-Alpha-Tocopherol Supplementation on Oxidative Damage, Total Antioxidant Protection and the Sex-Specific Responses of Reproductive Effort and Lifespan to Dietary Manipulation in Australian Field Crickets (Teleogryllus commodus)" Antioxidants 4, no. 4: 768-792. https://doi.org/10.3390/antiox4040768





