Effect of Dielectric Properties of a Solvent-Water Mixture Used in Microwave-Assisted Extraction of Antioxidants from Potato Peels
Abstract
:1. Introduction
2. Experimental Section
2.1. Materials
2.2. Equipment and Apparatus

2.3. MAE Extraction
2.4. Preparation of Potato Peel Extracts
2.5. Determination of Total Phenolic Compounds
2.6. Statistical Analysis
3. Results and Discussion
3.1. Effect of Temperature on Dielectric Properties of Methanol-Water Mixtures
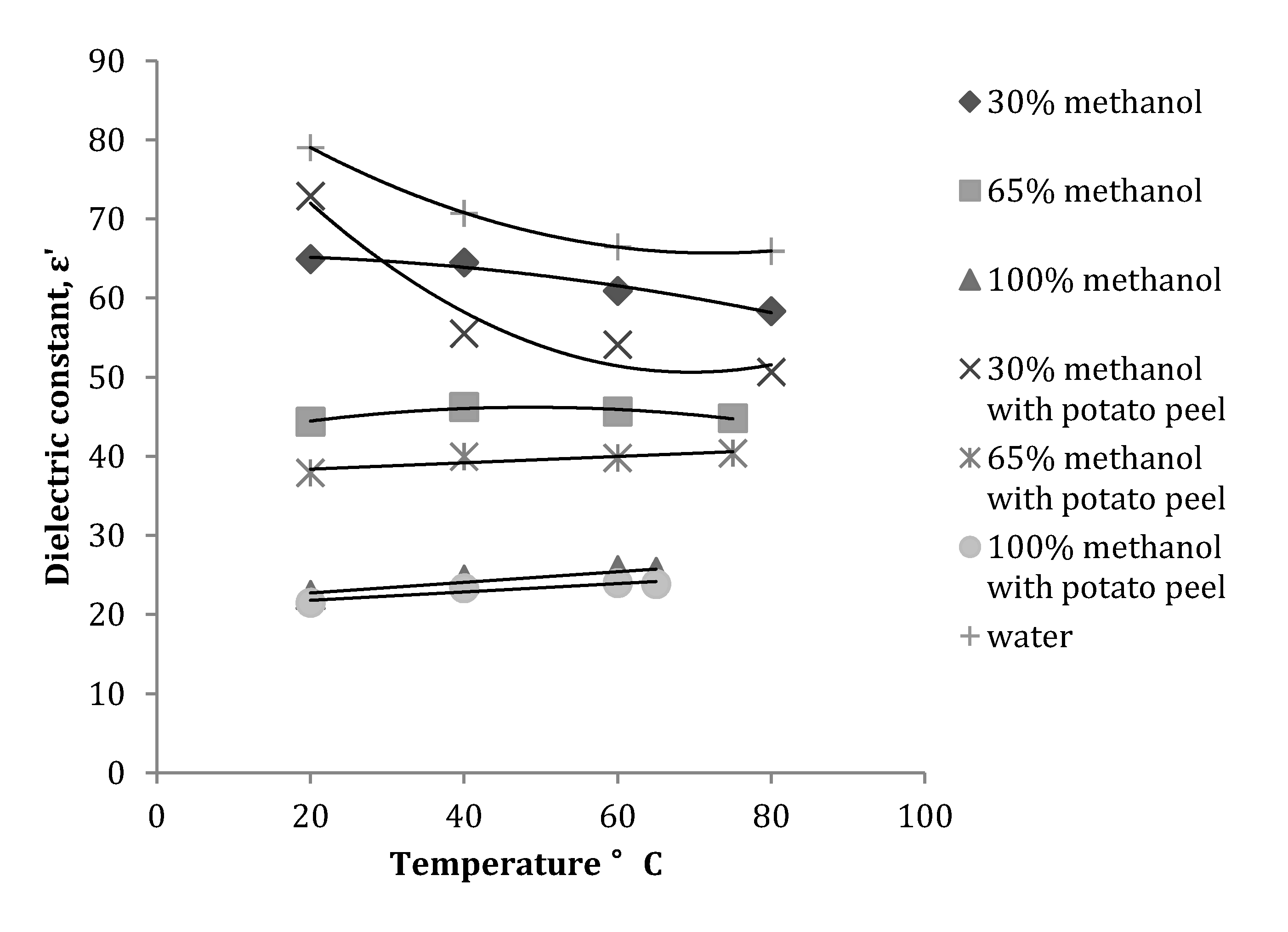

| Dielectric Properties | Plant material | Frequency (MHz) | Equation obtained | R2 |
|---|---|---|---|---|
| ε′ | Potato peel | 2450 | 78.65 − 0.51 Met − 0.11 Temp | 0.91 |
| ε″ | Potato peel | 2450 | 12.81 − 0.07 Temp | 0.26 |
| ε′ | Without peel | 2450 | 80.39 − 0.54 Met | 0.98 |
| ε″ | Without peel | 2450 | 19.48 − 0.20 Temp | 0.88 |

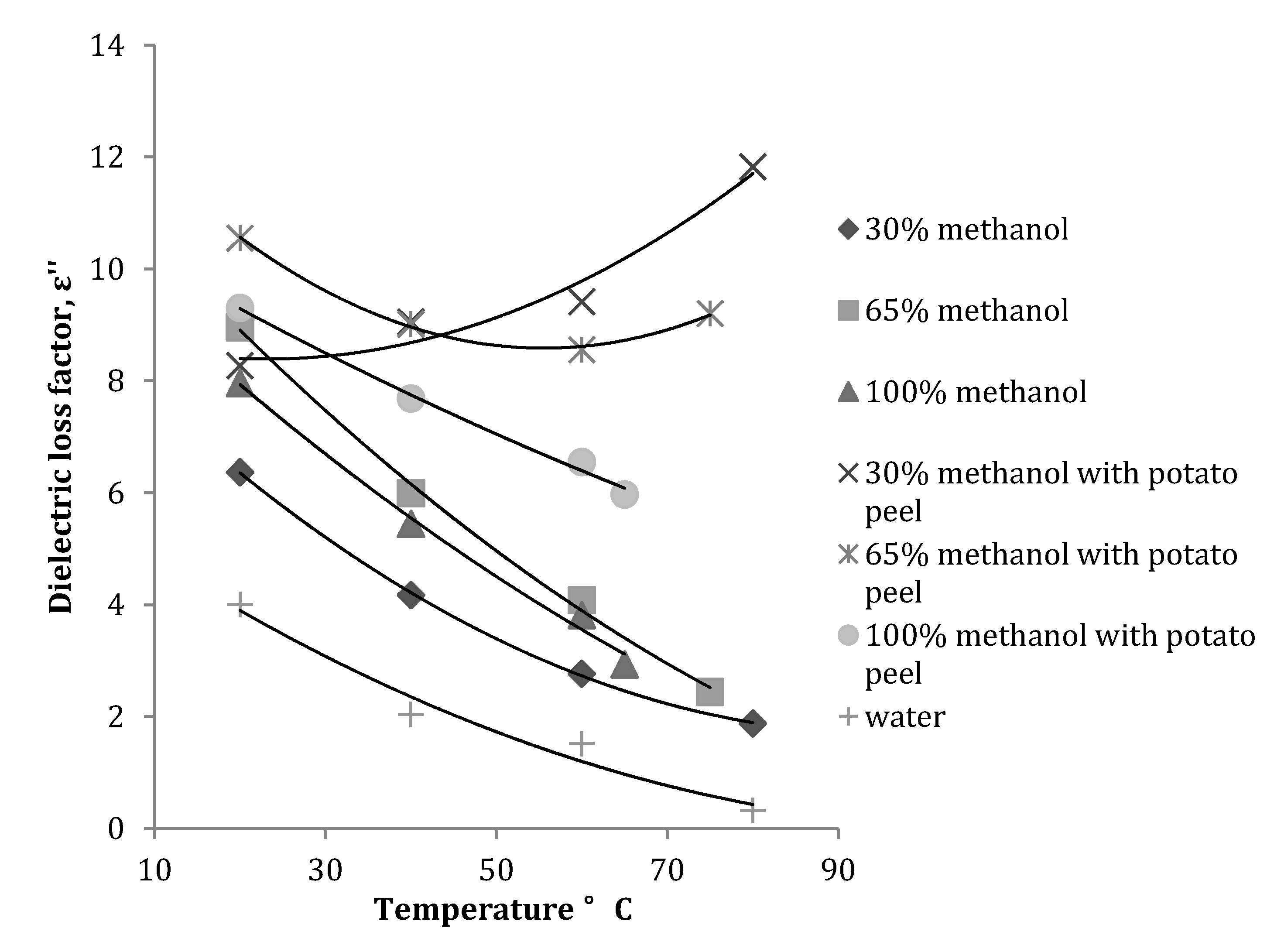
| Dielectric Properties | Plant material | Frequency (MHz) | Equation obtained | R2 |
|---|---|---|---|---|
| ε′ | Potato peel | 915 | 80.38 − 0.44 Met − 0.15 Temp | 0.97 |
| ε″ | Potato peel | 915 | 11.13 − 0.03 Met | 0.36 |
| ε′ | Without peel | 915 | 86.68 − 0.49 Met − 0.13 Temp | 0.98 |
| ε″ | Without peel | 915 | 8.60 + 0.012 Met − 0.096 Temp | 0.9 |
3.2. Effect of Temperature, Concentration and Presence of Plant Material on Dissipation Factor (tanδ)
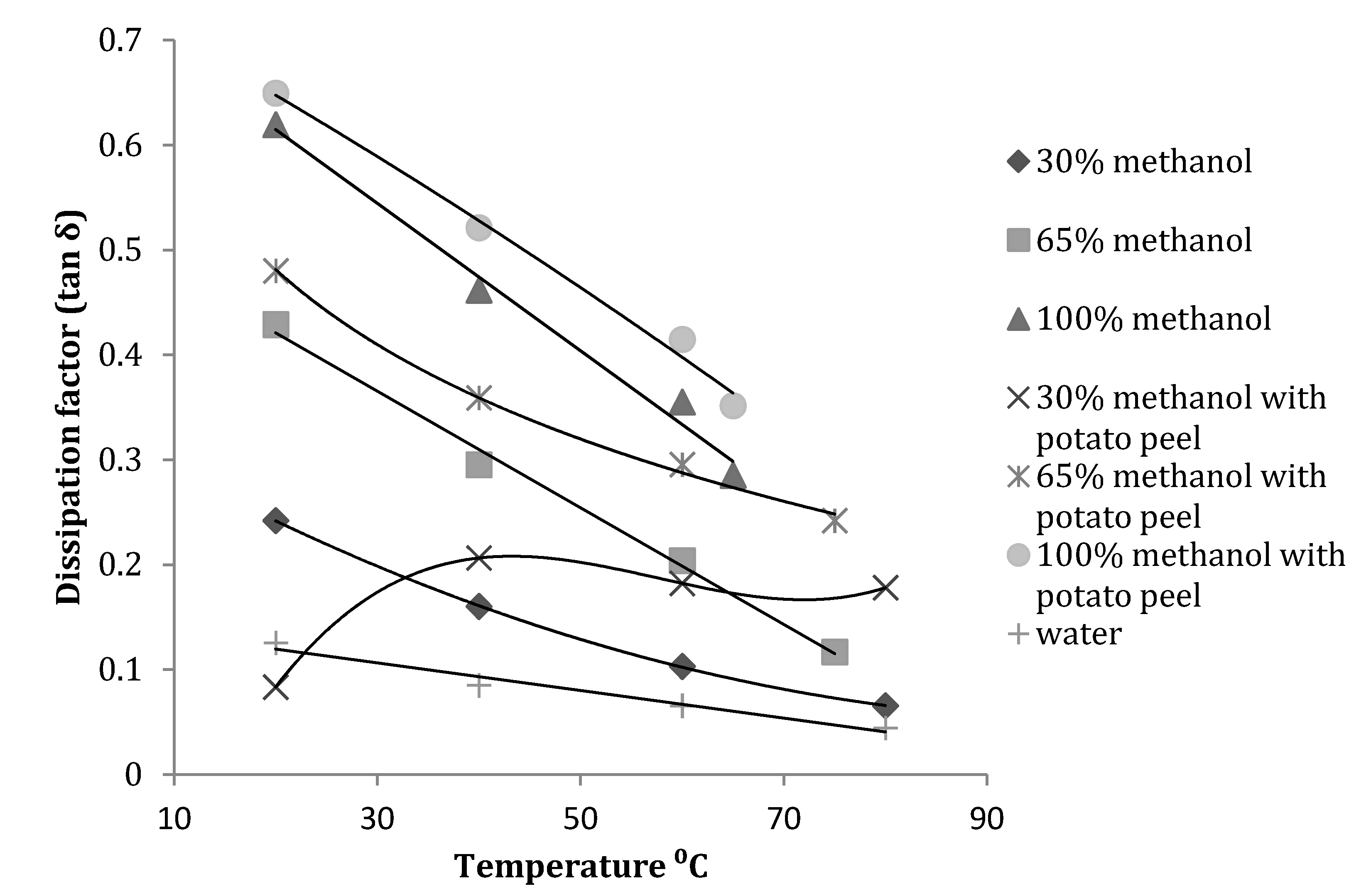

| Dissipation factor | Plant material | Frequency (MHz) | Equation obtained | R2 |
|---|---|---|---|---|
| tanδ | Potato peel | 915 | 0.12 + 0.00124 Met | 0.60 |
| tanδ | Without peel | 915 | 0.11 + 0.00144 Met − 0.002 Temp | 0.91 |
| tanδ | Potato peel | 2450 | 0.16 +0.005 Met − 0.003 Temp | 0.79 |
| tanδ | withoutpeel | 2450 | 0.26 + 0.004 Met − 0.005 Temp | 0.95 |
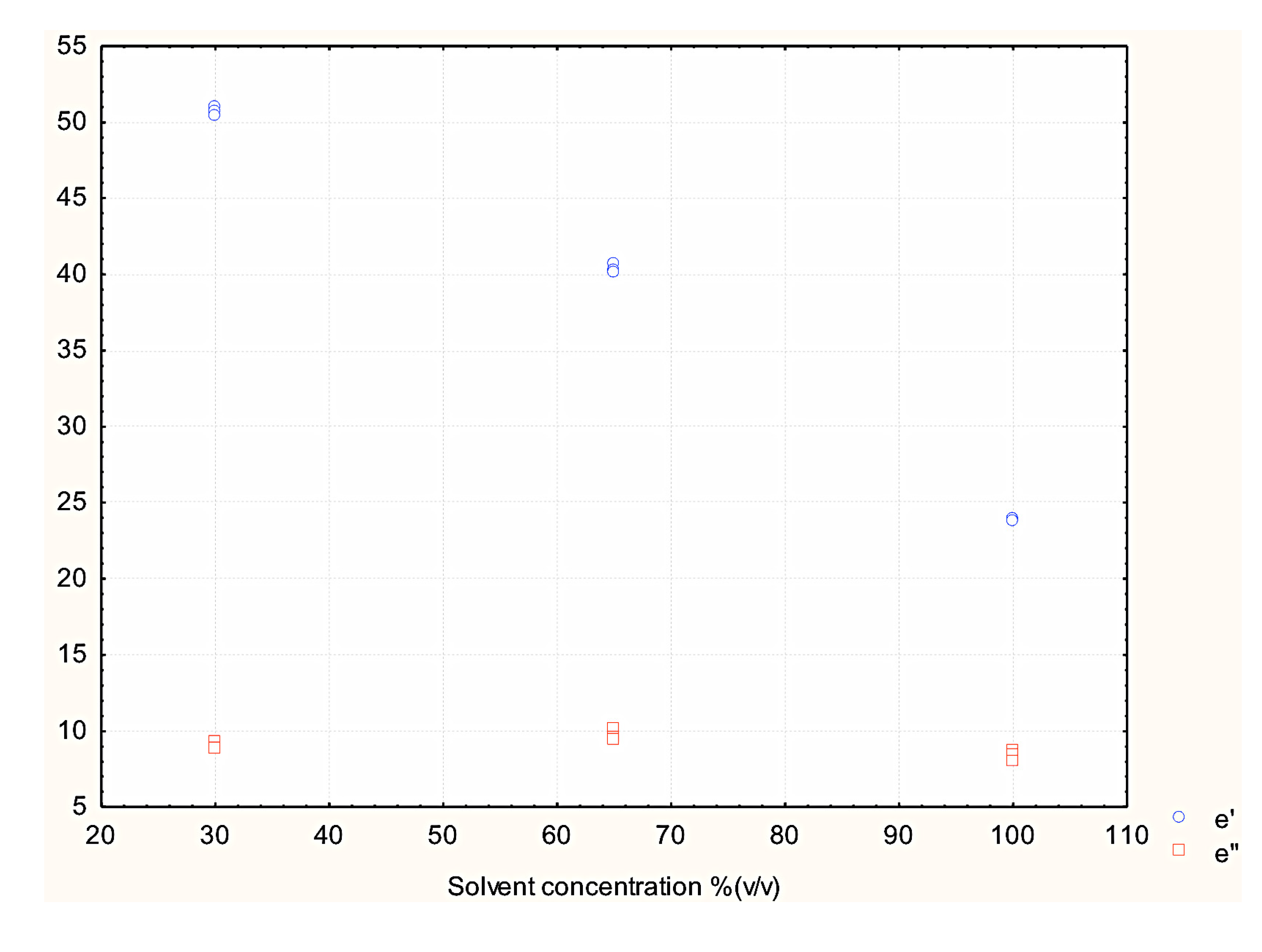
3.3. Effect of Dielectric Properties of Methanol Fraction and Time on MAE Extraction of Antioxidant
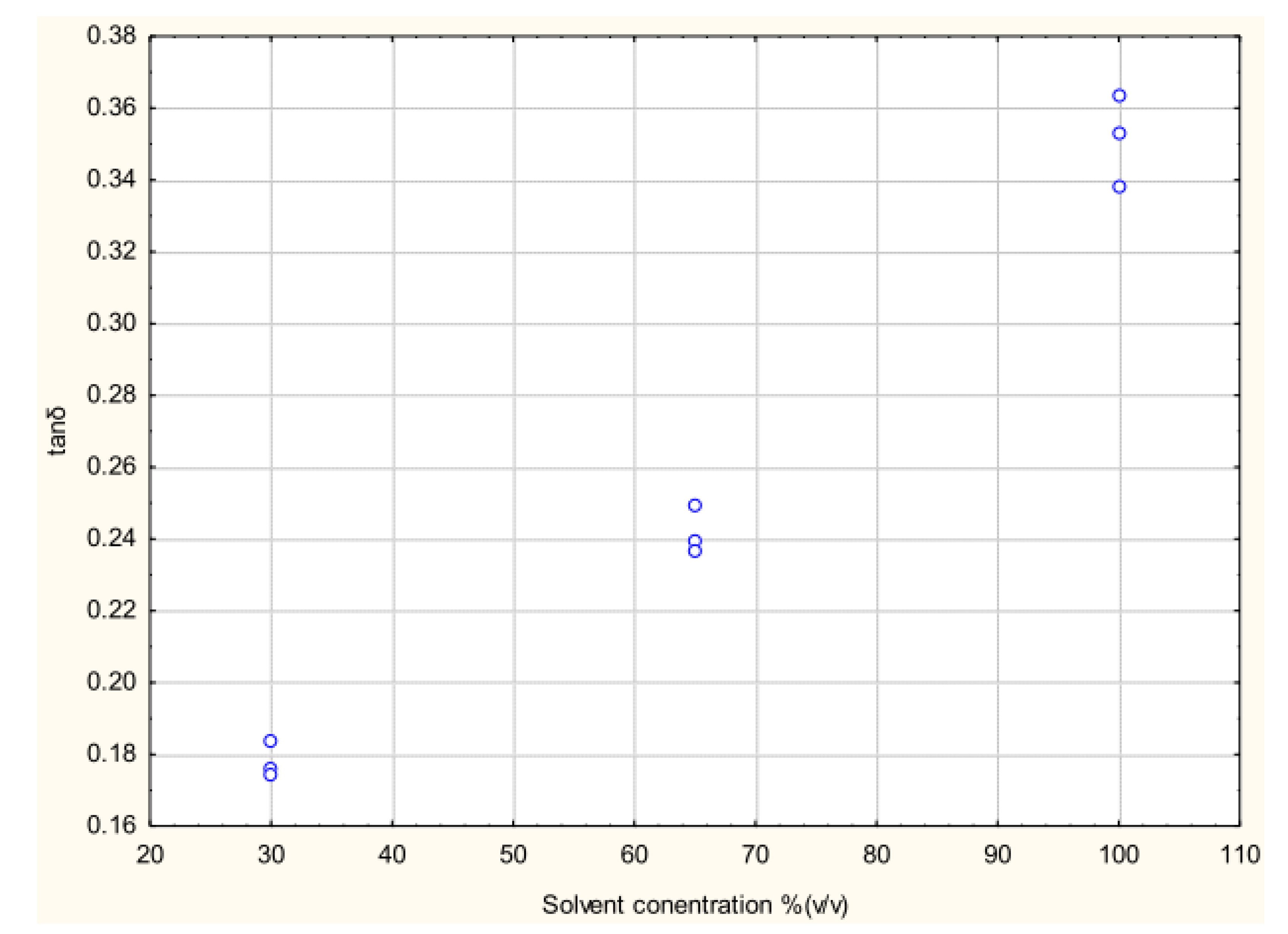
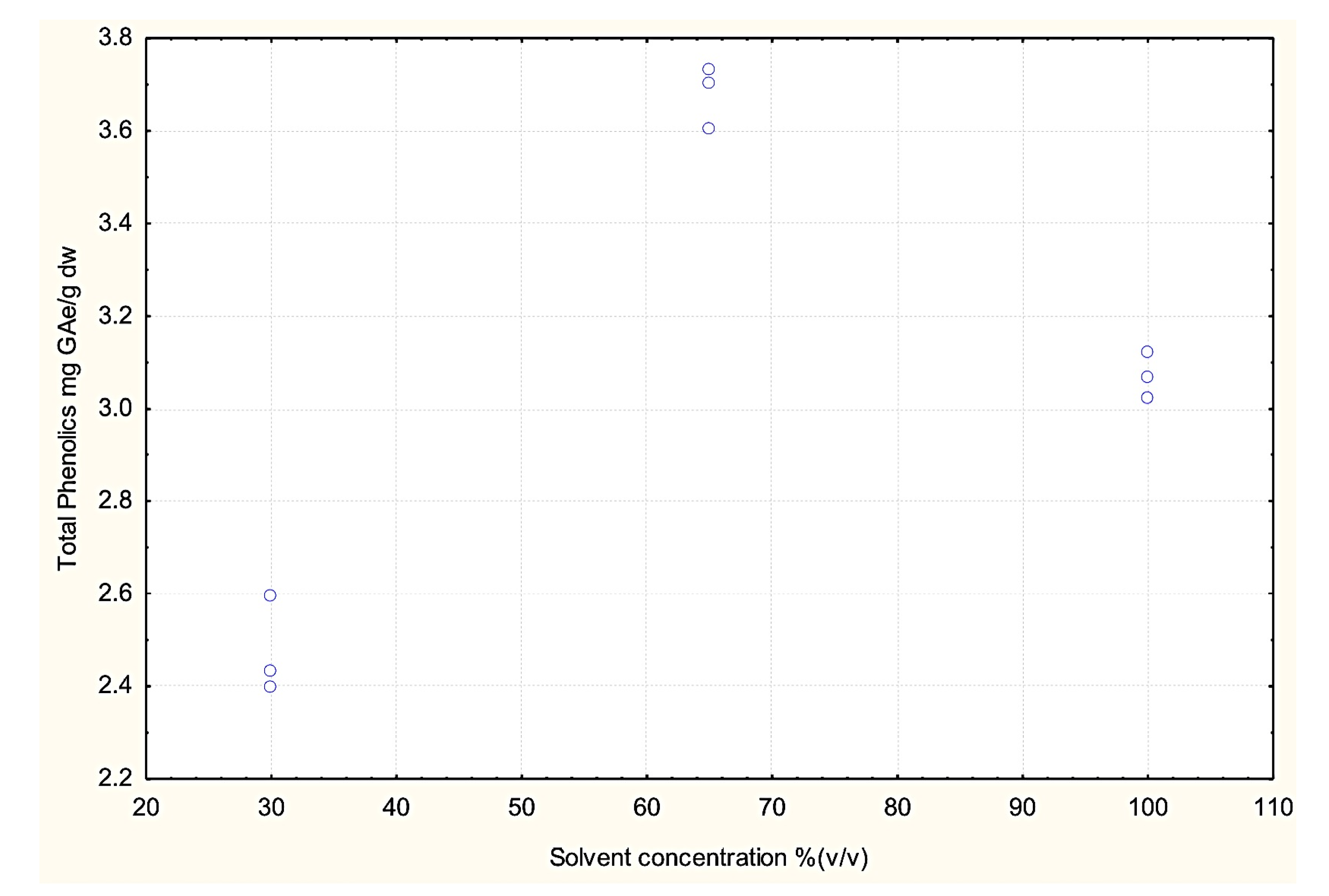
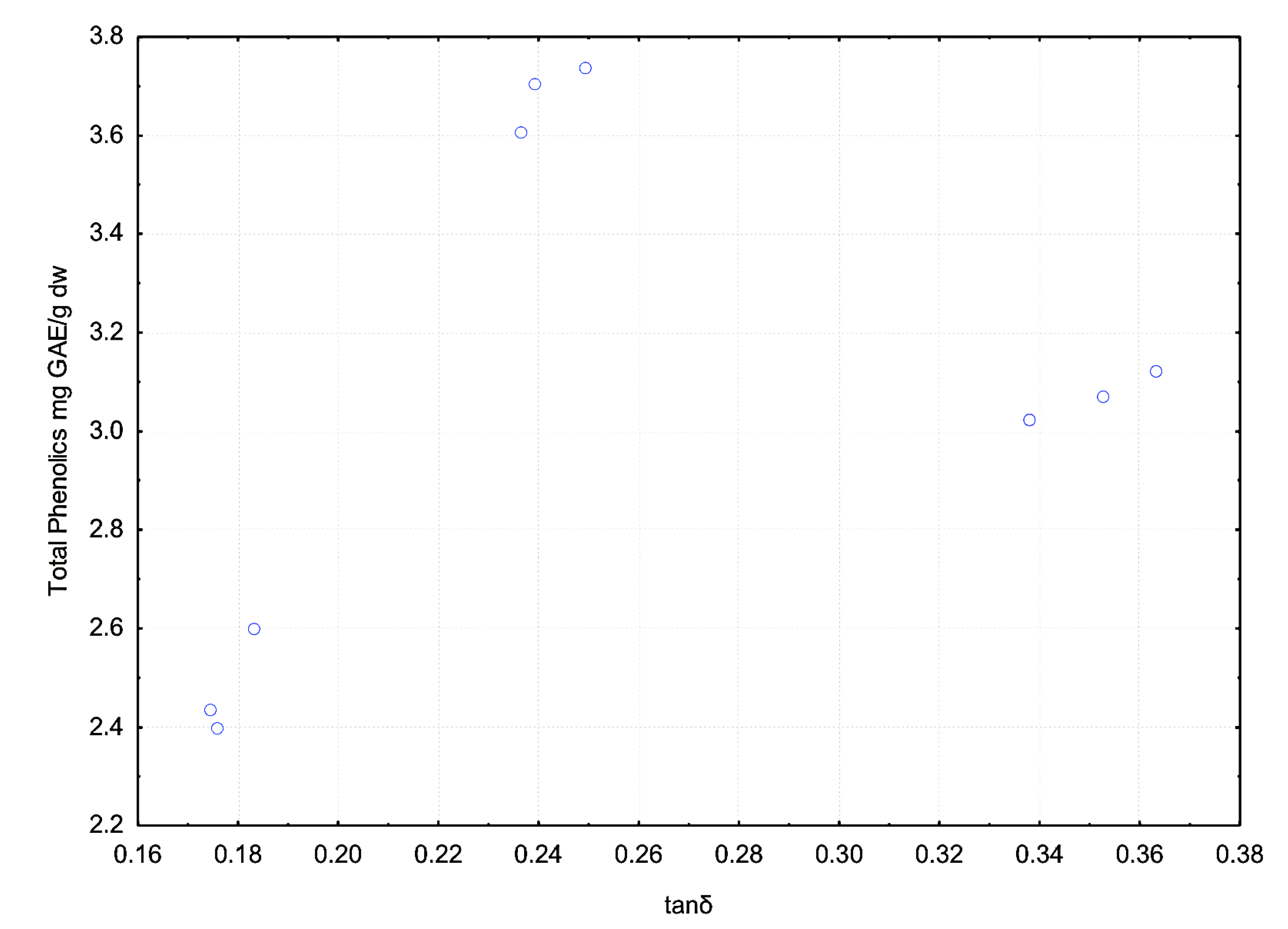

4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Al-Saikhan, M.S.; Howard, L.R.; Miller, J.C., Jr. Antioxidant activity and total phenolics in different genotypes of potato (Solanum tuberosum L.). J. Food Sci. 1995, 60, 341–343. [Google Scholar] [CrossRef]
- Singh, A.; Sabally, K.; Kubow, S.; Donnelly, D.J.; Gariepy, Y.; Orsat, V.; Raghavan, G.S.V. Microwave-Assisted Extraction of Phenolic Antioxidants from Potato Peels. Molecules 2011, 16, 2218–2232. [Google Scholar] [CrossRef]
- Duvernay, W.H.; Assad, J.M.; Sabliov, C.M.; Lima, M.; Xu, Z. Microwave extraction of antioxidant components from rice bran. Pharm. Eng. 2005, 25, 126–130. [Google Scholar]
- Tulasidas, T.N.; Raghavan, G.S.V.; van de Voort, F.; Girard, R. Dielectric properties of grapes and sugar solutions at 2.45 GHz. J. Microw. Power Electromagn. Energy 1995, 30, 117–123. [Google Scholar] [PubMed]
- Venkatesh, M.S.; Raghavan, G.S.V. An overview of microwave processing and dielectric properties of agri-food materials. Biosyst. Eng. 2004, 88, 1–18. [Google Scholar] [CrossRef]
- Bao, J.Z.; Swicord, M.L.; Davis, C.C. Microwave dielectric characterization of binary mixtures of water, methanol, and ethanol. J. Chem. Phys. 1996, 104, 4441–4450. [Google Scholar] [CrossRef]
- Barthel, J.; Bachhuber, K.; Buchner, R.; Hetzenauer, H. Dielectric spectra of some common solvents in the microwave region. Dipolar aprotic solvents and amides. Chem. Phys. Lett. 1990, 167, 62–66. [Google Scholar] [CrossRef]
- Lou, J.; Hatton, T.A.; Laibinis, P.E. Effective dielectric properties of solvent mixtures at microwave frequencies. J. Phys. Chem. A 1997, 101, 5262–5268. [Google Scholar] [CrossRef]
- Singh, A. Optimization of Microwave-assisted Extraction of Antioxidants from Potato Peels. Master’s Thesis, McGill University, Sainte-Anne-de-Bellevue, Quebec, Canada, 2010. [Google Scholar]
- Veggi, P.; Martinez, J.; Meireles, M.A. Fundamentals of Microwave Extraction. In Microwave-Assisted Extraction for Bioactive Compounds: Theory and Practice; Chemat, F., Cravotto, G., Eds.; Springer: New York, NY, USA, 2013; pp. 15–52. [Google Scholar]
- Alfaro, M.J.; Bélanger, J.M.R.; Padilla, F.C.; Paré, J.R.J. Influence of solvent, matrix dielectric properties, and applied power on the liquid-phase microwave-assisted processes (MAP™) extraction of ginger (Zingiber officinale). Food Res. Int. 2003, 36, 499–504. [Google Scholar] [CrossRef]
- Chen, T.; Sun, X.; Xiao, W.; Liu, X.; Zhang, W.; Ma, K.; Zhu, Y. Optimization of microwave-assisted extraction of solanesol from potato leaves and stems. Med. Chem. Res. 2009, 19, 732–742. [Google Scholar]
- Nemes, S.M.; Orsat, V. Microwave-Assisted Extraction of Secoisolariciresinol Diglucoside-Method Development. Food Bioprocess Technol. 2009, 4, 1219–1227. [Google Scholar] [CrossRef]
- Shah, S.; Richter, R.C.; Kingston, H.M.S. Microwave-assisted organic extraction and evaporation: An integrated approach. LC-GC N. Am. 2002, 20, 280–286. [Google Scholar]
- Williams, O.J.; Raghavan, V.G.S.; Orsat, V.; Dai, J. Microwave-Assisted Extraction of Capsaicinoids from Capsicum Species. In Presented at ASAE Annual International Meeting/CIGR XVth World Congress, Chicago, IL, USA, 28–31 July; 2002. [Google Scholar]
- Williams, O.J.; Raghavan, G.S.V.; Orsat, V.; Dai, J. Microwave-assisted extraction of capsaicinoids from capsicum fruit. J. Food Biochem. 2004, 28, 113–122. [Google Scholar] [CrossRef]
- Talebi, M.; Ghassempour, A.; Talebpour, Z.; Rassouli, A.; Dolatyari, L. Optimization of the extraction of paclitaxel from Taxus baccata L. by the use of microwave energy. J. Sep. Sci. 2004, 27, 1130–1136. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Singh, A.; Nair, G.R.; Liplap, P.; Gariepy, Y.; Orsat, V.; Raghavan, V. Effect of Dielectric Properties of a Solvent-Water Mixture Used in Microwave-Assisted Extraction of Antioxidants from Potato Peels. Antioxidants 2014, 3, 99-113. https://doi.org/10.3390/antiox3010099
Singh A, Nair GR, Liplap P, Gariepy Y, Orsat V, Raghavan V. Effect of Dielectric Properties of a Solvent-Water Mixture Used in Microwave-Assisted Extraction of Antioxidants from Potato Peels. Antioxidants. 2014; 3(1):99-113. https://doi.org/10.3390/antiox3010099
Chicago/Turabian StyleSingh, Ashutosh, Gopu Raveendran Nair, Pansa Liplap, Yvan Gariepy, Valerie Orsat, and Vijaya Raghavan. 2014. "Effect of Dielectric Properties of a Solvent-Water Mixture Used in Microwave-Assisted Extraction of Antioxidants from Potato Peels" Antioxidants 3, no. 1: 99-113. https://doi.org/10.3390/antiox3010099






