Antioxidant Activity and Phenolic Content of Streblus asper Leaves from Various Drying Methods
Abstract
:1. Introduction
2. Experimental Section
2.1. Chemicals
2.2. Collection of Plant Materials
2.3. Drying Methods
2.4. Samples Extractions
2.5. In Vitro Antioxidant Activity
Antioxidant Activity Assay
2.6. Total Phenolics Content
2.7. Total Flavonoids Content
2.8. Statistical Analysis
3. Results and Discussion
3.1. Radical Scavenging Assays (RSA)
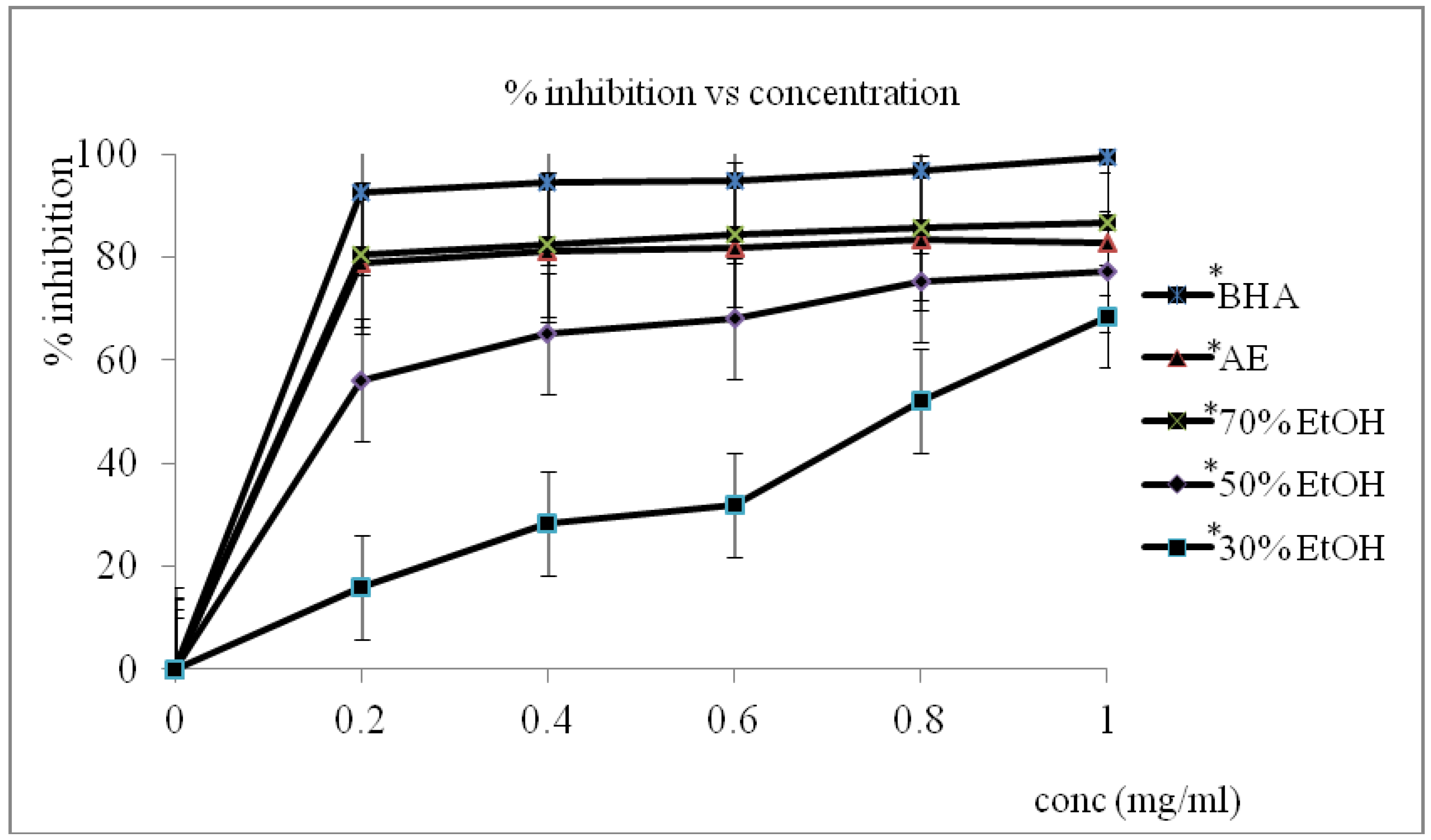
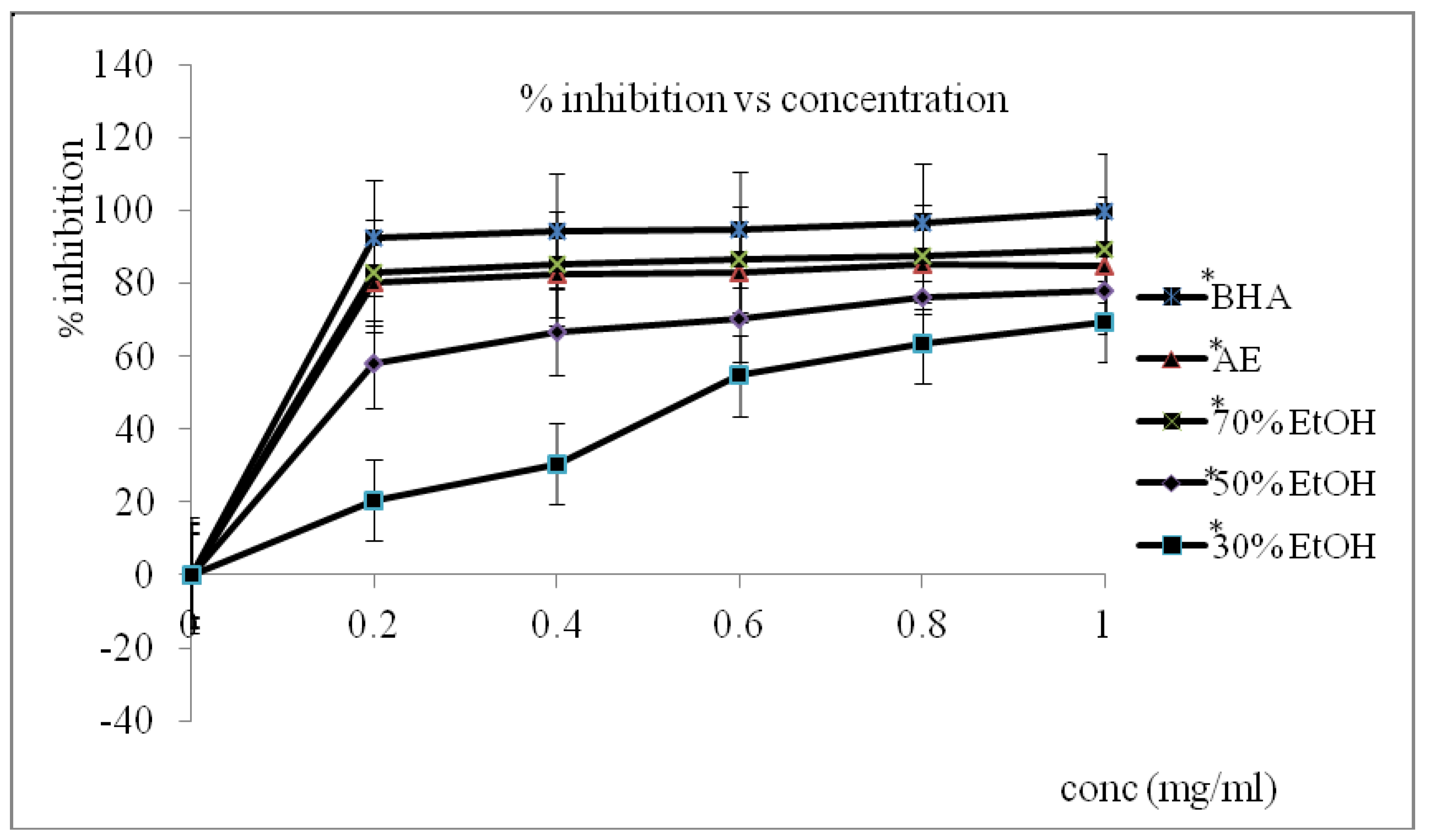
3.2. Total Phenolic Content
| Extraction solvent (mL) | Total phenolic content (mgGAE/g) | |
|---|---|---|
| * OD | * FD | |
| AE | 223.95 ± 0.04 a | 226.80 ± 0.03 b |
| 70% EtOH | 296.37 ± 0.05 a | 302.85 ± 0.03 b |
| 50% EtOH | 155.66 ± 0.02 a | 163.17 ± 0.01 b |
| 30% EtOH | 146.64 ± 0.03 a | 153.09 ± 0.02 b |
3.3. Total Flavonoids Content
| Extraction solvent (mL) | Total flavonoid content (mgQE/g) | |
|---|---|---|
| * OD | * FD | |
| AE | 10.26 ± 0.04 a | 15.38 ± 0.05 b |
| 70% EtOH | 22.28 ± 0.03 a | 22.70 ± 0.02 b |
| 50% EtOH | 19.16 ± 0.03 a | 19.74 ± 0.05 b |
| 30% EtOH | 8.87 ± 0.02 a | 9.92 ± 0.05 b |
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Wongkham, S.; Laupattarakasaem, P.; Pienthaweechai, K.; Areejitranusorn, P.; Wongkham, C.; Techanitiswad, T. Antimicrobial activity of Streblus asper leaf extract. Phytother. Res. 2001, 15, 119–121. [Google Scholar] [CrossRef]
- Taweechaisupapong, P.; Klanrit, P.; Singhara, S.; Pitiphat, W.; Wongklam, S. Inhibitory effect of Streblus asper leaf extract on adhesion of Candida albicans to dentene acrylic. J. Ethnopharmacol. 2006, 106, 414–417. [Google Scholar] [CrossRef]
- Rastogi, S.; Kulshreshtha, D.K.; Rawat, A.K.S. Streblus asper Lour. (Shakhotaka): A review of its chemical, pharmacological and ethnomedicinal properties. Evid. Based Complement. Altern. Med. 2006, 3, 217–222. [Google Scholar] [CrossRef]
- Gaitonde, B.B.; Vaz, A.X.; Patel, J.R. Chemical and pharmacological study of root bark of Streblus asper Linn. Indian J. Med. Sci. 1964, 18, 191–199. [Google Scholar] [PubMed]
- Zhang, X.S.; Wu, Z.Y.; Zao, Z.Y. The Plant Index of China; Scientific Press: Beijing, China, 1985; Volume 23, p. 38. [Google Scholar]
- Ebrahimzadeh, M.A.; Nabavi, S.F.; Nabavi, S.M. Antioxidant activities of methanol extract of Sambucus ebulus L. flower. J. Biol. Sci. 2009, 12, 447–450. [Google Scholar]
- Arulpriya, P.; Lalitha, P.; Hemalatha, S. Antioxidant activities of the extracts of the aerial roots of Pothos aurea. Der Chem. Sin. 2010, 1, 73–79. [Google Scholar]
- Koleva, I.I.; Van Beek, T.A.; Linssen, J.P.; deGroot, A.; Evstatieva, L.N. Screening of plant extracts for antioxidant activity: A comparative study on three testing methods. Phytochem. Anal. 2002, 13, 8–17. [Google Scholar] [CrossRef]
- Balestrin, L.; Dias, J.F.G.; Miguel, O.G.; Dall’Stella, D.S.G.; Miguel, M.D. Contribuição ao estudo fitoquímico de Dorstenia multiformis Miquel (Moraceae) comabordagem em atividade antioxidante. (in Portuguese). Rev. Bras. Farmacogn. 2008, 18, 230–235. [Google Scholar] [CrossRef]
- Stauth, D. Studies Force New View on Biology of Flavonoids. Available online: http://www.sciencedaily.com/releases/2007/03/070305140834.htm (accessed on 12 March 2012).
- Hossan, S.; Hanif, A.; Agarwala, B.; Sarwar, S.; Karim, M.; Taufiq-Ur-Rahman, M.; Jahan, R.; Rahmatullah, M. Traditional use of medicinal plants in bangladesh to treat urinary tract infections and sexually transmitted diseases. Ethnobot. Res. Appl. 2010, 8, 61–74. [Google Scholar]
- Velioglu, Y.S.; Mazza, G.; Gao, L.; Oomah, B.D. Antioxidant activity and total phenolics in selected fruits, vegetables, and grain products. J. Agric. Food Chem. 1998, 46, 4113–4117. [Google Scholar] [CrossRef]
- Kwon, Y.I.; Vattem, D.V.; Shetty, K. Evaluation of clonal herb of Lamiaceae species for management of diabetes and hypertension. Asia Pac. J. Clin. Nutr. 2006, 15, 107–118. [Google Scholar] [PubMed]
- Chew, Y.L.; Goh, J.K.; Lim, Y.Y. Assessment of in vitro antioxidant capacity and polyphenolic composition of selected medicinal herbs from Leguminosae family in Peninsular Malaysia. Food Chem. 2009, 119, 373–378. [Google Scholar]
- Sakanaka, S.; Tachibana, Y.; Okada, Y. Preparation and antioxidant properties of extracts of Japanese persimmon leaf tea (kakinoha-cha). Food Chem. 2005, 89, 569–575. [Google Scholar] [CrossRef]
- Lobo, V.; Patil, A.; Phatak, A.; Chandra, N. Free radicals, antioxidants and functional foods: Impact on human health. Pharmacogn. Rev. 2010, 4, 2968–2972. [Google Scholar]
- Lim, Y.Y.; Murtijaya, J. Antioxidant properties of Phyllanthus amarus extracts as affected by different drying methods. Food Sci. Technol. 2007, 40, 1664–1669. [Google Scholar] [CrossRef]
- Wong, P.Y.Y.; Kitts, D.D. Studies on the dual antioxidant and antibacterial properties of parsley (Petroselinum crispum) and cilantro (Coriandrum sativum) extracts. Food Chem. 2006, 97, 505–515. [Google Scholar] [CrossRef]
- Paul Raj, K.; Irudayaraj, V.; Johnson, M.; Patric Raja, D. Phytochemical and anti-bacterial activity of epidermal glands extract of Christella parasitica (L.) H. Lev. Asian Pac. J. Trop. Biomed. 2011, 1, 8–11. [Google Scholar] [CrossRef]
- Chan, E.W.C.; Lim, Y.Y.; Wong, S.K.; Lim, K.K.; Tan, S.P.; Lianto, F.S.; Yong, M.Y. Effects of different drying methods on the antioxidant properties of leaves and tea of ginger species. Food Chem. 2009, 113, 166–172. [Google Scholar] [CrossRef]
- Abascal, K.; Ganora, L.; Yarnell, E. The effect of freeze-drying and its implications for botanicalmedicine: A review. Phytother. Res. 2005, 19, 655–660. [Google Scholar] [CrossRef]
- Oktay, M.; Gulcin, I.; Kufrevioglu, O.I. Determination of in vitro antioxidant activity of fennel (Foeniculum vulgare) seed extracts. Food. Sci. Technol. 2003, 36, 263–271. [Google Scholar]
- Nsimba, R.Y.; Kikuzaki, H.; Konishi, Y. Antioxidant activity of various extracts fractions of Chenopodium quinoa and Amaranthus spp. seeds. Food Chem. 2008, 106, 760–766. [Google Scholar] [CrossRef]
- Zielisnki, H.; Koslowska, H. Antioxidant activity and total phenolics in selected cereal grains and their different morphological fractions. J. Agric. Food Chem. 2000, 48, 2008–2016. [Google Scholar] [CrossRef]
- Yan, S.W.; Asmah, R. Comparison of total phenolic contents and antioxidant activities of turmeric leaf, pandan leaf and torch ginger flower. Int. Food Res. J. 2010, 17, 417–423. [Google Scholar]
- Kaur, C.; Kapoor, H.C. Antioxidant activity and total phenolic content of some Asian vegetables. Int. J. Food Sci. Technol. 2002, 37, 153–161. [Google Scholar] [CrossRef]
- Salah, N.; Miller, N.J.; Pagange, G.; Tijburg, L.; Bolwell, G.P.; Rice, E.; Evans, C. Polyphenolic flavonols as scavengers of aqueous phase radicals as chain breaking antioxidant. Arch. Biochem. Biophys. 1995, 2, 339–346. [Google Scholar]
- Del-Rio, A.; Obdululio, B.G.; Casfillo, J.; Marin, F.G.; Ortuno, A. Uses and Properties of citrus flavonoids. J. Agric. Food Chem. 1997, 45, 4505–4515. [Google Scholar] [CrossRef]
- Okwu, D.E. Phytochemicals and Vitamin content of indigenous spices of Southeastern Nigeria. J. Sustain. Agric. Environ. 2004, 6, 30–37. [Google Scholar]
- Mbaebie, B.O.; Edeoga, H.O.; Afolayan, A.J. Phytochemical analysis and antioxidants activities of aqueous stem bark extract of Schotia latifolia Jacq. Asian Pac. J. Trop. Biomed. 2012, 2, 118–124. [Google Scholar] [CrossRef]
- Andrade, D.; Gil, C.; Breitenfeld, L.; Domingues, F.; Duarte, A.P. Bioactive extracts from Cistus ladanifer and Arbutus unedo L. Ind. Crops Prod. 2009, 30, 165–167. [Google Scholar] [CrossRef]
- Manga, H.M.; Brkic, D.; Marie, D.E.P.; Quetin-Leclercq, J. In vivo anti-inflammatory activity of Alchonear cordifolia. J. Ethnopharmacol. 2004, 92, 209–214. [Google Scholar] [CrossRef]
- Schuier, M.; Sies, H.; Billek, B.; Fischer, H. Cocoa-related flavonoids inhibit CFTR-mediated chloride transport across T84 human colon epithelia. J. Nutr. 2005, 135, 2320–2325. [Google Scholar] [PubMed]
- Runrawee, T. Samunpaithainaru; Ummarin printing and publishing: Bangkok, Thailand, 1999. [Google Scholar]
- Wongkham, S.; Pienthaweechai, K.; Laupattarakasaem, P.; Areejitranusorn, P.; Wongkham, C.; Jantamongkol, K.; Leungpailin, S.; Techanitisawad, T. Bactericidal Activity of Streblus asper. In Proceedings of the Second Thai–French Symposium on Plant Molecular Biology; INRA: Bordeaux, France, 1996; pp. 363–374. [Google Scholar]
- Peluso, M.R. Flavonoids attenuate cardiovascular disease, inhibit phosphodiesterase, and modulate lipid homeostasis in adipose tissue and liver. Exp. Biol. Med. 2006, 231, 1287–1299. [Google Scholar]
- Hertog, M.G.; Feskens, E.J.; Hollman, P.C. Dietary antioxidant flavonoids and risk of coronary heart disease: The zutphen elderly study. Lancet 1993, 342, 1007–1011. [Google Scholar] [CrossRef]
- Manzetti, A.R.; Reichstein, T. Die Glykoside von Streblus asper Lour. 3. Mitteilung Untersuchung der stark wasserloslichen Anteile. Helv. Chim. Acta 1964, 47, 2303–2309. [Google Scholar] [CrossRef]
- Tomaino, A.; Cimino, F.; Zimbalatti, V.; Venuti, V.; Sulfaro, V.; de Pasquale, A. Influence of heating on antioxidant activity and the chemical composition of some spice essential oils. Food Chem. 2005, 89, 549–554. [Google Scholar] [CrossRef]
- Mueller-Harvey, I. Analysis of hydrosable tannins. Anim. Feed Sci. Technol. 2001, 91, 3–20. [Google Scholar] [CrossRef]
- Lin Tein, M.; Durance, T.D.; Scaman, C.H. Characterization of vacuum microwave, air and freeze dried carrot slices. Food Res. Int. 1998, 3, 111–117. [Google Scholar]
- Maskan, M. Drying, shrinkage and rehydration characteristics of kiwifruits during hot air and microwave drying. J. Food Eng. 2001, 48, 177–182. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ibrahim, N.M.; Mat, I.; Lim, V.; Ahmad, R. Antioxidant Activity and Phenolic Content of Streblus asper Leaves from Various Drying Methods. Antioxidants 2013, 2, 156-166. https://doi.org/10.3390/antiox2030156
Ibrahim NM, Mat I, Lim V, Ahmad R. Antioxidant Activity and Phenolic Content of Streblus asper Leaves from Various Drying Methods. Antioxidants. 2013; 2(3):156-166. https://doi.org/10.3390/antiox2030156
Chicago/Turabian StyleIbrahim, Nor Mawarti, Ishak Mat, Vuanghao Lim, and Ruzita Ahmad. 2013. "Antioxidant Activity and Phenolic Content of Streblus asper Leaves from Various Drying Methods" Antioxidants 2, no. 3: 156-166. https://doi.org/10.3390/antiox2030156
APA StyleIbrahim, N. M., Mat, I., Lim, V., & Ahmad, R. (2013). Antioxidant Activity and Phenolic Content of Streblus asper Leaves from Various Drying Methods. Antioxidants, 2(3), 156-166. https://doi.org/10.3390/antiox2030156




