Effect of Chronic Administration of Nickel on Affective and Cognitive Behavior in Male and Female Rats: Possible Implication of Oxidative Stress Pathway
Abstract
:1. Introduction
2. Material & Methods
2.1. Animals and Experimental Conditions
- 1st group: rats control receiving daily an intraperitoneal injection of Nacl 0.9%.
- 2nd group: rats receiving daily a dose of 0.25 mg/kg of Ni.
- 3rd group: rats receiving daily a dose of 0.5 mg/kg of Ni.
- 4th group: rats receiving daily 1 mg/kg of Ni.
2.2. Neurobehavioral Tests
2.3. Anxiety-Like Measurement
2.4. Cognitive Measurement
2.5. Biochemical Examination
2.6. Statistical Analysis
3. Results
3.1. Effect of Nickel on the Levels of Anxiety-Like Measured in the OFT
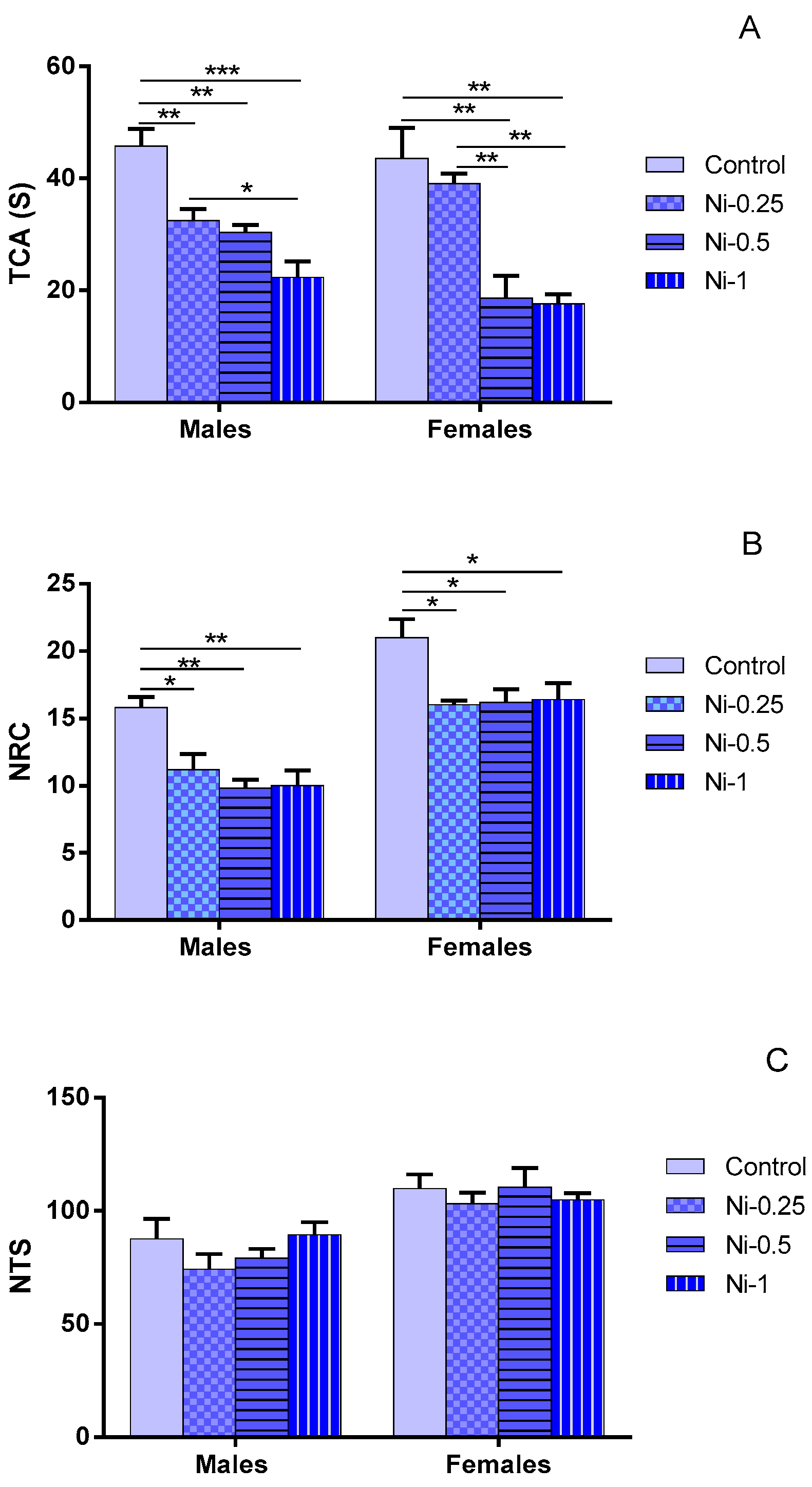
3.1.1. Time Spent in the Central Area (TCA)
3.1.2. Number of Returns to the Center (NRC)
3.1.3. Number of Total Squares (NTS)
3.2. Effect of Ni on Anxiety Levels Measured in Elevated Plus Maze Test (EPM)
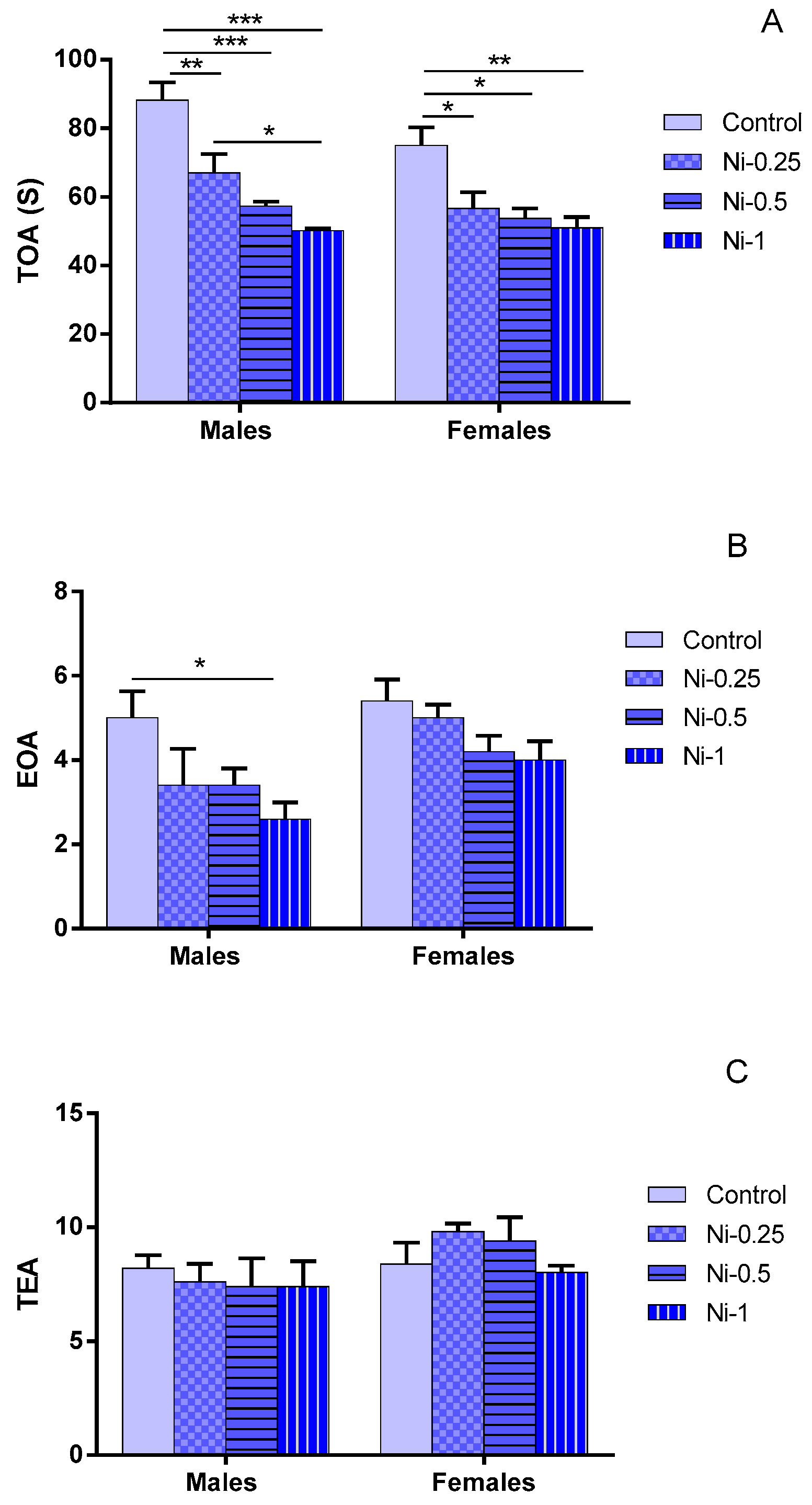
3.2.1. Time Spent in Open Arms (TOA)
3.2.2. Entry to Open Arms (EOA)
3.2.3. Total Entries in Arms (TEA)
3.3. Effect of Nickel on Depressive-Like Performances Measured by Forced Swimming Test (FST)
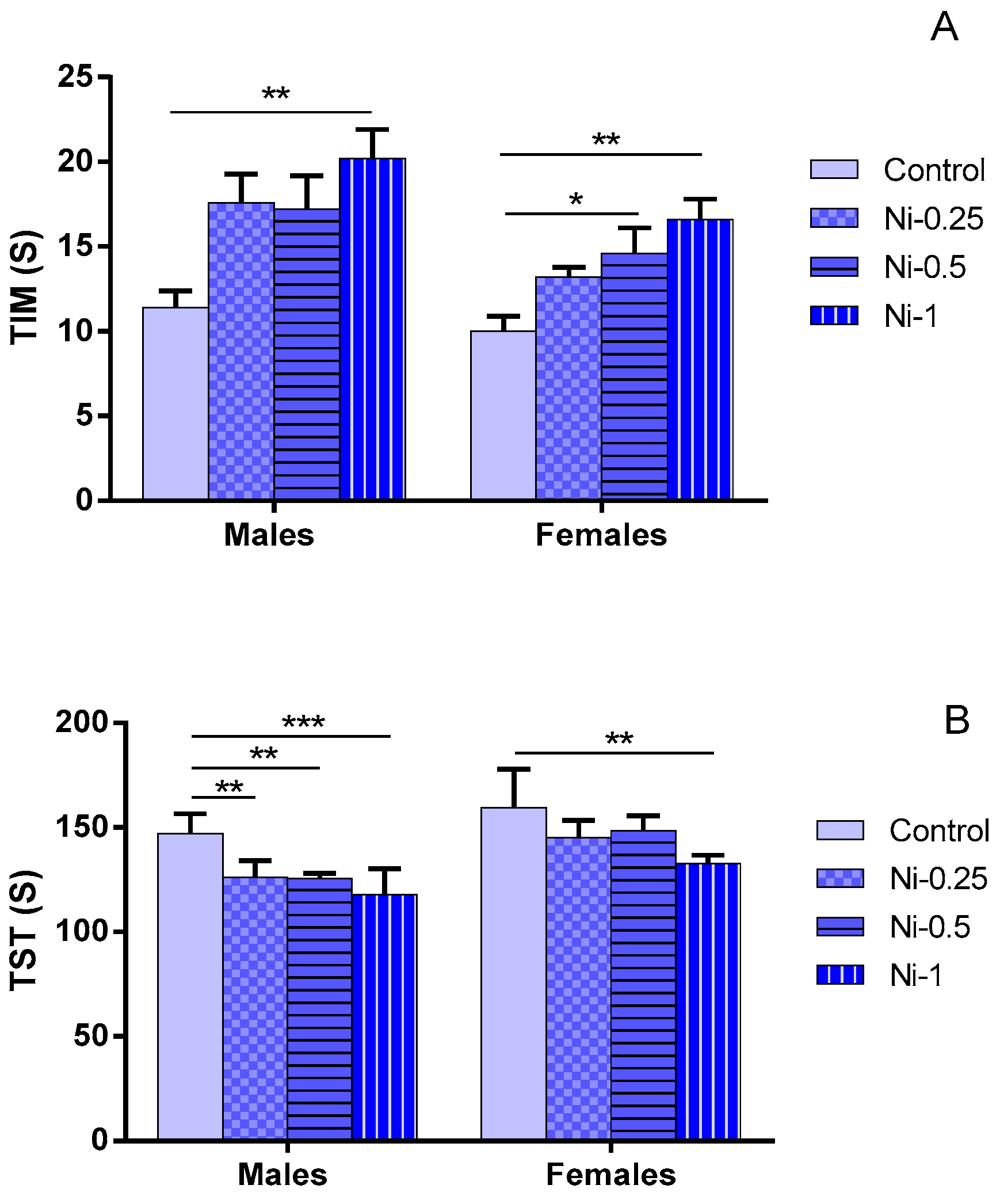
3.3.1. Immobility Time (TIM)
3.3.2. Struggling Time (TST)
3.4. Nickel Effect on Memory
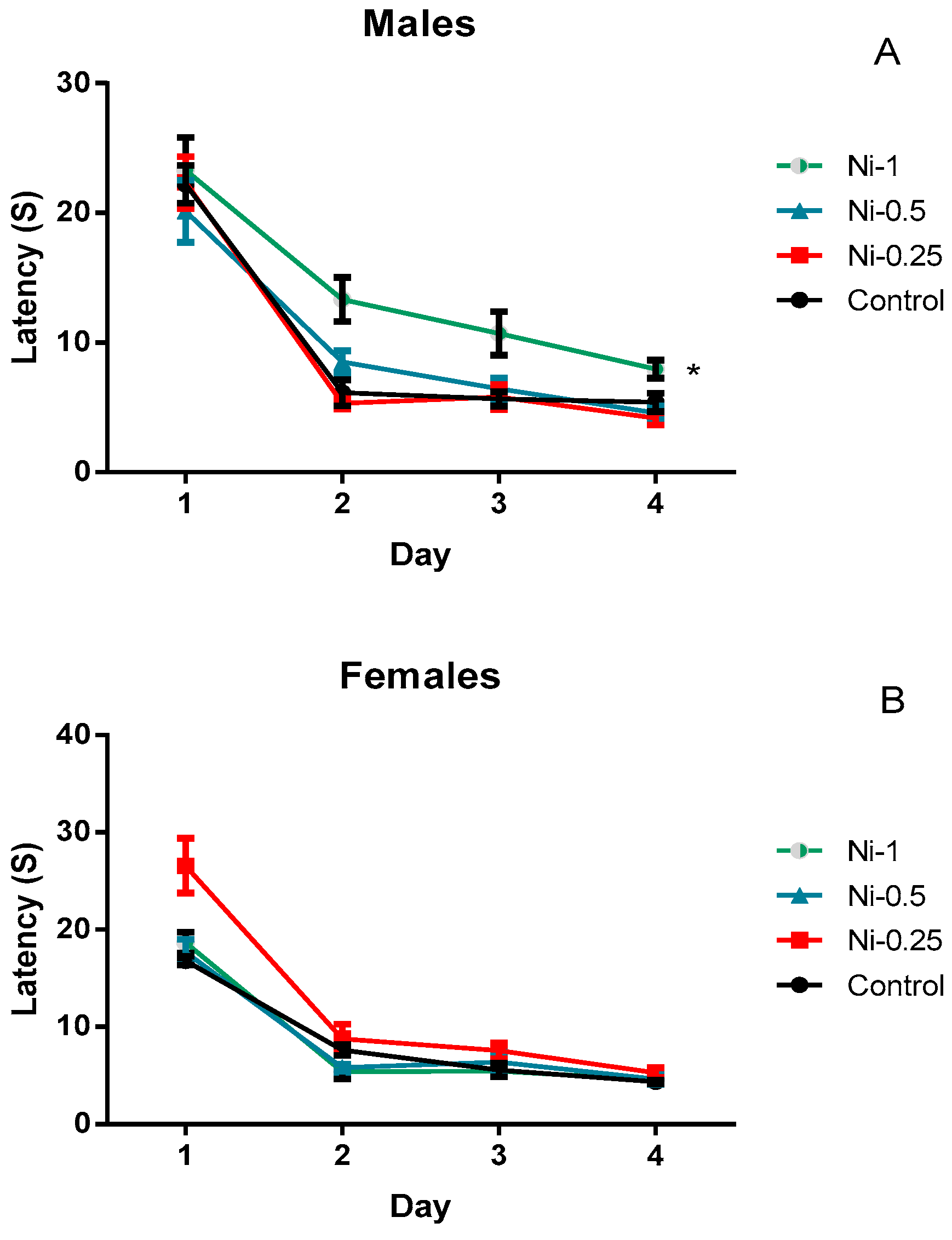
3.4.1. Morris Water Maze
Spatial Learning
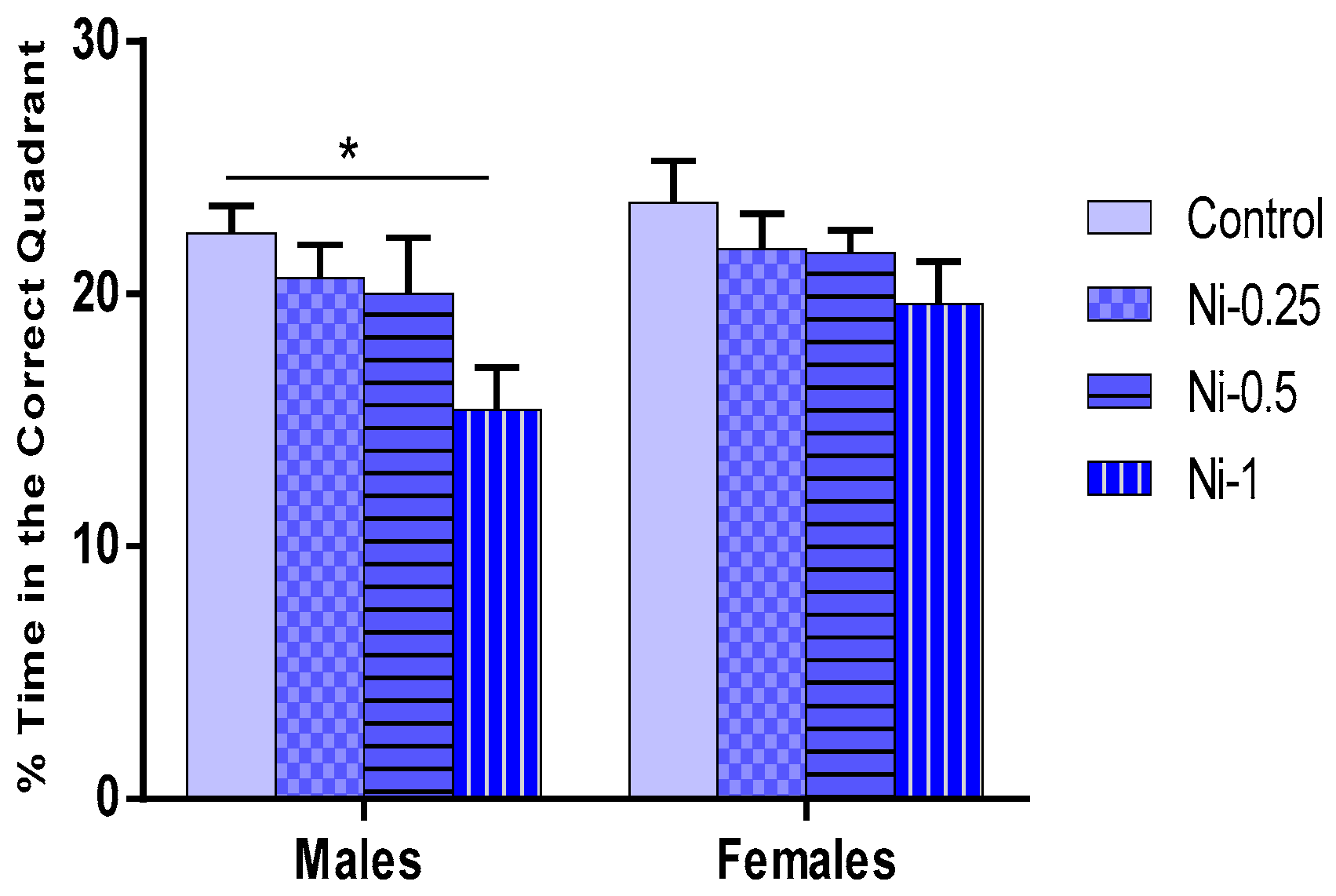
Percentage Time Spent in the Correct Quadrant During the Probe Test
3.5. Nickel Effect on Oxidative Stress
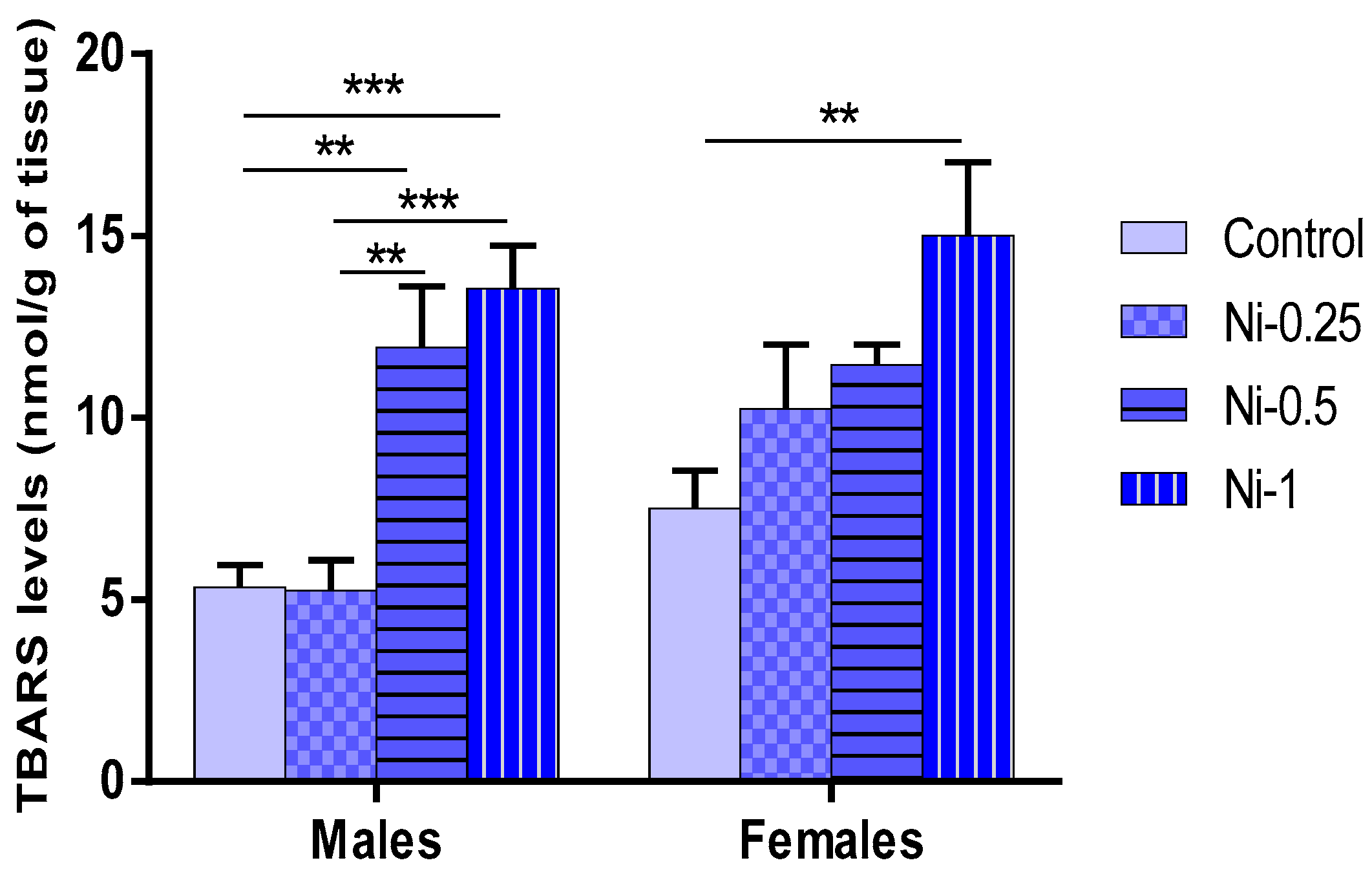
3.5.1. Lipid Peroxidation (LPO) in Hippocampus
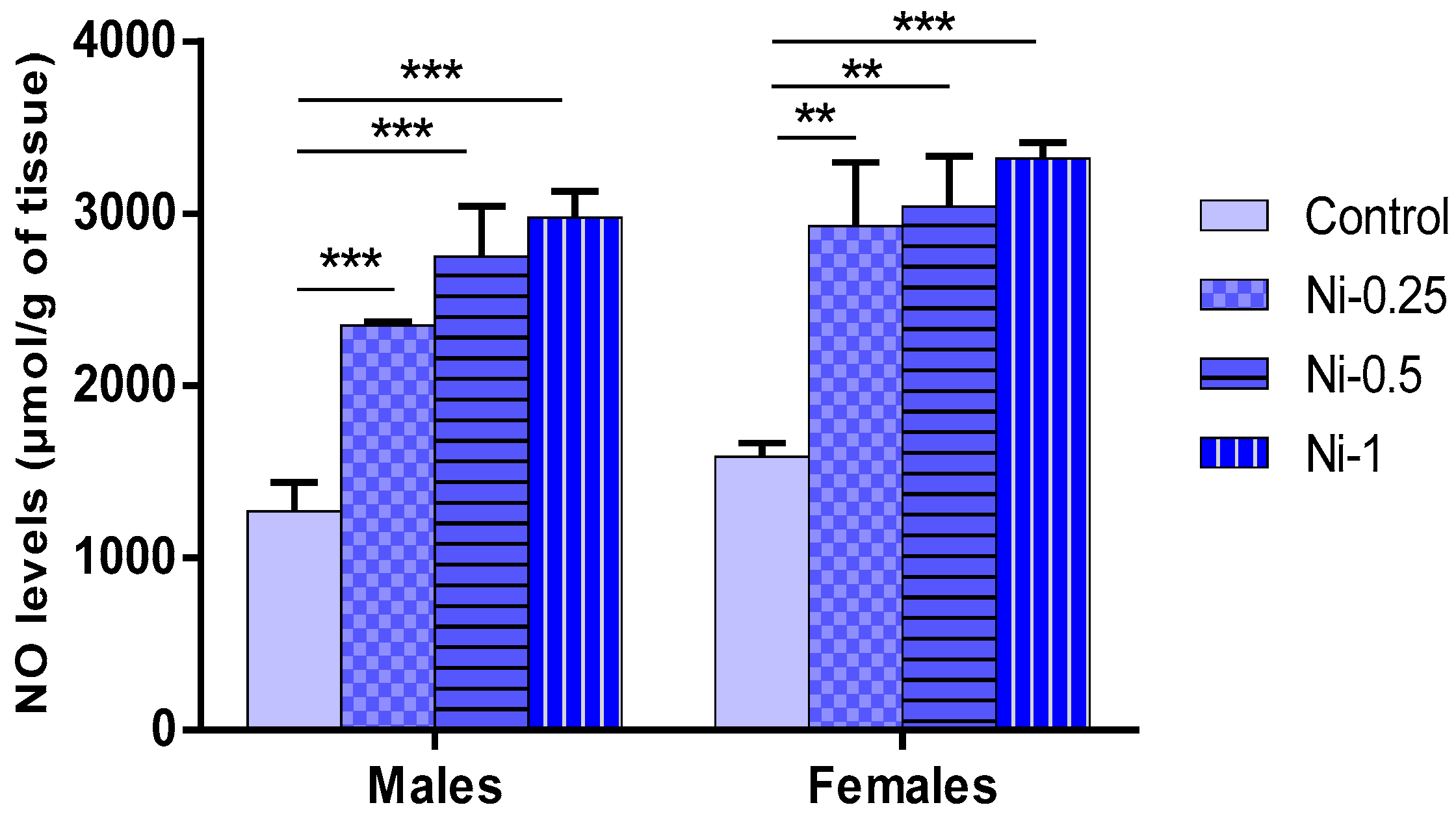
3.5.2. NO Concentrations in Hippocampus
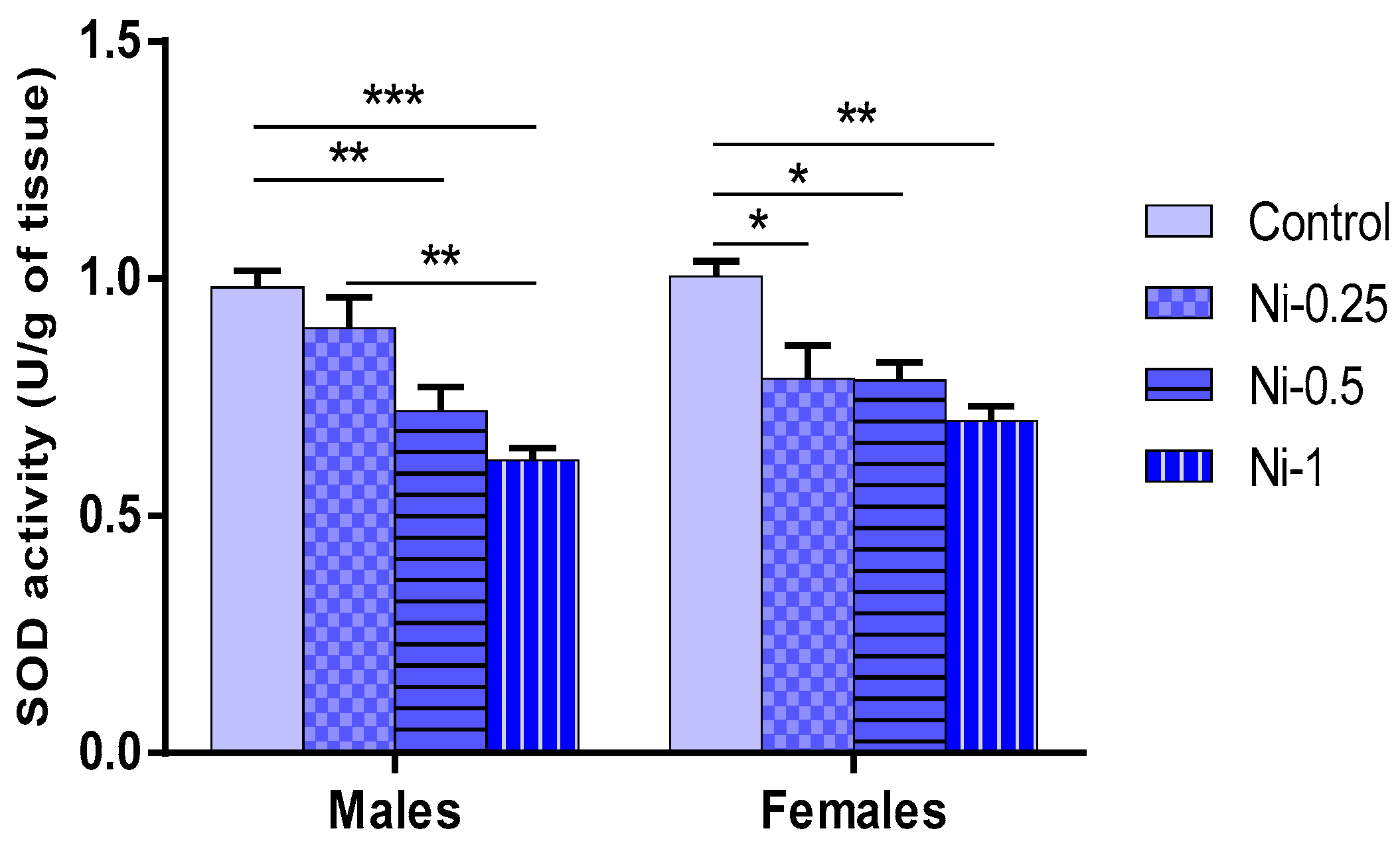
3.5.3. Superoxide Dismutase (SOD) Activity in Hippocampus
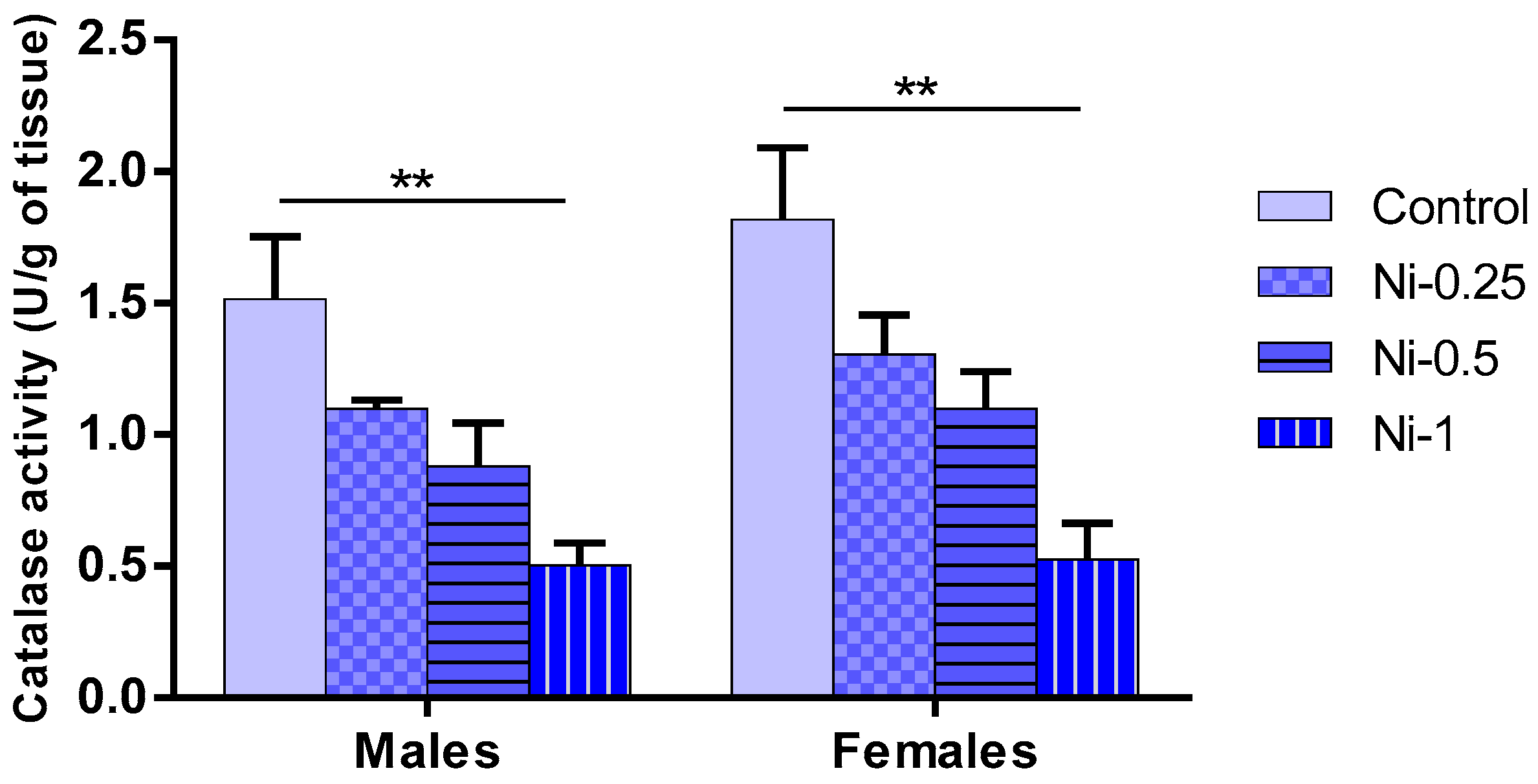
3.5.4. Catalase (CAT) Activity in Hippocampus
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Spears, J.W. Nickel as a “newer trace element” in the nutrition of domestic animals. J. Anim. Sci. 1984, 59, 823–835. [Google Scholar] [CrossRef] [PubMed]
- Marzec, Z. Alimentary chromium, nickel, and selenium intake of adults in Poland estimated by analysis and calculations using the duplicate portion technique. Mol. Nutr. Food Res. 2004, 48, 47–52. [Google Scholar] [CrossRef] [PubMed]
- Samal, L.; Mishra, C. Significance of Nickel in Livestock Health and Production. Int. J. Agro Vet. Med. Sci. 2011, 5, 349–361. [Google Scholar] [CrossRef]
- Denkhaus, E.; Salnikow, K. Nickel essentiality, toxicity, and carcinogenicity. Crit. Rev. Oncol. Hematol. 2002, 42, 35–56. [Google Scholar] [CrossRef]
- Costa, M.; Salnikow, K.; Sutherland, J.E.; Broday, L.; Peng, W.; Zhang, Q.; Kluz, T. The role of oxidative stress in nickel and chromate genotoxicity. Mol. Cell. Biochem. 2002, 234–235, 265–275. [Google Scholar] [CrossRef] [PubMed]
- Das, K.K.; Das, S.N.; Dhundasi, S.A. Nickel, its adverse health effects & oxidative stress. Indian J. Med. Res. 2008, 128, 412–425. [Google Scholar] [PubMed]
- Xu, S.C.; He, M.D.; Zhong, M.; Zhang, Y.W.; Wang, Y.; Yang, L.; Yang, J.; Yu, Z.P.; Zhou, Z. Melatonin protects against Nickel-induced neurotoxicity in vitro by reducing oxidative stress and maintaining mitochondrial function. J. Pineal Res. 2010, 49, 86–94. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.; Shi, H.; Dai, Z.; Mei, X. Variations of sediment toxicity in a tidal Estuary: A case study of the South Passage, Changjiang (Yangtze) Estuary. Chemosphere 2015, 128, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Henriksson, J.; Tallkvist, J.; Tjälve, H. Uptake of nickel into the brain via olfactory neurons in rats. Toxicol. Lett. 1997, 91, 153–162. [Google Scholar] [CrossRef]
- Chen, H.; Chen, X.; Hong, X.; Liu, C.; Huang, H.; Wang, Q.; Chen, S.; Chen, H.; Yang, K.; Sun, Q. Maternal exposure to ambient PM2.5 exaggerates fetal cardiovascular maldevelopment induced by homocysteine in rats. Environ. Toxicol. 2017, 32, 877–889. [Google Scholar] [CrossRef] [PubMed]
- He, M.D.; Xu, S.C.; Zhang, X.; Wang, Y.; Xiong, J.C.; Zhang, X.; Lu, Y.H.; Zhang, L.; Yu, Z.P.; Zhou, Z. Disturbance of aerobic metabolism accompanies neurobehavioral changes induced by nickel in mice. Neurotoxicology 2013, 38, 9–16. [Google Scholar] [CrossRef] [PubMed]
- He, M.D.; Xu, S.C.; Lu, Y.H.; Li, L.; Zhong, M.; Zhang, Y.W.; Wang, Y.; Li, M.; Yang, J.; Zhang, G.B.; et al. l-carnitine protects against nickel-induced neurotoxicity by maintaining mitochondrial function in Neuro-2a cells. Toxicol. Appl. Pharmacol. 2011, 253, 38–44. [Google Scholar] [CrossRef] [PubMed]
- Jia, C.; Roman, C.; Hegg, C.C. Nickel sulfate induces location-dependent atrophy of mouse olfactory epithelium: Protective and proliferative role of purinergic receptor activation. Toxicol. Sci. 2010, 115, 547–556. [Google Scholar] [CrossRef] [PubMed]
- David, A.; Lobner, D. In vitro cytotoxicity of orthodontic archwires in cortical cell cultures. Eur. J. Orthod. 2004, 26, 421–426. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nation, J.R.; Hare, M.F.; Baker, D.M.; Clark, D.E.; Bourgeois, A.E. Dietary administration of nickel: Effects on behavior and metallothionein levels. Physiol. Behav. 1985, 34, 349–353. [Google Scholar] [CrossRef]
- Fatehyab, S.; Hasan, M.; Hasan, M.Z.; Anwar, J. Effect of Nickel on the Levels of Dopamine, Noradrenaline and Serotonin in Different Regions of the Rat Brain. Acta Pharmarol. Toxicol. 1980, 47, 318–320. [Google Scholar]
- Seiden, L.S.; Dykstra, L.A. Psychopharmacology: A Biochemical and Behavioral Approach; Van Nostrand: New York, NY, USA, 1977; pp. 213–242. [Google Scholar]
- Kahloula, K.; Eddine, D.; Adli, H.; Slimani, M.; Terras, H.; Achour, S. Effet de l’exposition chronique au nickel sur les fonctions neurocomportementales chez les rats Wistar pendant la période de développement Effect of nickel chronic exposure on the neurobehavioral functions in Wistar rats during the development period. Toxicol. Anal. Clin. 2014, 26, 186–192. [Google Scholar] [CrossRef]
- Kita, H.; Van der Kloot, W. Action of Co and Ni at the frog neuromuscular junction. Nat. New Biol. 1973, 245, 52–53. [Google Scholar] [CrossRef] [PubMed]
- Slotkin, T.A.; Seidler, F.J. Protein kinase C is a target for diverse developmental neurotoxicants: Transcriptional responses to chlorpyrifos, diazinon, dieldrin and divalent nickel in PC12 cells. Brain Res. 2009, 1263, 23–32. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- M’Bemba-Meka, P.; Lemieux, N.; Chakrabarti, S.K. Role of oxidative stress, mitochondrial membrane potential, and calcium homeostasis in nickel subsulfide-induced human lymphocyte death in vitro. Sci. Total Environ. 2006, 369, 21–34. [Google Scholar] [CrossRef] [PubMed]
- Rao, M.V.; Chawla, S.L.; Sharma, S.R. Protective role of vitamin E on nickel and/or chromium induced oxidative stress in the mouse ovary. Food Chem. Toxicol. 2009, 47, 1368–1371. [Google Scholar] [CrossRef] [PubMed]
- Bouayed, J.; Rammal, H.; Soulimani, R. Oxidative stress and anxiety: Relationship and cellular pathways. Oxid. Med. Cell. Longev. 2009, 2, 63–67. [Google Scholar] [CrossRef] [PubMed]
- Rego, A.C.; Santos, M.S.; Oliveira, C.R. Oxidative Stress, Hypoxia, and Ischemia-Like Conditions Increase the Release of Endogenous Amino Acids by Distinct Mechanisms in Cultured Retinal Cells. J. Neurochem. 1996, 66, 2506–2516. [Google Scholar] [CrossRef] [PubMed]
- Braughler, J.M. Lipid Peroxidation-Induced Inhibition of γ-Aminobutyric Acid Uptake in Rat Brain Synaptosomes: Protection by Glucocorticoids. J. Neurochem. 1985, 44, 1282–1288. [Google Scholar] [CrossRef] [PubMed]
- Hwang, O. Role of Oxidative Stress in Parkinson’ s Disease. Exp. Neurol. 2013, 22, 11–17. [Google Scholar]
- Cheignon, C.; Tomas, M.; Faller, P.; Hureau, C.; Collin, F. Oxidative stress and the amyloid beta peptide in Alzheimer’s Disease. Redox Biol. 2017. [Google Scholar] [CrossRef] [PubMed]
- Bouayed, J.; Bohn, T. Exogenous antioxidants—Double-edged swords in cellular redox state Health beneficial effects at physiologic doses versus deleterious effects at high doses. Oxid. Med. Cell. Longev. 2010, 3, 228–237. [Google Scholar] [CrossRef] [PubMed]
- Balmus, I.M.; Ciobica, A.; Antioch, I.; Dobrin, R.; Timofte, D. Oxidative Stress Implications in the Affective Disorders: Main Biomarkers, Animal Models Relevance, Genetic Perspectives, and Antioxidant Approaches. Oxid. Med. Cell. Longev. 2016, 2016, 3975101. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Lee, S.; Han, C.; Patkar, A.A.; Masand, P.S.; Pae, C. Oxidative/nitrosative stress and antidepressants: Targets for novel antidepressants. Prog. Neuropsychopharmacol. Biol. Psychiatry 2013, 46, 224–235. [Google Scholar] [CrossRef] [PubMed]
- Dean, O.; Bush, A.I.; Berk, M.; Copolov, D.L.; Van Den Buuse, M. Glutathione depletion in the brain disrupts short-term spatial memory in the Y-maze in rats and mice. Behav. Brain Res. 2009, 198, 258–262. [Google Scholar] [CrossRef] [PubMed]
- Jyostna, V.; Sudhakar, P. Neurobehavioral Alterations in Cadmium Exposed Rats. Int. J. Recent Sci. Res. 2016, 7, 9418–9424. [Google Scholar]
- Kaoud, H.; Kamel, M.M.; Abdel-Razek, A.H.; Kamel, G.M.; Ahmed, K.A. Neurobehavioural, neurochemical and neuromorphological effects of cadmium in male rats. J. Am. Sci. 2010, 6, 189–202. [Google Scholar]
- Haider, S.; Anis, L.; Batool, Z.; Sajid, I.; Naqvi, F.; Khaliq, S.; Ahmed, S. Short term cadmium administration dose dependently elicits immediate biochemical, neurochemical and neurobehavioral dysfunction in male rats. Metab. Brain Dis. 2014, 30, 83–92. [Google Scholar] [CrossRef] [PubMed]
- Carola, V.; D’Olimpio, F.; Brunamonti, E.; Mangia, F.; Renzi, P. Evaluation of the elevated plus-maze and open-field tests for the assessment of anxiety-related behaviour in inbred mice. Behav. Brain Res. 2002, 134, 49–57. [Google Scholar] [CrossRef]
- Gentsch, C.; Lichtsteiner, M.; Feer, H. Open field and elevated plus-maze: A behavioural comparison between spontaneously hypertensive (SHR) and Wistar-Kyoto (WKY) rats and the effects of chlordiazepoxide. Behav. Brain Res. 1987, 25, 101–107. [Google Scholar] [CrossRef]
- Alicia, A.; Cheryl, A. The use of the elevated plus maze as an assay of anxiety-related behavior in rodents. Nat. Protoc. 2007, 2, 322–328. [Google Scholar] [CrossRef]
- Naranjo-Rodriguez, E.B.; Osornio, A.O.; Hernandez-Avitia, E.; Mendoza-Fernandez, V.; Escobar, A. Anxiolytic-like actions of melatonin, 5-metoxytryptophol, 5-hydroxytryptophol and benzodiazepines on a conflict procedure. Prog Neuro-Psychopharmacol. Biol. Psychiatry 2000, 24, 117–129. [Google Scholar] [CrossRef]
- Porsolt, R.D.; Anton, G.; Blavet, N.; Jalfre, M. Behavioural despair in rats: A new model sensitive to antidepressant treatments. Eur. J. Pharmacol. 1978, 47, 379–391. [Google Scholar] [CrossRef]
- Benabid, N.; Mesfioui, A.; Ouichou, A. Effects of photoperiod regimen on emotional behaviour in two tests for anxiolytic activity in Wistar rat. Brain Res. Bull. 2008, 75, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Morris, R. Spatial localization does not require the presence of local cues. Learn. Motiv. 1981, 12, 239–260. [Google Scholar] [CrossRef]
- Morris, R. Developments of a water-maze procedure for studying spatial learning in the rat. J. Neurosci. Method 1984, 11, 47–60. [Google Scholar] [CrossRef]
- Draper, H.H.; Hadley, M. Malondialdehyde determination as index of lipid Peroxidation. Methods Enzymol. 1990, 186, 421–431. [Google Scholar] [CrossRef] [PubMed]
- Freitas, R.M.; Sousa, F.C.F.; Vasconcelos, S.M.M.; Viana, G.S.B.; Fonteles, M.M.F. Pilocarpine-induced status epilepticus in rats: Lipid peroxidation level, nitrite formation, GABAergic and glutamatergic receptor alterations in the hippocampus, striatum and frontal cortex. Pharmacol. Biochem. Behav. 2004, 78, 327–332. [Google Scholar] [CrossRef] [PubMed]
- Bryan, N.S.; Grisham, M.B. Methods to detect nitric oxide and its metabolites in biological samples. Free Radic. Biol. Med. 2007, 43, 645–657. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chao, C.C.; Hu, S.; Molitor, T.W.; Shaskan, E.G.; Peterson, P.K. Activated microglia mediate neuronal cell injury via a nitric oxide mechanism. J. Immunol. 1992, 149, 2736–2741. [Google Scholar] [PubMed]
- Beauchamp, C.; Fridovich, I. Superoxide dismutase: Improved assays and an assay applicable to acrylamide gels. Anal. Biochem. 1971, 44, 276–287. [Google Scholar] [CrossRef]
- Aebi, H. Catalase in Vitro. Methods Enzymol. 1984, 105, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Kamel, M.M.; El Razek, A.H.A.; Ahmed, K.A.; Kamel, G.M. Exposure of adult male rats to cadmium: Assessment of sexual behaviour, fertility, aggression as well as anxiety like behaviour with special reference to biochemical and pathological alterations. Life Sci. J. 2011, 8, 106–119. [Google Scholar]
- Slotkin, T.A.; Seidler, F.J. Developmental neurotoxicants target neurodifferentiation into the serotonin phenotype: Chlorpyrifos, diazinon, dieldrin and divalent nickel. Toxicol. Appl. Pharmacol. 2008, 233, 211–219. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jameson, R.R.; Seidler, F.J.; Qiao, D.; Slotkin, T.A. Chlorpyrifos affects phenotypic outcomes in a model of mammalian neurodevelopment: Critical stages targeting differentiation PC12 cells. Environ. Health Perspect. 2006, 114, 667–672. [Google Scholar] [CrossRef] [PubMed]
- Slotkin, T.A.; MacKillop, E.A.; Ryde, I.T.; Tate, C.A.; Seidler, F.J. Screening for developmental neurotoxicity using PC12 cells: Comparisons of organophosphates with a carbamate, an organochlorine, and divalent nickel. Environ. Health Perspect. 2007, 115, 93–101. [Google Scholar] [CrossRef] [PubMed]
- Liapi, C.; Zarros, A.; Theocharis, S. Short-Term Exposure to Nickel Alters the Adult Rat Brain Antioxidant Status and the Activities of Crucial Membrane-Bound Enzymes: Neuroprotection by L-Cysteine. Biol. Trace Elem. Res. 2011, 143, 1673–1681. [Google Scholar] [CrossRef] [PubMed]
- Mata, M.; Fink, D.J.; Gainer, H.; Smith, C.B.; Davidsen, L.; Savaki, H.; Schwartz, W.J.; Sokoloff, L. Activity-dependent Energy Metabolism in Rat Posterior Pituitary Primarily Reflects Sodium Pump Activity. J. Neurochem. 1980, 34, 213–215. [Google Scholar] [CrossRef] [PubMed]
- Hernandez, J. Brain Na+,K+-ATPase activity possibly regulated by a specific serotonin receptor. Brain Res. 1987, 408, 399–402. [Google Scholar] [CrossRef]
- Amudha, K.; Shagirtha, K.; Elangovan, P. Naringin improves nickel-induced alterations of acetylcholinesterase, adenosine triphosphatases, and oxidative stress in brain of rats. J. Nat. Prod. Biomed. Res. 2015, 1, 21–28. [Google Scholar]
- Skutella, T.; Nitsch, R. New molecules for hippocampal development. Trends Neurosci. 2001, 24, 107–113. [Google Scholar] [CrossRef]
- Lalonde, R. The neurobiological basis of spontaneous alternation. Neurosci. Biobehav. Rev. 2002, 26, 91–104. [Google Scholar] [CrossRef]
- Kiraly, E.; Jones, D.G. Dendritic spine changes in rat hippocampal pyramidal cells after postnatal lead treatment: A Golgi study. Exp. Neurol. 1982, 77, 236–239. [Google Scholar] [CrossRef]
- Alfano, D.P.; Petit, T.L. Neonatal lead exposure alters the dendritic development of hippocampal dentate granule cells. Exp. Neurol. 1982, 75, 275–288. [Google Scholar] [CrossRef]
- Decker, M.W.; McGaugh, J.L. The role of interactions between the cholinergic system and other neuromodulatory systems in learing and memory. Synapse 1991, 7, 151–168. [Google Scholar] [CrossRef] [PubMed]
- Świergosz-Kowalewska, R.; Molenda, P.; Halota, A. Effects of chemical and thermal stress on acetylcholinesterase activity in the brain of the bank vole, Myodes glareolus. Ecotoxicol. Environ. Saf. 2014, 106, 204–212. [Google Scholar] [CrossRef] [PubMed]
- Pérez, J.; Monteiro, M.S.; Quintaneiro, C.; Soares, A.M.V.M.; Loureiro, S. Characterization of cholinesterases in Chironomus riparius and the effects of three herbicides on chlorpyrifos toxicity. Aquat. Toxicol. 2013, 144–145, 296–302. [Google Scholar] [CrossRef] [PubMed]
- Fisher, J.L. A histidine residue in the extracellular N-terminal domain of the GABAA receptor α5 subunit regulates sensitivity to inhibition by zinc. Neuropharmacology 2002, 42, 922–928. [Google Scholar] [CrossRef]
- Staruschenko, A.; Dorofeeva, N.A.; Bolshakov, K.V.; Stockand, J.D. Subunit-dependent cadmium and nickel inhibition of acid-sensing ion channels. Dev. Neurobiol. 2007, 67, 97–107. [Google Scholar] [CrossRef] [PubMed]
- Kang, H.W.; Moon, H.J.; Joo, S.H.; Lee, J.H. Histidine residues in the IS3–IS4 loop are critical for nickel-sensitive inhibition of the Cav2.3 calcium channel. FEBS Lett. 2007, 581, 5774–5780. [Google Scholar] [CrossRef] [PubMed]
- Kang, H.W.; Park, J.Y.; Jeong, S.W.; Kim, J.A.; Moon, H.J.; Perez-Reyes, E.; Lee, J.H. A molecular determinant of nickel inhibition in Cav3.2 T-type calcium channels. J. Biol. Chem. 2006, 281, 4823–4830. [Google Scholar] [CrossRef] [PubMed]
- Gavazzo, P.; Mazzolini, M.; Tedesco, M.; Marchetti, C. Nickel differentially affects NMDA receptor channels in developing cultured rat neurons. Brain Res. 2006, 1078, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Marchetti, C.; Gavazzo, P. Subunit-dependent effects of nickel on NMDA receptor channels. Mol. Brain Res. 2003, 117, 139–144. [Google Scholar] [CrossRef]
- Paoletti, P.; Neyton, J. NMDA receptor subunits: Function and pharmacology. Curr. Opin. Pharmacol. 2007, 7, 39–47. [Google Scholar] [CrossRef] [PubMed]
- Cull-Candy, S.; Brickley, S.; Farrant, M. NMDA receptor subunits: Diversity, development and disease. Curr. Opin. Neurobiol. 2001, 11, 327–335. [Google Scholar] [CrossRef]
- Choi, Y.B.; Lipton, S.A. Identification and mechanism of action of two histidine residues underlying high-affinity Zn2+ inhibition of the NMDA receptor. Neuron 1999, 23, 171–180. [Google Scholar] [CrossRef]
- Gavazzo, P.; Guida, P.; Zanardi, I.; Marchetti, C. Molecular determinants of multiple effects of nickel on NMDA receptor channels. Neurotox. Res. 2009, 15, 38–48. [Google Scholar] [CrossRef] [PubMed]
- Song, X.; Fiati Kenston, S.S.; Kong, L.; Zhao, J. Molecular mechanisms of nickel induced neurotoxicity and chemoprevention. Toxicology 2017, 392, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Ates, B.; Orun, I.; Talas, Z.S.; Durmaz, G.; Yilmaz, I. Effects of sodium selenite on some biochemical and hematological parameters of rainbow trout (Oncorhynchus mykiss Walbaum, 1792) exposed to Pb2+ and Cu2+. Fish Physiol. Biochem. 2008, 34, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Misra, M.; Rodriguez, R.E.; Kasprzak, K.S. Nickel induced lipid peroxidation in the rat: Correlation with nickel effect on antioxidant defense systems. Toxicology 1990, 64, 1–17. [Google Scholar] [CrossRef]
- Guan, F.; Zhang, D.; Wang, X.; Chen, J. Nitric oxide and bcl-2 mediated the apoptosis induced by nickel(II) in human T hybridoma cells. Toxicol. Appl. Pharmacol. 2007, 221, 86–94. [Google Scholar] [CrossRef] [PubMed]
- Gisone, P.; Boveris, A.D.; Dubner, D.; Perez, M.R.; Robello, E.; Puntarulo, S. Early Neuroprotective Effect of Nitric Oxide in Developing Rat Brain Irradiated in Utero. Neurotoxicolog 2003, 24, 245–253. [Google Scholar] [CrossRef]
- Hall, E.D.; Detloff, M.R.; Johnson, K.; Kupina, N.C.; Al, H.E.T. Peroxynitrite-Mediated Protein Nitration and Lipid Peroxidation in a Mouse Model of Traumatic Brain Injury. J. Neurotrauma 2004, 21, 9–20. [Google Scholar] [CrossRef] [PubMed]
- Sevanian, A.; Hochstein, P. Mechanisms and consequences of lipid peroxidation in biological systems. Annu. Rev. Nutr. 1985, 5, 365–390. [Google Scholar] [CrossRef] [PubMed]
- Pacifici, E.H.K.; McLeod, L.L.; Sevanian, A. Lipid hydroperoxide-induced peroxidation and turnover of endothelial cell phospholipids. Free Radic. Biol. Med. 1994, 17, 297–309. [Google Scholar] [CrossRef]
- Comporti, M. Lipid peroxidation and cellular damage in toxic liver injury. Lab. Investig. 1985, 53, 599–623. [Google Scholar] [PubMed]
- Al-Mutairi, D.A.; Craik, J.D.; Batinic-Haberle, I.; Benov, L.T. Induction of oxidative cell damage by photo-treatment with zinc N-methylpyridylporphyrin. Free Radic. Res. 2007, 41, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Stark, G. Functional consequences of oxidative membrane damage. J. Membr. Biol. 2005, 205, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Goel, A.; Dani, V.; Dhawan, D.K. Protective effects of zinc on lipid peroxidation, antioxidant enzymes and hepatic histoarchitecture in chlorpyrifos-induced toxicity. Chem. Biol. Interact. 2005, 156, 131–140. [Google Scholar] [CrossRef] [PubMed]
- Emerit, J.; Klein, J.M.; Coutellier, A.; Congy, F. Free radicals and lipid peroxidation in cell biology: Physiopathologic prospects. Pathol. Biol. 1991, 39, 316–327. [Google Scholar] [PubMed]
- Khanna, R.S.; Negi, R.; Pande, D.; Khanna, S.; Khanna, H.D. Markers of Oxidative Stress in Generalized Anxiety Psychiatric Disorder: Therapeutic Implications. J. Stress Physiol. Biochem. 2012, 8, 32–38. [Google Scholar]
- Abdalla, F.H.; Schmatz, R.; Cardoso, A.M.; Carvalho, F.B.; Baldissarelli, J.; de Oliveira, J.S.; Rosa, M.M.; Gonçalves Nunes, M.A.; Rubin, M.A.; da Cruz, I.B.; et al. Quercetin protects the impairment of memory and anxiogenic-like behavior in rats exposed to cadmium: Possible involvement of the acetylcholinesterase and Na+,K+-ATPase activities. Physiol. Behav. 2014, 135, 152–167. [Google Scholar] [CrossRef] [PubMed]
- Abu-Taweel, G.M.; Ajarem, J.S.; Ahmad, M. Protective Effect of Curcumin on Anxiety, Learning Behavior, Neuromuscular Activities, Brain Neurotransmitters and Oxidative Stress Enzymes in Cadmium Intoxicated Mice. J. Behav. Brain Sci. 2013, 3, 74–84. [Google Scholar] [CrossRef]
- Serafini, G.; Hayley, S.; Pompili, M.; Dwivedi, Y.; Brahmachari, G.; Girardi, P.; Amore, M. Hippocampal Neurogenesis, Neurotrophic Factors and Depression: Possible Therapeutic Targets? CNS Neurol. Disord. Drug Targets 2014, 13, 1708–1721. [Google Scholar] [CrossRef] [PubMed]
- Cole, J.; Costafreda, S.G.; Mcguf, P.; Fu, C.H.Y. Hippocampal atrophy in first episode depression: A meta-analysis of magnetic resonance imaging studies. J. Affect. Disord. 2011, 134, 483–487. [Google Scholar] [CrossRef] [PubMed]
- Frodl, T.; Schaub, A.; Banac, S.; Charypar, M.; Jäger, M.; Kümmler, P.; Bottlender, R.; Zetzsche, T.; Born, C.; Leinsinger, G.; et al. Reduced hippocampal volume correlates with executive dysfunctioning in major depression. Psychiatry Neurosci. 2006, 31, 316–323. [Google Scholar]
- Frodl, T.; Carballedo, A.; Hughes, M.M.; Saleh, K.; Fagan, A.; Skokauskas, N.; McLoughlin, D.M.; Meaney, J.; O’Keane, V.; Connor, T.J. Reduced expression of glucocorticoid-inducible genes GILZ and SGK-1: High IL-6 levels are associated with reduced hippocampal volumes in major depressive disorder. Transl. Psychiatry 2012, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, H.D.; Duman, R.S. The role of neurotrophic factors in adult hippocampal neurogenesis, antidepressant treatments and animal models of depressive-like behavior. Behav. Pharmacol. 2007, 18, 391–418. [Google Scholar] [CrossRef] [PubMed]
- Boldrini, M.; Hen, R.; Underwood, M.D.; Rosoklija, G.B.; Dwork, A.J.; Mann, J.J.; Arango, V. Hippocampal Angiogenesis and Progenitor Cell Major Depression. Biol. Psychiatry 2012, 72, 562–571. [Google Scholar] [CrossRef] [PubMed]
- Sanna, M.D.; Monti, M.; Casella, L.; Roggeri, R.; Galeotti, N.; Morbidelli, L. Neuronal effects of a nickel-piperazine/NO donor complex in rodents. Pharmacol. Res. 2015, 99, 162–173. [Google Scholar] [CrossRef] [PubMed]
- Yildiz, F.; Ulak, G.; Erden, B.F.; Gacar, N. Anxiolytic-like effects of 7-nitroindazole in the rat plus-maze test. Pharmacol. Biochem. Behav. 2000, 65, 199–202. [Google Scholar] [CrossRef]
- Yildiz, F.; Erden, B.F.; Ulak, G.; Utkan, T.; Gacar, N. Antidepressant-like effect of 7-nitroindazole in the forced swimming test in rats. Psychopharmacology 2000, 149, 41–44. [Google Scholar] [CrossRef] [PubMed]
- Guimarães, F.S.; Beijamini, V.; Moreira, F.A.; Aguiar, D.C.; De Lucca, A.C.B. Role of nitric oxide in brain regions related to defensive reactions. Neurosci. Biobehav. Rev. 2005, 29, 1313–1322. [Google Scholar] [CrossRef] [PubMed]
- Calixto, A.V.; Duarte, F.S.; Duzzioni, M.; Nascimento Häckl, L.P.; Faria, M.S.; De Lima, T.C.M. Role of ventral hippocampal nitric oxide/cGMP pathway in anxiety-related behaviors in rats submitted to the elevated T-maze. Behav. Brain Res. 2010, 207, 112–117. [Google Scholar] [CrossRef] [PubMed]
- De Oliveira, C.L.; Del Bel, E.A.; Guimarães, F.S. Effects of L-NOARG on plus-maze performance in rats. Pharmacol. Biochem. Behav. 1996, 56, 55–59. [Google Scholar] [CrossRef]
- Spiacci, A.; Kanamaru, F.; Guimares, F.S.; Oliveira, R.M.W. Nitric oxide-mediated anxiolytic-like and antidepressant-like effects in animal models of anxiety and depression. Pharmacol. Biochem. Behav. 2008, 88, 247–255. [Google Scholar] [CrossRef] [PubMed]
- Dhir, A.; Kulkarni, S.K. Nitric oxide and major depression. Nitric Oxide 2011, 24, 125–131. [Google Scholar] [CrossRef] [PubMed]
- Flicker, C.; Dean, R.L.; Watkins, D.L.; Fisher, S.K.; Bartus, R.T. Behavioral and neurochemical effects following neurotoxic lesions of a major cholinergic input to the cerebral cortex in the rat. Pharmacol. Biochem. Behav. 1983, 18, 973–981. [Google Scholar] [CrossRef]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lamtai, M.; Chaibat, J.; Ouakki, S.; Zghari, O.; Mesfioui, A.; El Hessni, A.; Rifi, E.-H.; Marmouzi, I.; Essamri, A.; Ouichou, A. Effect of Chronic Administration of Nickel on Affective and Cognitive Behavior in Male and Female Rats: Possible Implication of Oxidative Stress Pathway. Brain Sci. 2018, 8, 141. https://doi.org/10.3390/brainsci8080141
Lamtai M, Chaibat J, Ouakki S, Zghari O, Mesfioui A, El Hessni A, Rifi E-H, Marmouzi I, Essamri A, Ouichou A. Effect of Chronic Administration of Nickel on Affective and Cognitive Behavior in Male and Female Rats: Possible Implication of Oxidative Stress Pathway. Brain Sciences. 2018; 8(8):141. https://doi.org/10.3390/brainsci8080141
Chicago/Turabian StyleLamtai, Mouloud, Jihane Chaibat, Sihame Ouakki, Oussama Zghari, Abdelhalem Mesfioui, Aboubaker El Hessni, El-Housseine Rifi, Ilias Marmouzi, Azzouz Essamri, and Ali Ouichou. 2018. "Effect of Chronic Administration of Nickel on Affective and Cognitive Behavior in Male and Female Rats: Possible Implication of Oxidative Stress Pathway" Brain Sciences 8, no. 8: 141. https://doi.org/10.3390/brainsci8080141





