Promoting Motor Function by Exercising the Brain
Abstract
:1. Introduction
2. Neuro-Physiological Processes Associated with Brain Activity When Moving
2.1. Organization of the Motor System for Voluntary Movements
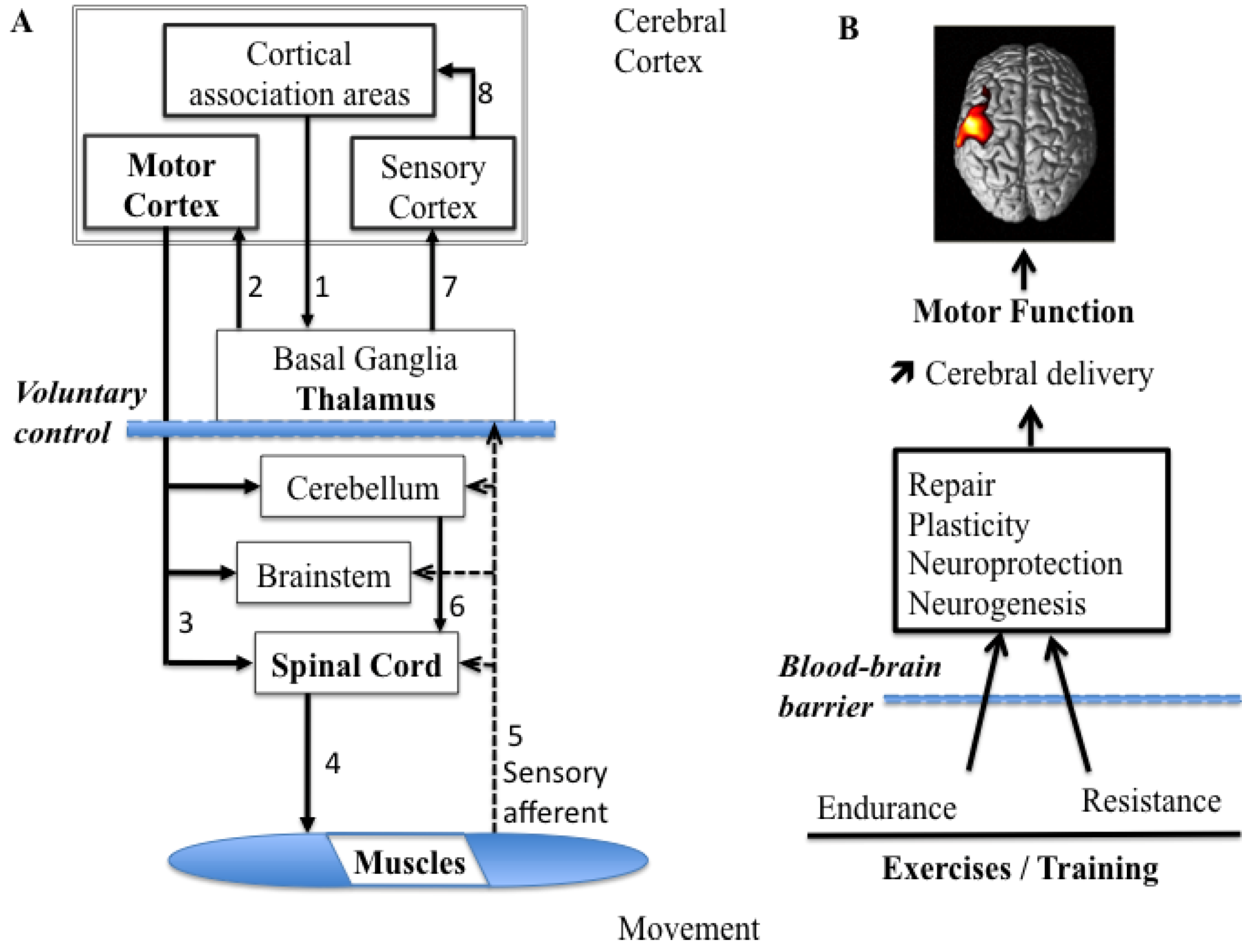
2.2. Optical Imaging of the Brain Function

2.3. Cerebrovascular Changes during Brain Activation
3. Brain’s Activation Patterns during Exercise and Training
3.1. Acute Changes Following Exercise
3.1.1. Endurance Exercise
3.1.2. Brain Regions Associated with Muscle Fatigue
3.2. Chronic Adaptations and Training Effect
4. Associations between Brain Activity Changes and Motor Function Recovery
4.1. Reorganization of Brain Activation after Brain Injury
4.2. Exercise-Induced Neuroprotection
4.3. NIRS for Monitoring Training Programs
5. Conclusion
Conflict of Interest
References
- Cotman, C.W.; Berchtold, N.C. Exercise: A behavioral intervention to enhance brain health and plasticity. Trends Neurosci. 2002, 25, 295–301. [Google Scholar] [CrossRef]
- Caspersen, C.J.; Powell, K.E.; Christenson, G.M. Physical activity, exercise, and physical fitness: Definitions and distinctions for health-related research. Public Health Rep. 1985, 100, 126–131. [Google Scholar]
- Dishman, R.K.; Berthoud, H.R.; Booth, F.W.; Cotman, C.W.; Edgerton, V.R.; Fleshner, M.R.; Gandevia, S.C.; Gomez-Pinilla, F.; Greenwood, B.N.; Hillman, C.H.; et al. Neurobiology of exercise. Obesity (Silver Spring) 2006, 14, 345–356. [Google Scholar] [CrossRef]
- Mattson, M.P.; Maudsley, S.; Martin, B. BDNF and 5-HT: A dynamic duo in age-related neuronal plasticity and neurodegenerative disorders. Trends Neurosci. 2004, 27, 589–594. [Google Scholar] [CrossRef]
- Schinder, A.F.; Poo, M. The neurotrophin hypothesis for synaptic plasticity. Trends Neurosci. 2000, 23, 639–645. [Google Scholar] [CrossRef]
- Churchill, J.D.; Galvez, R.; Colcombe, S.; Swain, R.A.; Kramer, A.F.; Greenough, W.T. Exercise, experience and the aging brain. Neurobiol. Aging 2002, 23, 941–955. [Google Scholar] [CrossRef]
- Orlandi, G.; Murri, L. Transcranial Doppler assessment of cerebral flow velocity at rest and during voluntary movements in young and elderly healthy subjects. Int. J. Neurosci. 1996, 84, 45–53. [Google Scholar] [CrossRef]
- Mosso, A. Ueber den Kreislauf des Blutes im Menschlichen Gehirn; Verlag von Veit: Leipzig, Germany, 1881. [Google Scholar]
- Roy, C.S.; Sherrington, C.S. On the regulation of the blood-supply of the brain. J. Physiol. (Lond.) 1890, 11, 85–108. [Google Scholar]
- Seifert, T.; Secher, N.H. Sympathetic influence of cerebral blood flow and metabolism during exercise in humans. Prog. Neurobiol. 2011, 95, 406–426. [Google Scholar] [CrossRef]
- Dustman, R.E.; Emmerson, R.Y.; Shearer, D.E. Physical activity, age, and cognitive neurophysiological function. J. Aging Phys. Act. 1994, 2, 143–181. [Google Scholar]
- Cui, X.; Bray, S.; Bryant, D.M.; Glover, G.H.; Reiss, A.L. A quantitative comparison of NIRS and fMRI across multiple cognitive tasks. Neuroimage 2011, 54, 2808–2821. [Google Scholar] [CrossRef]
- Obrig, H.; Hirth, C.; Junge-Hülsing, J.G.; Döge, C.; Wolf, T.; Dirnagl, U.; Villringer, A. Cerebral oxygenation changes in response to motor stimulation. J. Appl. Physiol. 1996, 81, 1174–1183. [Google Scholar]
- Perrey, S. Non-invasive NIR spectroscopy of human brain function during exercise. Methods 2008, 45, 289–299. [Google Scholar] [CrossRef]
- Perrey, S. NIRS for measuring cerebral hemodynamic responses during exercise. In Functional Neuroimaging in Exercise and Sport Sciences, 1st; Boecker, H., Hillman, C.H., Scheef, L., Strüder, H.K., Eds.; Springer: New York, NY, USA, 2012; pp. 335–349. [Google Scholar]
- Dudley, G.A.; Fleck, S.J. Strength and endurance training. Are they mutually exclusive? Sports Med. 1987, 4, 79–85. [Google Scholar] [CrossRef]
- Fleck, S.J.; Kraemer, W.J. Designing Resistance Training Programs, 1st ed; Human Kinetics Books: Champaign, IL, USA, 1987. [Google Scholar]
- Burns, J.M.; Cronk, B.B.; Anderson, H.S.; Donnely, J.E.; Thomas, G.P.; Harsha, A.; Brooks, W.M.; Swerdlow, R.H. Cardiorespiratory fitness and brain atrophy in early Alzheimer disease. Neurology 2008, 71, 210–216. [Google Scholar] [CrossRef]
- Brisswalter, J.; Arcelin, R.; Audiffren, M.; Delignieres, D. Influence of physical exercise on simple reaction time: Effects of physical fitness. Percept. Mot. Skills 1997, 85, 1019–1027. [Google Scholar] [CrossRef]
- Tomporowski, P.D.; Cureton, K.; Armstrong, L.E.; Kane, G.M.; Sparling, P.B.; Millard-Stafford, M. Short-term effects of aerobic exercise on executive processes and emotional reactivity. Int. J. Sport Exerc. Psychol. 2005, 3, 131–146. [Google Scholar] [CrossRef]
- Davranche, K.; Burle, B.; Audiffren, M.; Hasbroucq, T. Information processing during physical exercise: A chronometric and electromyographic study. Exp. Brain Res. 2005, 165, 532–540. [Google Scholar] [CrossRef]
- Dietrich, A. Functional neuroanatomy of altered states of consciousness: The transient hypofrontality hypothesis. Conscious. Cogn. 2003, 12, 231–256. [Google Scholar] [CrossRef]
- Tenenbaum, G.; Yuval, R.; Elbaz, G.; Gar-Eli, M.; Weinberg, R. The relationship between cognitive characteristics and decision making. Can. J. Appl. Physiol. 1993, 18, 48–62. [Google Scholar] [CrossRef]
- Arent, S.M.; Landers, D.M. Arousal, anxiety, and performance: A reexamination of the Inverted-U hypothesis. Res. Q. Exerc. Sport 2003, 74, 436–444. [Google Scholar]
- Brisswalter, J.; Collardeau, M.; Arcelin, R. Effects of acute physical exercise characteristics on cognitive performance. Sports Med. 2002, 32, 555–566. [Google Scholar] [CrossRef]
- Enoka, R. Neuromechanics of Human Movement, 4th ed; Human Kinetics Books: Champaign, IL, USA, 2008. [Google Scholar]
- Winges, S.A.; Santello, M. Common input to motor units of digit flexors during multi-digit grasping. J. Neurophysiol. 2004, 92, 3210–3220. [Google Scholar] [CrossRef]
- Dum, R.P.; Strick, PL. Spinal cord terminations of the medial wall motor areas in macaque monkeys. J. Neurosci. 1996, 16, 6513–6525. [Google Scholar]
- Carroll, T.J.; Selvanayagam, V.S.; Riek, S.; Semmler, J.G. Neural adaptations to strength training: Moving beyond transcranial magnetic stimulation and reflex studies. Acta Physiol. (Oxf.) 2011, 202, 119–140. [Google Scholar] [CrossRef]
- Tinazzi, M.; Farina, S.; Tamburin, S.; Facchini, S.; Fiaschi, A.; Restivo, D.; Berardelli, A. Task-dependent modulation of excitatory and inhibitory functions within the human primary motor cortex. Exp. Brain Res. 2003, 150, 222–229. [Google Scholar]
- Perez, M.A.; Lungholt, B.K.; Nyborg, K.; Nielsen, J.B. Motor skill training induces changes in the excitability of the leg cortical area in healthy humans. Exp. Brain Res. 2004, 159, 197–205. [Google Scholar] [CrossRef]
- Pearce, A.J.; Kidgell, D.J. Corticomotor excitability during precision motor tasks. J. Sci. Med. Sport 2009, 12, 280–283. [Google Scholar] [CrossRef] [Green Version]
- Carey, J.R.; Greer, K.R.; Grunewald, T.K.; Steele, J.L.; Wiemiller, J.W.; Bhatt, E.; Nagpal, A.; Lungu, O.; Auerbach, E.J. Primary motor area activation during precision-demanding versus simple finger movement. Neurorehabil. Neural Repair. 2006, 20, 361–370. [Google Scholar] [CrossRef]
- Biswal, B.B.; Mennes, M.; Zuo, X-N.; Gohel, S.; Kelly, C.; Smith, S.M.; Beckmann, C.F.; Adelstein, J.S.; Buckner, R.L.; Colcombe, S.; et al. Toward discovery science of human brain function. Proc. Natl. Acad. Sci. USA 2010, 107, 4734–4739. [Google Scholar]
- Siebner, H.R.; Bergmann, T.O.; Bestmann, S.; Massimini, M.; Johansen-Berg, H.; Mochizuki, H.; Bohning, D.E.; Boorman, E.D.; Groppa, S.; Miniussi, C.; et al. Consensus paper: Combining transcranial stimulation with neuroimaging. Brain Stimul. 2009, 2, 58–80. [Google Scholar] [CrossRef]
- Strangman, G.; Goldstein, R.; Rauch, S.L.; Stein, J. Near-infrared spectroscopy and imaging for investigating stroke rehabilitation: Test-retest reliability and review of the literature. Arch. Phys. Med. Rehabil. 2006, 87, S12–S19. [Google Scholar]
- Steinbrink, J.; Villringer, A.; Kempf, F.; Haux, D.; Boden, S.; Obrig, H. Illuminating the BOLD signal: Combined fMRI-fNIRS studies. Magn. Reson. Imaging 2006, 24, 495–505. [Google Scholar] [CrossRef]
- Kirilina, E.; Jelzow, A.; Heine, A.; Niessing, M.; Wabnitz, H.; Brühl, R.; Ittermann, B.; Jacobs, A.M.; Tachtsidis, I. The physiological origin of task-evoked systemic artefacts in functional near infrared spectroscopy. Neuroimage 2012, 15, 70–81. [Google Scholar]
- Villringer, A.; Dirgnal, U. Coupling of brain activity and cerebral blood flow: Basis of functional neuroimaging. Cerebrovasc. Brain Metab. Rev. 1995, 7, 240–276. [Google Scholar]
- Iwasaki, K.; Ogawa, Y.; Shibata, S.; Aoki, K. Acute exposure to normobaric mild hypoxia alters dynamic relationships between blood pressure and cerebral blood flow at very low frequency. J. Cereb. Blood Flow Metab. 2007, 27, 776–784. [Google Scholar]
- Atkins, E.R.; Brodie, F.G.; Rafelt, S.E.; Panerai, R.B.; Robinson, T.G. Dynamic cerebral autoregulation is compromised acutely following mild ischaemic stroke but not transient ischaemic attack. Cerebrovasc. Dis. 2010, 29, 228–235. [Google Scholar] [CrossRef]
- Ogoh, S.; Ainslie, P.N. Cerebral blood flow during exercise: mechanisms of regulation. J. Appl. Physiol. 2009, 107, 1370–1380. [Google Scholar] [CrossRef]
- Rupp, T.; Perrey, S. Prefrontal cortex oxygenation and neuromuscular responses to exhaustive exercise. Eur. J. Appl. Physiol. 2008, 102, 153–163. [Google Scholar]
- Leff, D.R.; Orihuela-Espina, F.; Elwell, C.E.; Athanasiou, T.; Delpy, D.T.; Darzi, A.W.; Yang, G.Z. Assessment of the cerebral cortex during motor task behaviours in adults: A systematic review of functional near infrared spectroscopy (fNIRS) studies. Neuroimage 2011, 54, 2922–2936. [Google Scholar] [CrossRef]
- Lin, P.Y.; Chen, J.J.; Lin, S.I. The cortical control of cycling exercise in stroke patients: An fNIRS study. Hum. Brain Mapp. 2012. [Google Scholar] [CrossRef]
- Buckworth, J.; Dishman, R.K. Exercise Psychology; Human Kinetics Books: Champaign, IL, USA, 2002. [Google Scholar]
- Liu-Ambrose, T.; Nagamatsu, L.S.; Voss, M.W.; Khan, K.M.; Handy, T.C. Resistance training and functional plasticity of the aging brain: A 12-month randomized controlled trial. Neurobiol. Aging 2012, 33, 1690–1698. [Google Scholar] [CrossRef]
- Cassilhas, R.C.; Viana, V.A.; Grassmann, V.; Santos, R.T.; Santos, R.F.; Tufik, S.; Mello, M.T. The impact of resistance exercise on the cognitive function of the elderly. Med. Sci. Sports Exerc. 2007, 39, 1401–1407. [Google Scholar] [CrossRef]
- Yarrow, J.F.; White, L.J.; McCoy, S.C.; Borst, S.E. Training augments resistance exercise induced elevation of circulating brain derived neurotrophic factor (BDNF). Neurosci. Lett. 2010, 479, 161–165. [Google Scholar] [CrossRef]
- Smith, S.A.; Mitchell, J.H.; Garry, M.G. The mammalian exercise pressor reflex in health and disease. Exp. Physiol. 2006, 91, 89–102. [Google Scholar]
- Willimason, J.W.; Fadel, P.J.; Mitchell, J.H. New insights into central cardiovascular control during exercise in humans: A central command update. Exp. Physiol. 2006, 91, 51–58. [Google Scholar]
- Madsen, P.L.; Sperling, B.K.; Warming, T.; Schmidt, J.F.; Secher, N.H.; Wildschiødtz, G.; Holm, S.; Lassen, N.A. Middle cerebral artery blood velocity and cerebral blood flow and O2 uptake during dynamic exercise. J. Appl. Physiol. 1993, 74, 245–250. [Google Scholar]
- Secher, N.H.; Seifert, T.; van Lieshout, J.J. Cerebral blood flow and metabolism during exercise: Implications for fatigue. J. Appl. Physiol. 2008, 104, 306–314. [Google Scholar]
- Querido, J.S.; Sheel, A.W. Regulation of cerebral blood flow during exercise. Sports Med. 2007, 37, 765–782. [Google Scholar] [CrossRef]
- Delp, M.D.; Armstrong, R.B.; Godfrey, D.A.; Laughlin, M.H.; Ross, C.D.; Wilkerson, M.K. Exercise increases blood flow to locomotor, vestibular, cardiorespiratory and visual regions of the brain in miniature swine. J. Physiol. 2001, 533, 849–859. [Google Scholar] [CrossRef]
- Dalsgaard, M.K.; Quistorff, B.; Danielsen, E.R.; Selmer, C.; Vogelsang, T.; Secher, N.H. A reduced cerebral metabolic ratio in exercise reflects metabolism and not accumulation of lactate within the human brain. J. Physiol. 2004, 554, 571–578. [Google Scholar]
- Bereczki, D.; Wei, L.; Acuff, V.; Gruber, K.; Tajima, A.; Patlak, C.; Fenstermacher, J. Technique-dependent variations in cerebral microvessel blood volumes and hematocrits in the rat. J. Appl. Physiol. 1992, 73, 918–924. [Google Scholar]
- Fox, P.T.; Raichle, M.E. Focal physiological uncoupling of cerebral blood flow and oxidative metabolism during somatosensory stimulation in human subjects. Proc. Natl. Acad. Sci. USA 1986, 83, 1140–144. [Google Scholar] [CrossRef]
- Hoshi, Y.; Kobayashi, N.; Tamura, M. Interpretation of near-infrared spectroscopy signals: A study with a newly developed perfused rat brain model. J. Appl. Physiol. 2001, 90, 1657–1662. [Google Scholar]
- Rooks, C.R.; Thom, N.J.; McCully, K.K.; Dishman, R.K. Effects of incremental exercise on cerebral oxygenation measured by near-infrared spectroscopy: A systematic review. Prog. Neurobiol. 2010, 92, 134–150. [Google Scholar] [CrossRef]
- Bhambhani, Y.; Malik, R.; Mookerjee, S. Cerebral oxygenation declines at exercise intensities above the respiratory compensation threshold. Respir. Physiol. Neurobiol. 2007, 156, 196–202. [Google Scholar] [CrossRef]
- Timinkul, A.; Kato, M.; Omori, T.; Deocaris, C.C.; Ito, A.; Kizuka, T.; Sakairi, Y.; Nishijima, T.; Asada, T.; Soya, H. Enhancing effect of cerebral blood volume by mild exercise in healthy young men: A near-infrared spectroscopy study. Neurosci. Res. 2008, 61, 242–248. [Google Scholar] [CrossRef]
- Rissanen, A.P.; Tikkanen, H.O.; Koponen, A.S.; Aho, J.M.; Hägglund, H.; Lindholm, H.; Peltonen, J.E. Alveolar gas exchange and tissue oxygenation during incremental treadmill exercise, and their associations with blood O2 carrying capacity. Front. Physiol. 2012, 3, 265. [Google Scholar]
- Szubski, C.; Burtscher, M.; Loscher, W.N. The effects of short-term hypoxia on motor cortex excitability and neuromuscular activation. J. Appl. Physiol. 2006, 101, 1673–1677. [Google Scholar]
- Roach, R.C.; Hackett, P.H. Frontiers of hypoxia research: Acute mountain sickness. J. Exp. Biol. 2001, 204, 3161–3170. [Google Scholar]
- Nybo, L.; Rasmussen, P. Inadequate cerebral oxygen delivery and central fatigue during strenuous exercise. Exerc. Sport Sci. Rev. 2007, 35, 110–118. [Google Scholar]
- Verges, S.; Rupp, T.; Jubeau, M.; Wuyam, B.; Esteve, F.; Levy, P.; Perrey, S.; Millet, G.Y. Cerebral perturbations during exercise in hypoxia. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2012, 302, R903–R916. [Google Scholar] [CrossRef] [Green Version]
- Liu, J.Z.; Dai, T.H.; Sahgal, V.; Brown, R.W.; Yue, G.H. Nonlinear cortical modulation of muscle fatigue: A functional MRI study. Brain Res. 2002, 957, 320–329. [Google Scholar] [CrossRef]
- Post, M.; Steens, A.; Renken, R.; Maurits, N.M.; Zijdewind, I. Voluntary activation and cortical activity during a sustained maximal contraction: An fMRI study. Hum. Brain Mapp. 2009, 30, 1014–1027. [Google Scholar] [CrossRef]
- Van Duinen, H.; Renken, R.; Maurits, N.; Zijdewind, I. Effects of motor fatigue on human brain activity, an fMRI study. Neuroimage 2007, 35, 1438–1449. [Google Scholar] [CrossRef]
- Benwell, N.M.; Mastaglia, F.L.; Thickbroom, G.W. Changes in the functional MR signal in motor and non-motor areas during intermittent fatiguing hand exercise. Exp. Brain Res. 2007, 182, 93–97. [Google Scholar] [CrossRef]
- Anwar, A.R.; Muthalib, M.; Perrey, S.; Galka, A.; Granert, O.; Wolff, S.; Deuschl, G.; Raethjen, J.; Heute, U.; Muthuraman, M. Directionality Analysis on Functional Magnetic Resonance Imaging during Motor Task Using Granger Causality. In Proceedings of Engineering in Medicine and Biology Society (EMBC)2012 Annual International Conference of the IEEE, San Diego, CA, USA, 28 August-1 September 2012; pp. 2287–2290.
- Jiang, Z.; Wang, X.-F.; Kisiel-Sajewicz, K.; Yan, J.H.; Yue, G.H. Strengthened functional connectivity in the brain during muscle fatigue. Neuroimage 2012, 60, 728–737. [Google Scholar] [CrossRef]
- Rupp, T.; Perrey, S. Effect of severe hypoxia on prefrontal cortex and muscle oxygenation responses at rest and during exhaustive exercise. Adv. Exp. Med. Biol. 2009, 645, 329–334. [Google Scholar] [CrossRef]
- Pereira, M.I.; Gomes, P.S.; Bhambhani, Y. Acute effects of sustained isometric knee extension on cerebral and muscle oxygenation responses. Clin. Physiol. Funct. Imaging 2009, 29, 300–308. [Google Scholar] [CrossRef]
- Matsuura, C.; Gomes, P.S.C.; Haykowsky, M.; Bhambhani, Y. Cerebral and muscle oxygenation changes during static and dynamic knee extensions to voluntary fatigue in healthy men and women: a near infrared spectroscopy study. Clin. Physiol. Funct. Imaging 2011, 31, 114–123. [Google Scholar]
- Gomes, P.S.; Matsuura, C.; Bhambhani, Y.N. Effects of hypoxia on cerebral and muscle haemodynamics during knee extensions in healthy subjects. Eur. J. Appl. Physiol. 2012, 113, 13–23. [Google Scholar]
- Sakatani, K.; Xie, Y.; Lichty, W.; Li, S.; Zuo, H. Language-activated cerebral blood oxygenation and hemodynamic changes of the left prefrontal cortex in poststroke aphasic patients: A near-infrared spectroscopy study. Stroke 1998, 29, 1299–1304. [Google Scholar] [CrossRef]
- Chen, S.; Sakatani, K.; Lichty, W.; Ning, P.; Zhao, S.; Zuo, H. Auditory-evoked cerebral oxygenation changes in hypoxic-ischemic encephalopathy of newborn infants monitored by near infrared spectroscopy. Early Hum. Dev. 2002, 67, 113–121. [Google Scholar] [CrossRef]
- Miyazawa, T.; Horiuchi, M.; Ichikawa, D.; Sato, K.; Tanaka, N.; Bailey, D.M.; Ogoh, S. Kinetics of exercise-induced neural activation; interpretive dilemma of altered cerebral perfusion. Exp. Physiol. 2011, 97, 219–227. [Google Scholar]
- Lange-Assechenfeldt, C.; Kjoda, G. Alzheimer’s disease, cerebrovascular dysfunction and the benefits of exercise: From vessels to neurons. Exp. Gerontol. 2008, 43, 499–504. [Google Scholar] [CrossRef]
- McCloskey, D.P.; Adamo, D.S.; Anderson, B.J. Exercise increases metabolic capacity in the motor cortex and striatum, but not in the hippocampus. Brain Res. 2001, 891, 168–175. [Google Scholar] [CrossRef]
- Johnson, R.A.; Mitchell, G.S. Exercise-induced changes in hippocampal brain-derived neurotrophic factor and neurotrophin-3: Effects of rat strain. Brain Res. 2003, 983, 108–114. [Google Scholar] [CrossRef]
- Pereira, A.C.; Huddleston, D.E.; Brickman, A.M.; Sosunov, A.A.; Hen, R.; McKhann, G.M.; Sloan, R.; Gage, F.H.; Brown, T.R.; Small, S.A. An in vivo correlate of exercise-induced neurogenesis in the adult dentate gyrus. Proc. Natl. Acad. Sci. USA 2007, 104, 5638–5643. [Google Scholar]
- Ogunshola, O.O.; Stewart, W.B.; Mihalcik, V.; Solli, T.; Madri, J.A.; Ment, L.R. Neuronal VEGF expression correlates with angiogenesis in postnatal developing rat brain. Brain Res. Dev. Brain Res. 2000, 119, 139–153. [Google Scholar] [CrossRef]
- Kleim, J.A.; Cooper, N.R.; vandenBerg, P.M. Exercise induces angiogenesis but dose not alter movement representations within rat motor cortex. Brain Res. 2002, 934, 1–6. [Google Scholar] [CrossRef]
- Swain, R.A.; Harris, A.B.; Wiener, E.C.; Dutka, M.V.; Morris, H.D.; Theien, B.E.; Konda, S.; Engberg, K.; Lauterbur, P.C.; Greenough, W.T. Prolonged exercise induces angiogenesis and increases cerebral blood volume in primary motor cortex of the rat. Neuroscience 2003, 117, 1037–1046. [Google Scholar] [CrossRef]
- Ding, Y.; Li, J.; Luan, X.; Ding, Y.H.; Lai, Q.; Rafols, J.A.; Phillis, J.W.; Clark, J.C.; Diaz, F.G. Exercise pre-conditioning reduces brain damage in ischemic rats that may be associated with regional angiogenesis and cellular overexpression of neurotrophin. Neuroscience 2004, 124, 583–591. [Google Scholar] [CrossRef]
- Li, J.; Ding, Y.H.; Rafols, J.A.; Lai, Q.; McAllister, J.P.; Ding, Y. Increased astrocyte proliferation in rats after running exercise. Neurosci. Lett. 2005, 386, 160–164. [Google Scholar] [CrossRef]
- Ainslie, P.N.; Cotter, J.D.; George, K.P.; Lucas, S.; Murrell, C.; Shave, R.; Thomas, K.N.; Williams, M.J.; Atkinson, G. Elevation in cerebral blood flow velocity with aerobic fitness throughout healthy human ageing. J. Physiol. 2008, 586, 4005–400. [Google Scholar] [CrossRef]
- Hooker, S.P.; Sui, X.; Colabianchi, N.; Vena, J.; Laditka, J.; LaMonte, M.J.; Blair, S.N. Cardiorespiratory fitness as a predictor of fatal and nonfatal stroke in asymptomatic women and men. Stroke 2008, 39, 2950–2957. [Google Scholar] [CrossRef]
- Larson, E.B.; Wang, L.; Bowen, J.D.; McCormick, W.C.; Teri, L.; Crane, P.; Kukull, W. Exercise is associated with reduced risk for incident dementia among persons 65 years of age and older. Ann. Intern. Med. 2006, 144, 73–81. [Google Scholar]
- Bundo, M.; Inao, S.; Nakamura, A.; Kato, T.; Ito, K.; Tadokoro, M.; Kabeya, R.; Sugimoto, T.; Kajita, Y.; Yoshida, J. Changes of neural activity correlate with the severity of cortical ischemia in patients with unilateral major cerebral artery occlusion. Stroke 2002, 33, 61–66. [Google Scholar] [CrossRef]
- Frost, S.B.; Barbay, S.; Friel, K.M.; Plautz, E.J.; Nudo, R.J. Reorganization of remote cortical regions after ischemic brain injury: A potential substrate for stroke recovery. J. Neurophysiol. 2003, 89, 3205–3214. [Google Scholar] [CrossRef]
- Plate, K.H. Mechanism of angiogenesis in the brain. J. Neuropathol. Exp. Neurol. 1999, 58, 313–320. [Google Scholar] [CrossRef]
- Kolb, B.; Gibb, R. Possible anatomical basis of recovery of function after neonatal frontal lesions in rats. Behav. Neurosci. 1993, 107, 799–811. [Google Scholar] [CrossRef]
- Ward, N.S.; Brown, M.M.; Thompson, A.J.; Frackowiak, R.S. Neural correlates of motor recovery after stroke: A longitudinal fMRI study. Brain 2003, 126, 2476–2496. [Google Scholar] [CrossRef]
- Krupinski, J.; Kaluza, J.; Kumar, P.; Kumar, S.; Wang, J.M. Role of angiogenesis in patients with cerebral ischemic stroke. Stroke 1994, 25, 1794–1798. [Google Scholar] [CrossRef]
- Cramer, S.C.; Nelles, G.; Benson, R.R.; Kaplan, J.D.; Parker, R.A.; Kwong, K.K.; Kennedy, D.N.; Finklestein, S.P.; Rosen, B.R. A functional MRI study of subjects recovered from hemiparetic stroke. Stroke 1997, 28, 2518–2527. [Google Scholar] [CrossRef]
- Isaacs, K.R.; Anderson, B.J.; Alcantara, A.A.; Black, J.E.; Greenough, W.T. Exercise and the brain: angiogenesis in the adult rat cerebellum after vigorous physical activity and motor skill learning. J. Cereb. Blood Flow Metab. 1992, 12, 10–119. [Google Scholar]
- Hayes, K.; Sprague, S.; Guo, M.; Davis, W.; Friedman, A.; Kumar, A.; Jimenez, D.F.; Ding, Y. Forced, not voluntary, exercise effectively induces neuroprotection in stroke. Acta Neuropathol. 2008, 115, 289–296. [Google Scholar] [CrossRef]
- Tajiri, N.; Yasuhara, T.; Shingo, T.; Kondo, A.; Yuan, W.; Kadota, T.; Wang, F.; Baba, T.; Tayra, J.T.; Morimoto, T.; et al. Exercise exerts neuroprotective effects on Parkinson’s disease model of rats. Brain Res. 2010, 1310, 200–207. [Google Scholar] [CrossRef]
- Petzinger, G.M.; Fisher, B.E.; van Leeuwen, J.E.; Vukovic, M.; Akopian, G.; Meshul, C.K.; Holschneider, D.P.; Nacca, A.; Walsh, J.P.; Jakowec, M.W. Enhancing neuroplasticity in the basal ganglia: The role of exercise in Parkinson’s disease. Mov. Disord. 2010, 25, S141–S145. [Google Scholar]
- Kinni, H.; Guo, M.; Ding, J.Y.; Konakondla, S.; Dornbos, D., III; Tran, R.; Guthikonda, M.; Ding, Y. Cerebral metabolism after forced or voluntary physical exercise. Brain Res. 2011, 1388, 48–55. [Google Scholar] [CrossRef]
- Alberts, J.L.; Linder, S.M.; Penko, A.L.; Lowe, M.J.; Philipps, M. It is not about the bike, it is about the pedaling: Forced exercise and Parkinson’s disease. Exerc. Sport Sci. Rev. 2011, 39, 177–186. [Google Scholar]
- Ding, Y.; Li, J.; Lai, Q.; Azam, S.; Rafols, J.A.; Diaz, F.G. Functional improvement after motor training is correlated with synaptic plasticity in rat thalamus. Neurol. Res. 2002, 24, 829–836. [Google Scholar] [CrossRef]
- Saitou, H.; Yanagi, H.; Hara, S.; Tsuchiya, S.; Tomura, S. Cerebral blood volume and oxygenationamong poststroke hemiplegic patients: Effects of 13 rehabilitation tasks measured by near-infrared spectroscopy. Arch. Phys. Med. Rehabil. 2000, 81, 1348–1356. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Perrey, S. Promoting Motor Function by Exercising the Brain. Brain Sci. 2013, 3, 101-122. https://doi.org/10.3390/brainsci3010101
Perrey S. Promoting Motor Function by Exercising the Brain. Brain Sciences. 2013; 3(1):101-122. https://doi.org/10.3390/brainsci3010101
Chicago/Turabian StylePerrey, Stephane. 2013. "Promoting Motor Function by Exercising the Brain" Brain Sciences 3, no. 1: 101-122. https://doi.org/10.3390/brainsci3010101




