RETRACTED: A Si-αTCP Scaffold for Biomedical Applications: An Experimental Study Using the Rabbit Tibia Model
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparation and Characterization of the Si-TCP Scaffolds
2.2. Animals and Surgical Procedure
2.3. Histological and Histomorphometric Analysis
2.4. Statistical Analysis
3. Results
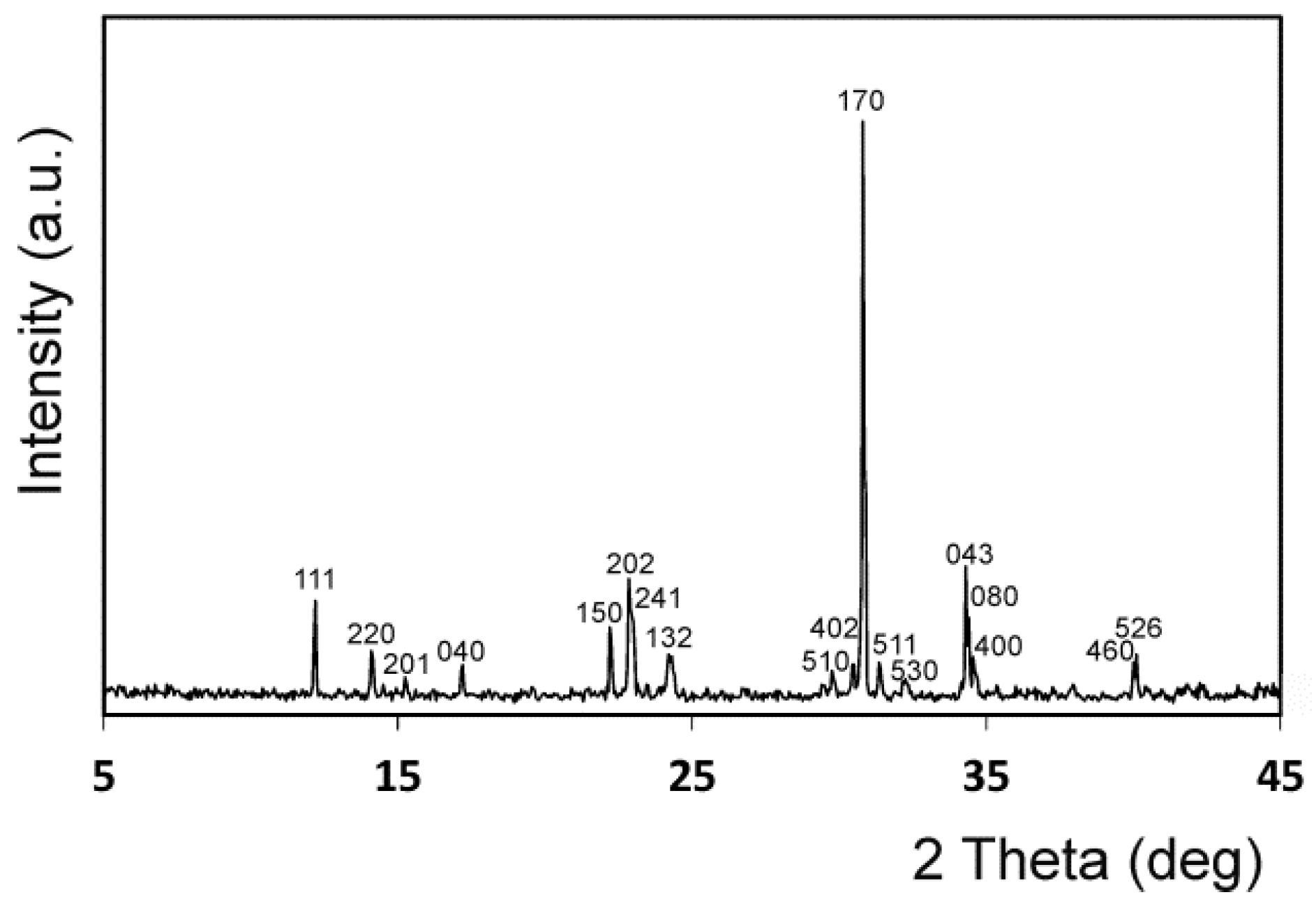
3.1. Implant Characterization
3.2. In Vivo Implant Characterization
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ros-Tarraga, P.; Mazon, P.; Rodriguez, M.A.; Meseguer-Olmo, L.; De Aza, P.N. Novel resorbable and osteoconductive calcium silicophosphate scaffold induced bone formation. Materials 2016, 9, 785. [Google Scholar] [CrossRef]
- Rabadan-Ros, R.; Velásquez, P.A.; Meseguer-Olmo, L.; De Aza, P.N. Morphological and structural study of a novel porous nurse’s a ceramic with osteoconductive properties for tissue engineering. Materials 2016, 9, 474. [Google Scholar] [CrossRef]
- Karageorgiou, V.; Kaplan, D. Porosity of 3D biomaterial scaffolds and osteogenesis. Biomaterials 2005, 26, 5474–5491. [Google Scholar] [CrossRef] [PubMed]
- De Mulder, E.L.W.; Buma, P.; Hannink, G. Anisotropic Porous Biodegradable Scaffolds for Musculoskeletal Tissue Engineering. Materials 2009, 2, 1674–1696. [Google Scholar] [CrossRef]
- Gerhardt, L.C.; Boccaccini, A.R. Bioactive Glass and Glass-Ceramic Scaffolds for Bone Tissue Engineering. Materials 2010, 3, 3867–3910. [Google Scholar] [CrossRef]
- Ros-Tarraga, P.; Rabadan-Ros, R.; Murciano, A.; Meseguer-Olmo, L.; De Aza, P.N. Assessment of Effects of Si-Ca-P Biphasic Ceramic on the Osteogenic Differentiation of a Population of Multipotent Adult Human Stem Cells. Materials 2016, 9, 969. [Google Scholar] [CrossRef]
- Buerger, M.J. Crystallographic Aspects of Phase Transformations, in Phase Transformations in Solids; Smoluchowski, R., Meyer, R.J.E., Weyl, W.A., Eds.; John Wiley & Sons Inc.: New York, NY, USA, 1951; pp. 183–211. [Google Scholar]
- Elliot, C. Structure and Chemistry of the Apatites and Other Calcium Orthophosphates, 3rd ed.; Elsevier Science: Amsterdam, The Netherlands, 1994. [Google Scholar]
- Carrodeguas, R.G.; De Aza, S. α-tricalcium phosphate: Synthesis, properties and biological applications. Acta Biomater. 2011, 7, 3536–3546. [Google Scholar] [CrossRef] [PubMed]
- Dorozhkin, S.V. Calcium orthophosphate cements for biomedical application. J. Mater. Sci. 2008, 43, 3028–3057. [Google Scholar] [CrossRef]
- Bohner, M. Calcium orthophosphates in medicine: From ceramics to calcium phosphate cements. Injury 2000, 31 (Suppl. D), 37–47. [Google Scholar] [CrossRef]
- Morejón-Alonso, L.; Bareiro, O.J.; Carrodeguas, R.G.; dos Santos, L.A. Bioactive composite bone cement based on α-tricalcium phosphate/tricalcium silicate. J. Biomed. Mater. Res. Part B Appl. Biomater. 2012, 100, 94–102. [Google Scholar] [CrossRef] [PubMed]
- Martinez, I.M.; Velasquez, P.A.; De Aza, P.N. The sub-system α-TCPss-Silicocarnotite within the binary system Ca3(PO4)2–Ca2SiO4. J. Am. Ceram. Soc. 2012, 95, 1112–1117. [Google Scholar] [CrossRef]
- Martinez, I.M.; Velasquez, P.A.; De Aza, P.N. Synthesis and stability of α-tricalcium phosphate doped with dicalcium silicate in the system Ca3(PO4)2–Ca2SiO4. Mater. Charact. 2010, 61, 761–767. [Google Scholar] [CrossRef]
- Martinez, I.M.; Velasquez, P.A.; Meseguer-Olmo, L.; De Aza, P.N. Production and study of in vitro behaviour of monolithic α-Tricalcium Phosphate based ceramics in the system Ca3(PO4)2–Ca2SiO4. Ceram. Int. 2011, 37, 2527–2535. [Google Scholar] [CrossRef]
- Martinez, I.M.; Velasquez, P.A.; Meseguer-Olmo, L.; Bernabeu-Esclapez, A.; De Aza, P.N. Preparation and characterization of novel bioactive α-Tricalcium Phosphate doped with Dicalcium Silicate ceramics. Mater. Sci. Eng. C 2012, 32, 878–886. [Google Scholar] [CrossRef]
- Meseguer-Olmo, L.; Aznar-Cervantes, S.; Mazón, P.; De Aza, P.N. In vitro behaviour of adult mesenchymal stem cells of human bone marrow origin seeded on a novel bioactive ceramics in the Ca2SiO4-Ca3(PO4)2 system. J. Mater. Sci. Mater. Med. 2012, 23, 3003–3014. [Google Scholar] [CrossRef] [PubMed]
- Velasquez, P.; Luklinska, Z.B.; Meseguer-Olmo, L.; Mate-Sanchez de Val, J.E.; Delgado-Ruiz, R.A.; Calvo-Guirado, J.L.; Ramirez-Fernandez, M.P.; de Aza, P.N. αTCP ceramic doped with Dicalcium Silicate for bone regeneration applications prepared by powder metallurgy method. In vitro and in vivo studies. J. Biomed. Mater. Res. A 2013, 101, 1943–1954. [Google Scholar] [CrossRef] [PubMed]
- Mate-Sanchez de Val, J.E.; Calvo-Guirado, J.L.; Delgado-Ruiz, R.A.; Ramirez-Fernandez, M.P.; Martinez, I.M.; Granero-Marin, J.M.; Negri, B.; Chiva-Garcia, F.; Martinez-Gonzalez, J.M.; De Aza, P.N. New block graft of α-TCP with silicon in critical size defects in rabbits: Chemical characterization, histological, histomorphometric and micro-CT study. Ceram. Int. 2012, 38, 1563–1570. [Google Scholar] [CrossRef]
- Mate-Sanchez de Val, J.E.; Calvo-Guirado, J.L.; Delgado-Ruiz, R.A.; Ramirez-Fernandez, M.P.; Negri, B.; Abboud, M.; Martinez, I.M.; De Aza, P.N. Physical properties, mechanical behavior, and electron microscopy study of a new α-tcp block graft with silicon in an animal model. J. Biomed. Mater. Res. A 2012, 100, 3446–3454. [Google Scholar] [CrossRef] [PubMed]
- De Aza, P.N.; Luklinska, Z.B.; Mate-Sanchez de Val, J.E.; Calvo-Guirado, J.L. Biodegradation process of α-tricalcium phosphate and α-tricalcium phosphate solid solution bioceramics in vivo: A comparative study. Microsc. Microanal. 2013, 19, 1350–1357. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Leong, K.F.; Du, Z.; Chua, C.K. The design of scaffolds for use in tissue engineering. Part I. Traditional factors. Tissue Eng. 2001, 7, 679–689. [Google Scholar] [CrossRef] [PubMed]
- De Aza, A.H.; Velasquez, P.; Alemany, M.I.; Pena, P.; De Aza, P.N. In situ bone-like apatite formation from a Bioeutectic® ceramic in SBF dynamic flow. J. Am. Ceram. Soc. 2007, 90, 1200–1207. [Google Scholar] [CrossRef]
- Rezwan, K.; Chen, Q.Z.; Blaker, J.J.; Boccaccini, A.R. Biodegradable and bioactive porous polymer/inorganic composite scaffolds for bone tissue engineering. Biomaterials 2006, 27, 3413–3431. [Google Scholar] [CrossRef] [PubMed]
- Tabata, Y. Biomaterial technology for tissue engineering applications. J. R. Soc. Interface 2009, 6, S311–S324. [Google Scholar] [CrossRef] [PubMed]
- Santos, M.I.; Reis, R.L. Vascularization in Bone Tissue Engineering: Physiology, Current Strategies, Major Hurdles and Future Challenges. Macromol. Biosci. 2010, 10, 12–27. [Google Scholar] [CrossRef] [PubMed]
- Chiu, L.L.Y.; Radisic, M. Scaffolds with covalently immobilized VEGF and Angiopoietin-1 for vascularization of engineered tissues. Biomaterials 2010, 31, 226–241. [Google Scholar] [CrossRef] [PubMed]
- Smith, I.O.; Ren, F.; Baumann, M.J.; Case, E.D. Confocal laser scanning microscopy as a tool for imaging cancellous bone. J. Biomed. Mater. Res. B Appl. Biomater. 2006, 79, 185–192. [Google Scholar] [CrossRef] [PubMed]
- Temenoff, J.S.; Mikos, A.G. Biomaterials: The Intersection of Biology and Materials Science; Pearson International Edition: London, UK, 2008; p. 471. [Google Scholar]
- Honda, M.; Fujimi, T.; Izumi, S.; Izawa, K.; Aizawa, M.; Morisue, H. Topographical analyses of proliferation and differentiation of osteoblasts in micro- and macropores of apatite-fiber scaffold. J. Biomed. Mater. Res. A 2010, 94, 937–944. [Google Scholar] [CrossRef] [PubMed]
- Yunos, D.M.; Bretcanu, O.; Boccaccini, A.R. Polymer-bioceramic composites for tissue engineering scaffolds. J. Mater. Sci. 2008, 43, 4433–4442. [Google Scholar] [CrossRef]
- Baino, F.; Ferraris, M.; Bretcanu, O.; Verné, E.; Vitale-Brovarone, C. Optimization of composition, structure and mechanical strength of bioactive 3-D glass-ceramic scaffolds for bone substitution. J. Biomater. Appl. 2013, 27, 872–890. [Google Scholar] [CrossRef] [PubMed]
- Baino, F.; Caddeo, F.S.; Novajra, G.; Vitale-Brovarone, C. Using porous bioceramic scaffolds to model healthy and osteoporotic bone. J. Eur. Ceram. Soc. 2016, 36, 2175–2182. [Google Scholar] [CrossRef]
- International Organization for Standardization. ISO 14801—Dentistry—Implants—Dynamic Loading Test for Endosseous Dental Implants, 3rd ed.; International Organization for Standardization: London, UK, 2016. [Google Scholar]
- Hulbert, S.F.; Morrison, S.J.; Klawitter, J.J. Tissue reaction to three ceramics of porous and non-porous structures. J. Biomed. Mater. Res. 1972, 6, 347–374. [Google Scholar] [CrossRef] [PubMed]
- Hollister, S.J.; Maddox, R.D.; Taboas, J.M. Optimal design and fabrication of scaffolds to mimic tissue properties and satisfy biological constraints. Biomaterials 2002, 23, 4095–4103. [Google Scholar] [CrossRef]
- Tadic, D.; Epple, M. Mechanically stable implants of synthetic bone mineral by cold isostatic. Biomaterials 2003, 24, 4565–4571. [Google Scholar] [CrossRef]
- Hench, L.L. The story of Bioglass®. J. Mater. Sci. Mater. Med. 2006, 17, 967–978. [Google Scholar] [CrossRef] [PubMed]
- Carrodeguas, R.G.; de Aza, A.H.; Jimenez, J.; de Aza, P.N.; Pena, P.; López-Bravo, A.; de Aza, S. Preparation and in vitro characterization of wollastonite doped tricalcium phosphate bioceramics. Key Eng. Mater. 2008, 361–363, 237–240. [Google Scholar] [CrossRef]
- Pernelle, K.; Imbert, L.; Bosser, C.; Auregan, J.C.; Cruel, M.; Ogier, A. Microscale mechanical and mineral heterogeneity of human cortical bone governs osteoclast activity. Bone 2017, 94, 42–49. [Google Scholar] [CrossRef] [PubMed]
- Carrodeguas, R.G.; De Aza, A.H.; Turrillas, X.; Pena, P.; De Aza, S. New approach to the β→α polimorphic transformation in magnesium-substituted tricalcium phosphate and its practical implications. J. Am. Ceram. Soc. 2008, 91, 1281–1286. [Google Scholar] [CrossRef]
- García-Paez, I.H.; Garcıa Carrodeguas, R.; de Aza, A.H.; Baudin, C.; Pena, P. Effect of Mg and Si co-substitution on microstructure and strength of tricalcium phosphate ceramics. J. Mech. Behav. Biomed. Mater. 2014, 30, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Zilm, M.; Thomson, S.D.; Wei, M.A. Comparative Study of the Sintering Behavior of Pure and Manganese-Substituted Hydroxyapatite. Materials 2015, 8, 6419–6436. [Google Scholar] [CrossRef]
- Pina, S.; Ferreira, J.M.F. Brushite-Forming Mg-, Zn- and Sr-Substituted Bone Cements for Clinical Applications. Materials 2010, 3, 519–535. [Google Scholar] [CrossRef]
- O’Donnell, M.D.; Hill, R.G. Influence of strontium and the importance of glass chemistry and structure when designing bioactive glasses for bone regeneration. Acta Biomater. 2010, 6, 2382–2385. [Google Scholar] [CrossRef] [PubMed]
- Cannillo, V.; Sola, A. Potassium-based composition for a bioactive glass. Ceram Int. 2009, 35, 3389–3393. [Google Scholar] [CrossRef]
- Ros-Tárraga, P.; Murciano, A.; Mazón, P.; Gehrke, S.A.; De Aza, P.N. In vitro behaviour of sol-gel interconnected porous scaffolds of doped wollasonite. Ceram. Int. 2017, 43, 11034–11038. [Google Scholar] [CrossRef]
- Aina, V.; Malavasi, G.; Fiorio Pla, A.; Munaron, L.; Morterra, C. Zinc-containing bioactive glasses: Surface reactivity and behaviour towards endothelial cells. Acta Biomater. 2009, 5, 1211–1222. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.; Lin, K.; Wang, Z.; Chang, J.; Wang, L.; Lu, J. Reconstruction of calvarial defect of rabbits using porous calcium silicate bioactive ceramics. Biomaterials 2008, 29, 2588–2596. [Google Scholar] [CrossRef] [PubMed]
- Serena, S.; Caballero, A.; De Aza, P.N.; Sainz, M.A. New evaluation of the in vitro response of silicocarnotite monophasic material. Ceram. Int. 2015, 41, 9411–9419. [Google Scholar] [CrossRef]
- Lugo, G.J.; Mazón, P.; De Aza, P.N. Phase transitions in single phase Si-Ca-P-based ceramic under thermal treatment. J. Eur. Ceram. Soc. 2015, 35, 3693–3700. [Google Scholar] [CrossRef]




| Test Group | Control | |||||||
|---|---|---|---|---|---|---|---|---|
| % | 15 Days | 30 Days | 60 Days | p Values * | p Values ** | 15 Days | 30 Days | 60 Days |
| BIC | 54.34 ± 0.32 (54.34) | 60.33 ± 0.13 (60.33) | 68.32 ± 1.21 ** (68.32) * | 0.014 | 0.038 | 0.00 ± 0.0 | 0.00 ± 0.0 | 0.00 ± 0.0 |
| New Bone | 48.83 ± 1.32 (48.83) | 52.26 ± 0.95 * (52.26) | 60.11 ± 2.01 ** (60.11) * | 0.028 | 0.011 | 26.07 ± 0.05 (26.07) | 26.26 ± 0.43 * (26.26) | 27.10 ± 0.32 ** (27.10) * |
| Residual | 32.18 ± 1.75 (32.18) | 29.94 ± 13 * (29.94) | 23.75 ± 0.85 (23.75) | 0.037 | 0.029 | 0.00 ± 0.0 | 0.00 ± 0.0 | 0.00 ± 0.0 |
| Defect Closure | 58.63 ± 1.03 (58.63) | 66.24 ± 4.06 (66.24) | 79.01 ± 94 ** (79.01) * | 0.015 | 0.014 | 10.87 ± 0.23 (10.87) | 25.56 ± 0.43 * (25.56) | 28.12 ± 0.32 ** (28.12) * |
| Resorption Rate | 35.93 ± 0.32 (25.93) | 42.14 ± 1.63 (32.14) | 53.13 ± 2.47 (40.13) * | 0.023 | 0.026 | 0.00 ± 0.0 | 0.00 ± 0.0 | 0.00 ± 0.0 |
| Connective Tissue | 18.37 ± 1.20 (18.37) | 17.30 ± 3.01 (17.30) | 16.14 ± 1.33 (16.14) | 0.036 | 0.022 | 0.00 ± 0.0 | 0.00 ± 0.0 | 0.00 ± 0.0 |
| (wt %) | O | Ca | P | Si | Ca/P Ratio |
|---|---|---|---|---|---|
| Implant/Scaffold | |||||
| 15 days | 44.73 ± 0.12 (44.73) | 35.01 ± 0.25 (32.01) | 20.25 ± 0.46 (20.25) | 0.11 ± 1.62 (0.11) | 1.74 ± 0.86 (1.74) |
| 30 days | 45.43 ± 0.13 (45.43) | 32.12 ± 0.24 (32.14) | 22.36 ± 0.43 (22.36) | 0.09 ± 1.83 (0.09) | 1.44 ± 0.84 (1.44) |
| 60 days | 48.47 ± 0.12 (48.47) | 29.62 ± 0.26 (29.62) | 21.84 ± 0.35 (21.84) | 0.07 ± 1.82 (0.07) | 1.36 ± 0.76 (1.36) |
| Bone Interfase | |||||
| 15 days | 50.68 ± 0.11 (50.68) | 32.95 ± 0.31 (32.95) | 16.32 ± 0.53 (16.32) | 0.05 ± 1.63 (0.05) | 2.02 ± 0.95 (2.02) |
| 30 days | 55.88 ± 0.10 (55.88) | 30.23 ± 0.29 (30.23) | 13.85 ± 0.54 (13.85) | 0.04 ± 1.33 (0.04) | 2.18 ± 0.84 (2.18) |
| 60 days | 59.15 ± 0.12 (59.15) | 28.62 ± 0.32 (28.62) | 12.21 ± 0.75 (12.21) | 0.02 ± 1.25 (0.02) | 2.34 ± 0.96 (2.34) |
| New bone | |||||
| 15 days | 59.69 ± 0.15 (59.69) | 26.78 ± 0.16 (26.78) | 13.5 ± 0.62 (13.50) | 0.02 ± 1.63 (0.02) | 1.98 ± 0.83 (1.98) |
| 30 days | 60.56 ± 0.09 (60.56) | 25.42 ± 0.24 (25.42) | 14.01 ± 0.53 (14.01) | 0.01 ± 1.71 (0.01) | 1.81 ± 0.91 (1.81) |
| 60 days | 63.38 ± 0.09 (63.38) | 24.36 ± 0.15 (24.36) | 12.25 ± 0.74 (12.25) | 0.01 ± 1.03 (0.01) | 1.98 ± 0.97 (1.98) |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
De Aza, P.N.; Rodríguez, M.A.; Gehrke, S.A.; Maté-Sánchez de Val, J.E.; Calvo-Guirado, J.L. RETRACTED: A Si-αTCP Scaffold for Biomedical Applications: An Experimental Study Using the Rabbit Tibia Model. Appl. Sci. 2017, 7, 706. https://doi.org/10.3390/app7070706
De Aza PN, Rodríguez MA, Gehrke SA, Maté-Sánchez de Val JE, Calvo-Guirado JL. RETRACTED: A Si-αTCP Scaffold for Biomedical Applications: An Experimental Study Using the Rabbit Tibia Model. Applied Sciences. 2017; 7(7):706. https://doi.org/10.3390/app7070706
Chicago/Turabian StyleDe Aza, Piedad N., Miguel A. Rodríguez, Sergio A. Gehrke, José E. Maté-Sánchez de Val, and Jose L. Calvo-Guirado. 2017. "RETRACTED: A Si-αTCP Scaffold for Biomedical Applications: An Experimental Study Using the Rabbit Tibia Model" Applied Sciences 7, no. 7: 706. https://doi.org/10.3390/app7070706
APA StyleDe Aza, P. N., Rodríguez, M. A., Gehrke, S. A., Maté-Sánchez de Val, J. E., & Calvo-Guirado, J. L. (2017). RETRACTED: A Si-αTCP Scaffold for Biomedical Applications: An Experimental Study Using the Rabbit Tibia Model. Applied Sciences, 7(7), 706. https://doi.org/10.3390/app7070706








