CERN-MEDICIS (Medical Isotopes Collected from ISOLDE): A New Facility
Abstract
:1. Introduction
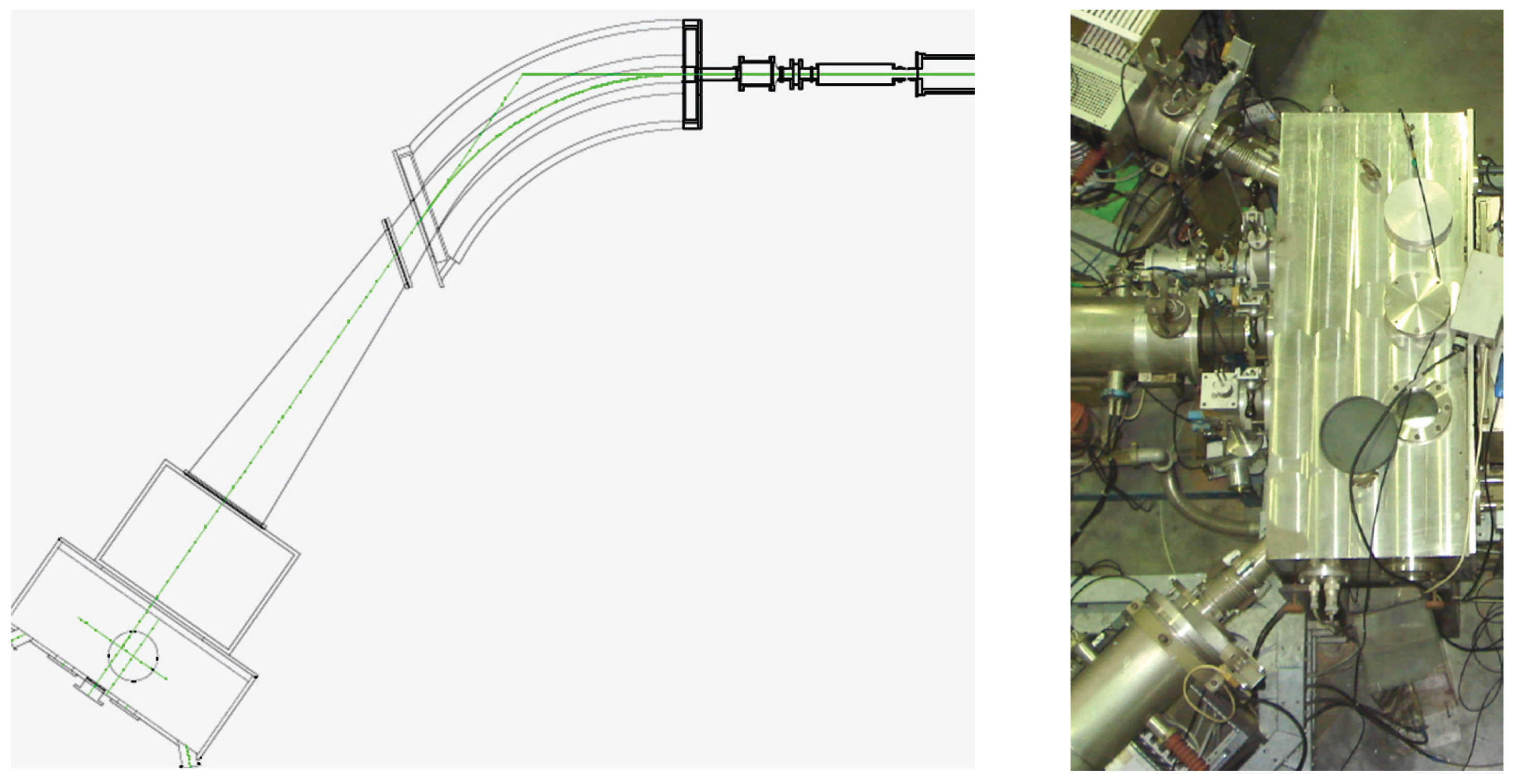
2. CERN-MEDICIS Facility
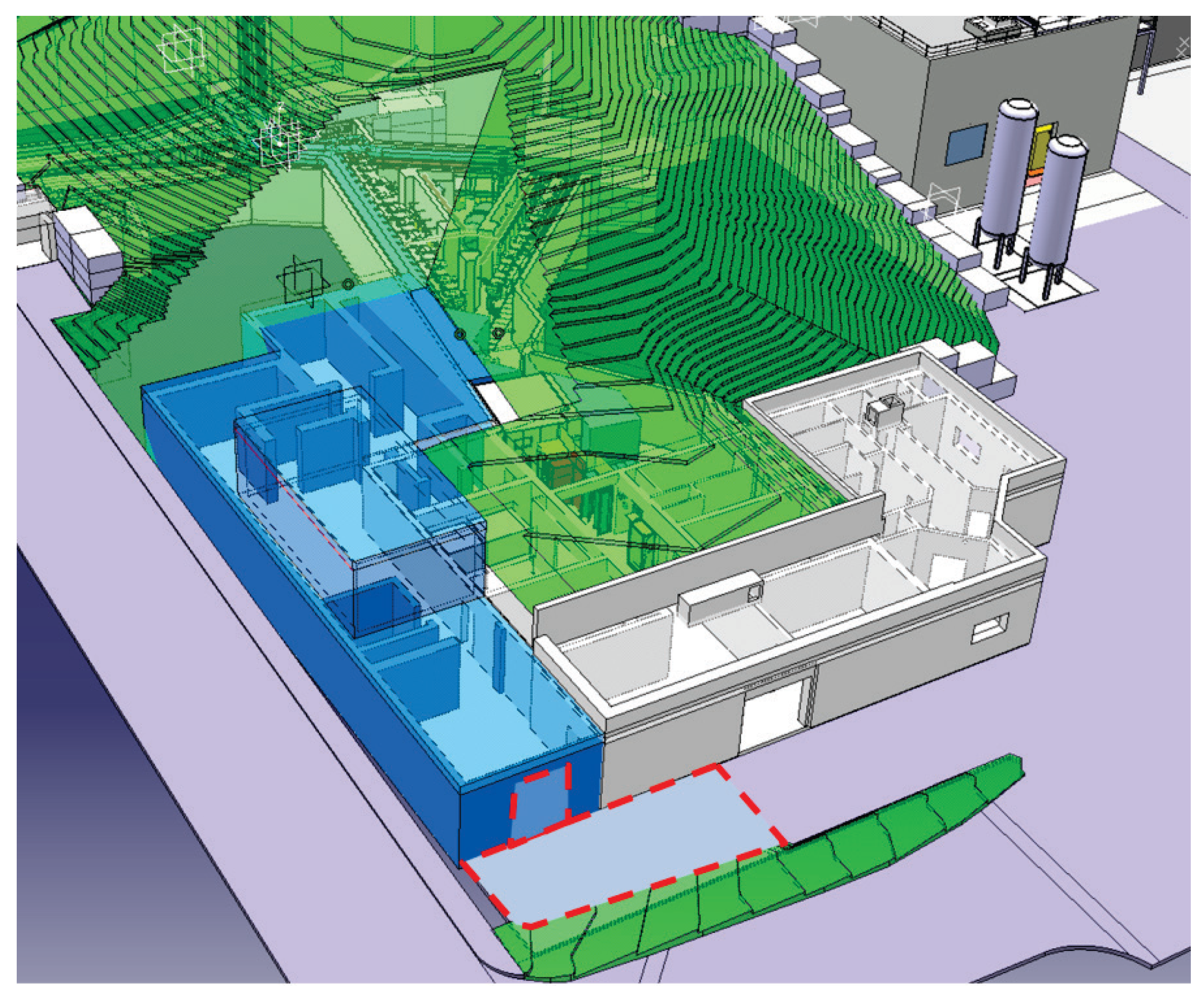
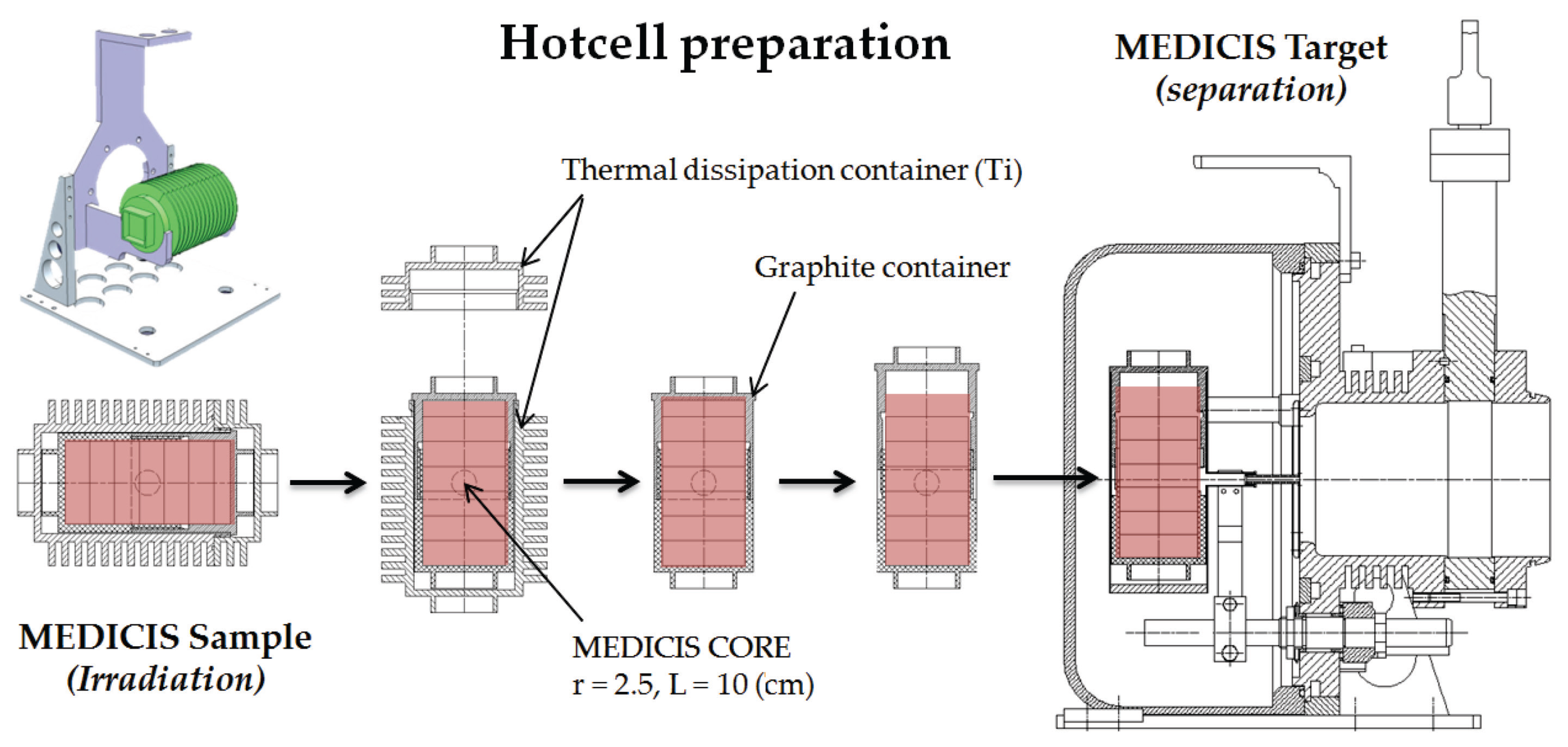
- A rail conveyor system (RCS) based on an industrial system, modified to bring the CERN-MEDICIS sample from the ISOLDE irradiation area to a shielded decay area. Once there, a robot acquired from the KUKA company (Augsburg, Germany) completes the target transport cycle.
- A heavy concrete hot-cell, for target preparation, and a bunker equipped with a mass separator, provided by the Katholieke Universiteit (KU) Leuven, for radioactive isotope mass separation.
| Target material | ISOLDE | CERN-MEDICIS |
|---|---|---|
| Ti | 0.80 | 2.5 |
| Y2O3 | 1.7 | 2.3 |
| Ta | 2.0 | 6.0 |
| UC2 | 3.5 | 7.0 |
| Medical Application | Isotope half-life | Parent isotope beam | Target ion source | ISOLDE † | RIB εext ** (%) | CERN-MEDICIS † | CERN-MEDICIS 2GeV 6 µA | Comments | ||||
| In-target | In-target ActivityEOB (Bq) | Extracted Activity#EOB (Bq) | Possible gain εext (%) | Extracted Activity#EOB (Bq) | ||||||||
| Production rate (pps) | ActivityEOB (Bq) | |||||||||||
| α,β therapy/SPECT/ | 213Bi 45.6 m | 225Ac | UCX-Re | 1.5 E9 * | 7.2 E8 | 221Fr 10 | 2.8 E8 | 2.8 E7 | 50 | 4.2 E8 | Only mass separation | |
| α,β therapy | 212Bi 60.6 m | 224Ac | UCX-Re | 1.5 E9* | 1.4 E9 | 220Fr 10 | 1.7 E9 | 1.7 E8 | 50 | 2.5 E9 | Only mass separation | |
| β therapy | 177Lu 6.7 d | 177Lu RILIS/VD | Ta-Re/Re-VD5 | 3.3 E9 | 7.4 E8 | 177Lu 1 | 6.4 E8 | 6.4 E6 | 20 | 1.7 E8 | Chemical purification | |
| Auger therapy | 166Yb 56.7 h | 166Yb | Ta-Re | 1.4 E10 | 5.4 E10 | 166Yb 5 | 4.1 E10 | 2.1 E9 | 20 | 1.1 E10 | Chemical purification | |
| β therapy | 166Ho 25.8 h | 166Ho | Ta-Re | 1.4 E7 | 1.2 E7 | 166Ho 5 | 9.6 E6 | 4.8 E5 | 20 | 6.0 E6 | Chemical purification | |
| β therapy/Auger therapy | 161Tb 6.9 d | 161Tb | UCX-Re | 2.1 E7 | 2.7 E7 | 161Tb 5 | 1.9 E7 | 9.5 E5 | 20 | 5.4 E6 | Chemical purification | |
| PET | 156Tb 5.35 d | 156Tb | Ta-Re | 2.5 E8 | 8.9 E7 | 156Tb 1 | 5.5 E7 | 5.5 E5 | 20 | 1.3 E7 | Chemical purification | |
| SPECT/CT diagnosis | 155Tb 5.33 d | 155Dy/Tb | Ta-Re | 3.2 E9/7.4 E8 | 7.9 E9 | 155Dy 1 | 5.3 E9 | 5.3 E7 | 20 | 6.8 E8 | RILIS Dy | |
| β therapy | 153Sm 46.8 h | 153Sm | UCX-Re | 1.5E8 | 2.2 E9 | 153Sm 5 | 2.8 E9 | 1.4 E8 | 20 | 1.0 E9 | Chemical purification | |
| PET/CT | 152Tb 17.5 h | 152Dy/Tb | Ta-Re | 1.3 E10/3.3 E9 | 5.6 E10 | 152Dy 1 | 3.7 E10 | 3.7 E8 | 20 | 2.2 E10 | RILIS Dy | |
| α therapy | 149Tb 4.1 h | 149Tb | Ta-Re | 1.1 E10 | 6.0 E10 | 149Tb 1 | 3.8 E10 | 3.8 E8 | 20 | 2.4 E10 | Chemical purification | |
| 140Pr-PET/Auger therapy | 140Nd 3.4 d | 140Nd | Ta-Re | 1.8 E9 | 2.0 E10 | 140Nd 5 | 1.2 E10 | 6.0 E8 | 20 | 4.0 E9 | Chemical purification | |
| β therapy | 89Sr 50.5 d | 89Sr | UCX-Re | 1.2 E10 | 2.3 E9 | 89Sr 5 | 2.0 E9 | 1.0 E8 | 20 | 2.7 E9 | 5.4 E8 | Only mass separation |
| PET | 82Sr 25.5 d | 82Sr | UCX-Re | 3.6 E10 | 4.6 E9 | 82Sr 5 | 1.7 E9 | 8.5 E7 | 20 | 2.0 E9 | 4.0 E8 | Only mass separation |
| β therapy | 77As 38.8 h | 77As | UCX-VD5 | 5.7 E9 | 1.1 E10 | 77As 5 | 5.8 E9 | 2.9 E8 | 20 | 9.4 E9 | 1.4 E9 | Chemical purification |
| PET | 74As 17.8 d | 74As | Y2O3 -VD5 | 6.5 E9 | 1.2 E9 | 74As 5 | 3.8 E8 | 1.9 E7 | 20 | 4.5 E8 | 9.0 E7 | Chemical purification |
| PET | 72As 26.0 d | 72As | Y2O3-VD5 | 1.6 E10 | 2.8 E10 | 72As 5 | 9.1 E9 | 4.6 E8 | 20 | 1.5 E10 | 3.0 E9 | Chemical purification |
| PET | 71As 65.3 h | 71As | Y2O3-VD5 | 1.8 E10 | 1.8 E10 | 71As 5 | 5.9 E9 | 3.0 E8 | 20 | 8.0 E9 | 1.6 E9 | Chemical purification |
| β therapy | 67Cu 61.9 h | 67Cu | UCX-Re | 2.7 E9 | 3.4 E9 | 67Cu 7 | 1.5 E9 | 1.1 E8 | 20 | 2.7 E9 | 5.4 E8 | Chemical purification |
| PET, dosimetry, therapy | 64Cu 12.7 h | 64Cu | Y2O3-VD5 | 1.1 E10 | 2.3 E10 | 64Cu 5 | 7.1 E9 | 3.6 E8 | 20 | 2.1 E10 | 3.6 E9 | Chemical purification |
| PET | 61Cu 3.3 h | 61Cu | Y2O3-VD5 | 7.7 E9 | 1.7 E10 | 61Cu 5 | 5.1 E9 | 2.6 E8 | 20 | 2.1 E10 | 4.0 E9 | Only mass separation |
| β therapy | 47Sc 3.4 d | 47Sc | Ti | 6.4 E10 | 5.0 E10 | 47Sc 5 | 4.2 E10 | 2.1 E9 | 20 | 5.9 E10 | 1.2 E10 | Evaporation |
| PET | 44Sc 4.0 h | 44Sc | Ti | 4.4 E10 | 6.6 E10 | 44Sc 6.4 | 5.7 E10 | 2.9 E9 | 20 | 1.6 E11 | 3.2 E10 | Evaporation |
| PET | 11C 20.3 m | 11CO | NaF-LiF-VD5 ◊ | - | - | - 15 | - | 1.4 E9 | - | - | 4.2 E9 | Only mass separation |
| Operation Phase | Milestone | Date |
|---|---|---|
| Phase I | Commissioning: no beam | end 2015 |
| Phase II | Commissioning with beam and light targets to gain operational experience | 2016 |
| Phase II B | Isotope production with light targets | middle 2016 |
| Phase III | Extending to heavy targets up to tantalum | end 2016 |
| Phase IV | Collection of short-lived alpha emitters (e.g., 149Tb) | 2017 |
| Phase IV B | Operation with lasers | |
| Phase V | Operation with uranium targets/possible proton beam upgrade | 2018 |
3. Expected Production Rates

4. Past and Ongoing Biomedical Activities at ISOLDE/Biomedical Research Performed at ISOLDE
4.1. Past Activities
4.2. Ongoing Research
5. Perspectives
5.1. Newly Established International Collaboration
5.2. New Radioisotopes
5.3. New Bioconjugates and Chelators
5.4. New Approaches in Brachytherapy
6. Conclusions
Acknowledgments
Conflicts of Interest
References
- Stora, T.; Da Visitacao, S.F.; Mathot, S.; Bowen, P. Nanostructured Target for Isotope Production. U.S. Patent 20110235766 A1, 29 September 2009. [Google Scholar]
- Mendonca, T.M.; Hodak, R.; Ghetta, V.; Allibert, M.; Heuer, D.; Noah, E.; Cimmino, S.; Delonca, M.; Gottberg, A.; Kronberger, M.; et al. Production and release of ISOL beams from molten fluoride salt targets. Nucl. Instrum. Methods Phys. Res. Sect. B 2014. submitted for publication. [Google Scholar]
- Battistoni, G.; Muraro, S.; Sala, P.R.; Cerutti, F.; Ferrari, A.; Roesler, S.; Fassò, A.; Ranft, J. The FLUKA code: Description and benchmarking. In Proceedings of the Hadronic Shower Simulation Workshop AIP Conference, Fermilab, Batavia, IL, USA, 6–8 September 2006; Albrow, M., Raja, R., Eds.; American Institute of Physics: Melville, New York, NY, USA, 2007; Volume 896, pp. 31–49. [Google Scholar]
- Ferrari, A.; Sala, P.R.; Fassò, A.; Ranft, J. FLUKA: A Multi-Particle Transport Code; CERN-2005-10; CERN: Geneva, Switzerland, 2005. [Google Scholar]
- Access to the Yield information. Available online: https://oraweb.cern.ch/pls/isolde/query_tgt (accessed on 24 September 2013).
- Stora, T.; Noah, E.; Hodak, R.; Hirsh, T.Y.; Hass, M.; Kumar, V.; Singh, K. Sergey Vaintraub, Pierre Delahaye, Hanna Franberg-Delahaye, Marie-Genevieve Saint-Laurent and Gerard Lhersonneau. A high intensity 6He beam for the β-beam neutrino oscillation facility. EPL 2012, 98, 32001. [Google Scholar] [CrossRef]
- Morley, T.J.; Dodd, M.; Gagnon, K.; Hanemaayer, V.; Wilson, J.; McQuarrie, S.A.; English, W.; Ruth, T.J.; Benard, F.; Schaffer, P. An automated module for the separation and purification of cyclotron-produced 99mTcO4. Nuclear Med. Biol. 2012, 39, 551–559. [Google Scholar] [CrossRef]
- Beyer, G.J. Radioactive ion beams for biomedical research and nuclear medical application. Hyperfine Interact. 2000, 129, 529–553. [Google Scholar] [CrossRef]
- Beyer, G.J.; Münze, R.; Fromm, W.D.; Franke, W.G.; Henke, H.; Khalkin, V.A.; Lebedev, N.A. Spallation-produced thulium-167 for medical applications. Med. Radionucl. Imaging IAEA 1980, 1, 587–598. [Google Scholar]
- Beyer, G.J.; Ravn, H.L.; Huang, Y.; ISOLDE collaboration. A new type of 81Rb-81mKr generator for medical use. J. Appl. Radiat. Isot. 1984, 35, 1075–1076. [Google Scholar] [CrossRef]
- Beyer, G.J.; Bergmann, R. Workshop on Endoradionuclide Therapy, Dresden, Germany, 30 October–2 November 1990.
- Beyer, G.J.; Offord, R.; Künzi, G.; Aleksandrova, Y.; Ravn, U.; Jahn, S.; Backe, J.; Tenblad, O.; Lindroos, M.; ISOLDE Collaboration. The influence of EDTMP-concentration on the biodistribution of radio-lanthanides and 225-Ac in tumor-bearing mice. Nucl. Med. Biol. 1997, 24, 367–372. [Google Scholar] [CrossRef]
- Proposal to the ISOLDE committee, CERN/ISC-4/P42 (ISOLDE experiment ISC330). Available online: https://cds.cern.ch/record/5299?ln=en (accessed on 14 April 2014).
- Proposal to the ISOLDE committee, CERN/ISC93-14/ISC/P48 (ISOLDE experiment ISC331). Available online: https://cds.cern.ch/record/5298?ln=en (accessed on 14 April 2014).
- Addendum to the proposal to the ISOLDE committee, CERN/ISC93-14/ISC/P48. Available online: http://cds.cern.ch/record/297279/ (accessed on 14 April 2014).
- Addendum 1 to ISOLDE experiments ISC330 and ISC331 from 4 May 1994. Available online: https://cds.cern.ch/record/294853/files/SC00000194.pdf (accessed on 14 April 2014).
- Pochon, S.; Buchegger, F.; Pélegrin, A.; Mach, J.P.; Offord, R.E.; Ryser, J.E.; Rose, K. A novel derivative of the chelon desferrioxamine for site specific conjugation to antibodies. Int. J. Cancer 1989, 43, 1188–1194. [Google Scholar] [CrossRef]
- Beyer, G.J.; Offord, R.E.; Künzi, G.; Jones, R.M.L.; Ravn, U.; Alexandrova, Y.; Werlen, R.C.; Mäcke, H.; Lindroos, M.; Jahn, S.; et al. Biokinetics of monoclonal antibodies labeled with radio-lanthanides and 225-Ac in xenografted nude mice: Preliminary results. J. Lab. Comp. Radiopharm. 1995, 37, 529–530. [Google Scholar]
- Beyer, G.J.; Offord, R.E.; Warlen, R.; Künzi, G.; Alexandrova, Y.; Mäcke, H.; ISOLDE collaboration. Comparison of the biokinetics of octreotide labeled with radio-lanthanides and 111In in tumor bearing rats. Eur. J. Nucl. Med. 1996, 23, 1029–1132. [Google Scholar]
- Beyer, G.J.; Morel, C.; Slosman, D.; Sakar, S.; Allen, B.J.; ISOLDE collaboration. Is Tb-152 suitable to monitor tissue doses in alpha therapy with Tb-149 using PET? Eur. J. Nucl. Med. 1998, 25, 837–1196. [Google Scholar] [CrossRef]
- Beyer, G.J.; Miederer, M.; Vranješ-Durić, S.; Čomor, J.J.; Künzi, G.; Hartley, O.; Senekowitsch-Schmidtke, R.; Soloviev, D.; Buchegger, F.; ISOLDE collaboration. Targeted alpha therapy in vivo: Direct evidence for single cancer cell kill using 149Tb-rituximab. Eur. J. Nucl. Med. Mol. Imaging 2004, 31, 547. [Google Scholar]
- Proposal to the ISOLDE committe CERN/97-14/ISC/P-90 (ISOLDE experiment IS363). Available online: https://cds.cern.ch/record/491305?ln=en (accessed on 14 April 2014).
- Addendum 1 to IS363 dated 31 January 2000. Available online: https://cds.cern.ch/record/430342?ln=en (accessed on 14 April 2014).
- Beyer, G.J. Medical application of Radioactive ion beams. In Presented at NuPEC, CERN, Geneva Switzerland, 10–12 October 2005.
- Müller, C.; Zhernosekov, K.; Köster, U.; Johnston, K.; Dorrer, H.; Hohn, A.; van der Walt, N.T.; Türler, A.; Schibli, R. A unique matched quadruplet of terbium radioisotopes for PET and SPECT and for α- and β-Radionuclide Therapy: An in vivo proof-of-concept study with a new receptor-targeted folate derivative. J. Nucl. Med. 2012, 53, 1951–1959. [Google Scholar] [CrossRef]
- Buchegger, F.; Tourwe, D.; Kosinski, M.; Viertl, D.; Stora, T.; Buehler, L.; Miralbell, R.; Prior, J. 68Ga-labeled NODAGA- and DOTA-neurotensin analogs for targeting breast, prostate and pancreas cancers. Available online: http://www.radiologiekongress.ch/cms/website.php?id=/de/attendees/program.htm (accessed on 14 April 2014).
- Butz, T.; Saibene, S.; Fraenzke, T.; Weber, M. A TDPAC-camera. Nucl. Instrum. Methods Phys. Res. Sect. A 1989, 284, 417–421. [Google Scholar] [CrossRef]
- Proposal to the ISOLDE committee CERN/ISC91-92/ISC/P20. Available online: https://cds.cern.ch/record/5746?ln=en (accessed on 14 April 2014).
- Butz, T.; Tröger, W.; Messerchmidt, A.; Thoenes, U.; Huber, R. 199mHg-derivatives of ascorbate oxidase and laccase: Selective depletion and blocking of Cu-sites. Hyperfine Interact 1993, 80, 1127–1132. [Google Scholar] [CrossRef]
- Tröger, W.; Lippert, C.; Schmidt, P.; Schmidt, U.; Butz, T.; Hoffman, R.; Zeppezauer, M.; ISOLDE collaboration. Hg-coordination studies of cysteine containing oligopeptides. Z. Naturforsch. A 1996, 51, 427–430. [Google Scholar]
- Tröger, W.; Lippert, C.; Butz, T.; Sigfriddsson, K.; Hannson, Ö.; Mclaughhlin, E.; Bauer, R.; Danielsen, E.; Hemmingsen, L.; Bjerrum, M.J.; ISOLDE collaboration. Small scale intramolecular flexibility in 111mCd-plastocyanin. Z. Naturforsch. A 1996, 51, 431–436. [Google Scholar]
- Ctortecka, B.; Tröger, W.; Mallion, S.; Butz, T.; Hoffmann, R. Hg-coordination studies of oligopeptides containing cysteine, histidine and tyrosine by 199mHg-TDPAC. Hyperfine Interact. 1999, 120–12, 737–743. [Google Scholar]
- Soldner, T.; Tröger, W.; Butz, T.; Blaha, P.; Schwarz, K.; ISOLDE collaboration. Measurement and calculation of electric field gradients in Hg-mercaptides. Z. Naturforsch. A 1998, 53, 404–410. [Google Scholar]
- Iranzo, O.; Thulstrup, P.W.; Ryu, S-b.; Hemmingsen, L.; Pecoraro, V.L. The application of 199Hg NMR and 199m Perturbed Angular Correlation (PAC) Spectroscopy to define the biological chemistry of Hg(II): A case study with designed two- and three-stranded coiled coils. Eur. J. Chem. 2007, 13, 9178–9190. [Google Scholar]
- Chakraborty, S.; Kravitz, J.Y.; Thulstrup, P.W.; Hemmingsen, L.; de Grado, W.F.; Pecoraro, V.L. Design of a three-helix bundle capable of binding heavy metals in a triscysteine environment. Angew. Chem. 2011, 50, 2049–2053. [Google Scholar] [CrossRef]
- Luczkowski, M.; Zeider, B.A.; Hinz, A.V.H.; Stachura, M.; Chakraborty, S.; Hemmingsen, L.; Huffman, D.L.; Pecoraro, V.L. Probing the coordination environment of the human copper chaperone HAH1: Characterization of Hg(II)-bridged homodimeric species in solution. Eur. J. Chem. 2013, 19, 9042–9049. [Google Scholar] [CrossRef]
- Gottberg, A.; Stachura, M.; Kowalska, M.; Bissell, M.L.; Arcisauskaite, V.; Blaum, K.; Helmke, A.; Johnston, K.; Kreim, K.; Larsen, F.H.; et al. Billion-fold sensitivity improvement in NMR spectroscopy of magnesium ions in solution. Angew. Chem. Unpublished work. 2014. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Dos Santos Augusto, R.M.; Buehler, L.; Lawson, Z.; Marzari, S.; Stachura, M.; Stora, T.; CERN-MEDICIS collaboration. CERN-MEDICIS (Medical Isotopes Collected from ISOLDE): A New Facility. Appl. Sci. 2014, 4, 265-281. https://doi.org/10.3390/app4020265
Dos Santos Augusto RM, Buehler L, Lawson Z, Marzari S, Stachura M, Stora T, CERN-MEDICIS collaboration. CERN-MEDICIS (Medical Isotopes Collected from ISOLDE): A New Facility. Applied Sciences. 2014; 4(2):265-281. https://doi.org/10.3390/app4020265
Chicago/Turabian StyleDos Santos Augusto, Ricardo Manuel, Leo Buehler, Zoe Lawson, Stefano Marzari, Monika Stachura, Thierry Stora, and CERN-MEDICIS collaboration. 2014. "CERN-MEDICIS (Medical Isotopes Collected from ISOLDE): A New Facility" Applied Sciences 4, no. 2: 265-281. https://doi.org/10.3390/app4020265
APA StyleDos Santos Augusto, R. M., Buehler, L., Lawson, Z., Marzari, S., Stachura, M., Stora, T., & CERN-MEDICIS collaboration. (2014). CERN-MEDICIS (Medical Isotopes Collected from ISOLDE): A New Facility. Applied Sciences, 4(2), 265-281. https://doi.org/10.3390/app4020265




