A Review of the Role of Amphiphiles in Biomass to Ethanol Conversion
Abstract
:1. Introduction
| Pretreatment | Substrate | Additive amount (g/g glucan) | Additive | Cellulase dose (FPU/g glucan) | Increase in Yg (%) | References |
|---|---|---|---|---|---|---|
| N/A | Avicel | 0.30 | PEG 6000 | 8.6 | 3.0 | Eckard et al. [36] |
| Avicel | 0.60 | Tween 20 | 7.5 | 64.0 | Kumar and Wyman [26] | |
| Avicel | 0.30 | Tween 20 | 8.6 | 12.0 | Eckard et al. [37] | |
| Avicel | 0.06 | PEG 4000 | 15.0 | 51.0 | Errikson et al. [27] | |
| Avicel | 0.50 | Casein | 15.0 | 0.0 | Eckard et al. [42] | |
| N/A | Newspaper | 0.60 | Tween 20 | 2.0 * | 55.0 | Wu and Ju [40] |
| Newspaper | 0.60 | Tween 80 | 2.0 * | 42.9 | Wu and Ju [40] | |
| Newspaper | 0.60 | F 68 | 2.0 * | 64.2 | Wu and Ju [40] | |
| Newspaper | 0.60 | F 88 | 2.0 * | 63.7 | Wu and Ju [40] | |
| AFEX | Corn stover | 0.60 | BSA | 7.5 | 11.6 | Kumar and Wyman [26] |
| Corn stover | 0.60 | Tween 20 | 7.5 | 10.3 | Kumar and Wyman [26] | |
| Corn stover | 0.60 | PEG 6000 | 7.5 | 6.5 | Kumar and Wyman [26] | |
| Corn stover | 0.50 | Casein | 25.0 | 17.4 | Eckard et al. [42] | |
| Corn stover | 2.50 | Casein | 25.0 | 23.7 | Eckard et al. [42] | |
| Dilute acid | Corn stover | 0.60 | BSA | 3.0 | 26.3 | Kumar and Wyman [26] |
| Corn stover | 1.40 | BSA | 15.0 | 12.1 | Yang and Wyman [35] | |
| Corn stover | 1.40 | BSA | 7.5 | 12.1 | Yang and Wyman [35] | |
| Corn stover | 0.60 | Tween 20 | 3.0 | 36.0 | Kumar and Wyman [26] | |
| Corn stover | 0.60 | PEG | 3.0 | 45.8 | Kumar and Wyman [26] | |
| Corn stover | 0.50 | Casein | 25.0 | 31.9 | Eckard et al. [42] | |
| Corn stover | 2.50 | Casein | 25.0 | 31.0 | Eckard et al. [42] | |
| Lime | Corn stover | 0.60 | Tween 20 | 7.5 | 20.0 | Kumar and Wyman [26] |
| Corn stover | 0.60 | BSA | 7.5 | 3.0 | Kumar and Wyman [26] | |
| Corn stover | 0.60 | PEG | 7.5 | 0.0 | Kumar and Wyman [26] | |
| Corn stover | 0.50 | Casein | 25.0 | 17.0 | Eckard et al. [42] | |
| Corn stover | 2.50 | Casein | 25.0 | 22.3 | Eckard et al. [42] | |
| Alkali | Corn stover | 0.15 | Triton X-100 | 15.0 | 0.0 | Eckard et al. [43] |
| Corn stover | 0.30 | Brij 30 | 15.0 | 0.0 | Eckard et al. [43] | |
| Corn stover | 0.47 | Tween 20 | 15.0 | 0.0 | Eckard et al. [43] | |
| Corn stover | 0.50 | Casein | 25.0 | 22.7 | Eckard et al. [42] | |
| Corn stover | 2.50 | Casein | 25.0 | 24.8 | Eckard et al. [42] | |
| Extrusion | Corn stover | 0.47 | Tween 20 | 8.6 | 27.5 | Eckard et al. [37] |
| Corn stover | 0.51 | PEG 6000 | 8.6 | 25.4 | Eckard et al. [36] | |
| Corn stover | 0.50 | Casein | 25.0 | 29.5 | Eckard et al. [42] | |
| Corn stover | 2.50 | Casein | 25.0 | 38.4 | Eckard et al. [42] | |
| Raw | Corn stover | 0.15 | BSA | 7.5 | 22.5 | Kumar and Wyman [26] |
| Corn stover | 0.30 | Tween 20 | 7.5 | 16.7 | Kumar and Wyman [26] | |
| Corn stover | 0.15 | PEG | 7.5 | 22.2 | Kumar and Wyman [26] | |
| Corn stover | 0.50 | Casein | 25.0 | 14.4 | Eckard et al. [42] | |
| Corn stover | 2.50 | Casein | 25.0 | 23.5 | Eckard et al. [42] |
1.1. Impact of Amphiphiles on Hydrolysis and Fermentation of Biomass
1.2. Impact of Amphiphiles on Enzyme Recycling
2. Mechanism of Action
2.1. Impact of Amphiphiles as Biomass Modifier
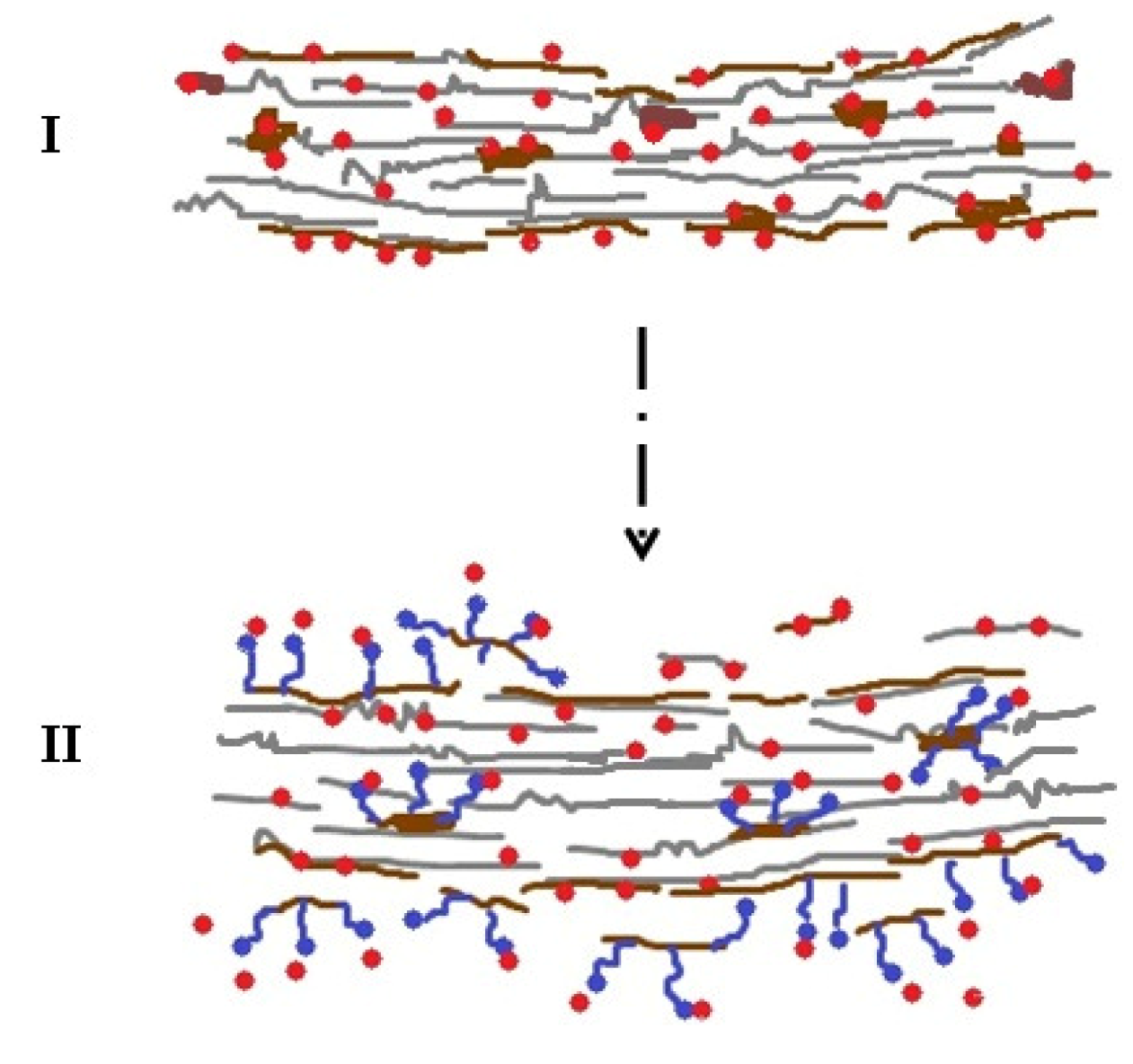
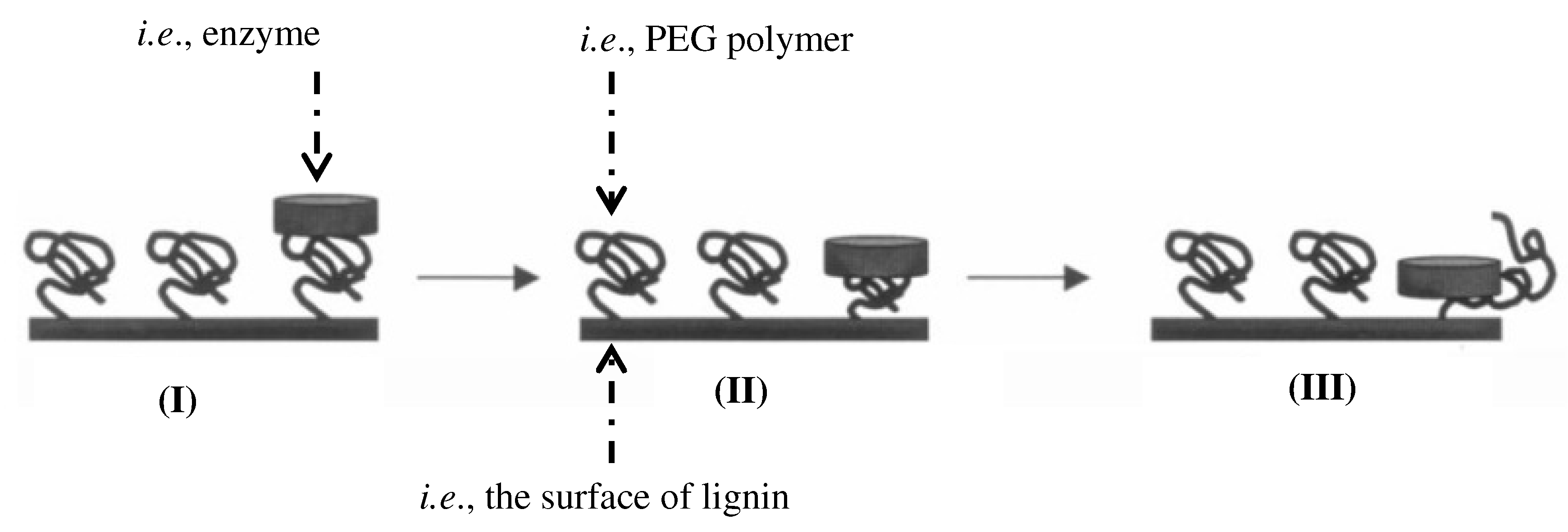
2.2. Impact of Amphiphiles on Enzyme Desorption
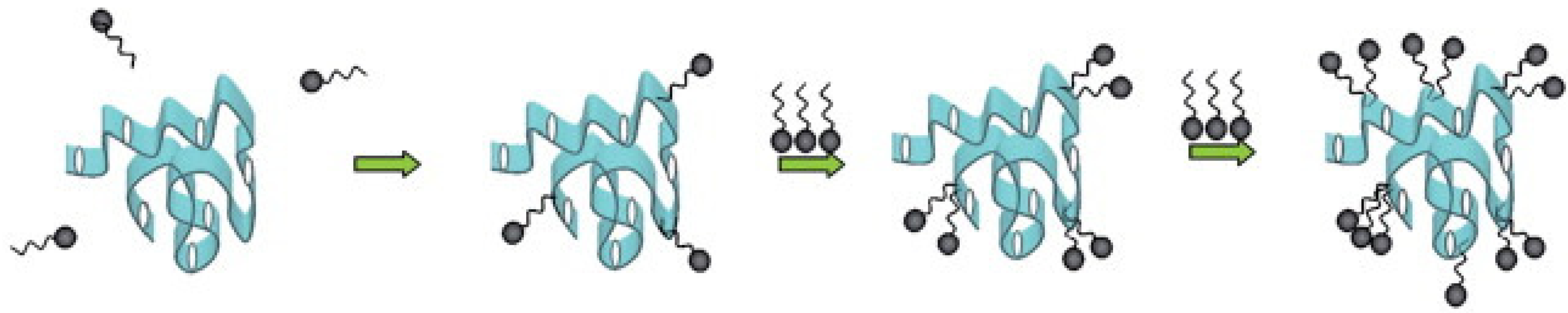
2.3. Impact of Amphiphiles on Enzyme Structure
2.4. Prohibition in Thermal Deactivation
3. Important Factors in Efficacy of Amphiphiles
3.1. Role of Lignin
3.2. Role of Amphiphiles Properties
3.3. Role of Amphiphiles Concentration
3.4. Role of Enzyme Loading
3.5. Role of Temperature
3.6. Role of Time
3.7. Role of Order of Addition
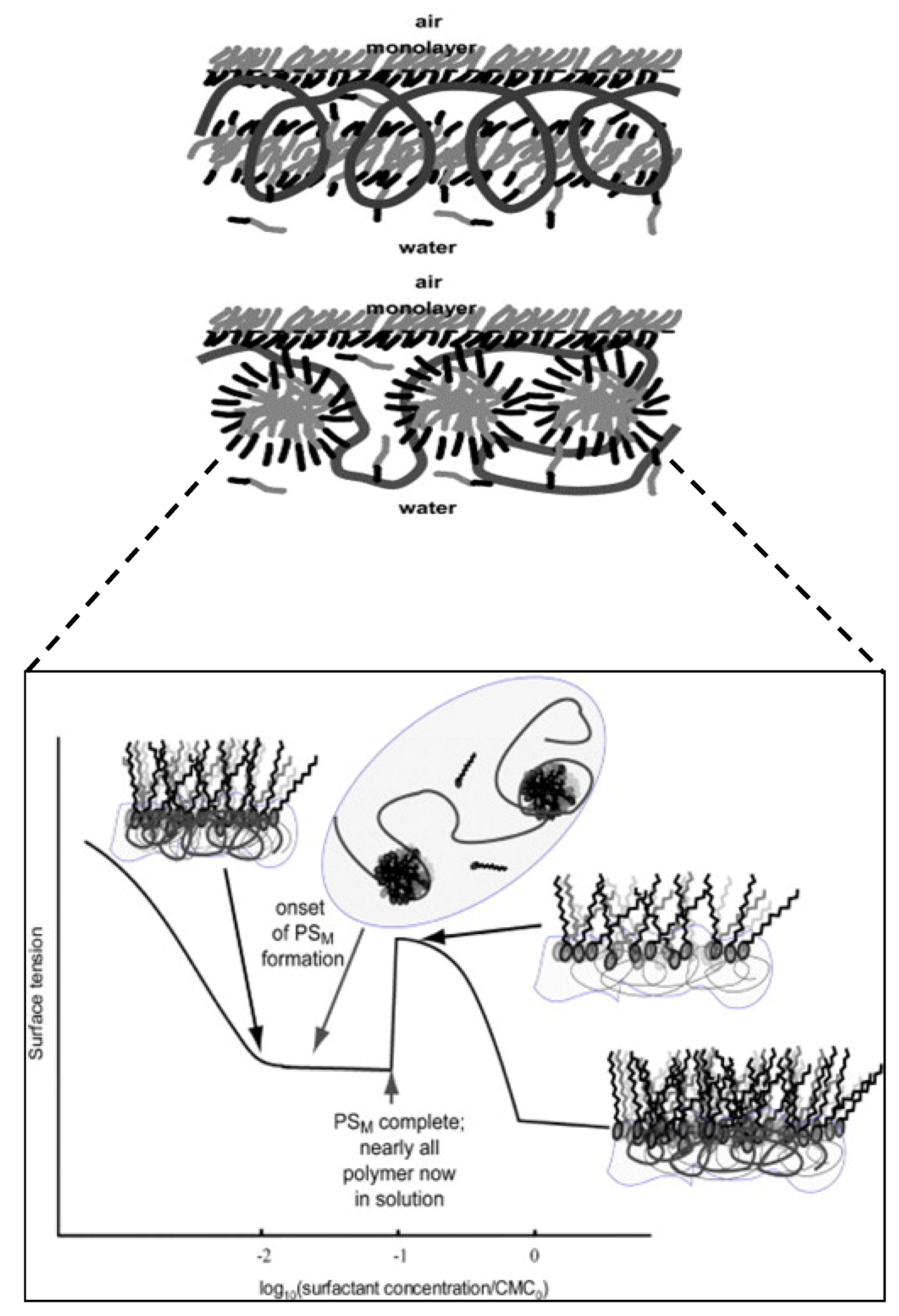
3.8. Role of Mixed Micelles and Polymeric Micelles
4. Conclusions
References
- Yang, B.; Wyman, C.E. BSA treatment to enhance enzymatic hydrolysis of cellulose in lignin containing substrates. Biotechnol. Bioeng. 2006, 94, 611–617. [Google Scholar]
- Ferreira, S.M.P.; Duarte, A.P.; Queiroz, J.A.; Domingues, F.C. Influence of buffer systems on Trichoderma reesei Rut C-30 morphology and cellulase production. Electron. J. Biotechnol. 2009, 12, 1–9. [Google Scholar]
- Ma, A.Z.; Hu, Q.; Qu, Y.B.; Bai, Z.H.; Liu, W.F.; Zhuang, G.Q. The enzymatic hydrolysis rate of cellulose decrease with irreversible adsorption of cellobiohydrolase I. Enzym. Microb. Technol. 2008, 42, 543–547. [Google Scholar] [CrossRef]
- Himmel, M.E.; Ruth, M.F.; Wyman, C.E. Cellulase for commodity products from cellulosic biomass. Curr. Opin. Biotechnol. 1999, 10, 358–364. [Google Scholar]
- Wingren, A.; Galbe, M.; Roslander, C.; Rudolf, A.; Zacch, G. Effect of reduction in yeast and enzyme concentrations in a simultaneous-saccharification-andfermentation-based bioethanol process. Technical and economic evaluation. Appl. Biochem. Biotechnol. 2005, 121, 485–499. [Google Scholar]
- Wyman, C.E. What is (and is not) vital to advancing cellulosic ethanol. Trends Biotechnol. 2007, 25, 153–157. [Google Scholar] [CrossRef]
- Aden, A.; Foust, T. Technoeconomic analysis of the dilute sulfuric acid and enzymatic hydrolysis process for the conversion of corn stover to ethanol. Cellulose 2009, 16, 535–545. [Google Scholar] [CrossRef]
- Lynd, L.R.; Laser, M.S.; Bransby, D.; Dale, B.E.; Davison, B.; Hamilton, R.; Himmel, M.; Keller, M.; McMillan, J.D.; Sheehan, J.; Wyman, C.E. How biotech can transform biofuels. Nat. Biotech. 2008, 26, 169–172. [Google Scholar]
- Dutta, A.; Dowe, N.; Ibsen, K.N.; Schell, D.J.; Aden, A. An economic comparison of different fermentation configurations to convert corn stover to ethanol using Z. mobilis and Saccharomyces. Biotechnol. Prog. 2010, 26, 64–72. [Google Scholar]
- Klein-Marcuschamer, D.; Oleskowicz-Popiel, P.; Simmons, B.A.; Blanch, H.W. The challenge of enzyme cost in the production of lignocellulosic biofuels. Biotechnol. Bioeng. 2011. [Google Scholar] [CrossRef]
- Kazi, F.K.; Fortman, J.A.; Anexm, R.P.; Hsum, D.D.; Adenm, A.; Duttam, A.; Kothandaraman, G. Techno-economic comparison of process technologies for biochemical ethanol production from corn stover. Fuel 2010, 89, S20–S28. [Google Scholar] [CrossRef]
- Li, J.; Li, S.-Z.; Fan, C.-Y.; Yan, Z.-P. The mechanism of polyethylene glycol (PEG) 4000 effect on enzymatic hydrolysis of lignocellulose. Colloid Surf. B 2011, 89, 203–120. [Google Scholar]
- Yang, J.; Zhang, X.P.; Yong, Q.; Yu, S.Y. Three-stage hydrolysis to enhance enzymatic saccharification of steam-exploded corn stover. Bioresour. Technol. 2010, 13, 4930–4935. [Google Scholar] [CrossRef]
- Sathitsuksanoh, N.; Zhu, Z.; Zhang, Y.-H.P. Cellulose solvent- and organic solvent-based lignocellulose fractionation enabled efficient sugar release from a variety of lignocellulosic feedstocks. Bioresour. Technol. 2012, 117, 228–233. [Google Scholar] [CrossRef]
- Qiu, Z.; Aita, G.M.; Walker, M.S. Effect of ionic liquid pretreatment on the chemical composition, structure and enzymatic hydrolysis of energy cane bagasse. Bioresour. Technol 2012, 117, 251–256. [Google Scholar] [CrossRef]
- Holtzapple, M.; Cognata, M.; Shu, Y.; Hendrickson, C. Inhibition of Trichoderma reesei cellulase by sugars and solvents. Biotechnol. Bioeng. 1990, 36, 275–287. [Google Scholar]
- Xiao, Z.Z.; Zhang, X.; Gregg, D.J.; Saddler, J.N. Effects of sugar inhibition on cellulases and beta-glucosidase during enzymatic hydrolysis of softwood substrates. Appl. Biochem. Biotechnol. 2004, 113, 1115–1126. [Google Scholar]
- Qing, Q.; Yang, B.; Wyman, C.E. Xylooligomers are strong inhibitors of cellulose hydrolysis by enzymes. Bioresour. Technol. 2010, 101, 9624–9630. [Google Scholar] [CrossRef]
- Kim, T.H.; Lee, Y.Y.; Sunwoo, C.; Kim, J.S. Pretreatment of corn stover by low liquid ammonia recycle percolation process. Appl. Biochem. Biotechnol. 2006, 133, 41–57. [Google Scholar] [CrossRef]
- Zhu, Y.M.; Kim, T.H.; Lee, Y.Y.; Chen, R.G.; Elander, R.T. Enzymatic production of xylooligosaccharides from corn stover and corn cobs treated with aqueous ammonia. Appl. Biochem. Biotechnol. 2006, 130, 586–598. [Google Scholar] [CrossRef]
- Ximenes, E.; Kim, Y.; Mosier, N.; Dien, B.; Ladisch, M. Deactivation of cellulases by phenols. Enzym. Microb. Technol. 2011, 48, 54–60. [Google Scholar] [CrossRef]
- Kurakake, M.; Ooshima, H.; Kato, J.; Harano, Y. Pretreatment of bagass by non-ionic surfactant for the enzymatic hydrolysis. Bioresour. Technol. 1994, 49, 247–251. [Google Scholar] [CrossRef]
- Sonati, S.; Appu, R.A.G. Kinetic and structural studies on the interaction of surfactants with lipoxygenase L1 from soybeans (glycine max). J. Agric. Food Chem. 1993, 41, 366–371. [Google Scholar] [CrossRef]
- Hayashi, Y.; Talukder, M.M.R.; Wu, J.; Takeyama, T.; Kawanishi, T.; Shimizu, N.J. Increased activity of Chromobacterium viscosum lipase in AOT reverse micelles in the presence of short chain methoxypolyethylene glycol. J. Chem. Technol. Biotechnol. 2001, 76, 844–850. [Google Scholar]
- Talunder, M.M.; Takayama, T.; Hayashi, Y.; Wu, J.C.; Kawanishi, T.; Shimizu, N.; Ogino, C. Improvement of enzyme activity and stability by adding of low molecular weight polyethylene glycol to sodium bis(2-ethyl-L-hexyl) sulfosuccinate/ isooctane reverse micelles. Appl. Biochem. Biotechnol. 2003, 110, 101–111. [Google Scholar] [CrossRef]
- Kumar, R.; Wyman, C.E. Effect of additives on the digestibility of corn stover solid following pretreatment by leading technologies. Biotechnol. Bioeng. 2008, 102, 1544–1557. [Google Scholar]
- Erriksson, T.; Karlsson, J.; Tjerland, F. Mechanism of surfactant effect in enzymatic hydrolysis of lignocellulose. Enzym. Microb. Technol. 2002, 3, 353–364. [Google Scholar]
- Borjesson, J.; Engqvist, M.; Slipos, B.; Tjerneld, F. Effect of polyethylene glycol on enzymatic hydrolysis and adsorption of cellulose enzymes to pretreated lignocellulose. Enzym. Microb. Technol. 2007, 41, 186–195. [Google Scholar] [CrossRef]
- Zheng, Y.; Pan, Z.; Zhang, R.; Wang, D.; Jenkins, B. Non-ionic surfactants and non-catalytic protein. Treatment on enzymatic hydrolysis of pretreated creeping wild ryegrass. Appl. Biochem. Biotechnol. 2008, 146, 231–248. [Google Scholar] [CrossRef]
- Rouimi, S.; Schorsch, C.; Valenitini, C.; Vaslin, S. Foam stability and interfacial properties of milk protein-surfactnt systems. Food Hydrocll. 2005, 19, 467–478. [Google Scholar] [CrossRef]
- Tu, M.; Chandra, R.P.; Sadller, J.N. Recycling cellulase during the hydrolysis of steam exploded and ethanol pretreated lodgepole pine. Biotechnol. Prog. 2007, 65, 1130–1137. [Google Scholar]
- Helle, S.S.; Duff, S.J.B.; Copper, D.G. Effect of surfactants on cellulose hydrolysis. Biotech. Bioeng. 1993, 42, 611–617. [Google Scholar]
- Castanon, M.; Wilke, C.R. Effect of the surfactant Tween 80 on enzymatic hydrolysis of newspaper. Biotechnol. Bioeng. 1981, 23, 1365–1372. [Google Scholar]
- Oshima, H.; Kurakake, M.; Kato, J.; Harano, Y. Enzymatic activity of cellulase adsorbed on cellulose and its change during hydrolysis. Appl. Biochem. Biotechnol. 1991, 31, 253–266. [Google Scholar] [CrossRef]
- Yang, B.; Wyman, C.E. Lignin Blockers and Uses Thereafter. U.S. Patent 7875444, 25 January 2011. [Google Scholar]
- Eckard, A.D.; Muthukumarappan, K.; Gibbons, W. Pretreatment of extruded corn stover with polyethylene glycol to enhance enzymatic hydrolysis: Optimization, kinetics, and mechanism of action. BioEnergy Res. 2011, 5, 424–438. [Google Scholar]
- Eckard, A.D.; Muthukumarappan, K.; Gibbons, W. Modeling of pretreatment condition of extrusion pretreated prairie cordgrass and corn stover with polyoxyethylen (20) sorbitan monolaurate. Appl. Biochem. Biotechnol. 2012, 167, 377–393. [Google Scholar] [CrossRef]
- Ballstros, J.; Olivia, M.; Carascoa, J. Simultaneous Saccharification and Fermentation of Steam-Exploded Poplar Biomass to Ethanol. Appl. Biochem. Biotechnol. 1998, 70–72, 369–381. [Google Scholar] [CrossRef]
- Alkasrawi, M.; Eriksson, T.; Börjesson, T.; Wingren, A.; Galbe, M.; Tjerneld, F.; Zacchi, G. The effect of Tween-20 on simultaneous saccharification and fermentation of softwood to ethanol. Enzym. Microb. Technol. 2003, 33, 71–78. [Google Scholar] [CrossRef]
- Wu, J.; Ju, L.K. Enhancing enzymatic saccharification of waste newsprint by surfactant addition. Biotechnol. Prog. 1998, 14, 649–652. [Google Scholar]
- Yang, B.; Lebanon, W.; Wyman, C.E. Non-Catalytic Additives to Enhance Biodegradation of Cellulosic Biomass. U.S. Patent Provisional Application filed 2009-640-1, 22 April 2009. [Google Scholar]
- Eckard, A.D.; Muthukumarappan, K.; Gibbons, W. Enhanced bioethanol production from pretreated corn stover via multi-positive effect of casein micelles. Bioresour. Technol. 2012. [Google Scholar] [CrossRef]
- Eckard, A.D.; Muthukumarappan, K.; Gibbons, W. Evaluation of the role of polymerized micelles on yield of hydrolysis, chemical changes of biomass and cellulase structure and adsorption. Bioenergy Res. 2013, in press.. [Google Scholar]
- Yang, B.; Deidre, M.W.; Charles, E.W. Changes in the enzymatic hydrolysis rate of Avicel cellulose with conversion. Biotechnol. Bioeng. 2006, 94, 1122–1128. [Google Scholar]
- Huyan, J.K.; Sung, B.K.; Chang, J.K. The effect of nonionic surfactants on the pretreatment and enzymatic hydrolysis of recycled newspaper. Biotechnol. Bioprocess Eng. 2007, 12, 147–151. [Google Scholar] [CrossRef]
- Tu, M.; Zhang, X.; Piace, M.; McFarlance, P.; Saddler, N. Effect of surfactants on separate hydrolysis fermentation and simultaneous saccharification fermentation of pretreated lodgepole pine. Biotechnol. Prog. 2009, 25, 1122–1129. [Google Scholar] [CrossRef]
- Taherzadeh, M.J.; Keikhosro, K. Enzyme-based hydrolysis proceses for ethanol from lignocellulosic material, a review. BioResources 2007, 2, 707–738. [Google Scholar]
- Parke, J.W.; Takahata, Y.; Kajuchi, T.; Akehata, T. Effects of non-ionic surfactant on enzymatic hydrolysis of used newspaper. Biotechnol. Bioeng. 1992, 39, 117–120. [Google Scholar]
- Sinitsyn, A.P.; Mitkevich, O.V.; Klesov, A.A. Inactivation of cellulolytic enzymes by stirring and their stabilization by cellulose. Prikladnaya Biokhimiya i Mikrobiologiya 1986, 22, 759–765. [Google Scholar]
- Palmqvist, E.; Hahn-Hagerdel, B.; Galbe, M.; Zacchi, G. The effect of water-soluble inhibitors from steam-pretreated willow on enzymatic hydrolysis and ethanol fermentation. Enzym. Microb. Technol. 1996, 19, 470–476. [Google Scholar] [CrossRef]
- Xue, Y.; Jameel, H.; Park, S. Strategies to recycle enzyme and their impact on hydrolysis for bioethanol production. Bioresources 2012, 7, 602–615. [Google Scholar]
- Eckard, A.D.; Muthukumarappan, K.; Gibbons, W. Enzyme recycling in a simultaneous & separate saccharification and fermentation of corn stover: Comparing polymeric micelles of surfactants and polypeptides. Bioresour. Technol. 2013, 132, 202–209. [Google Scholar]
- Kaar, W.K.; Holtzapple, M.T. Benefits from Tween during enzymatic hydrolysis of corn stover. Biotechnol. Bioeng. 1998, 59, 419–427. [Google Scholar]
- Seo, D.J.; Fujito, H.; Sakoda, A. Effects of a non-ionic surfactant, Tween 20, on adsorption /desorption of saccharification enzymes onto/from lignocelluloses and saccharification rate. Adsorption 2011, 17, 813–822. [Google Scholar] [CrossRef]
- Lindhoud, S. Polyelectrolyte Complex Micelles as Wrapping for Enzymes. Ph.D. Thesis, Wageningen Universiteit, Wageningen, The Netherlands, September 2009. [Google Scholar]
- Kim, M.H.; Lee, S.B.; Ryu, D.D.Y. Surface deactivation of cellulose and its prevention. Enzym. Microb. Technol. 1982, 4, 99–103. [Google Scholar] [CrossRef]
- Biasutti, M.A.; Abuin, E.B.; Silber, J.J.; Correa, N.M.; Lissi, E.A. Kinetics of reactions catalyzed by enzymes in solutions of surfactants. Adv. Colloid Interface Sci. 2008, 136, 1–24. [Google Scholar] [CrossRef]
- Liu, C.; Wyman, C.E. The effect of flow rate of compressed hot water on xylan, lignin, and total mass removal from corn stover. Ind. Eng. Chem. Res. 2003, 42, 5409–5416. [Google Scholar] [CrossRef]
- Yang, B.; Wyman, C.E. Effect of xylan and lignin removal by batch and flowthrough pretreatment on the enzymatic digestibility of corn stover cellulose. Biotechnol. Bioeng. 2004, 86, 88–95. [Google Scholar]
- Donohoe, B.S.; Decker, S.R.; Tucker, M.; Himmel, M.E.; Vinzant, T.B. Visualizing lignin coalescence and migration through maize cell walls following thermochemical pretreatment. Biotechnol. Bioeng. 2008, 101, 913–925. [Google Scholar]
- Penner, M.H. Expression and measurement of enzyme activity. Curr. Protocol. Food Anal. Chem. 2001. [Google Scholar] [CrossRef]
- Allen, C.; Dos Santos, C.N.; Johnstone, S.A.; Gallagher, R.; Janoff, A.S.; Chiu, G.N.C.; Mayer, L.D.; Shu, Y.; Webb, M.S.; Li, W.M.; Bally1, M.B. Controlling the physical behavior and biological performance of liposome formulations through use of surface grafted poly(ethylene glycol). Biosci. Rep. 2002, 22, 225–249. [Google Scholar] [CrossRef]
- Steels, B.M.; Leermakers, F.A.M.; Haynes, C.A. Analysis of compression of polymer mushrooms using self-consistent field theory. J. Chroma. B 2000, 743, 31–40. [Google Scholar] [CrossRef]
- Pribowo, A.; Arantes, V.; Saddler, J.N. The adsorption and enzyme activity profiles of specific Trichoderma reesei cellulase/xylanase components when hydrolyzing steam pretreated corn stover. Enzym. Microb. Technol. 2012, 50, 193–205. [Google Scholar]
- Chen, N.; Fan, J.B.; Xiang, J.; Chen, J.; Liang, Y. Enzymatic hydrolysis of microcrystalline cellulose in reverse micelles. 2007, 1764, 1029–1035. [Google Scholar]
- Lee, J.H.; Lee, H.B.; Andrade, J.D. Blood compatibility of polyethylene oxide surfaces. Prog. Polym. Sci. 1995, 20, 1043–1079. [Google Scholar] [CrossRef]
- Deshpande, M.V.; Eriksson, K.E.; Pettersson, L.G. An assay for selective determination of Exo-1,4,-Beta-glucanases in a mixture of cellulolytic enzymes. Anal. Biochem. 1984, 2, 481–487. [Google Scholar] [CrossRef]
- Kim, D.W.; Jang, Y.H.; Jeong, Y.K.; Son, K.H. Effect of a nonionic surfactant on the adsorption and kinetic mechanism for the hydrolysis of microcrystalline cellulose by endoglucanase I and exoglucanase II. Bull. Korean Chem. Soc. 1997, 18, 300–305. [Google Scholar]
- Steinhardt, J.; Reynolds, J.A. Multiple Equilibria in Proteins; Academic Press: New York, NY, USA, 1970. [Google Scholar]
- Liu, Y.; Guo, R. Interaction between casein and sodium dodecyl sulfate. J. Colloid Interface Sci. 2007, 315, 685–692. [Google Scholar] [CrossRef]
- Maldonado-Valderrama, J.; Rodríguez Patino, M. Interfacial rheology of protein–surfactant mixtures. Curr. Opin. Colloid Interface Sci. 2010, 15, 271–282. [Google Scholar] [CrossRef]
- Divne, C.; Stahlberg, J.; Teeri, T.T. High-resolution crystal structures reveal how a cellulose chain is bound in the 50A long tunnel of Cellobiohydrolase I from Trichoderma reesei. J. Mol. Biol. 1998, 275, 309–325. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, Y.; Tang, L. Effect of PEG 4000 on cellulose catalysis in the lignocellulose saccharification process. J. Chem. Technol. Biotechnol. 2010, 86, 115–120. [Google Scholar]
- Badley, R.A.; Carruthers, L.; Phillips, M.C. Hydrophobic free energy and the denaturation of proteins. Biochem. Biophys. Acta. 1997, 495, 110–113. [Google Scholar]
- Lu, Y.; Yang, B.; Gregg, D.; Saddler, J.N.; Mansfield, S.D. Cellulase adsorption and an evaluation of enzyme recycle during hydrolysis of steam-exploded softwood residues. Appl. Biochem. Biotechnol. 2002, 100, 641–654. [Google Scholar] [CrossRef]
- Sutcliffe, R.; Saddler, J.N. The role of lignin in the adsorption of cellulases during enzymatic treatment of lignocellulosic material. Biotechnol. Bioeng. 1986, 17, 749–762. [Google Scholar]
- Uribe, S.; Sampedro, J.G. Measuring solution viscosity and its effect on enzyme activity. Biol. Proced. Online 2003, 5, 108–115. [Google Scholar] [CrossRef]
- Jeon, S.I.; Andrade, J.D. Protein surface interactions in the presence of poly(ethylene oxide)2. Effects of protein size. J. Colloid Interface Sci. 1991, 142, 159–166. [Google Scholar] [CrossRef]
- Malmstren, M.; VanAlstine, J.M. Adsorption of poly (ethylene glycol) amphiphiles to form coatings which inhibit protein adsorption. J. Colloid Interface Sci. 1996, 177, 502–512. [Google Scholar] [CrossRef]
- Graham, D.E.; Phillips, M.C. Proteins at liquid interfaces. V. Shear properties. J. Colloid Interface Sci. 1980, 76, 240–250. [Google Scholar] [CrossRef]
- Dickinson, E. Adsorbed protein layers at fluid interfaces: Interactions, structure and surface rheology. Colloids Surf. B 1999, 15, 161–176. [Google Scholar] [CrossRef]
- Karlstrom, G. A new model for upper and lower critical solution temperature in poly ethylene oxide solusions. J. Phys. Chem. 1985, 89, 4962–4964. [Google Scholar] [CrossRef]
- Jacquilin, A.; Tnford, R.; Tanford, C. Binding of dodecyl sulfate to proteins at high binding ratios. Possible implications for the state of proteins in biological membranes. Proc. Natl. Acad. Sci. USA 1970, 66, 1002–1006. [Google Scholar] [CrossRef]
- Viparelli, P.; Alfani, F.; Cantarella, M. Models for enzyme superactivity in aqueous solutions of surfactants. Biochem. J. 1999, 344, 765–73. [Google Scholar] [CrossRef]
- Larsen, J.; Henning, J. Methods for Reducing Enzyme Consumption in Second Generation Bioethanol Fermentation in the Presence of Lignin. WO Patent WO/2009/095781, 8 June 2009. [Google Scholar]
- Tirosh, O.; Barenholz, Y.; Katzhender, J.; Priev, A. Hydration of polyethylene glycol-grafted liposomes. Biophys. J. 1998, 74, 1371–1379. [Google Scholar] [CrossRef]
- Lang, M.C.; Laupretre, F.; Noel, C.; Monnerie, L. Molecular motion of polyethylene oxide in dilute solutions studied by electron spin resonance and nuclear magnetic resonance. J. Chem. Soc. Faraday Trans. II 1979, 75, 349–355. [Google Scholar] [CrossRef]
- De Gennes, P.G. Conformation of polymers attached to an interface. Macromolecules 1980, 13, 1069–1075. [Google Scholar] [CrossRef]
- Harder, P.; Grunze, M.; Dahint, R.; Whitesides, G.M.; Laibinis, P.E. Molecular confor- mation in oligo(ethylene glycol)-terminated self-assembled monolayers on gold and silver surfaces determines their ability to resist protein adsorption. J. Phys. Chem. B 1998, 102, 426–436. [Google Scholar]
- Shashkina, J.A.; Philippova, O.E.; Zaroslov, Y.D.; Khokhlov, A.R.; Pryakhina, T.A.; Blagodatskikh, I.V. Rheology of viscoelastic solutions of cationic surfactant. Effect of added associating polymer. Langmuir 2005, 21, 1524–1530. [Google Scholar] [CrossRef]
- Harjinder, S.; Flanagan, J. Milk Proteins. In Handbook of Food Science, Technology and Engineering; Hui, W.H., Ed.; Taylor & Francis: New York, NY, USA, 2006. [Google Scholar]
- Jonstromer, M.; Strey, R. Non-ionic bilayers in dilution solutions-effect of additives. J. Phys. Chem. 1992, 96, 5993–6000. [Google Scholar] [CrossRef]
- Schomaker, R.; Strey, R. Effect of ionic surfactants on non-ionic bilayers-bending elasticity of weakly charged membrane. J. Phys. Chem. 1994, 98, 3908–3912. [Google Scholar] [CrossRef]
- Sivars, U.; BergfeltK, P.L.; Tjerland, F. Protein partitioning in weakly charged polymer-surfactant aqueous two-phase system. J. Chromatogr. B 1996, 680, 43–53. [Google Scholar] [CrossRef]
- Naem, A.; Khan, R.H. Characterization of molten globule state of cytochrome c at alkaline, native and acidic pH induced by butanol and SDS. Int. J. Biochem. Cell Biol. 2004, 36, 2281–2292. [Google Scholar] [CrossRef]
- Moore, B.M.; Flurkey, W.H. Sodium dodecyl sulfate activation of a plant polyphenoloxidase. J. Biol. Chem. 1990, 265, 4982–4988. [Google Scholar]
- Ahl, P.L.; Bhatia, S.K.; Meers, P.; Roberts, P.; Stevens, R.; Dause, R.; Perkins, W.R.; Janoff, A.S. Enhancement of the in vivo circulation lifetime of L-α line liposomes: Importance of liposomal aggregation versus complement opsonization. Biophys. Acta. 1997, 1329, 370–382. [Google Scholar] [CrossRef]
- Lu, K.W.; Pérez-Gil, J.; Taeusch, H.W. Kinematic viscosity of therapeutic pulmonary surfactants with added polymers. Biochim. Biophys. Acta 2009, 1788, 632–637. [Google Scholar] [CrossRef]
- Yamada, Y.; Kuboi, R.; Komasawa, I. Increased activity of Chromobacterium viscosum lipase in aerosol OT reverse micelles in the presence of nonionic surfactants. Biotechnol. Prog. 1993, 9, 468–472. [Google Scholar]
- Taylor, D.J.F.; Penfold, R.K.T. Polymer/surfactant interactions at the air/water interface. Adv. Colloid Interface Sci. 2007, 132, 69–110. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Eckard, A.D.; Muthukumarappan, K.; Gibbons, W. A Review of the Role of Amphiphiles in Biomass to Ethanol Conversion. Appl. Sci. 2013, 3, 396-419. https://doi.org/10.3390/app3020396
Eckard AD, Muthukumarappan K, Gibbons W. A Review of the Role of Amphiphiles in Biomass to Ethanol Conversion. Applied Sciences. 2013; 3(2):396-419. https://doi.org/10.3390/app3020396
Chicago/Turabian StyleEckard, Anahita Dehkhoda, Kasiviswanathan Muthukumarappan, and William Gibbons. 2013. "A Review of the Role of Amphiphiles in Biomass to Ethanol Conversion" Applied Sciences 3, no. 2: 396-419. https://doi.org/10.3390/app3020396
APA StyleEckard, A. D., Muthukumarappan, K., & Gibbons, W. (2013). A Review of the Role of Amphiphiles in Biomass to Ethanol Conversion. Applied Sciences, 3(2), 396-419. https://doi.org/10.3390/app3020396




