The Release of a Captive-Raised Female African Elephant (Loxodonta africana) in the Okavango Delta, Botswana
Abstract
:Simple Summary
Abstract
1. Introduction
2. Experimental Section
2.1. Nandipa’s History and Release
2.2. Data Collection
| Code | Social status |
|---|---|
| 1 | Alone, no other elephant within 500 m |
| 2 | In a small group of bulls, 1–5 males within 500 m of each other |
| 3 | In a large group of bulls, >5 within 500 m of each other |
| 4 1 | In a herd of male and female elephants within 500 m of each other |
| 5 | Within 500 m of the Abu herd |
| Code | Activity | Description |
|---|---|---|
| 1 | Sleeping | Standing in one place with eyes closed for longer than one minute while not feeding |
| 2 | Feeding | Chewing or using the trunk to manipulate food items |
| 3 | Drinking | Intake of water |
| 4 | Social behaviours | Focal elephant physically interacting with at least one other elephant |
| 4.1 | Greeting | Raises trunk to mouth of another elephant |
| 4.1.1 | Another elephant greets focal elephant | |
| 4.2/4.3 | Sparing/playing | Head to head contact and pushing between two or more elephants |
| 4.4 | Pushing from behind | Using tusks or resting trunk over back of the other elephant and pushing |
| 4.4.1 | Focal elephant is pushed from behind | |
| 4.5 | Displaying | Destruction of vegetation without eating, crashing through vegetation, headshaking |
| 4.5.1 | Another elephant is displaying | |
| 4.6 | Head over back | Standing or walking with head and/or trunk resting on back of another elephant |
| 4.6.1 | Another elephant with head and or trunk on back of the focal elephant | |
| 5 | Mud bathing/dusting | Collection of dust or mud with trunk and then throwing it over themselves |
| 6 | Walking | Moving purposefully at a steady pace |
| 7 | Walking while feeding | Moving at a steady pace while chewing or manipulating food items |
| 8 | Standing | Standing in one place with eyes open for longer than one minute while not feeding |
| 9 | Vocalizing | |
| 9.1 | Vocalization by focal individual | |
| 9.2 | Vocalization by known other | |
| 10 | Running | Moving at pace, generally when alarmed |
| 11 | Other | Focal elephant does another activity e.g., pushes over tree to eat |
| 11.1 | Another elephant does another activity |
2.3. Other Elephant Releases
2.4. Data Analysis
| Data set | Rainy season | Flood season | Dry season | Total |
|---|---|---|---|---|
| Total sightings | 58 | 68 | 29 | 155 |
| Social focal data | 17 | 21 | 5 | 43 |
| Focal data | 18 | 22 | 5 | 45 |
| Individual satellite fixes | 910 | 753 | 218 | 1,881 |
| Consecutive 24-h downloads | 268 | 195 | 60 | 523 |
3. Results
3.1. Social Behaviour
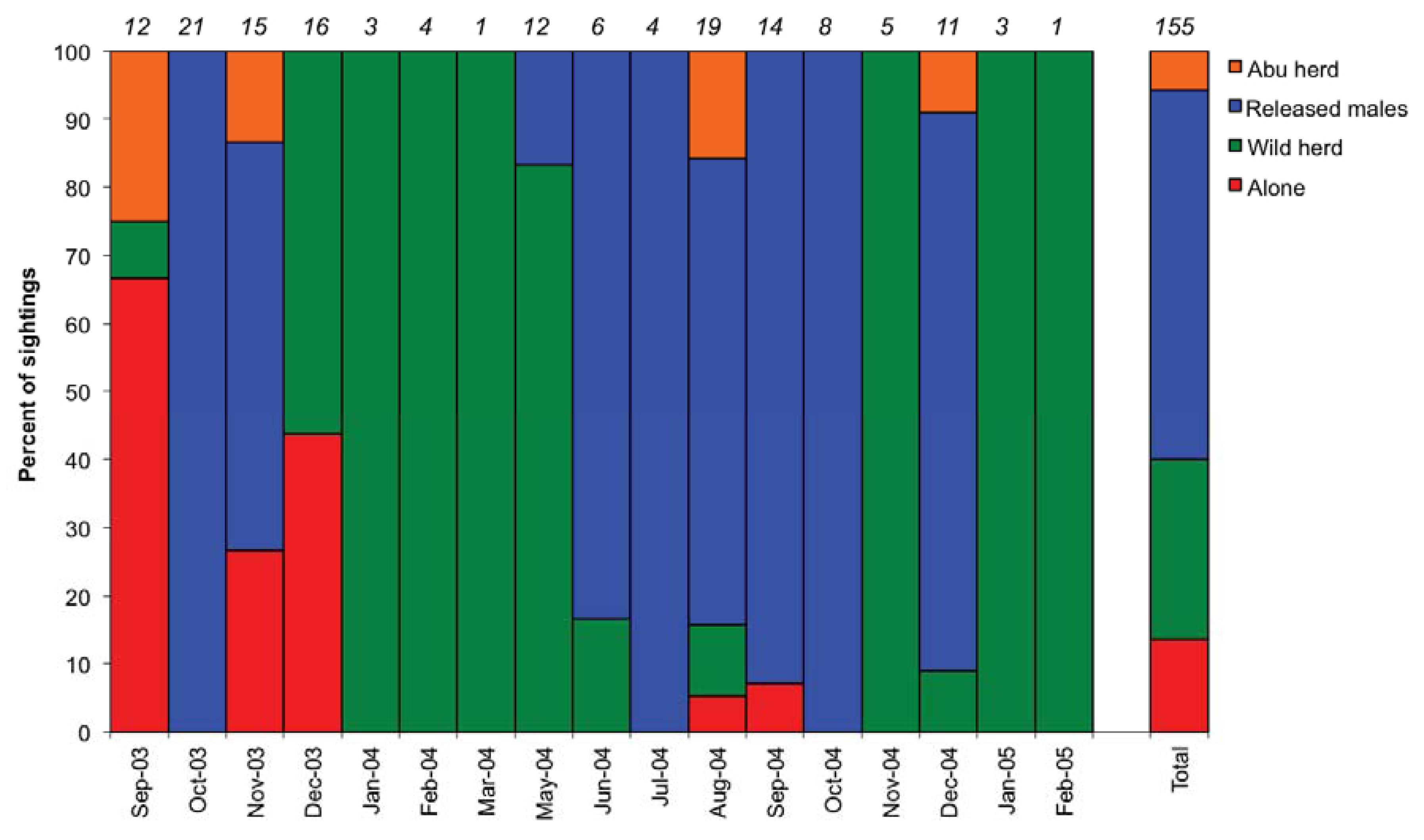
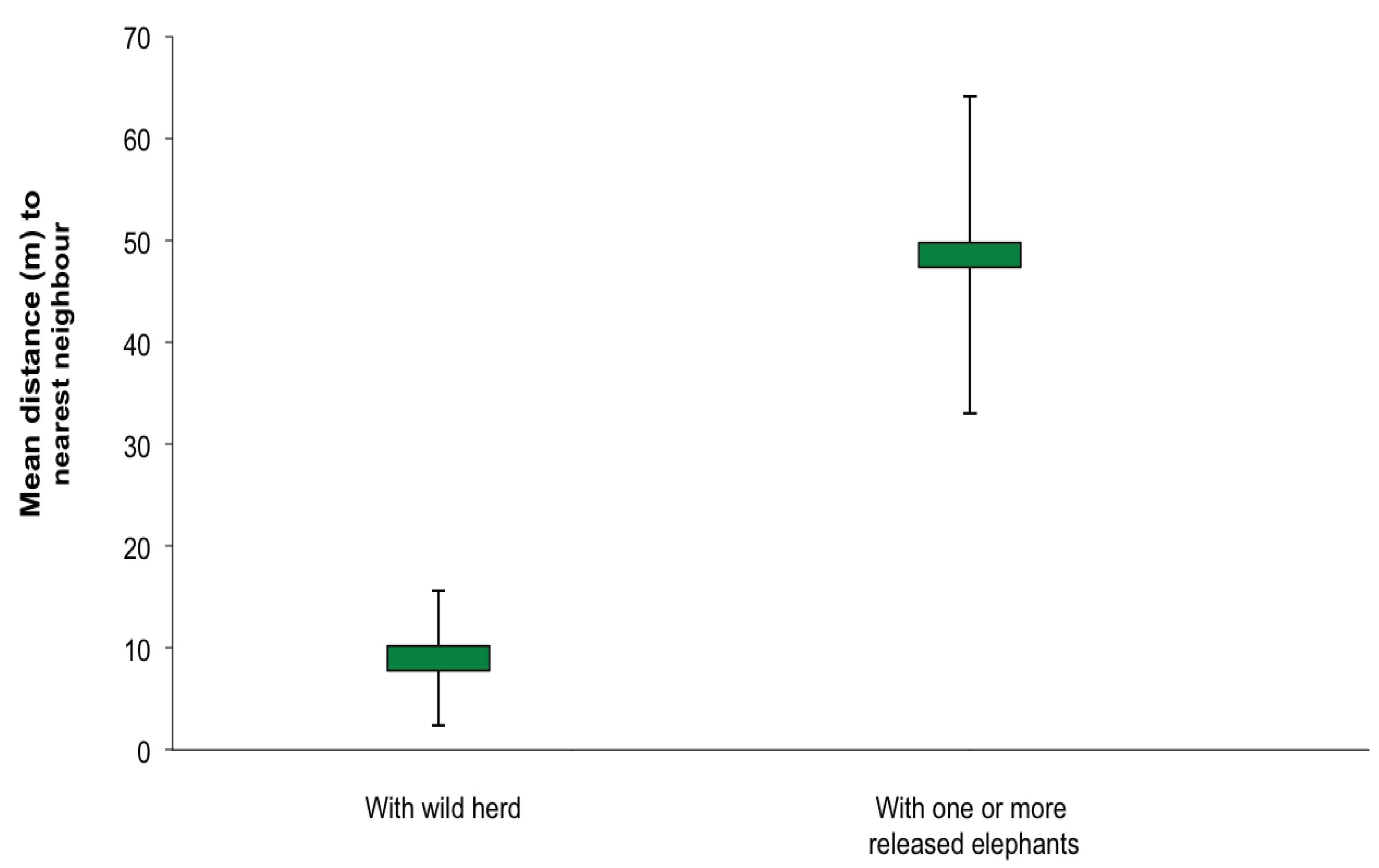
| Activity | Wild herd | Released males | Rainy | Flood | Dry |
|---|---|---|---|---|---|
| All social interactions | 0.20 ± 0.13 | 0.64 ± 0.27 | |||
| Greeting | 0.10 ± 0.10 | 0.24 ± 0.18 | |||
| Sparring | 0.10 ± 0.10 | 0.24 ± 0.24 | |||
| Vocalizations | 0.00 ± 0.00 | 0.61 ± 0.28 | |||
| Feeding | 52.9 ± 10.2 | 65.8 ± 4.9 | 58.8 ± 7.1 | 67.5 ± 6.4 | 62.8 ± 14.7 |
| Walking | 21.4 ± 5.7 | 13.0 ± 3.0 | 16.8 ± 4.3 | 15.0 ± 3.9 | 5.7 ± 5.7 |
| Sleeping | 12.9 ± 5.4 | 11.3 ± 4.1 | 13.5 ± 4.0 | 7.2 ± 4.6 | 25.7 ± 15.9 |
| Mudbathing/dusting | 2.9 ± 1.9 | 3.0 ± 1.2 | 3.4 ± 1.5 | 2.6 ± 1.5 | 2.9 ± 2.9 |
| Other activities | 1.4 ± 1.4 | 3.0 ± 1.6 | 0.8 ± 0.8 | 4.6 ± 2.4 | 0.0 ± 0.0 |
3.2. Home Range and Habitat Use
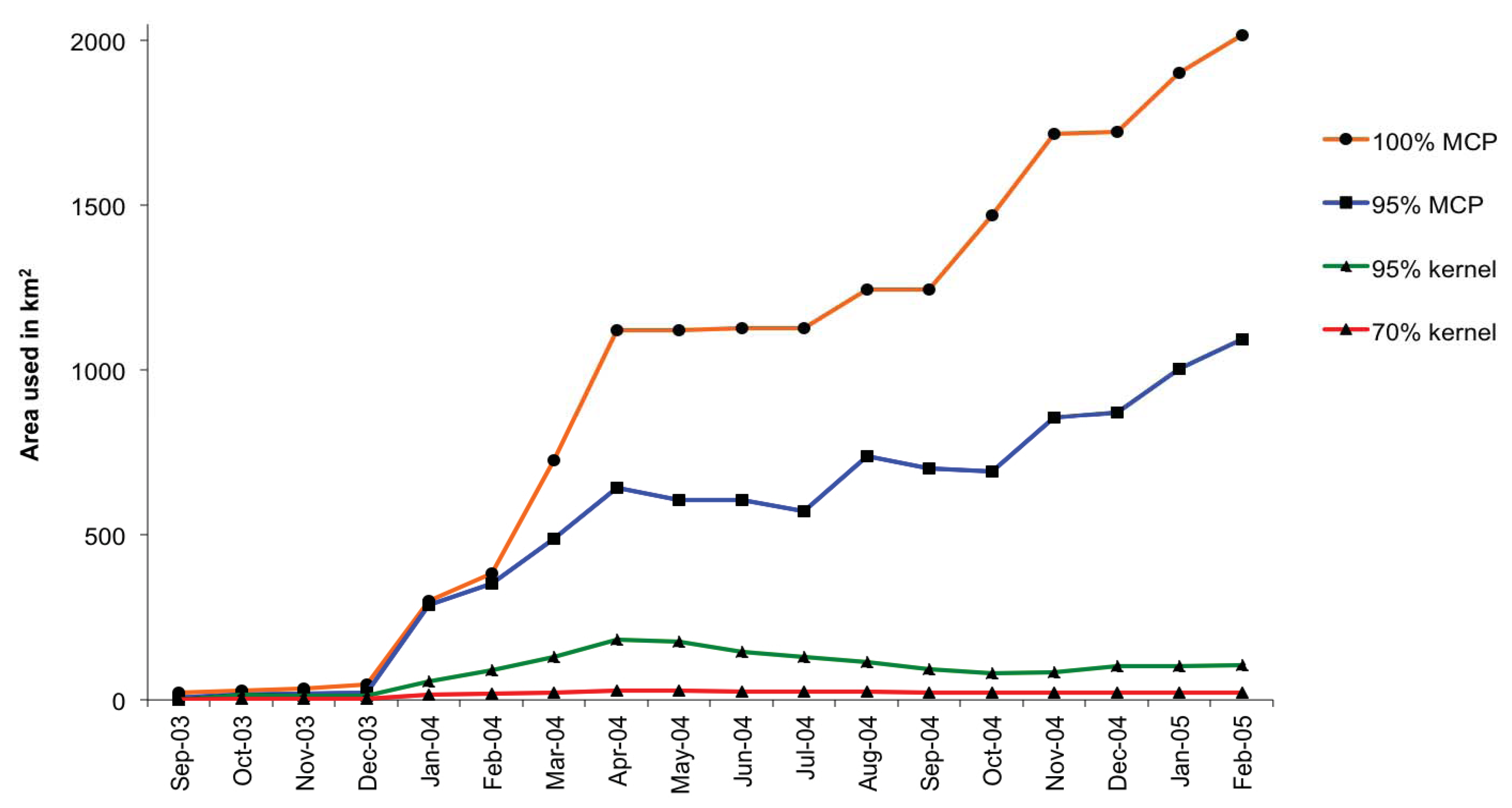
| Condition score 1,2 | Rainy season | Flood season | Dry season |
|---|---|---|---|
| 1 | 0 0 | 0 0 | 0 0 |
| 2 | 0 0 | 0 0 | 1 0 |
| 2.5 | 5 2 | 1 0 | 2 0 |
| 3 | 2 6 | 4 0 | 2 3 |
| 3.5 | 8 1 | 2 0 | 0 1 |
| 4 | 4 9 | 11 7 | 0 4 |
| 4.5 | 0 1 | 0 1 | 0 1 |
| 5 | 0 0 | 0 0 | 0 0 |
| Total | 19 19 | 18 8 | 5 9 |
3.3. Other Welfare Indicators
4. Discussion
4.1. Social Integration
4.2. Spatial and Habitat Use
4.3. Other Welfare Indicators
5. Conclusions
Conflict of Interest
Acknowledgements
References and Notes
- Charif, R.A.; Ramey, R.R.; Langbauer, W.R.; Payne, K.B.; Martin, R.B.; Brown, L.M. Spatial relationships and matrilineal kinship in African savanna elephant (Loxodonta africana) clans. Behav. Ecol. Sociobiol. 2005, 57, 327–338. [Google Scholar]
- Wittemyer, G.; Douglas-Hamilton, I.; Getz, W.M. The socioecology of elephants: Analysis of the processes creating multitiered social structures. Anim. Behav. 2005, 69, 1357–1371. [Google Scholar] [CrossRef]
- Archie, E.A.; Moss, C.J.; Alberts, S.C. The ties that bind: genetic relatedness predicts the fission and fusion of social groups in wild African elephants. Proc. Roy. Soc. Lond. B 2006, 273, 513–522. [Google Scholar] [CrossRef]
- Archie, E.A.; Moss, C.J.; Alberts, S.C. Friends and relations: Kinship and the nature of female elephant social relationships. In The Amboseli Elephants: A Long-Term Perspective on a Long-Lived Mammal; Moss, C.J., Croze, H., Lee, P.C., Eds.; University of Chicago Press: Chicago, IL, USA, 2011; pp. 238–245. [Google Scholar]
- Moss, C. Elephant Memories: Thirteen Years in the Life of an Elephant Family; Elm Tree Books: London, UK, 1988. [Google Scholar]
- Moss, C.J.; Lee, P.C. Female social dynamics: Fidelity and flexibility. In The Amboseli Elephants: A Long-Term Perspective on a Long-Lived Mammal; Moss, C.J., Croze, H., Lee, P.C., Eds.; University of Chicago Press: Chicago, IL, USA, 2011; pp. 205–223. [Google Scholar]
- Veasey, J. Concepts in the care and welfare of captive elephants. Int. Zoo Yearbook 2006, 40, 63–79. [Google Scholar] [CrossRef]
- Harris, M.; Sherwin, C.; Harris, S. The Welfare, Housing and Husbandry of Elephants in UK Zoos: Final Report. 2008. Available online: http://randd.defra.gov.uk/Default.aspx?Menu=Menu&Module=More&Location=None&Completed=1&ProjectID=13192 (accessed on 20 January 2013).
- Anon. Elephants Transferred. 2012. Available online: http://www.elephant.se/elephant_transfers.php?year=2012 (accessed on 20 January 2013).
- Clubb, R.; Rowcliffe, M.; Lee, P.; Mar, K.U.; Moss, C.; Mason, G.J. Compromised survivorship in zoo elephants. Science 2008, 322. [Google Scholar] [CrossRef]
- Evans, K.; Moore, R.; Harris, S. The social and ecological integration of captive-raised adolescent male African elephants (Loxodonta africana) into a wild population. PLoS One 2013, 8. [Google Scholar] [CrossRef]
- Moore, R.J.; Munnion, C. Back to Africa; Southern Book Publishers: Johannesburg, South Africa, 1989. [Google Scholar]
- Evans, K.E. The Behavioural Ecology and Movements of Adolescent Male African Elephant (Loxodonta africana) in the Okavango Delta, BOTSWANA. Ph.D. Thesis, Bristol University, Bristol, UK, 2006. [Google Scholar]
- Evans, K.E.; Harris, S. Adolescence in male African elephants, Loxodonta africana, and the importance of sociality. Anim. Behav. 2008, 76, 779–787. [Google Scholar] [CrossRef]
- Anderson, J.L. Restoring a wilderness: The reintroduction of wildlife to an African national Park. Int. Zoo Yearbook 1986, 24, 192–199. [Google Scholar] [CrossRef]
- Moore, R.J. Elephants for Africa; Abu Publications: Auckland Park, South Africa, 2000. [Google Scholar]
- Anon. Individual stories: Nina. Available online: http://www.bornfree.org.uk/uploads/media/board_13.pdf (accessed on 18 January 2012).
- Evans, K.E. University of Bristol: Bristol, UK, Unpublished data. 1995.
- Poole, J.H. Musth and Male-Male Competition in the African Elephant. Ph.D. Thesis, University of Cambridge, Cambridge, UK, 1982. [Google Scholar]
- Evans, K.E.; Harris, S. Sex differences in habitat use by African elephants (Loxodonta africana) in the Okavango Delta, Botswana: Is size really the deciding factor? Afr. J. Ecol. 2012, 50, 277–284. [Google Scholar]
- Jellema, A.; Ringrose, S.; Matheson, W. Vegetation Mapping in Northern Botswana; Unpublished Report; Harry Oppenheimer Okavango Research Centre: University of Botswana, Maun, Botswana, 2002. [Google Scholar]
- Aebischer, N.J.; Robertson, P.A. Practical aspects of compositional analysis as applied to pheasant habitat utilization. In Wildlife Telemetry: Remote Monitoring and Tracking of Animals; Priede, I.G., Swift, S.M., Eds.; Ellis Horwood: New York, NY, USA, 1992; pp. 285–293. [Google Scholar]
- Smith, P.G. Compos Analysis Version 5.0 User’s Guide. Smith Ecology Microsoft® Excel Tool for Compositional Analysis; Unpublished Report; Smith Ecology: Abergavenny, UK, 2003. [Google Scholar]
- Zar, J.H. Biostatistical Analysis; Prentice Hall: Upper Saddle River, NJ, USA, 1984. [Google Scholar]
- Sarrazin, F.; Barbault, R. Reintroduction: Challenges and lessons for basic ecology. Trends Ecol. Evol. 1996, 11, 474–478. [Google Scholar] [CrossRef]
- Mubalama, L. Population and distribution of elephants (Loxodonta africana africana) in the central sector of the Virunga National Park, eastern DRC. Pachyderm 2000, 28, 44–55. [Google Scholar]
- Mutinda, H.; Poole, J.H.; Moss, C.J. Decision making and leadership in using the ecosystem. In The Amboseli Elephants: A Long-Term Perspective on a Long-Lived Mammal; Moss, C.J., Croze, H., Lee, P.C., Eds.; University of Chicago Press: Chicago, IL, USA, 2011; pp. 246–259. [Google Scholar]
- Slotow, R.; Garai, M.E.; Reilly, B.; Page, B.; Carr, R.D. Population dynamics of elephants re-introduced to small fenced reserves in South Africa. S. Afr. J. Wildl. Res. 2005, 35, 23–32. [Google Scholar]
- Sanderson, I.T. The Dynasty of Abu: A History and Natural History of the Elephants and Their Relatives Past and Present; Cassell: London, UK, 1963. [Google Scholar]
- Chepko-Sade, B.D.; Sade, D.S. Patterns of group splitting within matrilineal kinship groups. Behav. Ecol. Sociobiol. 1979, 5, 67–86. [Google Scholar] [CrossRef]
- Goossens, B.; Setchell, J.M.; Tchidongo, E.; Dilambaka, E.; Vidal, C.; Ancrenaz, M.; Jamart, A. Survival, interactions with conspecifics and reproduction in 37 chimpanzees released into the wild. Biol. Conserv. 2005, 123, 461–475. [Google Scholar] [CrossRef]
- Poole, J.H.; Granli, P. Signals, gestures, and behavior of African elephants. In The Amboseli Elephants: A Long-Term Perspective on a Long-Lived Mammal; Moss, C.J., Croze, H., Lee, P.C., Eds.; University of Chicago Press: Chicago, IL, USA, 2011; pp. 109–124. [Google Scholar]
- Pinter-Wollman, N.; Isbell, L.A.; Hart, L.A. The relationship between social behaviour and habitat familiarity in African elephants (Loxodonta africana). Proc. Roy. Soc. B. 2009, 276, 1009–1014. [Google Scholar] [CrossRef]
- Western, D.; Lindsay, W.K. Seasonal herd dynamics of a savanna elephant population. Afr. J. Ecol. 1984, 22, 229–244. [Google Scholar] [CrossRef]
- Dunham, K.M. Movements of elephant cows in the unflooded Middle Zambezi Valley, Zimbabwe. Afr. J. Ecol. 1986, 24, 287–291. [Google Scholar] [CrossRef]
- Viljoen, P.J. Spatial distribution and movements of elephants (Loxodonta africana) in the northern Namib Desert region of the Kaokoveld, South West Africa/Namibia. J. Zool. 1989, 219, 1–19. [Google Scholar] [CrossRef]
- Lindeque, M.; Lindeque, P.M. Satellite tracking of elephants in northwestern Namibia. Afr. J. Ecol. 1991, 29, 196–206. [Google Scholar] [CrossRef]
- Tchamba, M.N.; Bauer, H.; De Iongh, H.H. Application of VHF-radio and satellite telemetry techniques on elephants in northern Cameroon. Afr. J. Ecol. 1995, 33, 335–346. [Google Scholar] [CrossRef]
- Thouless, C. Satellite tracking of elephants. In Studying Elephants; Kangwana, K., Ed.; African Wildlife Foundation: Nairobi, Kenya, 1996; pp. 120–125. [Google Scholar]
- De Villiers, P.A.; Kok, O.B. Home range, association and related aspects of elephants in the eastern Transvaal Lowveld. Afr. J. Ecol. 1997, 35, 224–236. [Google Scholar] [CrossRef]
- Blake, S.; Douglas-Hamilton, I.; Karesh, W.B. GPS telemetry of forest elephants in Central Africa: results of a preliminary study. Afr. J. Ecol. 2001, 39, 178–186. [Google Scholar] [CrossRef]
- Osborn, F.V. The concept of home range in relation to elephants in Africa. Pachyderm 2004, 37, 37–44. [Google Scholar]
- Galanti, V.; Preatoni, D.; Martinoli, A.; Wauters, L.A.; Tosi, G. Space and habitat use of the African elephant in the Tarangire-Manyara ecosystem, Tanzania: implications for conservation. Mamm. Biol. 2006, 71, 99–114. [Google Scholar]
- Grainger, M.; van Aarde, R.; Whyte, I. Landscape heterogeneity and the use of space by elephants in the Kruger National Park, South Africa. Afr. J. Ecol. 2005, 43, 369–375. [Google Scholar] [CrossRef]
- Gough, K.F.; Kerley, G.I.H. Demography and population dynamics in the elephants Loxodonta africana of Addo Elephant National Park, South Africa: Is there evidence of density dependent regulation? Oryx 2006, 40, 434–441. [Google Scholar] [CrossRef]
- Spinage, C. Elephants; Poyser: London, UK, 1994. [Google Scholar]
- Moss, C.J. The demography of an African elephant (Loxodonta africana) population in Amboseli, Kenya. J. Zool. 2001, 255, 145–156. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Evans, K.; Moore, R.J.; Harris, S. The Release of a Captive-Raised Female African Elephant (Loxodonta africana) in the Okavango Delta, Botswana. Animals 2013, 3, 370-385. https://doi.org/10.3390/ani3020370
Evans K, Moore RJ, Harris S. The Release of a Captive-Raised Female African Elephant (Loxodonta africana) in the Okavango Delta, Botswana. Animals. 2013; 3(2):370-385. https://doi.org/10.3390/ani3020370
Chicago/Turabian StyleEvans, Kate, Randall J. Moore, and Stephen Harris. 2013. "The Release of a Captive-Raised Female African Elephant (Loxodonta africana) in the Okavango Delta, Botswana" Animals 3, no. 2: 370-385. https://doi.org/10.3390/ani3020370





