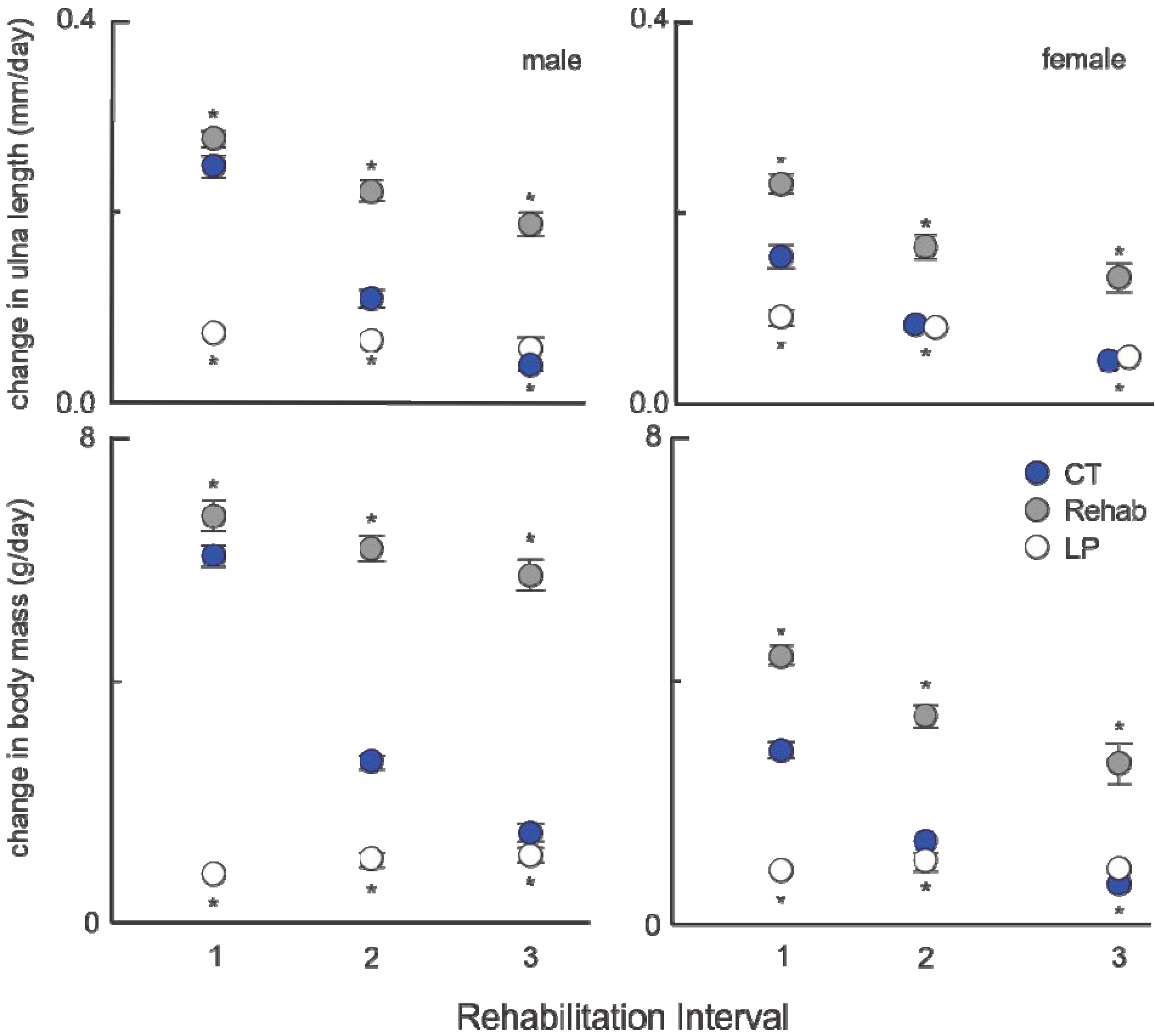
3.1. Consumption through Experimental Period
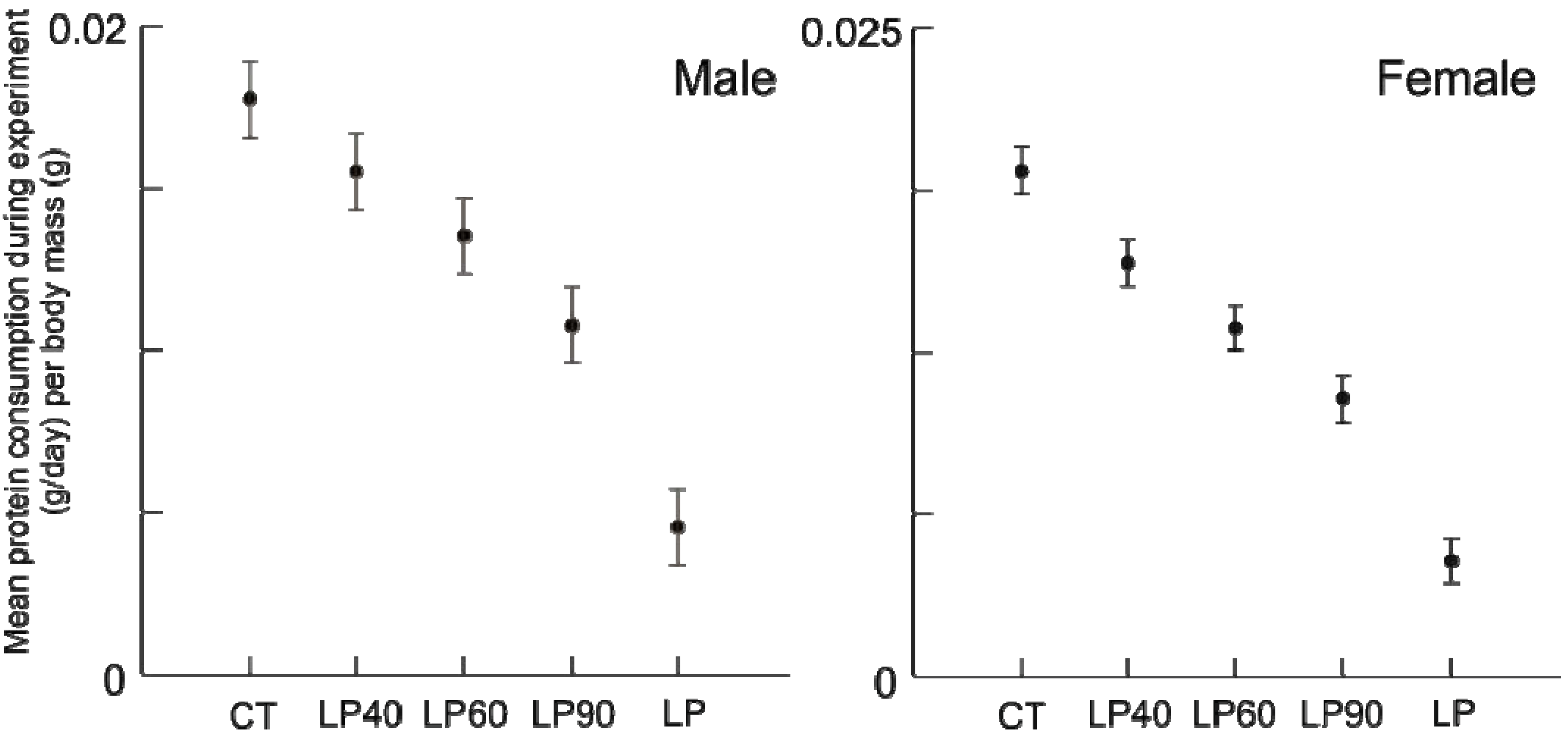
The amount of protein (
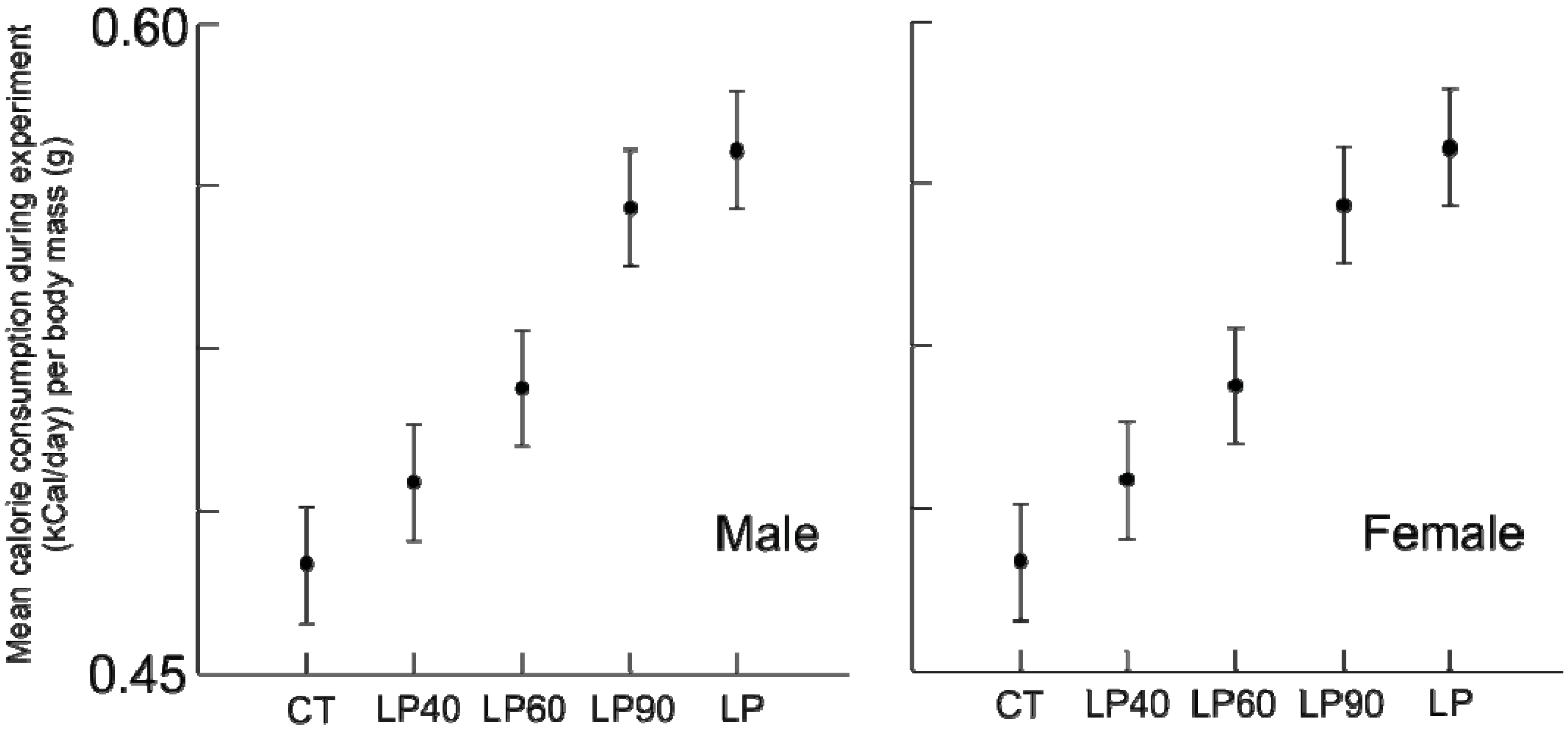
Figure 2) and calories (
Figure 3) consumed during the experiment differed among treatment groups, for both sexes (
p < 0.001). As expected,
post hoc hypothesis testing confirmed that the longer animals were offered the LP diet, the amount of protein consumed was less (all comparisons,
p ≤ 0.001), despite having access to additional food. Unlike protein consumption, calorie consumption was reduced. Despite being provided an isocaloric diet, animals fed reduced protein levels consumed greater calories than animals on the standard protein diet. Rehabilitation groups demonstrated a dose effect: animals with a shorter duration of protein malnutrition consumed fewer calories throughout the experiment than those malnourished longer (
post hoc testing,
p ≤ 0.001), with two exceptions: female LP90’s consumed nearly equal calories to chronic LP’s over the course of the experiment (
p = 0.479) and female LP40’s were statistically indistinct from chronic CT’s (
p = 0.054).
Figure 2.
Average standardized protein consumption (g/day/body mass(g)) for males and females for the duration of the experiment (±standard deviation).
Figure 2.
Average standardized protein consumption (g/day/body mass(g)) for males and females for the duration of the experiment (±standard deviation).
Figure 3.
Average standardized calorie consumption (kCal/day/body mass (g)) for males and females for the duration of the experiment (±standard deviation).
Figure 3.
Average standardized calorie consumption (kCal/day/body mass (g)) for males and females for the duration of the experiment (±standard deviation).
3.2. Consumption at Time of Rehabilitation (10 Day Averages)
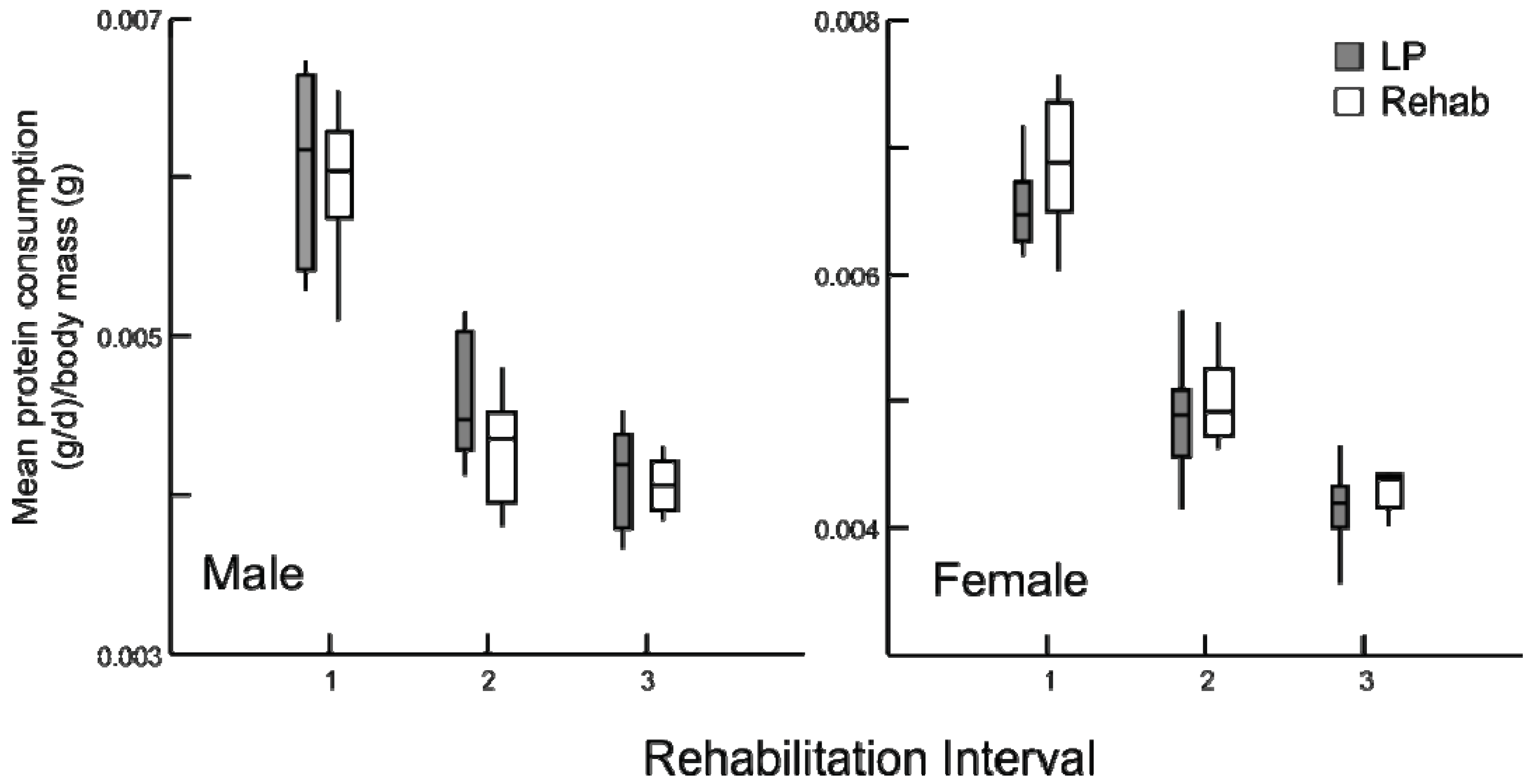
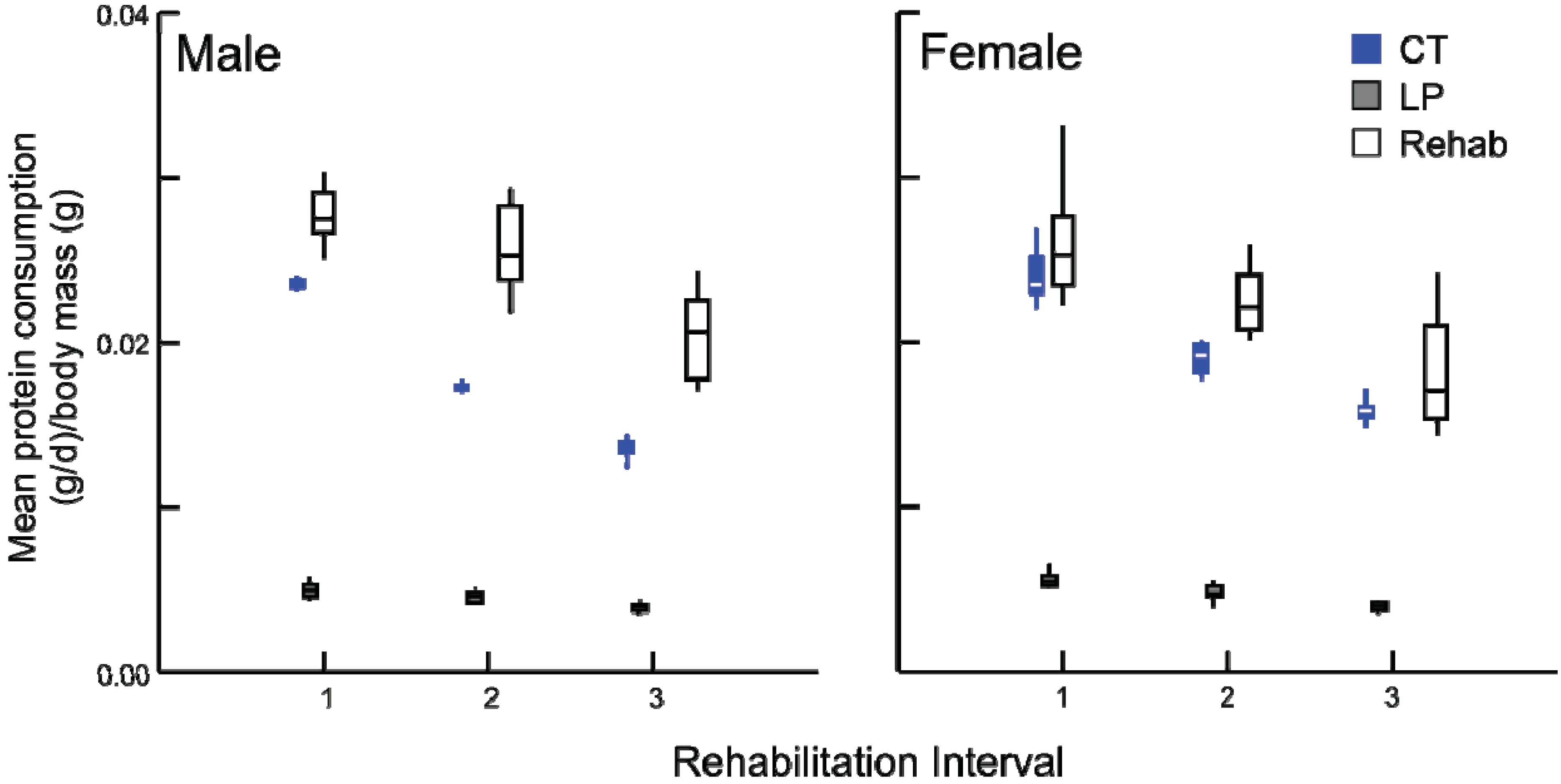
Following the change in diet, all rehabilitation animals altered the amount of protein and calories consumed. The individual paired consumption rates of protein were different across the dietary modification and among treatment groups (
p < 0.001). Prior to the switch in diet, all rehabilitated groups consumed the same amount of protein (
Figure 4) and calories (data not presented) per body mass as the chronic LP animals, demonstrating their efficacy as a malnutrition control.
Figure 4.
Daily average protein consumption (g/day) by body mass (g) for chronic LP and rehabilitation animals for 10 days prior to diet alteration. Rehabilitation intervals encompass the following durations: 1 = animals aged 30-39 days, 2 = 50–59 days, and 3 = 80–89 days (box plots with interquartile ranges indicated, p ≥ 0.5 for all comparisons).
Figure 4.
Daily average protein consumption (g/day) by body mass (g) for chronic LP and rehabilitation animals for 10 days prior to diet alteration. Rehabilitation intervals encompass the following durations: 1 = animals aged 30-39 days, 2 = 50–59 days, and 3 = 80–89 days (box plots with interquartile ranges indicated, p ≥ 0.5 for all comparisons).
After the dietary transition, the three rehabilitated groups consumed more protein than all LP animals and in some cases CT animals as well (
Figure 5). Specifically, rehabilitated males consumed more protein than age-matched CT individuals (all tests
p ≤ 0.001), but rehabilitated females consumed an equal amount of protein per body mass than did age-matched CT females (
p = 0.685). The response of calorie consumption was varied depending upon age and sex (
Figure 6). Regardless of age or sex, LP animals consumed more calories than CT animals (all comparisons
p ≤ 0.001). At rehabilitation males’ caloric intake was indistinguishable from LP amounts, but females immediately decreased their caloric consumption, and the magnitude of change decreases with age.
Figure 5.
Box plots illustrating daily average protein consumption (g/day) by body mass (g) for chronic CT, LP and rehabilitation animals for 10 days following diet alteration. Rehabilitation intervals encompass the following durations: 1 = animals aged 40–49 days, 2 = 60–69 days, and 3 = 90–99 days (all comparisons p ≤ 0.001, except females at interval 1, where p = 0.685).
Figure 5.
Box plots illustrating daily average protein consumption (g/day) by body mass (g) for chronic CT, LP and rehabilitation animals for 10 days following diet alteration. Rehabilitation intervals encompass the following durations: 1 = animals aged 40–49 days, 2 = 60–69 days, and 3 = 90–99 days (all comparisons p ≤ 0.001, except females at interval 1, where p = 0.685).
Figure 6.
Box plots, illustrating daily average calorie consumption (kCal/day) by body mass (g) for chronic CT, LP and rehabilitation animals for 10 days following diet alteration. Rehabilitation intervals encompass the following durations: 1 = animals aged 40–49 days, 2 = 60–69 days, and 3 = 90–99 days (all comparisons p ≤ 0.001, except between LP and rehabilitated males).
Figure 6.
Box plots, illustrating daily average calorie consumption (kCal/day) by body mass (g) for chronic CT, LP and rehabilitation animals for 10 days following diet alteration. Rehabilitation intervals encompass the following durations: 1 = animals aged 40–49 days, 2 = 60–69 days, and 3 = 90–99 days (all comparisons p ≤ 0.001, except between LP and rehabilitated males).
3.3. Discussion
Despite some individual variation, there were no differences in consumption among the LP and rehabilitation groups prior to the change in diet (
Figure 4). Therefore, differences in consumption immediately following the alteration of diet among the rehabilitation groups and LP animals provides an accurate assessment of pre- and post-rehabilitation consumption, even within a population of growing animals with daily, stochastic influences on satiation [
6].
Consumption amounts change with age and body mass, and this was confirmed in both CT and LP groups. It is very important to consider this effect, and this is why we included age in our models. It is not possible to separate age and protein malnutrition duration with our model, but we did find age to be a significant factor in our results. All animals decreased calorie and protein consumption as they aged, including the rehabilitated groups. Also, despite increased calorie consumption in the LP animals, long bones length and body mass were often smaller than in CT or rehabilitated animals [
1], although if given longer, those animals may have eventually attained larger sizes [
2,
3].
As protein was the manipulated factor, it was expected that protein consumption would change when the diet was altered. Although both males and females increased their protein consumption, it was interesting that males increased their consumption to greater than control levels, with a larger differential in the LP60 and LP90 groups. The significant interaction for male groups indicates that duration of protein malnutrition and/or age of animal matters more in males than in females. It is likely that this is the reason older rehabilitated males exceeded CT body mass by the end of the experiment [
1]. These data do not, however, shed light on why the ulna would exceed CT lengths (in both males and females), where other long bones attained targeted sizes. What it does indicate is that some plasticity in bone growth and body mass exists and that different tissues are able to respond to nutritional alteration independently [
3].
Protein content of the diet may influence how other nutrients are absorbed or processed. For instance, zinc and copper serum levels in children with protein energy malnutrition are lower than well-nourished children [
7] and studies in humans have shown reduced calcium absorption with low protein diets [
8] and our reduced protein diet contained a reduction in calcium carbonate because Dyets (Bethlehem, PA, USA) found problems associated with calcium depositions, particularly in the kidneys. However, protein complicates the absorption of polyphenols, as the amino acids show a biochemical affinity for these chemicals, making them less bioavailable [
9]. It is clear that fluctuating protein in the diet will alter the processing of other nutrients, but with complex results.
Animals unexpectedly consumed fewer calories per unit body mass immediately after receiving the standard protein diet. This suggests that satiation is at least partially controlled by protein. Despite the decreased calorie consumption concordant with diet change, there was an associated increase in growth rate in all groups and sexes. As calories were decreased, this elevated growth rate must be fueled by the increase in protein consumption. This has important implications for the growing animal. Prior to the diet alteration, the rats experienced what is known as kwashiorkor-style malnutrition in humans [
10]. This has different effects than calorie restriction, including disruption of the development of the nervous system, muscle wasting (including cardiac anomalies), atrophied intestinal mucosa, metabolic acidosis, fatty livers, and osteoporosis [
10,
11,
12], in addition to mild general stunting [
1,
2,
3,
4,
10]. The requirement for rehabilitative nutrition in this case focuses more on an increase in available amino acids, not an increase in calories. This has implications for interventions: rehabilitative diets should ameliorate the specific nutritional challenge and target the specific macronutrients needed to best support catch-up growth [
10].
This study has several limitations. First, rats do not have strict determinant growth as humans do and their protein requirements are higher than humans. We conducted no histology or metabolic analyses through the rehabilitation time to assess what was functionally altered in order to accommodate increased growth while the animal was self-selecting fewer calories.
The literature on catch-up and targeted growth is confusing. Differences seen are dependent on length of insult, timing of insult, magnitude of insult, type of insult, species insulted. Many claim that the confusion has to do with various experimental procedures and a lack of continuity of methods, data capture, and measured parameters. Although true, it could also be that the mechanisms generating catch-up and targeted growth are multivariate and influenced by many, potentially cumulative and/or confounding factors that our current model is not able to discern.