From in vitro to in vivo Models of Bacterial Biofilm-Related Infections
Abstract
:1. Introduction
2. In vitro Biofilm Models
- (i)
- Closed or static models, in which there are limited nutrients and aeration. This includes some of the most popular and successful models, such as the colony biofilm model and microtiter plates [23,24]. In addition, these models enable direct rapid quantification of biofilm mass (via stains like crystal violet, safranin and congo red) or viable cells (XTT reduction assay).
- (ii)
- Open or dynamic systems. The principle of these models is similar to continuous cultures, in which spent culture consisting of wastes, metabolic byproducts, dispersed and dead cells are constantly replaced by fresh medium. These methods generally allow the control of environmental parameters such as shear forces, and have been therefore extensively used to study the physical and chemical resistance of biofilms. However, they are in most instances less adapted to high throughput analysis and often demand specialized equipment and technical skills [22].
- (iii)
- Microcosms are more sophisticated models that aim to closely mimic in situ conditions. They often include several bacterial species and use material from the studied environment, for instance, addition of hydroxyapatite and saliva to model dental biofilms [25], or covering abiotic surfaces with human cells in order to mimic an in vivo situation [26]. Theoretically, both open and closed systems could be turned into microcosms. Microcosms include more environmental parameters and take into consideration the complexity and heterogeneity of natural settings.
| Method | Characteristics | Advantages | Uses of model | References |
|---|---|---|---|---|
| 1. Static systems | ||||
| Colony Biofilm | Colonies are grown over agar.
Maintains basic biofilm characteristics like structured environment and chemical gradients. | Reproducible and simple.
Amenable to high throughput screening. | Antibiotic susceptibility assessment
Morphotypes observation essentially upon polysaccharides production | [381,382] |
| Microtiter plate | Commonly used.
Bacteria attach to well surfaces. | Simple to run,
Amenable to high throughput screening. Suited to molecular genetic tests. | Evaluation of biofilm formation of strains, biofilm antibiotic tolerance and resistance, efficiency of antibiofilm/antimicrobial products | [23,383] |
| Biofilm Ring Test | Immobilization of magnetic beads
Results are automatized and analyzed by image analysis. | Allows for a rapid monitoring of biofilm formation.
Possibility to assess early adhesion events. Quick and automatic analysis. Does not involve washing and staining procedures. | Evaluation of biofilm formation of strains | [384] |
| Calgary Biofilm Device | Based on 96-well microtiter plate assay.
Includes a lid with 96 pegs on which biofilms develop. | Commercially available system.
Pegs can be removed individually without opening the system, and hence avoiding contaminations. Consistent shear force across all pegs. | Biofilm antibiotic resistance and tolerance, efficiency of antibiofilm/antimicrobial products.
Study biofilm development over time. | [385] |
| 2. Open systems | ||||
| Kadouri system | Based on microtiter plate assay but with constant renewal of media.
Minimum shear forces. | Formation of mature biofilm in microtiter plate wells, meaning a big amount of biomass that can be later used for microarrays and proteomics | Allows testing of multiple conditions and treatments. | [386] |
| Flow cell | Flat walled transparent chambers irrigated by culture media under the microscope.
Costly and expertise is needed. System is automatized and available for image analysis. | Enables for non-destructive real-time biofilm observation (Allows single cell visualization).
Excellent image quality | Evaluation of biofilm formation of strains, biofilm antibiotic tolerance, efficiency of antibiofilm/antimicrobial products. | [387] |
| CDC Biofilm reactors | Consists of eight polypropylene holders, accommodating 3 coupons each over which bacteria adhere, suspended from a lid surrounded by media | Commercially available system.
Easy sampling at different time points Reliable system. | Evaluation of biofilm formation, biofilm antibiotic resistance and tolerance. Test disinfectant efficiencies.
Study biofilm development over time. | [388] |
| Microfermentors | Chemostat-based
Biofilms develop over a removable spatula composed of different materials | Large mass of biofilm produced
Allows microscopic, genetic and biochemical analysis Different shear forces can be applied Can be easily turned into a microcosm | Easily converted into microcosms, by covering spatula with human cells.
Evaluation of antibiotic effects and biofilm formation ability of strains. | [389] |
| Modified Robbins Device | Ports sit in a linear array along a rectangular channel. In each port, a plug can be inserted. | Sampling plugs can be removed and replaced aseptically | Used to mimic throat conditions and evaluate the efficiency of different products in rubber tracheo-oesophageal prostheses | [390] |
| Drip flow reactor | Individual channels are introduced into polycarbonate blocks within which microscope slides may be placed.
Biofilms are grown in an angled way. | Low shear and high gas transfer.
Allows for both solid-liquid and solid-air biofilm establishment. | Wound biofilm model. Tested for antimicrobial efficiency [391], bacteriophage reduction of biofilms and other antibiofilm strategies.
Evaluation of disinfectant efficiencies. Study biofilm heterogeneity. | [392] |
| Microfluidic biochips | Biochip is embedded in an aluminium block in which temperature is controlled.
Has contactless dielectric microsensors. | Non-invasive technique.
Monitors dielectric changes of subcellular components within biofilm. Measures biofilm growth and development with sensitivity. | Useful to study population dynamics and quantitative cell analysis. | [393] |
| Constant depth film fermenter | Biofilms develop on polytetrafluoroethylene (PTFE) plugs.
Biofilm growth and depth is limited. Excess biofilm is removed. | Excess biofilm is removed (imitating mechanical biofilm removal like tongue effect or toothbrush). | Specially suited to study oral biofilms.
Tests of the effect of surface characteristic on biofilm formation. Antibiotic resistance tests. | [394,395] |
| Rotating Disc Reactor | Teflon rotor holding several (6 to 24) coupons over which biofilms will form. Rotor is embedded with a magnetic stir bar on the bottom and driven by a stirrer. | Liquid shear forces over the coupons can be varied. | Evaluate antimicrobial and antifouling treatments.
Also used to study multispecies biofilms. | [396] |
| BioFlux Device | 96 individual microfluidic channels fed with a pneumatic pump.
Shear can be controlled individually in each channel. | Low cost in reagents and energy supply.
High throughput analysis. Precise control of environmental conditions. Study of single cell behaviors within a community. | High throughput screening.
Evaluation of biofilm formation of strains, biofilm antibiotic tolerance and resistance, efficiency of antibiofilm/antimicrobial products | [397] |
| Annular reactors | Based on two concentric cylinders; an outer static one which acts as the wall of the vessel and the inner rotating cylinder. | Shear forces can be controlled.
Removable test coupons. | Evaluation of disinfection efficiencies.
Study the effect of shear forces. Specially suited to study aquatic biofilms | [398] |
| Sorbarod devices (SBF) | Sorbarod filter plugs with a cellulose matrix perfused with media. | Easy set up.
Substantial amounts of biomass. Growth rate control possible. Allows sampling of dispersed cells. | Used to evaluate long-time effects of antibiotics. | [399] |
| Perfused (membrane) biofilm fermenter | Cells are collected by pressure filtration in a cellulose acetate membrane.
Filter is the inverted into the base of a modified fermentor Filter is the perfused with fresh medium Newly formed and loosely attached cells are eluted with spent medium. | Allows growth-rate control bacteria or yeast
Adherent bacterial biomass is constant and proportional to the limiting nutrient concentrations | Used to evaluate antibiotic and fungicide efficiency | [400] |
| 3. Microcosms | ||||
| Reconstituted Human Epithelia (RHE) | Biofilms form on top of human keratinocytes derived from buccal mucosa. | Takes into account some host factors, such as receptor specificity. | Human cells - bacteria biofilms interactions.
Used to study oral biofilms. | [401] |
| Zürich Oral Biofilm-model | Biofilms form on hydroxyapatite disposed in 24-well microtiter plates | Can study population dynamics and antibiotic resistance and tolerance at the same time
Semi high throughput | Used to study oral biofilms. | [402] |
| Zürich Burn Biofilm-model | Polymicrobial biofilms are grown on gauze discs of DermaPlast recovered by a protein pellicle disposed on 24-welled microtiter plates. | Allows the study of structure of polymicrobial biofilms.
High repeatability. | Mimics biofilms development on burns.
Suitable to assess antimicrobial efficiencies. | [403] |
| Endothelial Cells Under Flow model | Biofilms forms on human microvascular endothelial cells attached to microscope slide, perfused with media, under an inverted fluorescent microscope. | Has a continuous flow of nutrients.
Biofilm development can be imaged and cells can be tracked. Takes into account shear forces in blood vessels. | Biofilm formation and dynamics on blood vessels and valves. | [404] |
| Airway Epithelial cell Model | Airway epithelial cells are disposed on collagen-coated membranes. | Allows formation of air-liquid biofilm formation. | Models chronic rhinosinusitis, cystic fibrosis
and other biofilm-related pulmonary infections. | [405] |
| Multiple Sorbarod device (MSD) | Modified SBF system with five replicate plugs. | Allows for multiple replicates. | Used to replicate oral microcosms, perfused with saliva and multispecies biofilms | [406] |
| Microfluidic Co-culture model | Microfluidic channels covered with HeLa cells over which biofilms form. | Analysis of host-bacteria interactions.
Real-time visualization of biofilm development. | Used to mimic gastrointestinal colonization.
Human cells – bacteria biofilms interactions. | [407] |
| 4. Ex vivo | ||||
| Root canal biofilms | Extracted tooth are embedded in silicone putty and irrigated. | Irrigation of dental surfaces.
Allows imaging. | Remove dental biofilms and root canal infections | [408] |
| Cardiac valve ex vivo model | Use of excised porcine heart valve. | Study initial bacteria and the valve tissue interactions.
Adapted to imaging (field emission scanning microscopy). | Evaluate progression of endocarditis | [409] |
| Candidiasis in vaginal mucosa | Rabbit vaginas are placed in 6-well tissue culture plates. | Optimal for microscopic evaluations (confocal and scanning). | Model of candidiasis | [27] |
| RWV Bioreactor | System able to grow 3D structures.
Bubble-free aeration: Maintains cell polarity, differentiation and extracellular matrix production: | Circumvents conventional monolayers limitations.
Minimizes mechanical cell damage. Microgravity conditions are maintained. | Has been used to model P. aeruginosa infection in lungs, Salmonella in gut and uropathogenic E. coli | [410,411,412] |
3. Ex vivo Biofilm Models
4. Non-Mammalian in vivo Models
| Organism | Size | Generation time | Temp. (°C) | Immune system | Follow-up of host infection | Relevant Model | Human Pathogens studied | References |
|---|---|---|---|---|---|---|---|---|
| Tetrahymena pyriformis | 20 × 40 μm | 7 hours | 22–26 | Unknown | Real-time through bacterial fluorescent markers | Biofilm grazing, Virulence and toxicity | Klebsiella pneumoniae, Legionella pneumophila, Vibrio cholerae | [413] |
| Acanthamoeba sp. | 15 to 35 μm | 6–10 hours | 19–25 | Macrophage analog | -- | Biofilm grazing, Phagocytosis, intracellular survival | L. pneumophila, Cryptococcus neoformans, Candida albicans, Methicillin-resistant Staphylococcus aureus (MRSA), V. cholerae | [414] |
| Dictyostellium discoideum (Slime mould) | 10–20 μm | 4–12 hours | 19–25 | Macrophage analog | Real-time through bacterial fluorescent markers | Biofilm grazing, Phagocytosis, intracellular survival | Pseudomonas aeruginosa, L. pneumophila, Listeria monocytogenes (intracellular pathogens) | [415] |
| Lemna minor (Duckweed) | 2–5 mm × 1.5–3.5 mm | 1 week | 28 | Unknown | ✗ | Biofilm formation and virulence | S. aureus, P. aeruginosa, Salmonella spp., Shigella spp., Yersinia spp. | [34] |
| Medicago sativa (Wounded alfalfa) | Seedlings | 3 months | 30 | Unknown | ✗ | Chronic bacterial lung infections, | P. aeruginosa, Burkholdheria cepacia | [416] |
| Arabidopsis thaliana (Thale cress) | 1 to 20–25 cm | 3 weeks | 20–25 | Analog pathwaysto MAPK | ✗ | Biofilm formation and virulence | Pseudomonas spp., S. aureus | [417] |
| Hirudo sp (Leach) | 15–40 mm (adult) | -- | 10–35 | Unknown | -- | Biofilm competition and gut colonization | Aeromonas spp. | [418] |
| Panagrellus redivivus (Sour paste nematode) | 1 mm × 50 μm | 3–5 days | 37 | Innate immunity (Toll-like receptor, MAPK) | -- | Biofilm formation, virulence, gut colonization | P. aeruginosa, Salmonella enterica, and S. aureus | [419] |
| Caenorhabditis elegans (Round worm) | 1 mm ×100 μm | 4–7 days | 22–27 | Innate immunity (Toll-like receptor, MAPK) | Real-time through bacterial fluorescent markers | Biofilm formation, virulence, gut colonization | Microbacterium nematophilum, Escherichia coli, Shigella flexneri, V. cholerae, Shewanella spp. Listeria spp., S. aureus, Streptococcus sp. | [420] |
| Galleria mellonella (Wax moth caterpillar) | 3 cm in length | -- | 30 | Innate immunity (Toll-like receptor, MAPK, NFκB) | -- | Biofilm formation and virulence | Pseudomonas spp., Proteus mirabilis, E. coli, Bacillus cereus, Bacillus thuringiensis, C. albicans, C. neoformans | [421] |
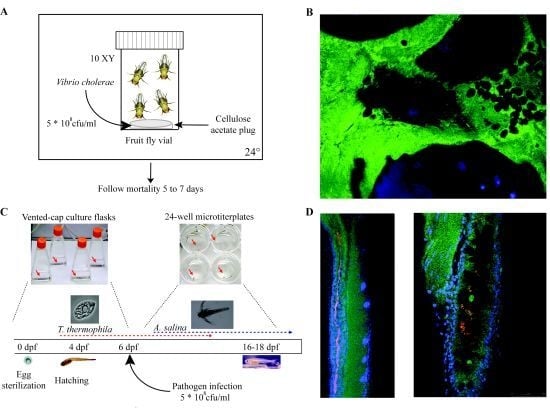
| Drosophila melanogaster (Fruit fly) | 3 mm | 10 days | 12–30 | Innate immunity (Toll-like receptor, Imd, MAPK, NFκB) | Real-time through fluorescent markers, LacZ fusions available | Biofilm formation, virulence, gut colonization | Wolbachia spp., Serratia marcescens, Erwinia spp., Pseudomonas entomophila, C. neoformans, Francisella novicida, L. monocytogenes, V. cholerae, C. albicans | [422] |
| Danio rerio (Zebrafish) | 3–5 mm (larvae)
6–6.5 cm (adult) | 3–4 months | 23–28 | Adaptive and innate | Real-time through fluorescent markers both on host and bacteria | Biofilm formation, virulence, gut colonization | Mycobacterium marinum, Oodinium, Microsporidia, E. coli, Pseudomonas spp., Salmonella spp. Vibrio spp. | [423] |

5. Tissue-Associated Biofilm Models
| Type of biofilm-related infection | Type of model | Animal | Microorganisms | Direct biofilm /chronic infection | Technical details | References |
|---|---|---|---|---|---|---|
| Tissue-related infections | ||||||
| CF lung infections | Agar-bead based infection model | Rats, mice, cats, guinea pigs and monkeys | P.a, S.a, H.i and B. cenocepacia | B | Intratracheal route of infection | [54,55,56,57,58,59] |
| Seaweed alginate microsphere infection | Rats, mice, guinea pigs | P.a | B | Intratracheal route of infection | [62,63,64] | |
| Agar-bead based model | Mice | P.a | B | Intravenous injections | [66,67,424,425] | |
| CF model (CFTR-/- mice) | Mice, pigs, ferrets | P.a, S.a and B. cepacia | B | Agar-bead based intranasal route of infection | [68,70,74,426] | |
| COPD associated infections | COPD/emphysema | Mice | H.i | B | Intranasal route of infection | [76] |
| Diffuse panbronchiolitis | Chronic diffuse panbronchiolitis | Mice | P.a | CI | Piece of intravenous catheter coated with P. aeruginosa | [80,81,82] |
| Urinary tract infections | Murine cystitis model | Mice | E.c, K.p | B | Transurethral catheter for inoculating bacteria in bladder | [89,91] |
| Rat model of chronic cystitis | Rats | E.c | CI | Transurethral catheter for inoculating bacteria in bladder | [86] | |
| Chronic bacterial prostatitis | Experimental model of chronic prostatitis | Rats | P.m, E.c | CI | Prostatic urethral injections | [115,117,118] |
| Urinary Stones or Struvites | Rat model of urolithiasis | Rats | P.m, U. urealyticum | B | Foreign body like zinc disc or chalk in bladder | [127,129,130,427] |
| Pyelonephritis | Urinary stone genesis model | Rats | P.m | B | Zinc discs in bladder | [127] |
| Intestinal Infections | Intestinal colonization model | Mice | C. rodentium | Oral dosing of bacteria | [141,142] | |
| Streptomycin-treated mouse model | Mice | E.c, Salmonella | B | Oral dosing of bacteria | [138,140] | |
| Gall Bladder Infections | Chronic infection model | Mice | Salmonella | CI | Oral infection | [145,146,147] |
| Chronic wounds infection | Needle scratch model, Skin abrasion | Mice | MRSA | B | Scratch with 28 gauge needle on skin to damage epidermis | [152] |
| Wound infection model | Mice | MSSA | B | Full-thickness wound is established through the panniculus carnosus on the back of animals | [158] | |
| Excisional wound model | Mice | S.a | B | Cuts made on the back of mice | [156] | |
| Ischemic wound model | Rats | P.a | B | Pressure-induced wounds | [159] | |
| Cutaneous wound healing model | Rabbits | P.a | B | Circular punch-wounds in ear | [428] | |
| Cutaneous porcine wound model | Pigs | S.a | B | Partial thickness wounds on paravertebral area using a modified electrokeratome | [157] | |
| Diabetic foot wound model | Mice | E.c, B. fragilis and C. perfringens | B | Leptin-receptor deficient mice injected in inner thigh | [161] | |
| Infective endocarditis | Catheter-induced IE | Rabbits | S.a | B | High inoculum of bacteria injected intravenously | [164,166] |
| Low-Grade bacteremia model of IE | Rats | L. lactis | B | Low-grade inoculum of bacteria injected intravenously | [169] | |
| Chronic otitis media | COM with effusion | Gerbils | H.i, S. pneumoniae | B | Injected percutaneously into the superior posterior chamber of the left middle ear | [429] |
| Chinchilla Model of COM | Chinchillas | H.i, P.a, group A Streptococcus | B | Bacteria is injected bilaterally via a transbullar approach | [183,186] | |
| Nonhuman primate model of COM | Cynomolugus macaques | P.a | B | Perforation of the tympanic membrane and inoculation of the middle ear | [190] | |
| COM | Rats | P.a | B | Intranasal inoculation using teflon cannula | [191] | |
| COM | Mice | S. pneumoniae | CI | Spontaneous OM development in plasminogen deficient mice | [192] | |
| Chronic rhinosinusitis | Chronic rhinosinusitis | Rabbits | S. pneumoniae | CI | Hole drilled into the dorsum of nose, cotton wool inserted and inoculated with 10.8 bacteria | [195] |
| Chronic rhinosinusitis | Mice | L. sakei, C. tuberculostearicum | CI | Intranasal inoculation | [197] | |
| Experimental rhinosinusitis biofilm model | Sheep | S.a | B | Ostium occluded and bacteria instilled | [198] | |
| Dental caries | Experimental caries | Hamsters | S. mutans | CI | Oral inoculation | [206] |
| Model of Cystic Fibrosis | Mice | S. mutans | B | Swabbing the oral cavity of CFTR knock out mice | [205] | |
| Model of periodontal disease | Rats | P. gingivalis | B | Topical administration of bacteria | [207] | |
| Periodontitis | Oral infection model | Mice | T. denticola, P. gingivalis | CI | Oral inoculation by gavage | [207,223] |
| Experimental periodontitis | Mice | F. nucleatum, P. gingivalis, T. forsythia | B | Oral gavage using a feeding needle | [222,224] | |
| Experimental periodontitis | Rabbits | P. gingivalis | B | Oral inoculation | [430,431] | |
| Osteomyelitis | Chronic osteomyelitis | Rabbits | S.a | CI | Injection in tibial metaphysis into the intramedullar cavity | [229] |
| Osteomyelitis model of biofilm | Rabbits | S.a | B | Injection in tibial metaphysis into the intramedullar cavity | [231,232] | |
| Experimental chronic osteomyelitis | Rats | S.a | CI | Hole is drilled into the medullar cavity, bacteria are injected into the bone | [233] | |
| Experimental model of osteomyelitis | Mice | S.a | CI | Bioluminescent strain of S. aureus is inoculated into the femurs of mice | [234] | |
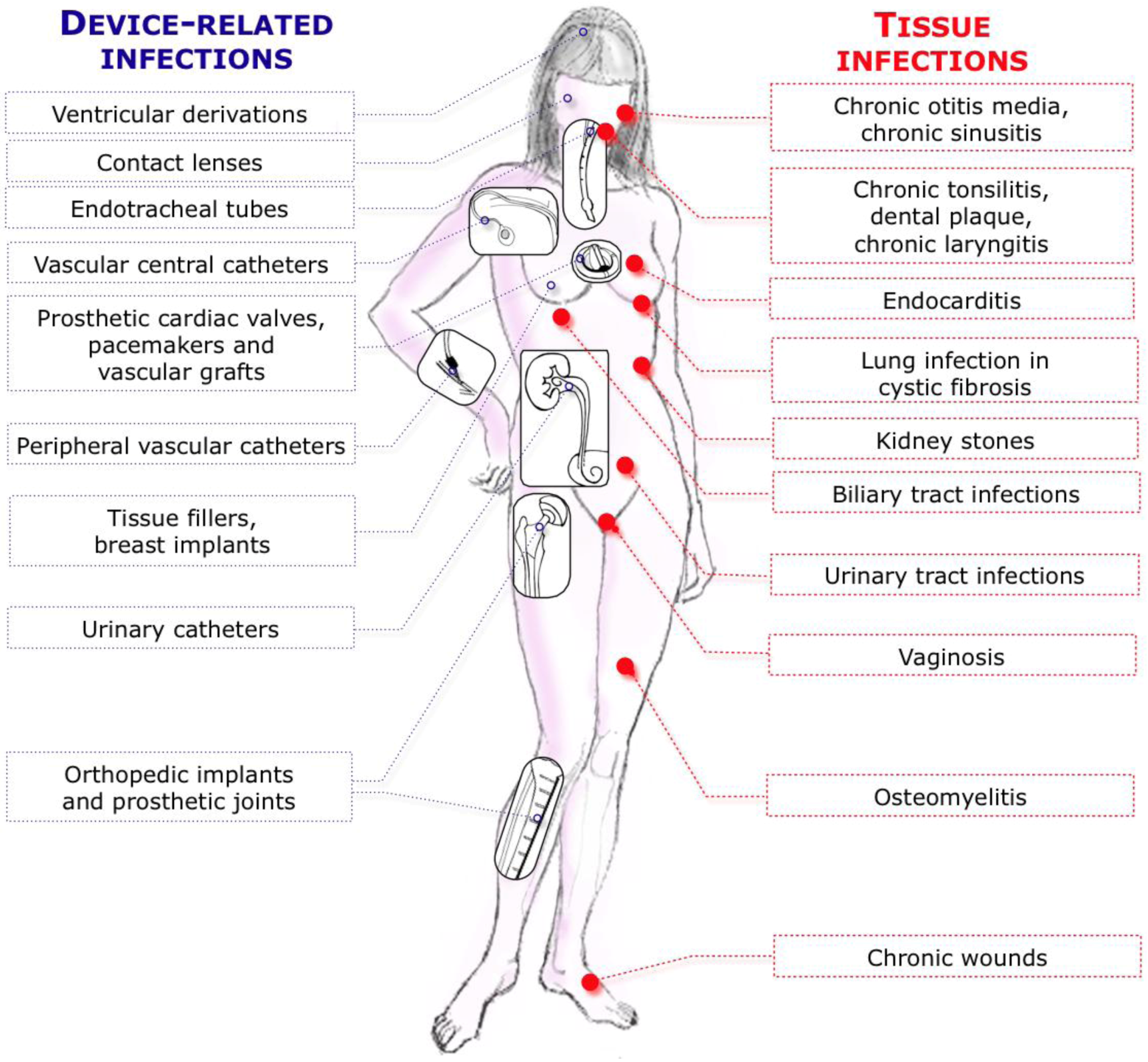
| Device related-infections | ||||||
| Vascular Catheter | CVC | Rats | S.e, S.a, C.a | B | Catheter tip in superior vena cava through jugular vein, tunneled subcutaneously and exits on the back. Use of restraint jacket | [239,245,432] |
| CVC | Rabbits | S.e, S.a | B | Catheter tip in superior vena cava through jugular vein, tunneled subcutaneously and exits on the back. Use of restraint jacket | [247,252,253] | |
| Totally implantable venous access port | Rats | S.a, S.e, P.a, E.c | B | Catheter tip in superior vena cava through jugular vein, tunneled subcutaneously and connected to a subcutaneous port | [256] | |
| Urinary tract catheters | Bladder glass bead (surgical) | Rats | E.c | B | Bead colonized by E. coli biofilm surgically inserted in the bladder. Urethra clamped 1h/day to reproduce vesico-ureteral reflux | [260] |
| Bladder pieces of catheter (surgical) | Rats | P.a | B | Surgical insertion of pieces of urinary catheter. Bacterial inoculation is made inside the bladder, after catheter insertion | [261] | |
| Mice | E.c | B | [262] | |||
| Bladder pieces of catheter (non surgical) | Rabbits | P.a, P.m | B | Use of urethral catheter and/or metal stylet in order to transurethrally insert pieces of catheter inside the animal bladder | [263,265] | |
| Rats | P.a | B | [433] | |||
| Mice | P.a, P.m, E.c, E. faecalis | B | [264,266] | |||
| Externalized urethral catheter | Rabbits | E.c | B | Urethral catheter inserted and connected to a urine collector via a closed system in order to mimic an externalized system | [269,270,271] | |
| Sheep | B | [272] | ||||
| Orthopedic implants | Foreign-body in tibia | Rabbits | S.a | B | Silicone rubber catheter inserted into the tibia and associated with sclerosing agent to induce aseptic necrosis | [277,434] |
| Foreign-body in tibia | Rabbits | S.a | B | Titanium cylinder or bone cement inserted into the tibia. Bacterial inoculation up to 4 months after foreign-body placement | [279,435] | |
| Electrode inserted in tibia | Rabbits | S.e | B | S. epidermidis is injected inside tibia through a hole. A stainless steel electrode is inserted inside medullar cavity + bone cement | [436] | |
| Devascularized bone and metal screws | Rabbits | B | A piece of diaphyseal radial bone is removed. Then, this devascularized bone is replaced inside the wound using metal screws | [280] | ||
| Spinal device model | Rabbits | S.a | B | Partial laminectomy followed by a wire implantation of the transverse processes of different vertebra (T13, L3, L6) | [437] | |
| Titanium wire inside tibia | Rats | S.a | B | Insertion in the medullar canal of a long titanium wire. Before the insertion of the foreign-body, S. aureus inoculum is injected | [282,283] | |
| Stainless steel pin inserted through tibia | Mice | S.a | B | Stainless steel pin incubated 20 minutes with bioluminescent S. aureus and then inserted transcortically through mice tibia | [281,438] | |
| Bone cement in tibia | Dogs | S.a | B | Removal of a cortical part of tibial metaphysis. Then, polymethylmethacrylate cement and S. aureus are inserted in bone pocket | [284] | |
| Cylindrical device in femoral canal | Dogs | S.e, S.a, E.c | B | Cylindrical device (made of stainless steel, cobalt chromium, polyethylene or polymethylmetacrylate) inserted inside femoral canal | [278] | |
| Intramedullar nail | Dogs | E.c, P.a | B | Mid-diaphyseal osteotomy and internal fixation with an intramedullar nail inoculated, before fixation, with E. coli and P. aeruginosa | [439] | |
| Fracture fixation stainless steel plates | Sheep | S.a | B | Membranes colonized by S. aureus biofilms are positioned on stainless steel plates and drilled on a cortical surface of sheep tibia | [440] | |
| Prosthetic joints | Hemiarthroplasty and bone cement | Rabbits | E.c | B | Knee hemiarthroplasty and acrylic bone cement followed by intraarticular E. coli injection | [287] |
| Total knee replacement | Rabbits | S.a | B | [288] | ||
| Hip stainless steel prosthesis | Rabbits | S.a | B | [441] | ||
| Silicone-elastomer implant | Rabbits | S.a | B | Partial knees arthroplasty using silicone-elastomer implants. At the end of surgical procedure, S. aureus is injected into the joint | [286] | |
| Pin inside femur with the tip in the joint | Mice | S.a | B | Stainless steel pin is inserted inside the femoral canal and the distal end of the pin protrudes inside the joint space | [289] | |
| Endotracheal tubes | Ventilated sheep | Sheep | Oral flora | B | Animals are intubated and ventilated for 24 to 96 hours before being sacrificed for endotracheal tubes analysis | [293,297] |
| Ventilated pig with induced pneumonia | Pigs | S.a | B | [294] | ||
| Ventilated dog | Dogs | P.a | B | [295] | ||
| Vascular grafts | Infrarenal aortic vascular graft | Dogs | S.e | B | Implantation of dacron prosthesis colonized by S. epidermidis on the infrarenal aorta of a dog leading to a prosthetic graft infection | [300,442,443] |
| Infrarenal aortic vascular graft | Pigs | S.a | B | Surgical implantation in the infrarenal abdominal aorta of vascular grafts colonized by S. aureus | [303] | |
| Tissue fillers | Breast implants | Pigs | S.e | B | Each pig received up to 6 miniature silicone gel-filled implants into submammary pockets + inoculated with S. epidermidis | [306,307] |
| Breast implants | Rats | S.a | B | [444] | ||
| Contact lenses | Damaged cornea and contact lenses | Mice | Fusarium spp. | B | Fusarium spp. are grown as biofilm on silicone hydrogel contact lenses and mouse cornea are damaged by scratches or abrasion | [311] |
| Dental implants | Titanium screw into hard palate | Rats | A. actinomycetemcomitans | B | Biofilm-inoculated titanium implants transmucosally placed into rat hard palate | [315] |
| Intrabuccal splints and disks | Humans | Oral flora | B | A removable fixation system applied on the mandibular region, inside the mouth of healthy volunteers. On these splints, disks made of various surfaces can be placed to study biofilm formation | [316] | |
| Subcutaneous models | Tissue cage | Mice, rats, hamsters, guinea pigs, ponies | S.a, S.e, A. radicidentis | B | Rigid tubes, mostly made of teflon and perforated with holes, sealed at each end, possibly filled with glass bead and usually inserted in the flank of animal. Tissue cage fluid can be collected by percutaneous aspiration | For review: [22] |
| Vascular catheter | Mice, rats, rabbits | S.e, S.a, E.c | B | Insertion of a 1-cm segment of vascular catheter in a subcutaneous space. At the end of the experiment, mice are euthanized, catheter segment is removed, vortexed in order to recover the biofilm | For review: [22,243] | |
| Cement disks | Rabbits | E.c | B | Acrylic bone cements shaped like disks and colonized by E. coli biofilm are surgically inserted into subcutaneous pockets on the back | [329] | |
| Pacing device | Rabbits | S.e, S. capitis, E. c and A. baumannii | B | Pacing device is inserted in subcutaneous pockets on the back of the animals. Bacterial contamination is made inside the pocket, at the end of the experiment | [330] | |
| Fabric to mimic cardiac valves | Guinea pigs, mice, rabbits | S.e, P.a | B | Various types of implants impregnated with antibiotics or not are incubated with a bacterial solution in order to allow biofilm formation. Afterwards, these devices are inserted into subcutaneous pockets | [331,332,334] | |
| Vascular grafts | Hamsters, mice, rats | S.e | B | Gore-tex implants colonized by S. epidermidis biofilm are inserted into subcutaneous pockets | [335,336,445] | |
| Polyethylene disks | Mice, rabbits | E.c, P.a | B | Subcutaneous implantation of polyethylene disks | [338] | |
| Beads | Rats | S.a | B | Polymethylmethacrylate beads loaded or not with various compounds are inserted in subcutaneous space | [339] | |
| Surgical mesh | Mice | S.a | B | Insertion of resorbable or non resorbable surgical meshes colonized by S. aureus biofilm inside subcutaneous pockets | [340,341] |
5.1. Lung Infections
5.1.1. Cystic Fibrosis (CF) Related Lung Infections
- Intratracheal route: Cash et al., were the first to describe a rat model to establish P. aeruginosa chronic lung infection, lasting up to 1 month, by immobilizing bacteria in agar beads [54]. This model was later adapted to mice, guinea pigs, cats, monkeys and to other pathogenic agents like S. aureus, Burkholderia cenocepacia and Haemophilus influenzae [54,55,56,57,58,59]. This model depicts human clinical pathologies such as bacterial persistence and airway inflammation [60]. Amongst very recent uses, it successfully evaluated the efficacy of liposomal bismuth-ethanediol loaded tobramycin against P. aeruginosa infection in rat lungs [61]. Another rat model developed by Pedersen et al., used bacteria embedded in seaweed alginate microspheres [62]. Amongst other advances, this model showed the role of Psl and Pel polysaccharides as a scaffold of biofilms formed by mucoid P. aeruginosa phenotype biofilms in mice [63]. In addition, to study pharmacokinetics/dynamics of colistin and imipenem, a neutropenic mouse model of lung biofilm infection was developed [64].
5.1.2. Other Lung Conditions
5.2. Urinary Tract Infection (UTI)
5.2.1. Cystitis
5.2.2. Chronic Bacterial Prostatitis (CBP)
5.2.3. Other UTI
5.3. Digestive Infections
5.3.1. Intestinal Infections
5.3.2. Gall Bladder Infections
5.4. Wounds Infections

5.5. Endocarditis
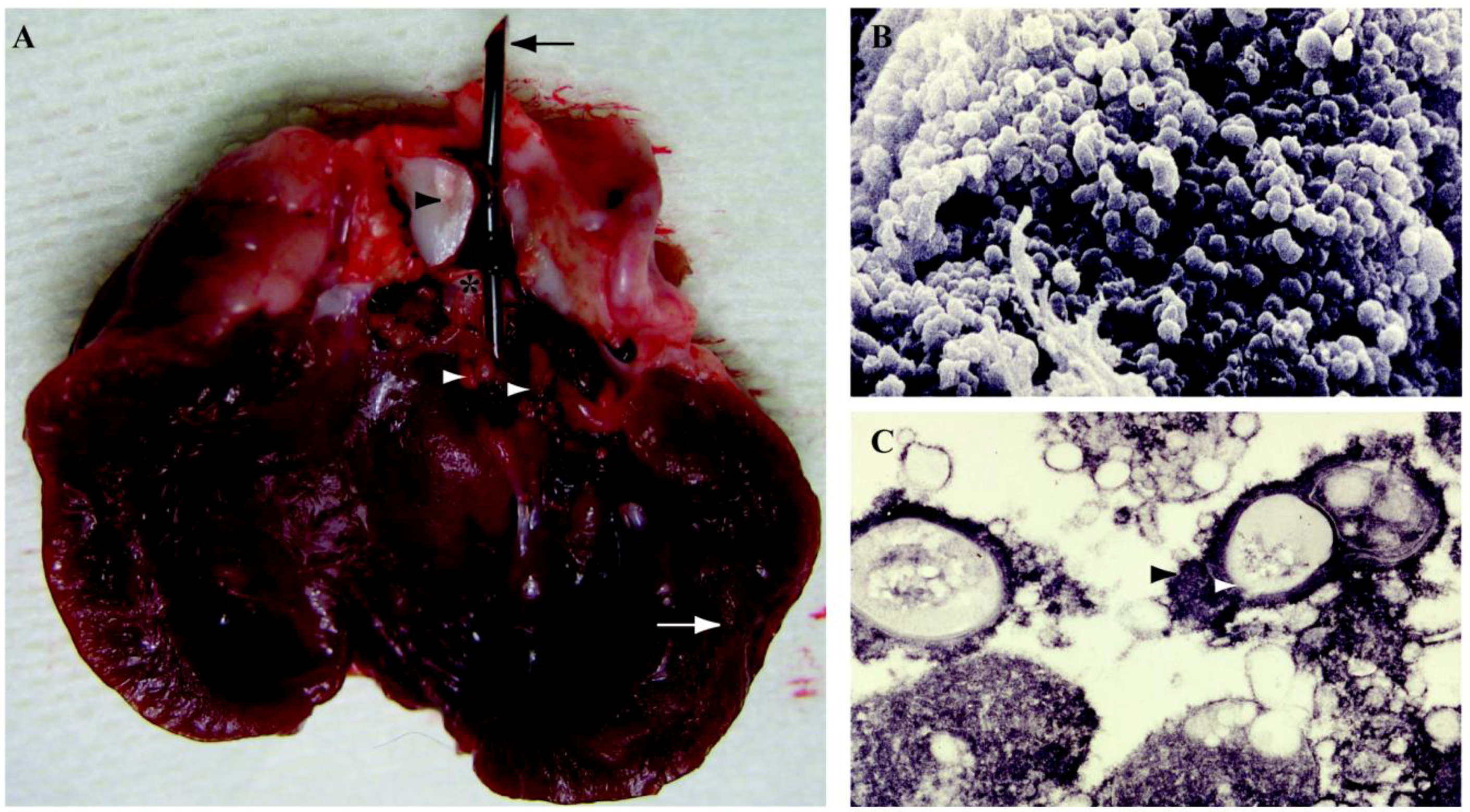
5.6. Ear, Nose, Throat Infections
5.6.1. Chronic Otitis Media (OM)
5.6.2. Chronic Rhinosinusitis (CRS)
5.7. Dental Biofilms
5.7.1. Dental Caries
5.7.2. Periodontitis
5.8. Other Biofilm-Related Infections
5.8.1. Osteomyelitis
6. In vivo Models of Device-Related Infections
6.1. Site-Specific Models
6.1.1. Vascular Catheters
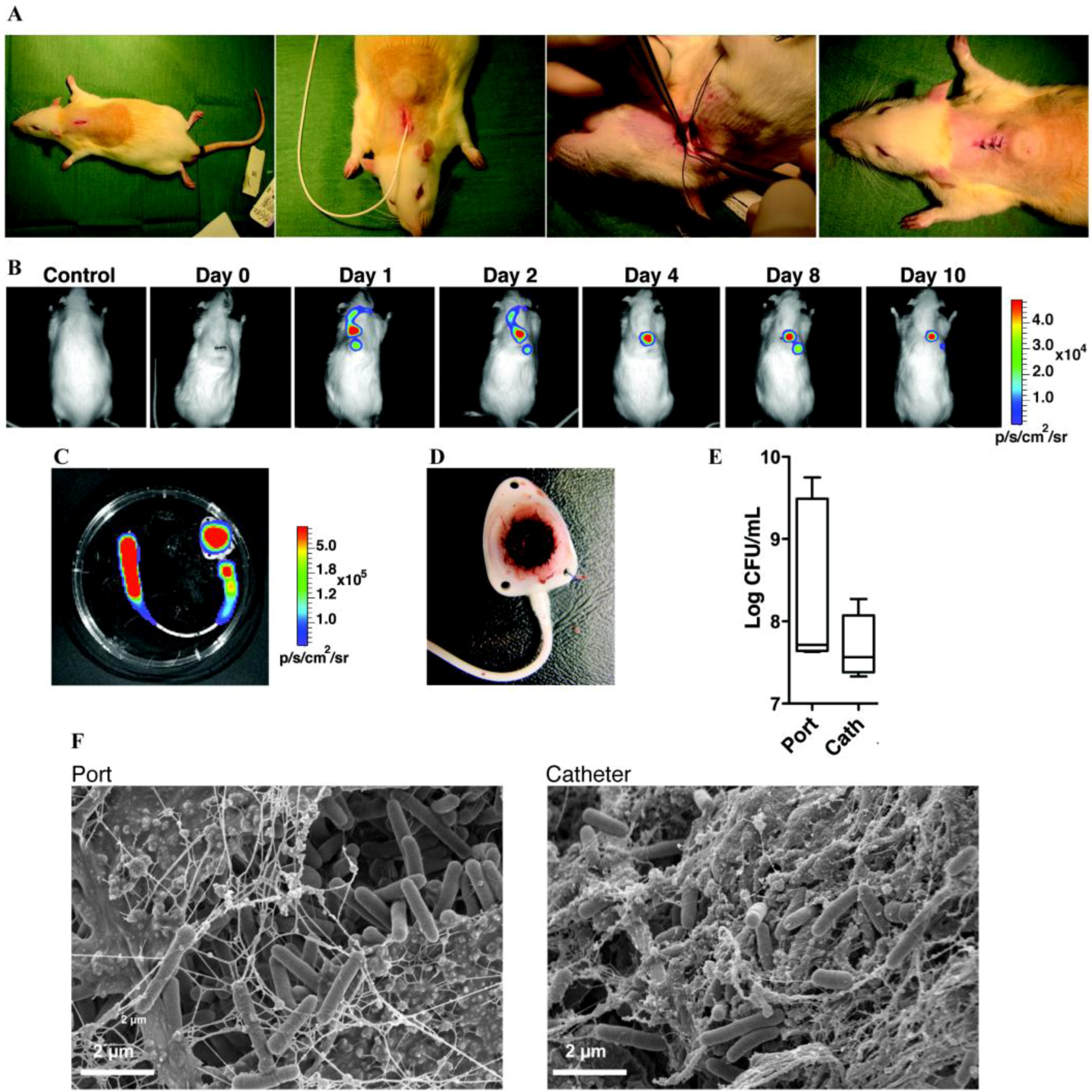
6.1.2. Urinary Catheters
6.1.3. Orthopedic Implants and Prosthetic Joints
6.1.4. Endotracheal Tubes

6.1.5. Vascular Grafts
6.1.6. Tissue Fillers
6.1.7. Contact Lenses
6.1.8. Dental Implants
6.2. Subcutaneous Foreign-Body Models
6.2.1. Tissue Cage
6.2.2. Vascular Catheters
6.2.3. Other Subcutaneous Models
7. Take-Home Messages and Future Directions
7.1. Pitfalls of in vivo Biofilm-Related Infection Models
| Type of biofilm-related infection | Animal model for the disease (References) | Implication of biofilms in clinics (References) |
|---|---|---|
| Tissue-related infections | ||
| Keratitis | Yes [446] | Yes [447] |
| Endophthalmitis | Yes [448] | Yes [449] |
| Chronic tonsillitis | Yes [450] | Yes [451] |
| Chronic laryngitis | No | Yes [452] |
| Bacterial Vaginosis | Yes [453] | Yes, reviewed [454] |
| Meningitidis | Yes [455] | Yes, discussed and reviewed in [456] |
| Device related-infections | ||
| Cochlear implants | No | Yes [457] |
| Voice prosthesis | No | Yes [458] |
| Neurological devices | No | Yes, reviewed in [459] |
| Penile prosthesis | No | Yes, reviewed in [460] |
| Biliary stent | Yes [461] | Yes [462] |
7.2. Under-Developed Aspects of Biofilm-Related Infections
7.2.1. Synergy between Biofilm Tolerance and Resistance Genes and Their Impact on Nosocomial Infections
7.2.2. A Switch towards Models to Study Biofilm Polymicrobial Infections?
7.2.3. What Can We Expect from Systems Biology, Computational Biology, Ecology or Experimental Evolution?
Acknowledgments
Conflict of Interest
References
- Henrici, A.T. Studies of freshwater bacteria: I. A direct microscopic technique. J. Bacteriol. 1933, 25, 277–287. [Google Scholar]
- Costerton, J.W.; Geesey, G.G.; Cheng, K.J. How bacteria stick. Sci. Am. 1978, 238, 86–95. [Google Scholar]
- Geesey, G.G.; Richardson, W.T.; Yeomans, H.G.; Irvin, R.T.; Costerton, J.W. Microscopic examination of natural sessile bacterial populations from an alpine stream. Can. J. Microbiol. 1977, 23, 1733–1736. [Google Scholar]
- Al-Azemi, A.; Fielder, M.D.; Abuknesha, R.A.; Price, R.G. Effects of chelating agent and environmental stresses on microbial biofilms: relevance to clinical microbiology. J. Appl. Microbiol. 2011, 110, 1307–1313. [Google Scholar]
- Hoiby, N.; Bjarnsholt, T.; Givskov, M.; Molin, S.; Ciofu, O. Antibiotic resistance of bacterial biofilms. Int. J. Antimicrob. Agents 2010, 35, 322–332. [Google Scholar]
- Iibuchi, R.; Hara-Kudo, Y.; Hasegawa, A.; Kumagai, S. Survival of Salmonella on a polypropylene surface under dry conditions in relation to biofilm-formation capability. J. Food Prot. 2010, 73, 1506–1510. [Google Scholar]
- Jabra-Rizk, M.A.; Falkler, W.A.; Meiller, T.F. Fungal biofilms and drug resistance. Emerg. Infect. Dis. 2004, 10, 14–19. [Google Scholar]
- McKew, B.A.; Taylor, J.D.; McGenity, T.J.; Underwood, G.J. Resistance and resilience of benthic biofilm communities from a temperate saltmarsh to desiccation and rewetting. Isme J. 2011, 5, 30–41. [Google Scholar]
- Costerton, J.W. The etiology and persistence of cryptic bacterial infections: a hypothesis. Rev. Infect. Dis. 1984, 6 Suppl 3, S608–S616. [Google Scholar]
- Costerton, J.W.; Irvin, R.T.; Cheng, K.J. The bacterial glycocalyx in nature and disease. Annu. Rev. Microbiol. 1981, 35, 299–324. [Google Scholar]
- Costerton, J.W.; Irvin, R.T.; Cheng, K.J. The role of bacterial surface structures in pathogenesis. Crit. Rev. Microbiol. 1981, 8, 303–338. [Google Scholar]
- Hoiby, N. Pseudomonas aeruginosa infection in cystic fibrosis. Diagnostic and prognostic significance of Pseudomonas aeruginosa precipitins determined by means of crossed immunoelectrophoresis. A survey. Acta Pathol. Microbiol. Scand. Suppl. 1977, 1–96. [Google Scholar]
- Hoiby, N.; Doring, G.; Schiotz, P.O. The role of immune complexes in the pathogenesis of bacterial infections. Annu. Rev. Microbiol. 1986, 40, 29–53. [Google Scholar]
- Lam, J.; Chan, R.; Lam, K.; Costerton, J.W. Production of mucoid microcolonies by Pseudomonas aeruginosa within infected lungs in cystic fibrosis. Infect. Immun. 1980, 28, 546–556. [Google Scholar]
- Potera, C. Forging a link between biofilms and disease. Science 1999, 283, 1837–1839. [Google Scholar]
- NIH RESEARCH ON MICROBIAL BIOFILMS. Available online: http://grants.nih.gov/grants/guide/pa-files/PA-03–047.html.
- Lebeaux, D.; Ghigo, J.M. Management of biofilm-associated infections: what can we expect from recent research on biofilm lifestyles? Med. Sci. (Paris) 2012, 28, 727–739. [Google Scholar] [CrossRef]
- Cremieux, A.C.; Carbon, C. Experimental models of bone and prosthetic joint infections. Clin. Infect. Dis. 1997, 25, 1295–1302. [Google Scholar]
- Morton, D.B. Ethical considerations in the use of animal models in infection. Clin. Microbiol. Infect. 1998, 4, 613–614. [Google Scholar]
- Russell, W.M.S.; Burch, R.L. The Principles of Humane Experimental Technique. Methuen & Co Ltd: London, UK., 1959. [Google Scholar]
- McBain, A.J. Chapter 4: In vitro biofilm models: an overview. Adv. Appl. Microbiol. 2009, 69, 99–132. [Google Scholar]
- Coenye, T.; Nelis, H.J. In vitro and in vivo model systems to study microbial biofilm formation. J. Microbiol. Methods 2010, 83, 89–105. [Google Scholar]
- Christensen, G.D.; Simpson, W.A.; Younger, J.J.; Baddour, L.M.; Barrett, F.F.; Melton, D.M.; Beachey, E.H. Adherence of coagulase-negative staphylococci to plastic tissue culture plates: a quantitative model for the adherence of staphylococci to medical devices. J. Clin. Microbiol. 1985, 22, 996–1006. [Google Scholar]
- Stepanovic, S.; Vukovic, D.; Hola, V.; Di Bonaventura, G.; Djukic, S.; Cirkovic, I.; Ruzicka, F. Quantification of biofilm in microtiter plates: overview of testing conditions and practical recommendations for assessment of biofilm production by staphylococci. APMIS 2007, 115, 891–899. [Google Scholar]
- Rudney, J.D.; Chen, R.; Lenton, P.; Li, J.; Li, Y.; Jones, R.S.; Reilly, C.; Fok, A.S.; Aparicio, C. A reproducible oral microcosm biofilm model for testing dental materials. J. Appl. Microbiol. 2012, 113, 1540–1553. [Google Scholar]
- Berry, R.E.; Klumpp, D.J.; Schaeffer, A.J. Urothelial cultures support intracellular bacterial community formation by uropathogenic Escherichia coli. Infect. Immun. 2009, 77, 2762–2772. [Google Scholar]
- Harriott, M.M.; Lilly, E.A.; Rodriguez, T.E.; Fidel, P.L., Jr.; Noverr, M.C. Candida albicans forms biofilms on the vaginal mucosa. Microbiology 2010, 156, 3635–3644. [Google Scholar]
- Lenton, P.; Rudney, J.; Chen, R.; Fok, A.; Aparicio, C.; Jones, R.S. Imaging in vivo secondary caries and ex vivo dental biofilms using cross-polarization optical coherence tomography. Dental materials 2012, 28, 792–800. [Google Scholar]
- Melican, K.; Sandoval, R.M.; Kader, A.; Josefsson, L.; Tanner, G.A.; Molitoris, B.A.; Richter-Dahlfors, A. Uropathogenic Escherichia coli P and Type 1 fimbriae act in synergy in a living host to facilitate renal colonization leading to nephron obstruction. PLoS Pathog 2011, 7, e1001298. [Google Scholar]
- Simmons, W.L.; Dybvig, K. Mycoplasma biofilms ex vivo and in vivo. FEMS Microbiol. Lett. 2009, 295, 77–81. [Google Scholar]
- Wolcott, R.D.; Rumbaugh, K.P.; James, G.; Schultz, G.; Phillips, P.; Yang, Q.; Watters, C.; Stewart, P.S.; Dowd, S.E. Biofilm maturity studies indicate sharp debridement opens a time- dependent therapeutic window. J. Wound Care 2010, 19, 320–328. [Google Scholar]
- Boman, H.G. Innate immunity and the normal microflora. Immunol. Rev. 2000, 173, 5–16. [Google Scholar]
- Lemaitre, B.; Ausubel, F. Animal models for host-pathogen interactions. Curr. Opin. Microbiol. 2008, 11, 249–250. [Google Scholar]
- Zhang, Y.; Hu, Y.; Yang, B.; Ma, F.; Lu, P.; Li, L.; Wan, C.; Rayner, S.; Chen, S. Duckweed (Lemna minor) as a model plant system for the study of human microbial pathogenesis. PLoS One 2010, 5, e13527. [Google Scholar]
- Diard, M.; Baeriswyl, S.; Clermont, O.; Gouriou, S.; Picard, B.; Taddei, F.; Denamur, E.; Matic, I. Caenorhabditis elegans as a simple model to study phenotypic and genetic virulence determinants of extraintestinal pathogenic Escherichia coli. Microbes and infection 2007, 9, 214–223. [Google Scholar]
- De Bentzmann, S.; Giraud, C.; Bernard, C.S.; Calderon, V.; Ewald, F.; Plesiat, P.; Nguyen, C.; Grunwald, D.; Attree, I.; Jeannot, K.; et al. Unique Biofilm signature, drug susceptibility and decreased virulence in Drosophila through the Pseudomonas aeruginosa two-component system PprAB. PLoS Pathog 2012, 8, e1003052. [Google Scholar] [CrossRef] [Green Version]
- Mulcahy, H.; Sibley, C.D.; Surette, M.G.; Lewenza, S. Drosophila melanogaster as an animal model for the study of Pseudomonas aeruginosa biofilm infections in vivo. PLoS Pathog 2011, 7, e1002299. [Google Scholar]
- Purdy, A.E.; Watnick, P.I. Spatially selective colonization of the arthropod intestine through activation of Vibriocholerae biofilm formation. Proc. Natl. Acad. Sci. USA 2011, 108, 19737–19742. [Google Scholar]
- Neely, M.N.; Pfeifer, J.D.; Caparon, M. Streptococcus-zebrafish model of bacterial pathogenesis. Infect. Immun. 2002, 70, 3904–3914. [Google Scholar]
- Rawls, J.F.; Mahowald, M.A.; Goodman, A.L.; Trent, C.M.; Gordon, J.I. In vivo imaging and genetic analysis link bacterial motility and symbiosis in the zebrafish gut. Proc. Natl. Acad. Sci. USA 2007, 104, 7622–7627. [Google Scholar]
- Rendueles, O.; Ferrieres, L.; Fretaud, M.; Begaud, E.; Herbomel, P.; Levraud, J.P.; Ghigo, J.M. A new zebrafish model of Oro-intestinal pathogen colonization reveals a key role for adhesion in protection by probiotic bacteria. PLoS Pathog 2012, 8, e1002815. [Google Scholar]
- Letamendia, A.; Quevedo, C.; Ibarbia, I.; Virto, J.M.; Holgado, O.; Diez, M.; Izpisua Belmonte, J.C.; Callol-Massot, C. Development and validation of an automated high-throughput system for zebrafish in vivo screenings. PLoS One 2012, 7, e36690. [Google Scholar]
- Ahlund, M.K.; Ryden, P.; Sjostedt, A.; Stoven, S. Directed screen of Francisellanovicida virulence determinants using Drosophila melanogaster. Infect. Immun. 2010, 78, 3118–3128. [Google Scholar]
- Kurz, C.L.; Chauvet, S.; Andres, E.; Aurouze, M.; Vallet, I.; Michel, G.P.; Uh, M.; Celli, J.; Filloux, A.; De Bentzmann, S.; et al. Virulence factors of the human opportunistic pathogen Serratia marcescens identified by in vivo screening. EMBO J. 2003, 22, 1451–1460. [Google Scholar] [CrossRef]
- Stoop, E.J.; Schipper, T.; Huber, S.K.; Nezhinsky, A.E.; Verbeek, F.J.; Gurcha, S.S.; Besra, G.S.; Vandenbroucke-Grauls, C.M.; Bitter, W.; van der Sar, A.M. Zebrafish embryo screen for mycobacterial genes involved in the initiation of granuloma formation reveals a newly identified ESX-1 component. Dis. Models Mechan. 2011, 4, 526–536. [Google Scholar]
- Joshua, G.W.; Karlyshev, A.V.; Smith, M.P.; Isherwood, K.E.; Titball, R.W.; Wren, B.W. A Caenorhabditis elegans model of Yersinia infection: biofilm formation on a biotic surface. Microbiology 2003, 149, 3221–3229. [Google Scholar]
- Silver, A.C.; Rabinowitz, N.M.; Küffer, S.; Graf, J. Identification of Aeromonas veronii genes required for colonization of the medicinal leech, Hirudo verbana. J. Bacteriol. 2007, 189, 6763–6772. [Google Scholar]
- Squiban, B.; Kurz, C.L. C. elegans: an all in one model for antimicrobial drug discovery. Curr. Drug Targets 2011, 12, 967–977. [Google Scholar] [CrossRef]
- Pradel, E.; Ewbank, J.J. Genetic models in pathogenesis. Ann. Rev. Genet. 2004, 38, 347–363. [Google Scholar]
- Cohen, T.S.; Prince, A. Cystic fibrosis: a mucosal immunodeficiency syndrome. Nat. Med. 2012, 18, 509–519. [Google Scholar]
- Hoiby, N.; Ciofu, O.; Bjarnsholt, T. Pseudomonas aeruginosa biofilms in cystic fibrosis. Future Microbiol. 2010, 5, 1663–1674. [Google Scholar]
- Lyczak, J.B.; Cannon, C.L.; Pier, G.B. Lung infections associated with cystic fibrosis. Clin. Microbiol. Rev. 2002, 15, 194–222. [Google Scholar]
- Singh, P.K.; Schaefer, A.L.; Parsek, M.R.; Moninger, T.O.; Welsh, M.J.; Greenberg, E.P. Quorum-sensing signals indicate that cystic fibrosis lungs are infected with bacterial biofilms. Nature 2000, 407, 762–764. [Google Scholar]
- Cash, H.A.; Woods, D.E.; McCullough, B.; Johanson, W.G., Jr.; Bass, J.A. A rat model of chronic respiratory infection with Pseudomonas aeruginosa. Am. Rev. Respir. Dis. 1979, 119, 453–459. [Google Scholar]
- Bernier, S.P.; Silo-Suh, L.; Woods, D.E.; Ohman, D.E.; Sokol, P.A. Comparative analysis of plant and animal models for characterization of Burkholderia cepacia virulence. Infect. Immun. 2003, 71, 5306–5313. [Google Scholar]
- Cheung, A.T.; Moss, R.B.; Leong, A.B.; Novick, W.J., Jr. Chronic Pseudomonas aeruginosa endobronchitis in rhesus monkeys: I. Effects of pentoxifylline on neutrophil influx. J. Med. Primatol. 1992, 21, 357–362. [Google Scholar]
- Starke, J.R.; Edwards, M.S.; Langston, C.; Baker, C.J. A mouse model of chronic pulmonary infection with Pseudomonas aeruginosa and Pseudomonas cepacia. Pediatr. Res. 1987, 22, 698–702. [Google Scholar]
- Thomassen, M.J.; Klinger, J.D.; Winnie, G.B.; Wood, R.E.; Burtner, C.; Tomashefski, J.F.; Horowitz, J.G.; Tandler, B. Pulmonary cellular response to chronic irritation and chronic Pseudomonas aeruginosa pneumonia in cats. Infect. Immun. 1984, 45, 741–747. [Google Scholar]
- Winnie, G.B.; Klinger, J.D.; Sherman, J.M.; Thomassen, M.J. Induction of phagocytic inhibitory activity in cats with chronic Pseudomonas aeruginosa pulmonary infection. Infect. Immun. 1982, 38, 1088–1093. [Google Scholar]
- van Heeckeren, A.M.; Schluchter, M.D. Murine models of chronic Pseudomonas aeruginosa lung infection. Lab. Anim. 2002, 36, 291–312. [Google Scholar]
- Alhariri, M.; Omri, A. Efficacy of liposomal bismuth-ethanedithiol-loaded tobramycin after intratracheal administration in rats with pulmonary Pseudomonas aeruginosa Infection. Antimicrob. Agents Chemother. 2013, 57, 569–578. [Google Scholar]
- Pedersen, S.S.; Shand, G.H.; Hansen, B.L.; Hansen, G.N. Induction of experimental chronic Pseudomonas aeruginosa lung infection with P. aeruginosa entrapped in alginate microspheres. APMIS 1990, 98, 203–211. [Google Scholar] [CrossRef]
- Yang, L.; Hengzhuang, W.; Wu, H.; Damkiaer, S.; Jochumsen, N.; Song, Z.; Givskov, M.; Hoiby, N.; Molin, S. Polysaccharides serve as scaffold of biofilms formed by mucoid Pseudomonas aeruginosa. FEMS Immunol. Med. Microbiol. 2012, 65, 366–376. [Google Scholar]
- Hengzhuang, W.; Wu, H.; Ciofu, O.; Song, Z.; Hoiby, N. In vivo pharmacokinetics/pharmacodynamics of colistin and imipenem in Pseudomonas aeruginosa biofilm infection. Antimicrob. Agents Chemother. 2011, 56, 2683–2690. [Google Scholar]
- Sawai, T.; Tomono, K.; Yanagihara, K.; Yamamoto, Y.; Kaku, M.; Hirakata, Y.; Koga, H.; Tashiro, T.; Kohno, S. Role of coagulase in a murine model of hematogenous pulmonary infection induced by intravenous injection of Staphylococcus aureus enmeshed in agar beads. Infect. Immun. 1997, 65, 466–471. [Google Scholar]
- Kihara, R.; Yanagihara, K.; Morinaga, Y.; Araki, N.; Nakamura, S.; Seki, M.; Izumikawa, K.; Kakeya, H.; Yamamoto, Y.; Tsukamoto, K.; et al. Potency of SMP-601, a novel carbapenem, in hematogenous murine bronchopneumonia caused by methicillin-resistant and vancomycin-intermediate Staphylococcus aureus. Antimicrob. Agents Chemother. 2008, 52, 2163–2168. [Google Scholar] [CrossRef]
- Yanagihara, K.; Kihara, R.; Araki, N.; Morinaga, Y.; Seki, M.; Izumikawa, K.; Kakeya, H.; Yamamoto, Y.; Yamada, Y.; Kohno, S.; et al. Efficacy of linezolid against Panton-Valentine leukocidin (PVL)-positive meticillin-resistant Staphylococcus aureus (MRSA) in a mouse model of haematogenous pulmonary infection. Int. J. Antimicrob. Agents 2009, 34, 477–481. [Google Scholar] [CrossRef]
- Clarke, L.L.; Grubb, B.R.; Gabriel, S.E.; Smithies, O.; Koller, B.H.; Boucher, R.C. Defective epithelial chloride transport in a gene-targeted mouse model of cystic fibrosis. Science 1992, 257, 1125–1128. [Google Scholar]
- Bragonzi, A. Murine models of acute and chronic lung infection with cystic fibrosis pathogens. Int. J. Med. Microbiol. 2010, 300, 584–593. [Google Scholar]
- Hoffmann, N.; Lee, B.; Hentzer, M.; Rasmussen, T.B.; Song, Z.; Johansen, H.K.; Givskov, M.; Hoiby, N. Azithromycin blocks quorum sensing and alginate polymer formation and increases the sensitivity to serum and stationary-growth-phase killing of Pseudomonas aeruginosa and attenuates chronic P. aeruginosa lung infection in Cftr(-/-) mice. Antimicrob. Agents Chemother. 2007, 51, 3677–3687. [Google Scholar] [CrossRef]
- Saiman, L.; Marshall, B.C.; Mayer-Hamblett, N.; Burns, J.L.; Quittner, A.L.; Cibene, D.A.; Coquillette, S.; Fieberg, A.Y.; Accurso, F.J.; Campbell, P.W., 3rd. Azithromycin in patients with cystic fibrosis chronically infected with Pseudomonas aeruginosa: a randomized controlled trial. JAMA J. Am. Med. Assoc. 2003, 290, 1749–1756. [Google Scholar]
- Bragonzi, A.; Farulla, I.; Paroni, M.; Twomey, K.B.; Pirone, L.; Lore, N.I.; Bianconi, I.; Dalmastri, C.; Ryan, R.P.; Bevivino, A. Modelling co-infection of the cystic fibrosis lung by Pseudomonas aeruginosa and Burkholderia cenocepacia reveals influences on biofilm formation and host response. PLoS One 2012, 7, e52330. [Google Scholar]
- Stoltz, D.A.; Meyerholz, D.K.; Pezzulo, A.A.; Ramachandran, S.; Rogan, M.P.; Davis, G.J.; Hanfland, R.A.; Wohlford-Lenane, C.; Dohrn, C.L.; Bartlett, J.A.; et al. Cystic fibrosis pigs develop lung disease and exhibit defective bacterial eradication at birth. Sci. Transl. Med. 2010, 2, 29ra31. [Google Scholar] [CrossRef]
- Keiser, N.W.; Engelhardt, J.F. New animal models of cystic fibrosis: what are they teaching us? Curr. Opin. Pulm. Med. 2011, 17, 478–483. [Google Scholar]
- Sethi, S.; Wrona, C.; Eschberger, K.; Lobbins, P.; Cai, X.; Murphy, T.F. Inflammatory profile of new bacterial strain exacerbations of chronic obstructive pulmonary disease. Am. J. Respir. Crit. Care Med. 2008, 177, 491–497. [Google Scholar]
- Pang, B.; Hong, W.; West-Barnette, S.L.; Kock, N.D.; Swords, W.E. Diminished ICAM-1 expression and impaired pulmonary clearance of nontypeable Haemophilus influenzae in a mouse model of chronic obstructive pulmonary disease/emphysema. Infect. Immun. 2008, 76, 4959–4967. [Google Scholar]
- Homma, H.; Yamanaka, A.; Tanimoto, S.; Tamura, M.; Chijimatsu, Y.; Kira, S.; Izumi, T. Diffuse panbronchiolitis. A disease of the transitional zone of the lung. Chest 1983, 83, 63–69. [Google Scholar]
- Kobayashi, H. Airway biofilms: implications for pathogenesis and therapy of respiratory tract infections. Treat. Respir. Med. 2005, 4, 241–253. [Google Scholar]
- Schultz, M.J. Macrolide activities beyond their antimicrobial effects: macrolides in diffuse panbronchiolitis and cystic fibrosis. J. Antimicrob. Chemother. 2004, 54, 21–28. [Google Scholar]
- Yanagihara, K.; Tomono, K.; Sawai, T.; Hirakata, Y.; Kadota, J.; Koga, H.; Tashiro, T.; Kohno, S. Effect of clarithromycin on lymphocytes in chronic respiratory Pseudomonas aeruginosa infection. Am. J. Respir. Crit. Care Med. 1997, 155, 337–342. [Google Scholar]
- Nagata, T.; Mukae, H.; Kadota, J.; Hayashi, T.; Fujii, T.; Kuroki, M.; Shirai, R.; Yanagihara, K.; Tomono, K.; Koji, T.; et al. Effect of erythromycin on chronic respiratory infection caused by Pseudomonas aeruginosa with biofilm formation in an experimental murine model. Antimicrob. Agents Chemother. 2004, 48, 2251–2259. [Google Scholar] [CrossRef]
- Yanagihara, K.; Tomono, K.; Sawai, T.; Kuroki, M.; Kaneko, Y.; Ohno, H.; Higashiyama, Y.; Miyazaki, Y.; Hirakata, Y.; Maesaki, S.; et al. Combination therapy for chronic Pseudomonas aeruginosa respiratory infection associated with biofilm formation. J. Antimicrob. Chemother. 2000, 46, 69–72. [Google Scholar] [CrossRef]
- Hannan, T.J.; Totsika, M.; Mansfield, K.J.; Moore, K.H.; Schembri, M.A.; Hultgren, S.J. Host-pathogen checkpoints and population bottlenecks in persistent and intracellular uropathogenic Escherichia coli bladder infection. FEMS Microbiol. Rev. 2012, 36, 616–648. [Google Scholar]
- Conway, P.H.; Cnaan, A.; Zaoutis, T.; Henry, B.V.; Grundmeier, R.W.; Keren, R. Recurrent urinary tract infections in children: risk factors and association with prophylactic antimicrobials. JAMA J. Am. Med. Assoc. 2007, 298, 179–186. [Google Scholar]
- Ronald, A. The etiology of urinary tract infection: traditional and emerging pathogens. Am. J. Med. 2002, 113 Suppl 1A, 14S–19S. [Google Scholar]
- Ozok, H.U.; Ekim, O.; Saltas, H.; Arikok, A.T.; Babacan, O.; Sagnak, L.; Topaloglu, H.; Ersoy, H. The preventive role of transurethral antibiotic delivery in a rat model. Drug Des. Dev. Ther. 2012, 6, 187–194. [Google Scholar]
- Hung, C.S.; Dodson, K.W.; Hultgren, S.J. A murine model of urinary tract infection. Nat. Protoc. 2009, 4, 1230–1243. [Google Scholar]
- Rosen, D.A.; Hooton, T.M.; Stamm, W.E.; Humphrey, P.A.; Hultgren, S.J. Detection of intracellular bacterial communities in human urinary tract infection. PLoS Med. 2007, 4, e329. [Google Scholar]
- Anderson, G.G.; Palermo, J.J.; Schilling, J.D.; Roth, R.; Heuser, J.; Hultgren, S.J. Intracellular bacterial biofilm-like pods in urinary tract infections. Science 2003, 301, 105–107. [Google Scholar]
- Blango, M.G.; Mulvey, M.A. Persistence of uropathogenic Escherichia coli in the face of multiple antibiotics. Antimicrob. Agents Chemother. 2010, 54, 1855–1863. [Google Scholar]
- Justice, S.S.; Hung, C.; Theriot, J.A.; Fletcher, D.A.; Anderson, G.G.; Footer, M.J.; Hultgren, S.J. Differentiation and developmental pathways of uropathogenic Escherichia coli in urinary tract pathogenesis. Proc. Natl. Acad. Sci. USA 2004, 101, 1333–1338. [Google Scholar]
- Cusumano, C.K.; Pinkner, J.S.; Han, Z.; Greene, S.E.; Ford, B.A.; Crowley, J.R.; Henderson, J.P.; Janetka, J.W.; Hultgren, S.J. Treatment and prevention of urinary tract infection with orally active FimH inhibitors. Sci. Transl. Med. 2011, 3, 109ra115. [Google Scholar]
- Hadjifrangiskou, M.; Kostakioti, M.; Chen, S.L.; Henderson, J.P.; Greene, S.E.; Hultgren, S.J. A central metabolic circuit controlled by QseC in pathogenic Escherichia coli. Mol. Microbiol. 2011, 80, 1516–1529. [Google Scholar]
- Alteri, C.J.; Smith, S.N.; Mobley, H.L. Fitness of Escherichia coli during urinary tract infection requires gluconeogenesis and the TCA cycle. PLoS Pathog. 2009, 5, e1000448. [Google Scholar]
- Hultgren, S.J.; Porter, T.N.; Schaeffer, A.J.; Duncan, J.L. Role of type 1 pili and effects of phase variation on lower urinary tract infections produced by Escherichia coli. Infect. Immun. 1985, 50, 370–377. [Google Scholar]
- Abraham, S.N.; Babu, J.P.; Giampapa, C.S.; Hasty, D.L.; Simpson, W.A.; Beachey, E.H. Protection against Escherichia coli-induced urinary tract infections with hybridoma antibodies directed against type 1 fimbriae or complementary D-mannose receptors. Infect. Immun. 1985, 48, 625–628. [Google Scholar]
- Wright, K.J.; Seed, P.C.; Hultgren, S.J. Development of intracellular bacterial communities of uropathogenic Escherichia coli depends on type 1 pili. Cell. Microbiol. 2007, 9, 2230–2241. [Google Scholar]
- Poggio, T.V.; La Torre, J.L.; Scodeller, E.A. Intranasal immunization with a recombinant truncated FimH adhesin adjuvanted with CpG oligodeoxynucleotides protects mice against uropathogenic Escherichia coli challenge. Can. J. Microbiol. 2006, 52, 1093–1102. [Google Scholar]
- Brumbaugh, A.R.; Mobley, H.L. Preventing urinary tract infection: progress toward an effective Escherichia coli vaccine. Expert Rev. Vaccines 2012, 11, 663–676. [Google Scholar]
- Sunden, F.; Hakansson, L.; Ljunggren, E.; Wullt, B. Escherichia coli 83972 bacteriuria protects against recurrent lower urinary tract infections in patients with incomplete bladder emptying. J. Urol. 2010, 184, 179–185. [Google Scholar]
- Watts, R.E.; Totsika, M.; Challinor, V.L.; Mabbett, A.N.; Ulett, G.C.; De Voss, J.J.; Schembri, M.A. Contribution of siderophore systems to growth and urinary tract colonization of asymptomatic bacteriuria Escherichia coli. Infect. Immun. 2012, 80, 333–344. [Google Scholar]
- Fischer, H.; Lutay, N.; Ragnarsdottir, B.; Yadav, M.; Jonsson, K.; Urbano, A.; Al Hadad, A.; Ramisch, S.; Storm, P.; Dobrindt, U.; et al. Pathogen specific, IRF3-dependent signaling and innate resistance to human kidney infection. PLoS Pathog. 2010, 6, e1001109. [Google Scholar] [CrossRef]
- Ragnarsdottir, B.; Jonsson, K.; Urbano, A.; Gronberg-Hernandez, J.; Lutay, N.; Tammi, M.; Gustafsson, M.; Lundstedt, A.C.; Leijonhufvud, I.; Karpman, D.; et al. Toll-like receptor 4 promoter polymorphisms: common TLR4 variants may protect against severe urinary tract infection. PLoS One 2010, 5, e10734. [Google Scholar] [CrossRef]
- Sivick, K.E.; Mobley, H.L. Waging war against uropathogenic Escherichia coli: winning back the urinary tract. Infect. Immun. 2010, 78, 568–585. [Google Scholar]
- Hansson, S.; Hanson, E.; Hjalmas, K.; Hultengren, M.; Jodal, U.; Olling, S.; Svanborg-Eden, C. Follicular cystitis in girls with untreated asymptomatic or covert bacteriuria. J. Urol. 1990, 143, 330–332. [Google Scholar]
- Schlager, T.A.; LeGallo, R.; Innes, D.; Hendley, J.O.; Peters, C.A. B cell infiltration and lymphonodular hyperplasia in bladder submucosa of patients with persistent bacteriuria and recurrent urinary tract infections. J. Urol. 2011, 186, 2359–2364. [Google Scholar]
- Wang, C.; Mendonsa, G.R.; Symington, J.W.; Zhang, Q.; Cadwell, K.; Virgin, H.W.; Mysorekar, I.U. Atg16L1 deficiency confers protection from uropathogenic Escherichia coli infection in vivo. Proc. Natl. Acad. Sci. USA 2012, 109, 11008–11013. [Google Scholar]
- Naber, K.G.; Bergman, B.; Bishop, M.C.; Bjerklund-Johansen, T.E.; Botto, H.; Lobel, B.; Jinenez Cruz, F.; Selvaggi, F.P. EAU guidelines for the management of urinary and male genital tract infections. Urinary Tract Infection (UTI) Working Group of the Health Care Office (HCO) of the European Association of Urology (EAU). Eur. Urol. 2001, 40, 576–588. [Google Scholar] [CrossRef]
- Nickel, J.C.; Costerton, J.W. Bacterial localization in antibiotic-refractory chronic bacterial prostatitis. Prostate 1993, 23, 107–114. [Google Scholar]
- Krieger, J.N.; Lee, S.W.; Jeon, J.; Cheah, P.Y.; Liong, M.L.; Riley, D.E. Epidemiology of prostatitis. Int. J. Antimicrob. Agents 2008, 31 Suppl 1, S85–90. [Google Scholar]
- Mazzoli, S. Biofilms in chronic bacterial prostatitis (NIH-II) and in prostatic calcifications. FEMS Immunol. Med. Microbiol. 2010, 59, 337–344. [Google Scholar]
- Potts, J.; Payne, R.E. Prostatitis: Infection, neuromuscular disorder, or pain syndrome? Proper patient classification is key. Cleve Clin. J. Med. 2007, 74 Suppl 3, S63–S71. [Google Scholar] [CrossRef]
- Garcia-Castillo, M.; Morosini, M.I.; Galvez, M.; Baquero, F.; del Campo, R.; Meseguer, M.A. Differences in biofilm development and antibiotic susceptibility among clinical Ureaplasma urealyticum and Ureaplasma parvum isolates. J. Antimicrob. Chemother. 2008, 62, 1027–1030. [Google Scholar]
- Soto, S.M.; Smithson, A.; Martinez, J.A.; Horcajada, J.P.; Mensa, J.; Vila, J. Biofilm formation in uropathogenic Escherichia coli strains: relationship with prostatitis, urovirulence factors and antimicrobial resistance. J. Urol. 2007, 177, 365–368. [Google Scholar]
- Kim, H.W.; Ha, U.S.; Woo, J.C.; Kim, S.J.; Yoon, B.I.; Lee, S.J.; Cho, Y.H. Preventive effect of selenium on chronic bacterial prostatitis. J. Infect. Chemother. 2012, 18, 30–34. [Google Scholar]
- Nelson, L.K.; D'Amours, G.H.; Sproule-Willoughby, K.M.; Morck, D.W.; Ceri, H. Pseudomonas aeruginosa las and rhl quorum-sensing systems are important for infection and inflammation in a rat prostatitis model. Microbiology 2009, 155, 2612–2619. [Google Scholar]
- Phan, V.; Belas, R.; Gilmore, B.F.; Ceri, H. ZapA, a virulence factor in a rat model of Proteus mirabilis-induced acute and chronic prostatitis. Infect. Immun. 2008, 76, 4859–4864. [Google Scholar]
- Kim, S.H.; Ha, U.S.; Lee, H.R.; Sohn, D.W.; Lee, S.J.; Kim, H.W.; Han, C.H.; Lee, C.B.; Cho, Y.H. Do Escherichia coli extract and cranberry exert preventive effects on chronic bacterial prostatitis? Pilot study using an animal model. J. Infect. Chemother. 2011, 17, 322–326. [Google Scholar]
- Lee, Y.S.; Han, C.H.; Kang, S.H.; Lee, S.J.; Kim, S.W.; Shin, O.R.; Sim, Y.C.; Cho, Y.H. Synergistic effect between catechin and ciprofloxacin on chronic bacterial prostatitis rat model. Int. J. Urol. 2005, 12, 383–389. [Google Scholar]
- Bichler, K.H.; Eipper, E.; Naber, K.; Braun, V.; Zimmermann, R.; Lahme, S. Urinary infection stones. Int. J. Antimicrob. Agents 2002, 19, 488–498. [Google Scholar]
- Hinman, F., Jr. Directional growth of renal calculi. J. Urol. 1979, 121, 700–705. [Google Scholar]
- Nemoy, N.J.; Staney, T.A. Surgical, bacteriological, and biochemical management of “infection stones”. JAMA J. Am. Med. Assoc. 1971, 215, 1470–1476. [Google Scholar] [CrossRef]
- Nickel, J.C.; Reid, G.; Bruce, A.W.; Costerton, J.W. Ultrastructural microbiology of infected urinary stone. Urology 1986, 28, 512–515. [Google Scholar]
- Broomfield, R.J.; Morgan, S.D.; Khan, A.; Stickler, D.J. Crystalline bacterial biofilm formation on urinary catheters by urease-producing urinary tract pathogens: a simple method of control. J. Future Med. Microbiol. 2009, 58, 1367–1375. [Google Scholar]
- Campieri, C.; Campieri, M.; Bertuzzi, V.; Swennen, E.; Matteuzzi, D.; Stefoni, S.; Pirovano, F.; Centi, C.; Ulisse, S.; Famularo, G.; et al. Reduction of oxaluria after an oral course of lactic acid bacteria at high concentration. Kidney Int. 2001, 60, 1097–1105. [Google Scholar] [CrossRef]
- Kwak, C.; Kim, H.K.; Kim, E.C.; Choi, M.S.; Kim, H.H. Urinary oxalate levels and the enteric bacterium Oxalobacter formigenes in patients with calcium oxalate urolithiasis. Eur. Urol. 2003, 44, 475–481. [Google Scholar]
- Nickel, J.C.; Olson, M.; McLean, R.J.; Grant, S.K.; Costerton, J.W. An ecological study of infected urinary stone genesis in an animal model. Br. J. Urol. 1987, 59, 21–30. [Google Scholar]
- Satoh, M.; Takeuchi, H.; Munakata, K.; Yoshida, O. Therapeutic effect of cefluprenam on polymicrobial urinary tract infection associated with Enterococcus faecalis, using the infectious urolithiasis model in rats. Kansenshogaku Zasshi 1998, 72, 371–378. [Google Scholar]
- Vermeulen, C.W.; Goetz, R. Experimental urolithiasis. IX. Influence of infection on stone growth in rats. J. Urol. 1954, 72, 761–769. [Google Scholar]
- Satoh, M.; Munakata, K.; Kitoh, K.; Takeuchi, H.; Yoshida, O. A newly designed model for infection-induced bladder stone formation in the rat. J. Urol. 1984, 132, 1247–1249. [Google Scholar]
- Nickel, J.C.; Costerton, J.W.; McLean, R.J.; Olson, M. Bacterial biofilms: influence on the pathogenesis, diagnosis and treatment of urinary tract infections. J. Antimicrob. Chemother. 1994, 33 (Suppl A), 31–41. [Google Scholar] [CrossRef]
- Lee, F.D.; Kraszewski, A.; Gordon, J.; Howie, J.G.; McSeveney, D.; Harland, W.A. Intestinal spirochaetosis. Gut 1971, 12, 126–133. [Google Scholar]
- Macfarlane, S.; Dillon, J.F. Microbial biofilms in the human gastrointestinal tract. J. Appl. Microbiol. 2007, 102, 1187–1196. [Google Scholar]
- Palestrant, D.; Holzknecht, Z.E.; Collins, B.H.; Parker, W.; Miller, S.E.; Bollinger, R.R. Microbial biofilms in the gut: visualization by electron microscopy and by acridine orange staining. Ultrastruct. Pathol. 2004, 28, 23–27. [Google Scholar]
- Swidsinski, A.; Weber, J.; Loening-Baucke, V.; Hale, L.P.; Lochs, H. Spatial organization and composition of the mucosal flora in patients with inflammatory bowel disease. J. Clin. Microbiol. 2005, 43, 3380–3389. [Google Scholar]
- Zoetendal, E.G.; Akkermans, A.D.; De Vos, W.M. Temperature gradient gel electrophoresis analysis of 16S rRNA from human fecal samples reveals stable and host-specific communities of active bacteria. Appl. Environ. Microbiol. 1998, 64, 3854–3859. [Google Scholar]
- Xavier, R.J.; Podolsky, D.K. Unravelling the pathogenesis of inflammatory bowel disease. Nature 2007, 448, 427–434. [Google Scholar]
- Bohnhoff, M.; Miller, C.P.; Martin, W.R. Resistance of the Mouse's Intestinal Tract to Experimental Salmonella Infection. Ii. Factors Responsible for Its Loss Following Streptomycin Treatment. J. Exp. Med. 1964, 120, 817–828. [Google Scholar] [CrossRef]
- Onderdonk, A.; Marshall, B.; Cisneros, R.; Levy, S.B. Competition between congenic Escherichia coli K-12 strains in vivo. Infect. Immun. 1981, 32, 74–79. [Google Scholar]
- Kaiser, P.; Diard, M.; Stecher, B.; Hardt, W.D. The streptomycin mouse model for Salmonella diarrhea: functional analysis of the microbiota, the pathogen's virulence factors, and the host's mucosal immune respons. Immunol. Rev. 2012, 245, 56–83. [Google Scholar]
- Eckmann, L. Animal models of inflammatory bowel disease: lessons from enteric infections. Ann. N. Y. Acad. Sci. 2006, 1072, 28–38. [Google Scholar]
- Nell, S.; Suerbaum, S.; Josenhans, C. The impact of the microbiota on the pathogenesis of IBD: lessons from mouse infection models. Nat. Rev. Microbiol. 2010, 8, 564–577. [Google Scholar]
- Dinbar, A.; Altmann, G.; Tulcinsky, D.B. The treatment of chronic biliary salmonella carriers. Am. J. Med. 1969, 47, 236–242. [Google Scholar]
- Lai, C.W.; Chan, R.C.; Cheng, A.F.; Sung, J.Y.; Leung, J.W. Common bile duct stones: a cause of chronic salmonellosis. Am. J. Gastroenterol. 1992, 87, 1198–1199. [Google Scholar]
- Sukupolvi, S.; Edelstein, A.; Rhen, M.; Normark, S.J.; Pfeifer, J.D. Development of a murine model of chronic Salmonella infection. Infect. Immun. 1997, 65, 838–842. [Google Scholar]
- Monack, D.M.; Bouley, D.M.; Falkow, S. Salmonella typhimurium persists within macrophages in the mesenteric lymph nodes of chronically infected Nramp1+/+ mice and can be reactivated by IFNgamma neutralization. J. Exp. Med. 2004, 199, 231–241. [Google Scholar]
- Crawford, R.W.; Reeve, K.E.; Gunn, J.S. Flagellated but not hyperfimbriated Salmonella enterica serovar Typhimurium attaches to and forms biofilms on cholesterol-coated surfaces. J. Bacteriol. 2010, 192, 2981–2990. [Google Scholar]
- Bjarnsholt, T.; Kirketerp-Moller, K.; Jensen, P.O.; Madsen, K.G.; Phipps, R.; Krogfelt, K.; Hoiby, N.; Givskov, M. Why chronic wounds will not heal: a novel hypothesis. Wound Repair. Regen. 2008, 16, 2–10. [Google Scholar]
- James, G.A.; Swogger, E.; Wolcott, R.; Pulcini, E.; Secor, P.; Sestrich, J.; Costerton, J.W.; Stewart, P.S. Biofilms in chronic wounds. Wound Repair. Regen. 2008, 16, 37–44. [Google Scholar]
- Seth, A.K.; Geringer, M.R.; Hong, S.J.; Leung, K.P.; Mustoe, T.A.; Galiano, R.D. In vivo modeling of biofilm-infected wounds: a review. J. Surg. Res. 2012, 178, 330–338. [Google Scholar]
- Thomson, C.H. Biofilms: do they affect wound healing? Int. Wound J. 2011, 8, 63–67. [Google Scholar] [CrossRef]
- Dai, T.; Tegos, G.P.; Zhiyentayev, T.; Mylonakis, E.; Hamblin, M.R. Photodynamic therapy for methicillin-resistant Staphylococcus aureus infection in a mouse skin abrasion model. Lasers Surg. Med. 2010, 42, 38–44. [Google Scholar]
- Dai, T.; Kharkwal, G.B.; Tanaka, M.; Huang, Y.Y.; Bil de Arce, V.J.; Hamblin, M.R. Animal models of external traumatic wound infections. Virulence 2011, 2, 296–315. [Google Scholar]
- Trøstrup, H.; Thomsen, K.; Christophersen, L.J.; Hougen, H.P.; Bjarnsholt, T.; Jensen, P.Ø.; Kirkby, N.; Calum, H.; Høiby, N.; Moser, C. Pseudomonas aeruginosa biofilm aggravates skin inflammatory response in BALB/c mice in a novel chronic wound model. Wound Repair Regen 2013, 21, 292–299. [Google Scholar] [CrossRef]
- Calum, H.; Moser, C.; Jensen, P.O.; Christophersen, L.; Maling, D.S.; van Gennip, M.; Bjarnsholt, T.; Hougen, H.P.; Givskov, M.; Jacobsen, G.K.; et al. Thermal injury induces impaired function in polymorphonuclear neutrophil granulocytes and reduced control of burn wound infection. Clin. Exp. Immunol. 2009, 156, 102–110. [Google Scholar] [CrossRef]
- Akiyama, H.; Kanzaki, H.; Tada, J.; Arata, J. Staphylococcus aureus infection on cut wounds in the mouse skin: experimental staphylococcal botryomycosis. J. Dermatol. Sci. 1996, 11, 234–238. [Google Scholar]
- Davis, S.C.; Ricotti, C.; Cazzaniga, A.; Welsh, E.; Eaglstein, W.H.; Mertz, P.M. Microscopic and physiologic evidence for biofilm-associated wound colonization in vivo. Wound Repair. Regen. 2008, 16, 23–29. [Google Scholar]
- Simonetti, O.; Cirioni, O.; Ghiselli, R.; Goteri, G.; Scalise, A.; Orlando, F.; Silvestri, C.; Riva, A.; Saba, V.; Madanahally, K.D.; et al. NAIII-inhibiting peptide enhances healing of wounds infected with methicillin-resistant Staphylococcus aureus. Antimicrob. Agents Chemother. 2008, 52, 2205–2211. [Google Scholar] [CrossRef]
- Nakagami, G.; Sanada, H.; Sugama, J.; Morohoshi, T.; Ikeda, T.; Ohta, Y. Detection of Pseudomonas aeruginosa quorum sensing signals in an infected ischemic wound: an experimental study in rats. Wound Repair. Regen. 2008, 16, 30–36. [Google Scholar]
- Citron, D.M.; Goldstein, E.J.; Merriam, C.V.; Lipsky, B.A.; Abramson, M.A. Bacteriology of moderate-to-severe diabetic foot infections and In vitro activity of antimicrobial agents. J. Clin. Microbiol. 2007, 45, 2819–2828. [Google Scholar]
- Mastropaolo, M.D.; Evans, N.P.; Byrnes, M.K.; Stevens, A.M.; Robertson, J.L.; Melville, S.B. Synergy in polymicrobial infections in a mouse model of type 2 diabetes. Infect. Immun. 2005, 73, 6055–6063. [Google Scholar]
- Donlan, R.M.; Costerton, J.W. Biofilms: survival mechanisms of clinically relevant microorganisms. Clin. Microbiol. Rev. 2002, 15, 167–193. [Google Scholar]
- Gotz, F. Staphylococcus and biofilms. Mol. Microbiol. 2002, 43, 1367–1378. [Google Scholar]
- Durack, D.T.; Beeson, P.B.; Petersdorf, R.G. Experimental bacterial endocarditis. 3. Production and progress of the disease in rabbits. Br. J. Exp. Pathol. 1973, 54, 142–151. [Google Scholar]
- Garrison, P.K.; Freedman, L.R. Experimental endocarditis I. Staphylococcal endocarditis in rabbits resulting from placement of a polyethylene catheter in the right side of the heart. Yale J. Biol. Med. 1970, 42, 394–410. [Google Scholar]
- Xiong, Y.Q.; Willard, J.; Yeaman, M.R.; Cheung, A.L.; Bayer, A.S. Regulation of Staphylococcus aureus alpha-toxin gene (hla) expression by agr , sarA , and sae In vitro and in experimental infective endocarditis. J. Infect. Dis. 2006, 194, 1267–1275. [Google Scholar] [CrossRef]
- Dubee, V.; Chau, F.; Arthur, M.; Garry, L.; Benadda, S.; Mesnage, S.; Lefort, A.; Fantin, B. The In vitro contribution of autolysins to bacterial killing elicited by amoxicillin increases with inoculum size in Enterococcus faecalis. Antimicrob. Agents Chemother. 2011, 55, 910–912. [Google Scholar]
- Frehel, C.; Hellio, R.; Cremieux, A.C.; Contrepois, A.; Bouvet, A. Nutritionally variant streptococci develop ultrastructural abnormalities during experimental endocarditis. Microbial. Pathog. 1988, 4, 247–255. [Google Scholar]
- Veloso, T.R.; Amiguet, M.; Rousson, V.; Giddey, M.; Vouillamoz, J.; Moreillon, P.; Entenza, J.M. Induction of experimental endocarditis by continuous low-grade bacteremia mimicking spontaneous bacteremia in humans. Infect. Immun. 2011, 79, 2006–2011. [Google Scholar]
- Veloso, T.R.; Chaouch, A.; Roger, T.; Giddey, M.; Vouillamoz, J.; Majcherczyk, P.; Que, Y.A.; Rousson, V.; Moreillon, P.; Entenza, J.M. Use of a human-like low-grade bacteremia model of experimental endocarditis to study the role of Staphylococcus aureus adhesins and platelet aggregation in early endocarditis. Infect. Immun. 2013, 81, 697–703. [Google Scholar]
- Ambrose, P.G.; Drusano, G.L.; Craig, W.A. In vivo activity of oritavancin in animal infection models and rationale for a new dosing regimen in humans. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2012, 54 Suppl 3, S220–228. [Google Scholar]
- Seidl, K.; Chen, L.; Bayer, A.S.; Hady, W.A.; Kreiswirth, B.N.; Xiong, Y.Q. Relationship of agr expression and function with virulence and vancomycin treatment outcomes in experimental endocarditis due to methicillin-resistant Staphylococcus aureus. Antimicrob. Agents Chemother. 2011, 55, 5631–5639. [Google Scholar]
- Tattevin, P.; Saleh-Mghir, A.; Davido, B.; Ghout, I.; Massias, L.; Garcia de la Maria, C.; Miro, J.M.; Perronne, C.; Laurent, F.; Cremieux, A.C. Comparison of six generic vancomycin products for treatment of methicillin-resistant Staphylococcus aureus experimental endocarditis in rabbits. Antimicrob. Agents Chemother. 2013, 57, 1157–1162. [Google Scholar]
- Hall-Stoodley, L.; Hu, F.Z.; Gieseke, A.; Nistico, L.; Nguyen, D.; Hayes, J.; Forbes, M.; Greenberg, D.P.; Dice, B.; Burrows, A.; et al. Direct detection of bacterial biofilms on the middle-ear mucosa of children with chronic otitis media. JAMA 2006, 296, 202–211. [Google Scholar] [CrossRef]
- Bakaletz, L.O. Bacterial biofilms in the upper airway - evidence for role in pathology and implications for treatment of otitis media. Paediatr. Respir. Rev. 2012, 13, 154–159. [Google Scholar]
- Bakaletz, L.O. Chinchilla as a robust, reproducible and polymicrobial model of otitis media and its prevention. Expert Rev. Vaccines 2009, 8, 1063–1082. [Google Scholar]
- Silva, R.C.; Dohar, J.E.; Hebda, P.A. Novel rat model of tympanostomy tube otorrhea. Int. J. Pediatr. Otorhinolaryngol. 2012, 76, 179–182. [Google Scholar]
- Trune, D.R.; Zheng, Q.Y. Mouse models for human otitis media. Brain Res. 2009, 1277, 90–103. [Google Scholar]
- Giebink, G.S. Immunoprophylaxis of otitis media. Adv. Exp. Med. Biol. 1991, 303, 149–158. [Google Scholar]
- Giebink, G.S.; Payne, E.E.; Mills, E.L.; Juhn, S.K.; Quie, P.G. Experimental otitis media due to Streptococcus pneumoniae: immunopathogenic response in the chinchilla. J. Infect. Dis. 1976, 134, 595–604. [Google Scholar]
- Juhn, S.K.; Giebink, G.S.; Hanson, D.G.; Paparella, M.M. Experimentally induced acute otitis media--an animal model. Arch. Otorhinolaryngol. 1977, 215, 95–96. [Google Scholar]
- Jurcisek, J.A.; Durbin, J.E.; Kusewitt, D.F.; Bakaletz, L.O. Anatomy of the nasal cavity in the chinchilla. Cells Tissues Organs 2003, 174, 136–152. [Google Scholar]
- Ehrlich, G.D.; Veeh, R.; Wang, X.; Costerton, J.W.; Hayes, J.D.; Hu, F.Z.; Daigle, B.J.; Ehrlich, M.D.; Post, J.C. Mucosal biofilm formation on middle-ear mucosa in the chinchilla model of otitis media. JAMA 2002, 287, 1710–1715. [Google Scholar]
- Post, J.C. Direct evidence of bacterial biofilms in otitis media. Laryngoscope 2001, 111, 2083–2094. [Google Scholar]
- Roberts, A.L.; Connolly, K.L.; Doern, C.D.; Holder, R.C.; Reid, S.D. Loss of the group A Streptococcus regulator Srv decreases biofilm formation in vivo in an otitis media model of infection. Infect. Immun. 2010, 78, 4800–4808. [Google Scholar]
- Byrd, M.S.; Pang, B.; Hong, W.; Waligora, E.A.; Juneau, R.A.; Armbruster, C.E.; Weimer, K.E.; Murrah, K.; Mann, E.E.; Lu, H.; et al. Direct evaluation of Pseudomonas aeruginosa biofilm mediators in a chronic infection model. Infect. Immun. 2011, 79, 3087–3095. [Google Scholar] [CrossRef]
- Bhutta, M.F. Mouse models of otitis media: strengths and limitations. Otolaryngol. Head Neck Surg. 2012, 147, 611–614. [Google Scholar]
- Briles, D.E.; Hollingshead, S.K.; Nabors, G.S.; Paton, J.C.; Brooks-Walter, A. The potential for using protein vaccines to protect against otitis media caused by Streptococcus pneumoniae. Vaccine 2000, 19 Suppl 1, S87–95. [Google Scholar]
- Holmes, A.R.; McNab, R.; Millsap, K.W.; Rohde, M.; Hammerschmidt, S.; Mawdsley, J.L.; Jenkinson, H.F. The pavA gene of Streptococcus pneumoniae encodes a fibronectin-binding protein that is essential for virulence. Mol. Microbiol. 2001, 41, 1395–1408. [Google Scholar]
- Dohar, J.E.; Hebda, P.A.; Veeh, R.; Awad, M.; Costerton, J.W.; Hayes, J.; Ehrlich, G.D. Mucosal biofilm formation on middle-ear mucosa in a nonhuman primate model of chronic suppurative otitis media. Laryngoscope 2005, 115, 1469–1472. [Google Scholar]
- Chaney, E.J.; Nguyen, C.T.; Boppart, S.A. Novel method for non-invasive induction of a middle-ear biofilm in the rat. Vaccine 2011, 29, 1628–1633. [Google Scholar]
- Eriksson, P.O.; Li, J.; Ny, T.; Hellstrom, S. Spontaneous development of otitis media in plasminogen-deficient mice. Int. J. Med. Microbiol. 2006, 296, 501–509. [Google Scholar]
- Ferguson, B.J.; Stolz, D.B. Demonstration of biofilm in human bacterial chronic rhinosinusitis. Am. J. Rhinol. 2005, 19, 452–457. [Google Scholar]
- Harvey, R.J.; Lund, V.J. Biofilms and chronic rhinosinusitis: systematic review of evidence, current concepts and directions for research. Rhinology 2007, 45, 3–13. [Google Scholar]
- Johansson, P.; Kumlien, J.; Carlsoo, B.; Drettner, B.; Nord, C.E. Experimental acute sinusitis in rabbits. A bacteriological and histological study. Acta Otolaryngol. 1988, 105, 357–366. [Google Scholar] [CrossRef]
- Westrin, K.M.; Norlander, T.; Stierna, P.; Carlsoo, B.; Nord, C.E. Experimental maxillary sinusitis induced by Bacteroides fragilis. A bacteriological and histological study in rabbits. Acta Otolaryngol. 1992, 112, 107–114. [Google Scholar]
- Abreu, N.A.; Nagalingam, N.A.; Song, Y.; Roediger, F.C.; Pletcher, S.D.; Goldberg, A.N.; Lynch, S.V. Sinus microbiome diversity depletion and Corynebacterium tuberculostearicum enrichment mediates rhinosinusitis. Sci. Transl. Med. 2012, 4, 151ra124. [Google Scholar]
- Ha, K.R.; Psaltis, A.J.; Tan, L.; Wormald, P.J. A sheep model for the study of biofilms in rhinosinusitis. Am. J. Rhinol. 2007, 21, 339–345. [Google Scholar]
- Le, T.; Psaltis, A.; Tan, L.W.; Wormald, P.J. The efficacy of topical antibiofilm agents in a sheep model of rhinosinusitis. Am. J. Rhinol. 2008, 22, 560–567. [Google Scholar]
- Huang, R.; Li, M.; Gregory, R.L. Bacterial interactions in dental biofilm. Virulence 2011, 2, 435–444. [Google Scholar]
- Kolenbrander, P.E.; Andersen, R.N.; Kazmerzak, K.; Wu, R.; Palmer, R.J., Jr. Spatial organization of oral bacteria in biofilms. Methods Enzymol. 1999, 310, 322–332. [Google Scholar]
- Kolenbrander, P.E. Multispecies communities: interspecies interactions influence growth on saliva as sole nutritional source. Int. J. Oral. Sci. 2011, 3, 49–54. [Google Scholar]
- Quivey, R.G., Jr.; Kuhnert, W.L.; Hahn, K. Adaptation of oral streptococci to low pH. Adv. Microb. Physiol. 2000, 42, 239–274. [Google Scholar]
- Bowen, W.H.; Schilling, K.; Giertsen, E.; Pearson, S.; Lee, S.F.; Bleiweis, A.; Beeman, D. Role of a cell surface-associated protein in adherence and dental caries. Infect. Immun. 1991, 59, 4606–4609. [Google Scholar]
- Catalan, M.A.; Scott-Anne, K.; Klein, M.I.; Koo, H.; Bowen, W.H.; Melvin, J.E. Elevated incidence of dental caries in a mouse model of cystic fibrosis. PLoS One 2011, 6, e16549. [Google Scholar]
- Fitzgerald, R.J.; Keyes, P.H. Demonstration of the etiologic role of streptococci in experimental caries in the hamster. J. Am. Dent. Assoc. 1960, 61, 9–19. [Google Scholar]
- Bainbridge, B.; Verma, R.K.; Eastman, C.; Yehia, B.; Rivera, M.; Moffatt, C.; Bhattacharyya, I.; Lamont, R.J.; Kesavalu, L. Role of Porphyromonas gingivalis phosphoserine phosphatase enzyme SerB in inflammation, immune response, and induction of alveolar bone resorption in rat. Infect. Immun. 2010, 78, 4560–4569. [Google Scholar]
- Okada, Y.; Hamada, N.; Kim, Y.; Takahashi, Y.; Sasaguri, K.; Ozono, S.; Sato, S. Blockade of sympathetic beta-receptors inhibits Porphyromonas gingivalis-induced alveolar bone loss in an experimental rat periodontitis model. Archives of oral biology 2010, 55, 502–508. [Google Scholar]
- Koo, H.; Duarte, S.; Murata, R.M.; Scott-Anne, K.; Gregoire, S.; Watson, G.E.; Singh, A.P.; Vorsa, N. Influence of cranberry proanthocyanidins on formation of biofilms by Streptococcus mutans on saliva-coated apatitic surface and on dental caries development in vivo. Caries Res. 2010, 44, 116–126. [Google Scholar] [CrossRef]
- Koo, H.; Schobel, B.; Scott-Anne, K.; Watson, G.; Bowen, W.H.; Cury, J.A.; Rosalen, P.L.; Park, Y.K. Apigenin and tt-farnesol with fluoride effects on S. mutans biofilms and dental caries. J. Dent. Res. 2005, 84, 1016–1020. [Google Scholar] [CrossRef]
- Assuma, R.; Oates, T.; Cochran, D.; Amar, S.; Graves, D.T. IL-1 and TNF antagonists inhibit the inflammatory response and bone loss in experimental periodontitis. J. Immunol. 1998, 160, 403–409. [Google Scholar]
- Graves, D.T.; Kang, J.; Andriankaja, O.; Wada, K.; Rossa, C., Jr. Animal models to study host-bacteria interactions involved in periodontitis. Front. Oral. Biol. 2012, 15, 117–132. [Google Scholar]
- Socransky, S.S.; Haffajee, A.D.; Cugini, M.A.; Smith, C.; Kent, R.L., Jr. Microbial complexes in subgingival plaque. J. Clin. Periodontol. 1998, 25, 134–144. [Google Scholar]
- Madden, T.E.; Caton, J.G. Animal models for periodontal disease. Methods Enzymol. 1994, 235, 106–119. [Google Scholar]
- Struillou, X.; Boutigny, H.; Soueidan, A.; Layrolle, P. Experimental animal models in periodontology: a review. Open Dent. J. 2010, 4, 37–47. [Google Scholar]
- Giannobile, W.V.; Finkelman, R.D.; Lynch, S.E. Comparison of canine and non-human primate animal models for periodontal regenerative therapy: results following a single administration of PDGF/IGF-I. J. Periodontol. 1994, 65, 1158–1168. [Google Scholar]
- Page, R.C.; Schroeder, H.E. Periodontitis in Man and Other Animals: A Comparative Review. Pub, S.K., Ed.; Basel, 1982; p. 300. [Google Scholar]
- Schou, S.; Holmstrup, P.; Reibel, J.; Juhl, M.; Hjorting-Hansen, E.; Kornman, K.S. Ligature-induced marginal inflammation around osseointegrated implants and ankylosed teeth: stereologic and histologic observations in cynomolgus monkeys (Macaca fascicularis). J. Periodontol. 1993, 64, 529–537. [Google Scholar]
- Yamasaki, A.; Nikai, H.; Niitani, K.; Ijuhin, N. Ultrastructure of the junctional epithelium of germfree rat gingiva. J. Periodontol. 1979, 50, 641–648. [Google Scholar]
- Baker, P.J.; Evans, R.T.; Roopenian, D.C. Oral infection with Porphyromonas gingivalis and induced alveolar bone loss in immunocompetent and severe combined immunodeficient mice. Arch. Oral. Biol. 1994, 39, 1035–1040. [Google Scholar]
- Myneni, S.R.; Settem, R.P.; Connell, T.D.; Keegan, A.D.; Gaffen, S.L.; Sharma, A. TLR2 signaling and Th2 responses drive Tannerella forsythia-induced periodontal bone loss. J. Immunol. 2011, 187, 501–509. [Google Scholar]
- Settem, R.P.; El-Hassan, A.T.; Honma, K.; Stafford, G.P.; Sharma, A. Fusobacterium nucleatum and Tannerella forsythia induce synergistic alveolar bone loss in a mouse periodontitis model. Infect. Immun. 2012, 80, 2436–2443. [Google Scholar]
- Lee, S.F.; Andrian, E.; Rowland, E.; Marquez, I.C. Immune response and alveolar bone resorption in a mouse model of Treponema denticola infection. Infect. Immun. 2009, 77, 694–698. [Google Scholar]
- Polak, D.; Wilensky, A.; Shapira, L.; Halabi, A.; Goldstein, D.; Weiss, E.I.; Houri-Haddad, Y. Mouse model of experimental periodontitis induced by Porphyromonas gingivalis/Fusobacterium nucleatum infection: bone loss and host response. J. Clin. Periodontol. 2009, 36, 406–410. [Google Scholar]
- Schwab, J.M.; Chiang, N.; Arita, M.; Serhan, C.N. Resolvin E1 and protectin D1 activate inflammation-resolution programmes. Nature 2007, 447, 869–874. [Google Scholar]
- Tsukayama, D.T. Pathophysiology of posttraumatic osteomyelitis. Clin. Orthop. Relat. Res. 1999, 360, 22–29. [Google Scholar]
- Gristina, A.G.; Oga, M.; Webb, L.X.; Hobgood, C.D. Adherent bacterial colonization in the pathogenesis of osteomyelitis. Science 1985, 228, 990–993. [Google Scholar]
- Marrie, T.J.; Costerton, J.W. Mode of growth of bacterial pathogens in chronic polymicrobial human osteomyelitis. J. Clin. Microbiol. 1985, 22, 924–933. [Google Scholar]
- Scheman, L.; Janot, M.; Lewin, P. The production of experimental osteomyelitis: prelimary report. JAMA 1941, 117, 1525–1529. [Google Scholar]
- Mader, J.T.; Shirtliff, M.E.; Bergquist, S.C.; Calhoun, J. Antimicrobial treatment of chronic osteomyelitis. Clin. Orthop. Relat. Res. 1999, 47–65. [Google Scholar]
- Brady, R.A.; Leid, J.G.; Camper, A.K.; Costerton, J.W.; Shirtliff, M.E. Identification of Staphylococcus aureus proteins recognized by the antibody-mediated immune response to a biofilm infection. Infect. Immun. 2006, 74, 3415–3426. [Google Scholar]
- Brady, R.A.; O'May, G.A.; Leid, J.G.; Prior, M.L.; Costerton, J.W.; Shirtliff, M.E. Resolution of Staphylococcus aureus biofilm infection using vaccination and antibiotic treatment. Infect. Immun. 2011, 79, 1797–1803. [Google Scholar]
- Poeppl, W.; Tobudic, S.; Lingscheid, T.; Plasenzotti, R.; Kozakowski, N.; Lagler, H.; Georgopoulos, A.; Burgmann, H. Daptomycin, fosfomycin, or both for treatment of methicillin-resistant Staphylococcus aureus osteomyelitis in an experimental rat mode. Antimicrob. Agents Chemother. 2011, 55, 4999–5003. [Google Scholar] [CrossRef]
- Funao, H.; Ishii, K.; Nagai, S.; Sasaki, A.; Hoshikawa, T.; Aizawa, M.; Okada, Y.; Chiba, K.; Koyasu, S.; Toyama, Y.; et al. Establishment of a real-time, quantitative, and reproducible mouse model of Staphylococcus osteomyelitis using bioluminescence imaging. Infect. Immun. 2012, 80, 733–741. [Google Scholar] [CrossRef]
- Marrie, T.J.; Nelligan, J.; Costerton, J.W. A scanning and transmission electron microscopic study of an infected endocardial pacemaker lead. Circulation 1982, 66, 1339–1341. [Google Scholar]
- Lewis, K. Persister cells, dormancy and infectious disease. Nat. Rev. Microbiol. 2007, 5, 48–56. [Google Scholar]
- Christensen, G.D.; Simpson, W.A.; Bisno, A.L.; Beachey, E.H. Experimental foreign body infections in mice challenged with slime-producing Staphylococcus epidermidis. Infect. Immun. 1983, 40, 407–410. [Google Scholar]
- Nickel, J.C.; Grant, S.K.; Costerton, J.W. Catheter-associated bacteriuria. An experimental study. Urology 1985, 26, 369–375. [Google Scholar]
- Rupp, M.E.; Ulphani, J.S.; Fey, P.D.; Mack, D. Characterization of Staphylococcus epidermidis polysaccharide intercellular adhesin/hemagglutinin in the pathogenesis of intravascular catheter-associated infection in a rat model. Infect. Immun. 1999, 67, 2656–2659. [Google Scholar]
- Zimmerli, W.; Waldvogel, F.A.; Vaudaux, P.; Nydegger, U.E. Pathogenesis of foreign body infection: description and characteristics of an animal model. J. Infect. Dis. 1982, 146, 487–497. [Google Scholar]
- Christensen, G.D.; Bisno, A.L.; Parisi, J.T.; McLaughlin, B.; Hester, M.G.; Luther, R.W. Nosocomial septicemia due to multiply antibiotic-resistant Staphylococcus epidermidis. Ann. Intern. Med. 1982, 96, 1–10. [Google Scholar]
- Christensen, G.D.; Simpson, W.A.; Bisno, A.L.; Beachey, E.H. Adherence of slime-producing strains of Staphylococcus epidermidis to smooth surfaces. Infect. Immun. 1982, 37, 318–326. [Google Scholar]
- Rupp, M.E.; Fey, P.D. In vivo models to evaluate adhesion and biofilm formation by Staphylococcus epidermidis. Methods Enzymol. 2001, 336, 206–215. [Google Scholar]
- Rupp, M.E.; Fey, P.D.; Heilmann, C.; Gotz, F. Characterization of the importance of Staphylococcus epidermidis autolysin and polysaccharide intercellular adhesin in the pathogenesis of intravascular catheter-associated infection in a rat model. J. Infect. Dis. 2001, 183, 1038–1042. [Google Scholar]
- Cirioni, O.; Giacometti, A.; Ghiselli, R.; Dell'Acqua, G.; Orlando, F.; Mocchegiani, F.; Silvestri, C.; Licci, A.; Saba, V.; Scalise, G.; et al. RNAIII-inhibiting peptide significantly reduces bacterial load and enhances the effect of antibiotics in the treatment of central venous catheter-associated Staphylococcus aureus infections. J. Infect. Dis. 2006, 193, 180–186. [Google Scholar] [CrossRef]
- Ebert, T.; Smith, S.; Pancari, G.; Wu, X.; Zorman, J.; Clark, D.; Cook, J.; Burns, C.; Antonello, J.M.; Cope, L.; et al. Development of a rat central venous catheter model for evaluation of vaccines to prevent Staphylococcus epidermidis and Staphylococcus aureus early biofilms. Hum. Vaccin 2011, 7, 630–638. [Google Scholar] [CrossRef]
- Hall, L.L.; DeLopez, O.H.; Roberts, A.; Smith, F.A. A procedure for chronic intravenous catheterization in the rabbit. Lab. Anim. Sci. 1974, 24, 79–83. [Google Scholar]
- Kokai-Kun, J.F.; Chanturiya, T.; Mond, J.J. Lysostaphin eradicates established Staphylococcus aureus biofilms in jugular vein catheterized mice. J. Antimicrob. Chemother. 2009, 64, 94–100. [Google Scholar] [CrossRef]
- Giacometti, A.; Cirioni, O.; Ghiselli, R.; Orlando, F.; Mocchegiani, F.; Silvestri, C.; Licci, A.; De Fusco, M.; Provinciali, M.; Saba, V.; et al. Comparative efficacies of quinupristin-dalfopristin, linezolid, vancomycin, and ciprofloxacin in treatment, using the antibiotic-lock technique, of experimental catheter-related infection due to Staphylococcus aureus. Antimicrob. Agents Chemother. 2005, 49, 4042–4045. [Google Scholar] [CrossRef]
- Van Praagh, A.D.; Li, T.; Zhang, S.; Arya, A.; Chen, L.; Zhang, X.X.; Bertolami, S.; Mortin, L.I. Daptomycin antibiotic lock therapy in a rat model of staphylococcal central venous catheter biofilm infections. Antimicrob. Agents Chemother. 2011, 55, 4081–4089. [Google Scholar]
- Capdevila, J.A.; Gavalda, J.; Fortea, J.; Lopez, P.; Martin, M.T.; Gomis, X.; Pahissa, A. Lack of antimicrobial activity of sodium heparin for treating experimental catheter-related infection due to Staphylococcus aureus using the antibiotic-lock technique. Clin. Microbiol. Infect. 2001, 7, 206–212. [Google Scholar]
- Fernandez-Hidalgo, N.; Gavalda, J.; Almirante, B.; Martin, M.T.; Onrubia, P.L.; Gomis, X.; Pahissa, A. Evaluation of linezolid, vancomycin, gentamicin and ciprofloxacin in a rabbit model of antibiotic-lock technique for Staphylococcus aureus catheter-related infection. J. Antimicrob. Chemother. 2010, 65, 525–530. [Google Scholar] [CrossRef]
- Raad, I.; Hachem, R.; Tcholakian, R.K.; Sherertz, R. Efficacy of minocycline and EDTA lock solution in preventing catheter-related bacteremia, septic phlebitis, and endocarditis in rabbit. Antimicrob. Agents Chemother. 2002, 46, 327–332. [Google Scholar]
- Del Pozo, J.L.; Rodil, R.; Aguinaga, A.; Yuste, J.R.; Bustos, C.; Montero, A.; Espinosa, G.; Garcia-Fernandez, N. Daptomycin lock therapy for grampositive long-term catheter-related bloodstream infections. Int. J. Clin. Pract. 2012, 66, 305–308. [Google Scholar]
- Raad, I.I.; Fang, X.; Keutgen, X.M.; Jiang, Y.; Sherertz, R.; Hachem, R. The role of chelators in preventing biofilm formation and catheter-related bloodstream infections. Curr. Opin. Infect. Dis. 2008, 21, 385–392. [Google Scholar]
- Chauhan, A.; Lebeaux, D.; Decante, B.; Kriegel, I.; Escande, M.C.; Ghigo, J.M.; Beloin, C. A rat model of central venous catheter to study establishment of long-term bacterial biofilm and related acute and chronic infections. PLoS One 2012, 7, e37281. [Google Scholar]
- Chauhan, A.; Lebeaux, D.; Ghigo, J.M.; Beloin, C. Full and broad-spectrum in vivo eradication of catheter-associated biofilms using gentamicin-EDTA antibiotic lock therapy. Antimicrob. Agents Chemother. 2012, 56, 6310–6318. [Google Scholar]
- Jones, R.F.; Young, P.S.; Marosszeky, J.E. Treatment of infection in the presence of an indwelling urethral catheter. Br. J. Urol. 1982, 54, 316–319. [Google Scholar]
- Nickel, J.C.; Gristina, A.G.; Costerton, J.W. Electron microscopic study of an infected Foley catheter. Can. J. Surg. 1985, 28, 50–51. [Google Scholar]
- Haraoka, M.; Matsumoto, T.; Takahashi, K.; Kubo, S.; Tanaka, M.; Kumazawa, J. Effect of prednisolone on ascending renal infection due to biofilm disease and lower urinary tract obstruction in rats. Urol. Res. 1995, 22, 383–387. [Google Scholar]
- Cirioni, O.; Ghiselli, R.; Silvestri, C.; Minardi, D.; Gabrielli, E.; Orlando, F.; Rimini, M.; Brescini, L.; Muzzonigro, G.; Guerrieri, M.; et al. Effect of the combination of clarithromycin and amikacin on Pseudomonas aeruginosa biofilm in an animal model of ureteral stent infection. J. Antimicrob. Chemother. 2011, 66, 1318–1323. [Google Scholar] [CrossRef]
- Allison, K.R.; Brynildsen, M.P.; Collins, J.J. Metabolite-enabled eradication of bacterial persisters by aminoglycosides. Nature 2011, 473, 216–220. [Google Scholar]
- Fung, L.C.; Mittelman, M.W.; Thorner, P.S.; Khoury, A.E. A novel rabbit model for the evaluation of biomaterial associated urinary tract infection. Can. J. Urol. 2003, 10, 2007–2012. [Google Scholar]
- Guiton, P.S.; Hung, C.S.; Hancock, L.E.; Caparon, M.G.; Hultgren, S.J. Enterococcal biofilm formation and virulence in an optimized murine model of foreign body-associated urinary tract infections. Infect. Immun. 2010, 78, 4166–4175. [Google Scholar]
- Cadieux, P.A.; Chew, B.H.; Knudsen, B.E.; Dejong, K.; Rowe, E.; Reid, G.; Denstedt, J.D. Triclosan loaded ureteral stents decrease Proteus mirabilis 296 infection in a rabbit urinary tract infection model. J. Urol. 2006, 175, 2331–2335. [Google Scholar]
- Kadurugamuwa, J.L.; Modi, K.; Yu, J.; Francis, K.P.; Purchio, T.; Contag, P.R. Noninvasive biophotonic imaging for monitoring of catheter-associated urinary tract infections and therapy in mice. Infect. Immun. 2005, 73, 3878–3887. [Google Scholar]
- Guiton, P.S.; Cusumano, C.K.; Kline, K.A.; Dodson, K.W.; Han, Z.; Janetka, J.W.; Henderson, J.P.; Caparon, M.G.; Hultgren, S.J. Combinatorial small-molecule therapy prevents uropathogenic Escherichia coli catheter-associated urinary tract infections in mice. Antimicrob. Agents Chemother. 2012, 56, 4738–4745. [Google Scholar]
- Morck, D.W.; Lam, K.; McKay, S.G.; Olson, M.E.; Prosser, B.; Ellis, B.D.; Cleeland, R.; Costerton, J.W. Comparative evaluation of fleroxacin, ampicillin, trimethoprimsulfamethoxazole, and gentamicin as treatments of catheter-associated urinary tract infection in a rabbit model. Int. J. Antimicrob. Agents 1994, 4 Suppl 2, S21–S27. [Google Scholar]
- Olson, M.E.; Nickel, J.C.; Khoury, A.E.; Morck, D.W.; Cleeland, R.; Costerton, J.W. Amdinocillin treatment of catheter-associated bacteriuria in rabbits. J. Infect. Dis. 1989, 159, 1065–1072. [Google Scholar]
- Hachem, R.; Reitzel, R.; Borne, A.; Jiang, Y.; Tinkey, P.; Uthamanthil, R.; Chandra, J.; Ghannoum, M.; Raad, I. Novel antiseptic urinary catheters for prevention of urinary tract infections: correlation of in vivo and In vitro test results. Antimicrob. Agents Chemother. 2009, 53, 5145–5149. [Google Scholar]
- Hazan, Z.; Zumeris, J.; Jacob, H.; Raskin, H.; Kratysh, G.; Vishnia, M.; Dror, N.; Barliya, T.; Mandel, M.; Lavie, G. Effective prevention of microbial biofilm formation on medical devices by low-energy surface acoustic waves. Antimicrob. Agents Chemother. 2006, 50, 4144–4152. [Google Scholar]
- Davis, C.P.; Shirtliff, M.E.; Scimeca, J.M.; Hoskins, S.L.; Warren, M.M. In vivo reduction of bacterial populations in the urinary tract of catheterized sheep by iontophoresis. J Urol. 1995, 154, 1948–1953. [Google Scholar]
- Pickard, R.; Lam, T.; MacLennan, G.; Starr, K.; Kilonzo, M.; McPherson, G.; Gillies, K.; McDonald, A.; Walton, K.; Buckley, B.; et al. Antimicrobial catheters for reduction of symptomatic urinary tract infection in adults requiring short-term catheterisation in hospital: a multicentre randomised controlled trial. Lancet 2012, 380, 1927–1935. [Google Scholar] [CrossRef] [Green Version]
- Rodet, A. Physiologie pathologique - étude expérimentale sur l’ostéomyelite infectieuse. C R Acad. Sci. 1885, 99, 569–571. [Google Scholar]
- Andriole, V.T.; Nagel, D.A.; Southwick, W.O. A paradigm for human chronic osteomyelitis. J. Bone Joint Surg. Am. 1973, 55, 1511–1515. [Google Scholar]
- Gristina, A.G.; Costerton, J.W. Bacterial adherence and the glycocalyx and their role in musculoskeletal infection. Orthop. Clin. North. Am. 1984, 15, 517–535. [Google Scholar]
- Mayberry-Carson, K.J.; Tober-Meyer, B.; Smith, J.K.; Lambe, D.W., Jr.; Costerton, J.W. Bacterial adherence and glycocalyx formation in osteomyelitis experimentally induced with Staphylococcus aureus. Infect. Immun. 1984, 43, 825–833. [Google Scholar]
- Petty, W.; Spanier, S.; Shuster, J.J.; Silverthorne, C. The influence of skeletal implants on incidence of infection. Experiments in a canine model. J. Bone Joint Surg. Am. 1985, 67, 1236–1244. [Google Scholar]
- Sanzen, L.; Linder, L. Infection adjacent to titanium and bone cement implants: an experimental study in rabbits. Biomaterials 1995, 16, 1273–1277. [Google Scholar]
- Evans, R.P.; Nelson, C.L.; Harrison, B.H. The effect of wound environment on the incidence of acute osteomyelitis. Clin. Orthop. Relat. Res. 1993, 289–297. [Google Scholar]
- Prabhakara, R.; Harro, J.M.; Leid, J.G.; Keegan, A.D.; Prior, M.L.; Shirtliff, M.E. Suppression of the inflammatory immune response prevents the development of chronic biofilm infection due to methicillin-resistant Staphylococcus aureus. Infect. Immun. 2011, 79, 5010–5018. [Google Scholar]
- Lucke, M.; Schmidmaier, G.; Sadoni, S.; Wildemann, B.; Schiller, R.; Haas, N.P.; Raschke, M. Gentamicin coating of metallic implants reduces implant-related osteomyelitis in rats. Bone 2003, 32, 521–531. [Google Scholar]
- Lucke, M.; Wildemann, B.; Sadoni, S.; Surke, C.; Schiller, R.; Stemberger, A.; Raschke, M.; Haas, N.P.; Schmidmaier, G. Systemic versus local application of gentamicin in prophylaxis of implant-related osteomyelitis in a rat model. Bone 2005, 36, 770–778. [Google Scholar]
- Fitzgerald, R.H., Jr. Experimental osteomyelitis: description of a canine model and the role of depot administration of antibiotics in the prevention and treatment of sepsis. J. Bone Joint Surg. Am. 1983, 65, 371–380. [Google Scholar]
- Fuchs, T.; Stange, R.; Schmidmaier, G.; Raschke, M.J. The use of gentamicin-coated nails in the tibia: preliminary results of a prospective study. Arch. Orthop. Trauma Surg. 2011, 131, 1419–1425. [Google Scholar]
- Belmatoug, N.; Cremieux, A.C.; Bleton, R.; Volk, A.; Saleh-Mghir, A.; Grossin, M.; Garry, L.; Carbon, C. A new model of experimental prosthetic joint infection due to methicillin-resistant Staphylococcus aureus: a microbiologic, histopathologic, and magnetic resonance imaging characterization. J. Infect. Dis. 1996, 174, 414–417. [Google Scholar] [CrossRef]
- Schurman, D.J.; Trindade, C.; Hirshman, H.P.; Moser, K.; Kajiyama, G.; Stevens, P. Antibiotic-acrylic bone cement composites. Studies of gentamicin and Palacos. J. Bone Joint Surg. Am. 1978, 60, 978–984. [Google Scholar]
- Blomgren, G.; Lindgren, U. Late hematogenous infection in total joint replacement: studies of gentamicin and bone cement in the rabbit. Clin. Orthop. Relat. Res. 1981, 244–248. [Google Scholar]
- Bernthal, N.M.; Stavrakis, A.I.; Billi, F.; Cho, J.S.; Kremen, T.J.; Simon, S.I.; Cheung, A.L.; Finerman, G.A.; Lieberman, J.R.; Adams, J.S.; et al. A mouse model of post-arthroplasty Staphylococcus aureus joint infection to evaluate in vivo the efficacy of antimicrobial implant coatings. PLoS One 2010, 5, e12580. [Google Scholar]
- Cremieux, A.C.; Mghir, A.S.; Bleton, R.; Manteau, M.; Belmatoug, N.; Massias, L.; Garry, L.; Sales, N.; Maziere, B.; Carbon, C. Efficacy of sparfloxacin and autoradiographic diffusion pattern of [14C]Sparfloxacin in experimental Staphylococcus aureus joint prosthesis infection. Antimicrob. Agents Chemother. 1996, 40, 2111–2116. [Google Scholar]
- Inglis, T.J.; Millar, M.R.; Jones, J.G.; Robinson, D.A. Tracheal tube biofilm as a source of bacterial colonization of the lung. J. Clin. Microbiol. 1989, 27, 2014–2018. [Google Scholar]
- Gil-Perotin, S.; Ramirez, P.; Marti, V.; Sahuquillo, J.M.; Gonzalez, E.; Calleja, I.; Menendez, R.; Bonastre, J. Implications of endotracheal tube biofilm in ventilator-associated pneumonia response: a state of concept. Crit. Care 2012, 16, R93. [Google Scholar]
- Berra, L.; De Marchi, L.; Yu, Z.X.; Laquerriere, P.; Baccarelli, A.; Kolobow, T. Endotracheal tubes coated with antiseptics decrease bacterial colonization of the ventilator circuits, lungs, and endotracheal tube. Anesthesiology 2004, 100, 1446–1456. [Google Scholar] [CrossRef]
- Fernandez-Barat, L.; Li Bassi, G.; Ferrer, M.; Bosch, A.; Calvo, M.; Vila, J.; Gabarrus, A.; Martinez-Olondris, P.; Rigol, M.; Esperatti, M.; et al. Direct analysis of bacterial viability in endotracheal tube biofilm from a pig model of methicillin-resistant Staphylococcus aureus pneumonia following antimicrobial therapy. FEMS Immunol. Med. Microbiol. 2012, 65, 309–317. [Google Scholar] [CrossRef]
- Olson, M.E.; Harmon, B.G.; Kollef, M.H. Silver-coated endotracheal tubes associated with reduced bacterial burden in the lungs of mechanically ventilated dogs. Chest 2002, 121, 863–870. [Google Scholar]
- Kollef, M.H.; Afessa, B.; Anzueto, A.; Veremakis, C.; Kerr, K.M.; Margolis, B.D.; Craven, D.E.; Roberts, P.R.; Arroliga, A.C.; Hubmayr, R.D.; et al. Silver-coated endotracheal tubes and incidence of ventilator-associated pneumonia: the NASCENT randomized trial. JAMA 2008, 300, 805–813. [Google Scholar] [CrossRef]
- Berra, L.; Curto, F.; Li Bassi, G.; Laquerriere, P.; Baccarelli, A.; Kolobow, T. Antibacterial-coated tracheal tubes cleaned with the Mucus Shaver : a novel method to retain long-term bactericidal activity of coated tracheal tubes. Intensive Care Med. 2006, 32, 888–893. [Google Scholar]
- Berra, L.; Coppadoro, A.; Bittner, E.A.; Kolobow, T.; Laquerriere, P.; Pohlmann, J.R.; Bramati, S.; Moss, J.; Pesenti, A. A clinical assessment of the Mucus Shaver: a device to keep the endotracheal tube free from secretions. Crit. Care Med. 2012, 40, 119–124. [Google Scholar]
- Fernandez-Barat, L.; Ferrer, M.; Sierra, J.M.; Soy, D.; Guerrero, L.; Vila, J.; Li Bassi, G.; Cortadellas, N.; Martinez-Olondris, P.; Rigol, M.; et al. Linezolid limits burden of methicillin-resistant Staphylococcus aureus in biofilm of tracheal tubes. Crit. Care Med. 2012, 40, 2385–2389. [Google Scholar] [CrossRef]
- Tollefson, D.F.; Bandyk, D.F.; Kaebnick, H.W.; Seabrook, G.R.; Towne, J.B. Surface biofilm disruption. Enhanced recovery of microorganisms from vascular prostheses. Arch. Surg. 1987, 122, 38–43. [Google Scholar] [CrossRef]
- Gahtan, V.; Esses, G.E.; Bandyk, D.F.; Nelson, R.T.; Dupont, E.; Mills, J.L. Antistaphylococcal activity of rifampin-bonded gelatin-impregnated Dacron grafts. J. Surg. Res. 1995, 58, 105–110. [Google Scholar]
- Gao, H.; Sandermann, J.; Prag, J.; Lund, L.; Lindholt, J.S. Rifampicin-soaked silver polyester versus expanded polytetrafluoro-ethylene grafts for in situ replacement of infected grafts in a porcine randomised controlled trial. Eur. J. Vasc. Endovasc. Surg. 2012, 43, 582–587. [Google Scholar]
- Aboshady, I.; Raad, I.; Shah, A.S.; Vela, D.; Dvorak, T.; Safi, H.J.; Buja, L.M.; Khalil, K.G. A pilot study of a triple antimicrobial-bonded Dacron graft for the prevention of aortic graft infection. J. Vasc. Surg. 2012, 56, 794–801. [Google Scholar]
- Lew, W.; Moore, W. Antibiotic-impregnated grafts for aortic reconstruction. Semin. Vasc. Surg. 2011, 24, 211–219. [Google Scholar]
- Virden, C.P.; Dobke, M.K.; Stein, P.; Parsons, C.L.; Frank, D.H. Subclinical infection of the silicone breast implant surface as a possible cause of capsular contracture. Aesthetic Plast Surg. 1992, 16, 173–179. [Google Scholar]
- Jacombs, A.; Allan, J.; Hu, H.; Valente, P.M.; Wessels, W.L.; Deva, A.K.; Vickery, K. Prevention of biofilm-induced capsular contracture with antibiotic-impregnated mesh in a porcine model. Aesthet Surg. J. 2012, 32, 886–891. [Google Scholar]
- Tamboto, H.; Vickery, K.; Deva, A.K. Subclinical (biofilm) infection causes capsular contracture in a porcine model following augmentation mammaplasty. Plast Reconstr. Surg. 2010, 126, 835–842. [Google Scholar]
- Slusher, M.M.; Myrvik, Q.N.; Lewis, J.C.; Gristina, A.G. Extended-wear lenses, biofilm, and bacterial adhesion. Arch. Ophthalmol. 1987, 105, 110–115. [Google Scholar] [CrossRef]
- Szczotka-Flynn, L.B.; Pearlman, E.; Ghannoum, M. Microbial contamination of contact lenses, lens care solutions, and their accessories: a literature review. Eye Contact Lens 2010, 36, 116–129. [Google Scholar] [CrossRef]
- Cole, N.; Hume, E.B.; Vijay, A.K.; Sankaridurg, P.; Kumar, N.; Willcox, M.D. In vivo performance of melimine as an antimicrobial coating for contact lenses in models of CLARE and CLPU. Invest Ophthalmol. Vis Sci. 2010, 51, 390–395. [Google Scholar]
- Sun, Y.; Chandra, J.; Mukherjee, P.; Szczotka-Flynn, L.; Ghannoum, M.A.; Pearlman, E. A murine model of contact lens-associated Fusarium keratitis. Invest. Ophthalmol. Vis. Sci. 2010, 51, 1511–1516. [Google Scholar]
- Wood, S.R.; Kirkham, J.; Marsh, P.D.; Shore, R.C.; Nattress, B.; Robinson, C. Architecture of intact natural human plaque biofilms studied by confocal laser scanning microscopy. J. Dental Res. 2000, 79, 21–27. [Google Scholar]
- Berglundh, T.; Lindhe, J.; Marinello, C.; Ericsson, I.; Liljenberg, B. Soft tissue reaction to de novo plaque formation on implants and teeth. An experimental study in the dog. Clin. Oral. Implants Res. 1992, 3, 1–8. [Google Scholar]
- Albouy, J.P.; Abrahamsson, I.; Berglundh, T. Spontaneous progression of experimental peri-implantitis at implants with different surface characteristics: an experimental study in dogs. J. Clin. Periodontol. 2012, 39, 182–187. [Google Scholar]
- Freire, M.O.; Sedghizadeh, P.P.; Schaudinn, C.; Gorur, A.; Downey, J.S.; Choi, J.H.; Chen, W.; Kook, J.K.; Chen, C.; Goodman, S.D.; et al. Development of an animal model for Aggregatibacteractinomycetemcomitans biofilm-mediated oral osteolytic infection: a preliminary study. J. Periodontol. 2011, 82, 778–789. [Google Scholar] [CrossRef]
- Rimondini, L.; Fare, S.; Brambilla, E.; Felloni, A.; Consonni, C.; Brossa, F.; Carrassi, A. The effect of surface roughness on early in vivo plaque colonization on titanium. J. Periodontol. 1997, 68, 556–562. [Google Scholar] [CrossRef]
- Scarano, A.; Piattelli, M.; Caputi, S.; Favero, G.A.; Piattelli, A. Bacterial adhesion on commercially pure titanium and zirconium oxide disks: an in vivo human study. J. Periodontol. 2004, 75, 292–296. [Google Scholar]
- Garrigos, C.; Murillo, O.; Lora-Tamayo, J.; Verdaguer, R.; Tubau, F.; Cabellos, C.; Cabo, J.; Ariza, J. Fosfomycin-Daptomycin and Other Fosfomycin Combinations as Alternative Therapies in Experimental Foreign-Body Infection by Methicillin-Resistant Staphylococcus aureus. Antimicrob. Agents Chemother. 2013, 57, 606–610. [Google Scholar]
- Zimmerli, W.; Widmer, A.F.; Blatter, M.; Frei, R.; Ochsner, P.E. Role of rifampin for treatment of orthopedic implant-related staphylococcal infections: a randomized controlled trial. Foreign-Body Infection (FBI) Study Group. JAMA 1998, 279, 1537–1541. [Google Scholar] [CrossRef]
- Zimmerli, W. Experimental models in the investigation of device-related infections. J. Antimicrob. Chemother. 1993, 31 Suppl D, 97–102. [Google Scholar]
- Beenken, K.E.; Dunman, P.M.; McAleese, F.; Macapagal, D.; Murphy, E.; Projan, S.J.; Blevins, J.S.; Smeltzer, M.S. Global gene expression in Staphylococcus aureus biofilms. J. Bacteriol. 2004, 186, 4665–4684. [Google Scholar]
- Thurlow, L.R.; Hanke, M.L.; Fritz, T.; Angle, A.; Aldrich, A.; Williams, S.H.; Engebretsen, I.L.; Bayles, K.W.; Horswill, A.R.; Kielian, T. Staphylococcus aureus biofilms prevent macrophage phagocytosis and attenuate inflammation in vivo. J. Immunol. 2011, 186, 6585–6596. [Google Scholar] [CrossRef]
- Weiss, E.C.; Zielinska, A.; Beenken, K.E.; Spencer, H.J.; Daily, S.J.; Smeltzer, M.S. Impact of sarA on daptomycin susceptibility of Staphylococcus aureus biofilms in vivo. Antimicrob. Agents Chemother. 2009, 53, 4096–4102. [Google Scholar] [CrossRef]
- Cobrado, L.; Silva-Dias, A.; Azevedo, M.M.; Pina-Vaz, C.; Rodrigues, A.G. In vivo antibiofilm effect of cerium, chitosan and hamamelitannin against usual agents of catheter-related bloodstream infections. J. Antimicrob. Chemother. 2013, 68, 126–130. [Google Scholar]
- Kadurugamuwa, J.L.; Sin, L.; Albert, E.; Yu, J.; Francis, K.; DeBoer, M.; Rubin, M.; Bellinger-Kawahara, C.; Parr Jr, T.R.; Contag, P.R. Direct continuous method for monitoring biofilm infection in a mouse model. Infect. Immun. 2003, 71, 882–890. [Google Scholar]
- Raad, I.; Darouiche, R.; Hachem, R.; Mansouri, M.; Bodey, G.P. The broad-spectrum activity and efficacy of catheters coated with minocycline and rifampin. J. Infect. Dis. 1996, 173, 418–424. [Google Scholar]
- Darouiche, R.O.; Raad, I.I.; Heard, S.O.; Thornby, J.I.; Wenker, O.C.; Gabrielli, A.; Berg, J.; Khardori, N.; Hanna, H.; Hachem, R.; et al. A comparison of two antimicrobial-impregnated central venous catheters. Catheter Study Group. N. Engl. J. Med. 1999, 340, 1–8. [Google Scholar] [CrossRef]
- Darouiche, R.O.; Mansouri, M.D.; Gawande, P.V.; Madhyastha, S. Antimicrobial and antibiofilm efficacy of triclosan and DispersinB combination. J. Antimicrob. Chemother. 2009, 64, 88–93. [Google Scholar]
- Ensing, G.T.; Roeder, B.L.; Nelson, J.L.; van Horn, J.R.; van der Mei, H.C.; Busscher, H.J.; Pitt, W.G. Effect of pulsed ultrasound in combination with gentamicin on bacterial viability in biofilms on bone cements in vivo. J. Appl. Microbiol. 2005, 99, 443–448. [Google Scholar] [CrossRef]
- Hansen, L.K.; Brown, M.; Johnson, D.; Palme II, D.F.; Love, C.; Darouiche, R. In vivo model of human pathogen infection and demonstration of efficacy by an antimicrobial pouch for pacing devices. Pacing Clin. Electrophysiol. 2009, 32, 898–907. [Google Scholar]
- Illingworth, B.L.; Tweden, K.; Schroeder, R.F.; Cameron, J.D. In vivo efficacy of silver-coated (Silzone) infection-resistant polyester fabric against a biofilm-producing bacteria, Staphylococcus epidermidis. J. Heart Valve Dis. 1998, 7, 524–530. [Google Scholar]
- Illingworth, B.; Bianco, R.W.; Weisberg, S. In vivo efficacy of silver-coated fabric against Staphylococcus epidermidis. J. Heart Valve Dis. 2000, 9, 135–141. [Google Scholar]
- Darouiche, R.O.; Meade, R.; Mansouri, M.; Raad, I.I. In vivo efficacy of antimicrobial-coated fabric from prosthetic heart valve sewing rings. J. Heart Valve Dis. 1998, 7, 639–646. [Google Scholar]
- Darouiche, R.O.; Mansouri, M.D.; Meade, R. In-vitro and in vivo activity of antimicrobial-coated prosthetic heart valve sewing cuffs. J. Heart Valve Dis. 2002, 11, 99–104. [Google Scholar]
- Nakamoto, D.A.; Rosenfield, M.L.; Haaga, J.R.; Merritt, K.; Sachs, P.B.; Hutton, M.C.; Graham, R.C.; Rowland, D.Y. Young Investigator Award. In vivo treatment of infected prosthetic graft material with urokinase: an animal model. J. Vasc. Interv. Radiol. 1994, 5, 549–552. [Google Scholar] [CrossRef]
- Garrison, J.R., Jr.; Henke, P.K.; Smith, K.R.; Brittian, K.R.; Lam, T.M.; Peyton, J.C.; Bergamini, T.M. In vitro and in vivo effects of rifampin on Staphylococcus epidermidis graft infections. ASAIO J. 1997, 43, 8–12. [Google Scholar]
- Cirioni, O.; Mocchegiani, F.; Cacciatore, I.; Vecchiet, J.; Silvestri, C.; Baldassarre, L.; Ucciferri, C.; Orsetti, E.; Castelli, P.; Provinciali, M.; Vivarelli, M.; Fornasari, E.; Giacometti, A. Quorum sensing inhibitor FS3-coated vascular graft enhances daptomycin efficacy in a rat model of staphylococcal infection. Peptides 2013, 40, 77–81. [Google Scholar] [CrossRef]
- Rediske, A.M.; Roeder, B.L.; Nelson, J.L.; Robison, R.L.; Schaalje, G.B.; Robison, R.A.; Pitt, W.G. Pulsed ultrasound enhances the killing of Escherichia coli biofilms by aminoglycoside antibiotics in vivo. Antimicrob. Agents Chemother. 2000, 44, 771–772. [Google Scholar] [CrossRef]
- Anguita-Alonso, P.; Giacometti, A.; Cirioni, O.; Ghiselli, R.; Orlando, F.; Saba, V.; Scalise, G.; Sevo, M.; Tuzova, M.; Patel, R.; et al. RNAIII-inhibiting-peptide-loaded polymethylmethacrylate prevents in vivo Staphylococcus aureus biofilm formation. Antimicrob. Agents Chemother. 2007, 51, 2594–2596. [Google Scholar] [CrossRef]
- Engelsman, A.F.; van Dam, G.M.; van der Mei, H.C.; Busscher, H.J.; Ploeg, R.J. In vivo evaluation of bacterial infection involving morphologically different surgical meshes. Ann. Surg. 2010, 251, 133–137. [Google Scholar]
- Daghighi, S.; Sjollema, J.; Jaspers, V.; de Boer, L.; Zaat, S.A.; Dijkstra, R.J.; van Dam, G.M.; van der Mei, H.C.; Busscher, H.J. Persistence of a bioluminescent Staphylococcus aureus strain on and around degradable and non-degradable surgical meshes in a murine model. Acta Biomater 2012, 8, 3991–3996. [Google Scholar]
- Klionsky, D.J.; Abdalla, F.C.; Abeliovich, H.; Abraham, R.T.; Acevedo-Arozena, A.; Adeli, K.; Agholme, L.; Agnello, M.; Agostinis, P.; Aguirre-Ghiso, J.A.; et al. Guidelines for the use and interpretation of assays for monitoring autophagy. Autophagy 2012, 8, 445–544. [Google Scholar] [CrossRef]
- Romero, D.; Kolter, R. Will biofilm disassembly agents make it to market? Trends Microbiol. 2011, 19, 304–306. [Google Scholar] [CrossRef]
- Von Herrath, M.G.; Nepom, G.T. Lost in translation: barriers to implementing clinical immunotherapeutics for autoimmunity. J. Exp. Med. 2005, 202, 1159–1162. [Google Scholar]
- Warren, H.S.; Fitting, C.; Hoff, E.; Adib-Conquy, M.; Beasley-Topliffe, L.; Tesini, B.; Liang, X.; Valentine, C.; Hellman, J.; Hayden, D.; et al. Resilience to bacterial infection: difference between species could be due to proteins in serum. J. Infect. Dis. 2009, 201, 223–232. [Google Scholar]
- Bolker, J. Model organisms: There's more to life than rats and flies. Nature 2012, 491, 31–33. [Google Scholar]
- Schnabel, J. Neuroscience: Standard model. Nature 2008, 454, 682–685. [Google Scholar]
- Kilkenny, C.; Browne, W.J.; Cuthill, I.C.; Emerson, M.; Altman, D.G. Improving bioscience research reporting: the ARRIVE guidelines for reporting animal research. PLoS Biol. 2010, 8, e1000412. [Google Scholar]
- Madsen, J.S.; Burmolle, M.; Hansen, L.H.; Sorensen, S.J. The interconnection between biofilm formation and horizontal gene transfer. FEMS Immunol. Med. Microbiol. 2012, 65, 183–195. [Google Scholar]
- Hennequin, C.; Aumeran, C.; Robin, F.; Traore, O.; Forestier, C. Antibiotic resistance and plasmid transfer capacity in biofilm formed with a CTX-M-15-producing Klebsiella pneumoniae isolate. J. Antimicrob. Chemother. 2012, 67, 2123–2130. [Google Scholar]
- Guerin, E.; Cambray, G.; Sanchez-Alberola, N.; Campoy, S.; Erill, I.; Da Re, S.; Gonzalez-Zorn, B.; Barbe, J.; Ploy, M.C.; Mazel, D. The SOS response controls integron recombination. Science 2009, 324, 1034. [Google Scholar]
- Hoffman, L.R.; D'Argenio, D.A.; MacCoss, M.J.; Zhang, Z.; Jones, R.A.; Miller, S.I. Aminoglycoside antibiotics induce bacterial biofilm formation. Nature 2005, 436, 1171–1175. [Google Scholar]
- Kohanski, M.A.; DePristo, M.A.; Collins, J.J. Sublethal antibiotic treatment leads to multidrug resistance via radical-induced mutagenesis. Mol. Cell 2010, 37, 311–320. [Google Scholar]
- Peters, B.M.; Jabra-Rizk, M.A.; O'May, G.A.; Costerton, J.W.; Shirtliff, M.E. Polymicrobial interactions: impact on pathogenesis and human disease. Clin. Microbiol. Rev. 2012, 25, 193–213. [Google Scholar]
- Wolcott, R.; Costerton, J.W.; Raoult, D.; Cutler, S.J. The polymicrobial nature of biofilm infection. Clin. Microbiol. Infect. Off. Publ. Eur. Soc. Clin. Microbiol. Infect. Dis. 2013, 19, 107–112. [Google Scholar]
- Frank, D.N.; Wilson, S.S.; St Amand, A.L.; Pace, N.R. Culture-independent microbiological analysis of foley urinary catheter biofilms. PLoS One 2009, 4, e7811. [Google Scholar]
- Larsen, M.K.; Thomsen, T.R.; Moser, C.; Hoiby, N.; Nielsen, P.H. Use of cultivation-dependent and -independent techniques to assess contamination of central venous catheters: a pilot study. BMC Clin. Pathol. 2008, 8, 10. [Google Scholar]
- Donelli, G.; Vuotto, C.; Cardines, R.; Mastrantonio, P. Biofilm-growing intestinal anaerobic bacteria. FEMS Immunol. Med. Microbiol. 2012, 65, 318–325. [Google Scholar] [CrossRef]
- Ramsey, M.M.; Rumbaugh, K.P.; Whiteley, M. Metabolite cross-feeding enhances virulence in a model polymicrobial infection. PLoS Pathog. 2011, 7, e1002012. [Google Scholar]
- Korgaonkar, A.; Trivedi, U.; Rumbaugh, K.P.; Whiteley, M. Community surveillance enhances Pseudomonas aeruginosa virulence during polymicrobial infection. Proc. Natl. Acad. Sci. USA 2013, 110, 1059–1064. [Google Scholar]
- Armbruster, C.E.; Hong, W.; Pang, B.; Weimer, K.E.; Juneau, R.A.; Turner, J.; Swords, W.E. Indirect pathogenicity of Haemophilus influenzae and Moraxella catarrhalis in polymicrobial otitis media occurs via interspecies quorum signaling. MBio 2010, 1, e00102. [Google Scholar]
- Dalton, T.; Dowd, S.E.; Wolcott, R.D.; Sun, Y.; Watters, C.; Griswold, J.A.; Rumbaugh, K.P. An in vivo polymicrobial biofilm wound infection model to study interspecies interactions. PLoS One 2011, 6, e27317. [Google Scholar]
- Kline, K.A.; Schwartz, D.J.; Gilbert, N.M.; Hultgren, S.J.; Lewis, A.L. Immune modulation by group B Streptococcus influences host susceptibility to urinary tract infection by uropathogenic Escherichia coli. Infect. Immun. 2012, 80, 4186–4194. [Google Scholar]
- Conrad, D.; Haynes, M.; Salamon, P.; Rainey, P.B.; Youle, M.; Rohwer, F. Cystic fibrosis therapy: a community ecology perspective. Am. J. Respir. Cell Mol. Biol. 2013, 48, 150–156. [Google Scholar]
- Costello, E.K.; Stagaman, K.; Dethlefsen, L.; Bohannan, B.J.; Relman, D.A. The application of ecological theory toward an understanding of the human microbiome. Science 2012, 336, 1255–1262. [Google Scholar]
- Reppas, A.I.; Spiliotis, K.G.; Siettos, C.I. Epidemionics: from the host-host interactions to the systematic analysis of the emergent macroscopic dynamics of epidemic networks. Virulence 2010, 1, 338–349. [Google Scholar]
- Zur Wiesch, P.A.; Kouyos, R.; Engelstadter, J.; Regoes, R.R.; Bonhoeffer, S. Population biological principles of drug-resistance evolution in infectious diseases. Lancet Infect. Dis. 2011, 11, 236–247. [Google Scholar]
- Kim, H.U.; Sohn, S.B.; Lee, S.Y. Metabolic network modeling and simulation for drug targeting and discovery. Biotechnol. J. 2012, 7, 330–342. [Google Scholar]
- Chavali, A.K.; D'Auria, K.M.; Hewlett, E.L.; Pearson, R.D.; Papin, J.A. A metabolic network approach for the identification and prioritization of antimicrobial drug targets. Trends Microbiol. 2012, 20, 113–123. [Google Scholar]
- Sigurdsson, G.; Fleming, R.M.; Heinken, A.; Thiele, I. A systems biology approach to drug targets in Pseudomonas aeruginosa biofilm. PLoS One 2012, 7, e34337. [Google Scholar]
- Fagerlind, M.G.; Webb, J.S.; Barraud, N.; McDougald, D.; Jansson, A.; Nilsson, P.; Harlen, M.; Kjelleberg, S.; Rice, S.A. Dynamic modelling of cell death during biofilm development. J. Theor. Biol. 2012, 295, 23–36. [Google Scholar]
- Ilie, O.; van Loosdrecht, M.C.; Picioreanu, C. Mathematical modelling of tooth demineralisation and pH profiles in dental plaque. J. Theor. Biol. 2012, 309, 159–175. [Google Scholar]
- Hindre, T.; Knibbe, C.; Beslon, G.; Schneider, D. New insights into bacterial adaptation through in vivo and in silico experimental evolution. Nat. Rev. Microbiol. 2012, 10, 352–365. [Google Scholar]
- Kawecki, T.J.; Lenski, R.E.; Ebert, D.; Hollis, B.; Olivieri, I.; Whitlock, M.C. Experimental evolution. Trends Ecol. Evol. 2012, 27, 547–560. [Google Scholar]
- Bianconi, I.; Milani, A.; Cigana, C.; Paroni, M.; Levesque, R.C.; Bertoni, G.; Bragonzi, A. Positive signature-tagged mutagenesis in Pseudomonas aeruginosa: tracking patho-adaptive mutations promoting airways chronic infection. PLoS Pathog. 2011, 7, e1001270. [Google Scholar]
- Hogardt, M.; Heesemann, J. Microevolution of Pseudomonas aeruginosa to a Chronic Pathogen of the Cystic Fibrosis Lung. Curr Top. Microbiol. Immunol. 2012, 2012, 91–118. [Google Scholar]
- Traverse, C.C.; Mayo-Smith, L.M.; Poltak, S.R.; Cooper, V.S. Tangled bank of experimentally evolved Burkholderia biofilms reflects selection during chronic infections. Proc. Natl. Acad. Sci. USA 2012, 110, E250–259. [Google Scholar]
- Yang, L.; Jelsbak, L.; Marvig, R.L.; Damkiaer, S.; Workman, C.T.; Rau, M.H.; Hansen, S.K.; Folkesson, A.; Johansen, H.K.; Ciofu, O.; et al. Evolutionary dynamics of bacteria in a human host environment. Proc. Natl. Acad. Sci. USA 2011, 108, 7481–7486. [Google Scholar] [CrossRef]
- Holmes, E.; Kinross, J.; Gibson, G.R.; Burcelin, R.; Jia, W.; Pettersson, S.; Nicholson, J.K. Therapeutic modulation of microbiota-host metabolic interactions. Sci. Transl. Med. 2012, 4, 137rv136. [Google Scholar]
- Mee, M.T.; Wang, H.H. Engineering ecosystems and synthetic ecologies. Mol. bioSystems 2012, 8, 2470–2483. [Google Scholar]
- Anderl, J.N.; Franklin, M.J.; Stewart, P.S. Role of antibiotic penetration limitation in Klebsiella pneumoniae biofilm resistance to ampicillin and ciprofloxacin. Antimicrob. Agents Chemother. 2000, 44, 1818–1824. [Google Scholar]
- Zuroff, T.R.; Bernstein, H.; Lloyd-Randolfi, J.; Jimenez-Taracido, L.; Stewart, P.S.; Carlson, R.P. Robustness analysis of culturing perturbations on Escherichia coli colony biofilm beta-lactam and aminoglycoside antibiotic tolerance. BMC Microbiol. 2010, 10, 185. [Google Scholar]
- Genevaux, P.; Muller, S.; Bauda, P. A rapid screening procedure to identify mini-Tn10 insertion mutants of Escherichia coli K-12 with altered adhesion properties. FEMS Microbiol. Lett. 1996, 142, 27–30. [Google Scholar]
- Chavant, P.; Gaillard-Martinie, B.; Talon, R.; Hébraud, M.; Bernardi, T. A new device for rapid evaluation of biofilm formation potential by bacteria. J. Microbiol. Methods 2007, 68, 605–612. [Google Scholar]
- Ceri, H.; Olson, M.E.; Stremick, C.; Read, R.R.; Morck, D.; Buret, A. The Calgary Biofilm Device: new technology for rapid determination of antibiotic susceptibilities of bacterial biofilms. J. Clin. Microbiol. 1999, 37, 1771–1776. [Google Scholar]
- Merritt, J.H.; Kadouri, D.E.; O'Toole, G.A. Growing and analyzing static biofilms. In Current Protocols in Microbiology; John Wiley & Sons, 2005; Vol. 1B1, pp. 1–17. [Google Scholar]
- Heydorn, A.; Nielsen, A.T.; Hentzer, M.; Sternberg, C.; Givskov, M.; Ersbøll, B.K.; Molin, S. Quantification of biofilm structures by the novel computer program COMSTAT. Microbiology (Reading, Engl) 2000, 146, 2395–2407. [Google Scholar]
- Donlan, R.M.; Piede, J.A.; Heyes, C.D.; Sanii, L.; Murga, R.; Edmonds, P.; El-Sayed, I.; El-Sayed, M.A. Model system for growing and quantifying Streptococcus pneumoniae biofilms in situ and in real time. Appl. Environ. Microbiol. 2004, 70, 4980–4988. [Google Scholar]
- Ghigo, J.M. Natural conjugative plasmids induce bacterial biofilm development. Nature 2001, 412, 442–445. [Google Scholar]
- McCoy, W.F.; Bryers, J.D.; Robbins, J.; Costerton, J.W. Observations of fouling biofilm formation. Can. J. Microbiol. 1981, 27, 910–917. [Google Scholar]
- Woods, J.; Boegli, L.; Kirker, K.R.; Agostinho, A.M.; Durch, A.M.; Delancey Pulcini, E.; Stewart, P.S.; James, G.A. Development and application of a polymicrobial, In vitro, wound biofilm model. J. Appl. Microbiol. 2012, 112, 998–1006. [Google Scholar]
- Xu, K.D.; Stewart, P.S.; Xia, F.; Huang, C.T.; McFeters, G.A. Spatial physiological heterogeneity in Pseudomonas aeruginosa biofilm is determined by oxygen availability. Appl. Environ. Microbiol. 1998, 64, 4035–4039. [Google Scholar]
- Richter, L.; Stepper, C.; Mak, A.; Reinthaler, A.; Heer, R.; Kast, M.; Bruckl, H.; Ertl, P. Development of a microfluidic biochip for online monitoring of fungal biofilm dynamics. Lab. Chip 2007, 7, 1723–1731. [Google Scholar]
- Peters, A.C.; Wimpenny, J.W. A constant-depth laboratory model film fermentor. Biotechnol. Bioeng. 1988, 32, 263–270. [Google Scholar]
- Wilson, M. Use of constant depth film fermentor in studies of biofilms of oral bacteria. Methods Enzymol. 1999, 310, 264–279. [Google Scholar]
- Pitts, B.; Willse, A.; McFeters, G.A.; Hamilton, M.A.; Zelver, N.; Stewart, P.S. A repeatable laboratory method for testing the efficacy of biocides against toilet bowl biofilms. J. Appl. Microbiol. 2001, 91, 110–117. [Google Scholar]
- Benoit, M.R.; Conant, C.G.; Ionescu-Zanetti, C.; Schwartz, M.; Matin, A. New device for high-throughput viability screening of flow biofilms. Appl. Environ. Microbiol. 2010, 76, 4136–4142. [Google Scholar]
- Lawrence, J.R.; Swerhone, G.D.; Neu, T.R. A simple rotating annular reactor for replicated biofilm studies. J. Microbiol. Methods 2000, 42, 215–224. [Google Scholar]
- Hodgson, A.E.; Nelson, S.M.; Brown, M.R.; Gilbert, P. A simple In vitro model for growth control of bacterial biofilms. J. Appl. Bacteriol. 1995, 79, 87–93. [Google Scholar]
- Gilbert, P.; Allison, D.G.; Evans, D.J.; Handley, P.S.; Brown, M.R. Growth rate control of adherent bacterial populations. Appl. Environ. Microbiol. 1989, 55, 1308–1311. [Google Scholar]
- Schaller, M.; Schafer, W.; Korting, H.C.; Hube, B. Differential expression of secreted aspartyl proteinases in a model of human oral candidosis and in patient samples from the oral cavity. Mol. Microbiol. 1998, 29, 605–615. [Google Scholar]
- Guggenheim, B.; Giertsen, E.; Schüpbach, P.; Shapiro, S. Validation of an In vitro biofilm model of supragingival plaque. J. Dental Res. 2001, 80, 363–370. [Google Scholar]
- Guggenheim, M.; Thurnheer, T.; Gmur, R.; Giovanoli, P.; Guggenheim, B. Validation of the Zurich burn-biofilm model. Burns J. Int. Soc. Burn Injuries 2011, 37, 1125–1133. [Google Scholar]
- Grubb, S.E.; Murdoch, C.; Sudbery, P.E.; Saville, S.P.; Lopez-Ribot, J.L.; Thornhill, M.H. Adhesion of Candidaalbicans to endothelial cells under physiological conditions of flow. Infect. Immun. 2009, 77, 3872–3878. [Google Scholar]
- Woodworth, B.A.; Tamashiro, E.; Bhargave, G.; Cohen, N.A.; Palmer, J.N. An In vitro model of Pseudomonas aeruginosa biofilms on viable airway epithelial cell monolayers. Am. J. Rhinol. 2008, 22, 235–238. [Google Scholar]
- McBain, A.J.; Sissons, C.; Ledder, R.G.; Sreenivasan, P.K.; De Vizio, W.; Gilbert, P. Development and characterization of a simple perfused oral microcosm. J. Appl. Microbiol. 2005, 98, 624–634. [Google Scholar]
- Kim, J.; Hegde, M.; Jayaraman, A. Microfluidic co-culture of epithelial cells and bacteria for investigating soluble signal-mediated interactions. J. Vis. Exp. 2010, 38, e1749. [Google Scholar]
- Huang, T.Y.; Gulabivala, K.; Ng, Y.L. A bio-molecular film ex vivo model to evaluate the influence of canal dimensions and irrigation variables on the efficacy of irrigation. Int. Endodontic J. 2008, 41, 60–71. [Google Scholar]
- Chuang-Smith, O.N.; Wells, C.L.; Henry-Stanley, M.J.; Dunny, G.M. Acceleration of Enterococcusfaecalis biofilm formation by aggregation substance expression in an ex vivo model of cardiac valve colonization. PLoS One 2010, 5, e15798. [Google Scholar]
- Carterson, A.J.; Honer zu Bentrup, K.; Ott, C.M.; Clarke, M.S.; Pierson, D.L.; Vanderburg, C.R.; Buchanan, K.L.; Nickerson, C.A.; Schurr, M.J. A549 lung epithelial cells grown as three-dimensional aggregates: alternative tissue culture model for Pseudomonas aeruginosa pathogenesis. Infect. Immun. 2005, 73, 1129–1140. [Google Scholar] [CrossRef]
- Nickerson, C.A.; Goodwin, T.J.; Terlonge, J.; Ott, C.M.; Buchanan, K.L.; Uicker, W.C.; Emami, K.; LeBlanc, C.L.; Ramamurthy, R.; Clarke, M.S.; et al. Three-dimensional tissue assemblies: novel models for the study of Salmonella enterica serovar Typhimurium pathogenesis. Infect. Immun. 2001, 69, 7106–7120. [Google Scholar] [CrossRef]
- Smith, Y.C.; Grande, K.K.; Rasmussen, S.B.; O'Brien, A.D. Novel three-dimensional organoid model for evaluation of the interaction of uropathogenic Escherichia coli with terminally differentiated human urothelial cells. Infect. Immun. 2006, 74, 750–757. [Google Scholar]
- Benghezal, M.; Adam, E.; Lucas, A.; Burn, C.; Orchard, M.G.; Deuschel, C.; Valentino, E.; Braillard, S.; Paccaud, J.P.; Cosson, P. Inhibitors of bacterial virulence identified in a surrogate host model. Cell Microbiol. 2007, 9, 1336–1342. [Google Scholar]
- Sandstrom, G.; Saeed, A.; Abd, H. Acanthamoeba-bacteria: a model to study host interaction with human pathogens. Curr. Drug Targets 2011, 12, 936–941. [Google Scholar]
- Annesley, S.J.; Fisher, P.R. Dictyostelium discoideum--a model for many reasons. Mol. Cell. Biochem. 2009, 329, 73–91. [Google Scholar]
- Silo-Suh, L.; Suh, S.J.; Sokol, P.A.; Ohman, D.E. A simple alfalfa seedling infection model for Pseudomonas aeruginosa strains associated with cystic fibrosis shows AlgT (sigma-22) and RhlR contribute to pathogenesis. Proc. Natl. Acad. Sci. USA 2002, 99, 15699–15704. [Google Scholar]
- Schlaich, N.L. Arabidopsis thaliana- the model plant to study host-pathogen interactions. Curr. Drug Targets 2011, 12, 955–966. [Google Scholar]
- Graf, J. Symbiosis of Aeromonas veronii biovar sobria and Hirudo medicinalis, the medicinal leech: a novel model for digestive tract associations. Infect. Immun. 1999, 67, 1–7. [Google Scholar]
- Laws, T.R.; Smith, S.A.; Smith, M.P.; Harding, S.V.; Atkins, T.P.; Titball, R.W. The nematode Panagrellus redivivus is susceptible to killing by human pathogens at 37 degrees C. FEMS Microbiol. Lett. 2005, 250, 77–83. [Google Scholar] [CrossRef]
- Marsh, E.K.; May, R.C. Caenorhabditis elegans, a model organism for investigating immunity. Appl. Environ. Microbiol. 2012, 78, 2075–2081. [Google Scholar]
- Jander, G.; Rahme, L.G.; Ausubel, F.M. Positive correlation between virulence of Pseudomonas aeruginosa mutants in mice and insects. J. Bacteriol. 2000, 182, 3843–3845. [Google Scholar]
- Kounatidis, I.; Ligoxygakis, P. Drosophila as a model system to unravel the layers of innate immunity to infection. Open Biol. 2012, 2, 120075. [Google Scholar]
- Kanther, M.; Rawls, J.F. Host-microbe interactions in the developing zebrafish. Curr. Opin. Immunol. 2010, 22, 10–19. [Google Scholar]
- Yanagihara, K.; Ohnishi, Y.; Morinaga, Y.; Nakamura, S.; Kurihara, S.; Seki, M.; Izumikawa, K.; Kakeya, H.; Yamamoto, Y.; Yamada, Y.; et al. Efficacy of ME1036 against meticillin-resistant Staphylococcus aureus and vancomycin-insensitive S. aureus in a model of haematogenous pulmonary infection. Int. J. Antimicrob. Agents 2008, 32, 401–404. [Google Scholar] [CrossRef]
- Yanagihara, K.; Seki, M.; Izumikawa, K.; Higashiyama, Y.; Miyazaki, Y.; Hirakata, Y.; Tomono, K.; Mizuta, Y.; Tsukamoto, K.; Kohno, S. Potency of DX-619, a novel des-F(6)-quinolone, in haematogenous murine bronchopneumonia caused by methicillin-resistant and vancomycin-intermediate Staphylococcus aureus. Int. J. Antimicrob. Agents 2006, 28, 212–216. [Google Scholar] [CrossRef]
- Rogers, C.S.; Hao, Y.; Rokhlina, T.; Samuel, M.; Stoltz, D.A.; Li, Y.; Petroff, E.; Vermeer, D.W.; Kabel, A.C.; Yan, Z.; et al. Production of CFTR-null and CFTR-DeltaF508 heterozygous pigs by adeno-associated virus-mediated gene targeting and somatic cell nuclear transfer. J. Clin. Invest. 2008, 118, 1571–1577. [Google Scholar] [CrossRef]
- Clerc, M.; Bebear, C.; Goursolle, M.; Aparicio, M. Calculi experimentally obtained in the rat by intrarenal injection of Ureaplasma urealyticum. Ann. Biol. Clin. (Paris) 1984, 42, 277–281. [Google Scholar]
- Gurjala, A.N.; Geringer, M.R.; Seth, A.K.; Hong, S.J.; Smeltzer, M.S.; Galiano, R.D.; Leung, K.P.; Mustoe, T.A. Development of a novel, highly quantitative in vivo model for the study of biofilm-impaired cutaneous wound healing. Wound Repair. Regen 2011, 19, 400–410. [Google Scholar] [CrossRef]
- Swords, W.E.; Moore, M.L.; Godzicki, L.; Bukofzer, G.; Mitten, M.J.; VonCannon, J. Sialylation of lipooligosaccharides promotes biofilm formation by nontypeable Haemophilus influenzae. Infect. Immun. 2004, 72, 106–113. [Google Scholar] [CrossRef]
- Hasturk, H.; Kantarci, A.; Goguet-Surmenian, E.; Blackwood, A.; Andry, C.; Serhan, C.N.; Van Dyke, T.E. Resolvin E1 regulates inflammation at the cellular and tissue level and restores tissue homeostasis in vivo. J. Immunol. 2007, 179, 7021–7029. [Google Scholar]
- Pouliot, M.; Clish, C.B.; Petasis, N.A.; Van Dyke, T.E.; Serhan, C.N. Lipoxin A(4) analogues inhibit leukocyte recruitment to Porphyromonas gingivalis: a role for cyclooxygenase-2 and lipoxins in periodontal disease. Biochemistry 2000, 39, 4761–4768. [Google Scholar]
- Andes, D.; Nett, J.; Oschel, P.; Albrecht, R.; Marchillo, K.; Pitula, A. Development and characterization of an in vivo central venous catheter Candida albicans biofilm model. Infect. Immun. 2004, 72, 6023–6031. [Google Scholar] [CrossRef]
- Kurosaka, Y.; Ishida, Y.; Yamamura, E.; Takase, H.; Otani, T.; Kumon, H. A non-surgical rat model of foreign body-associated urinary tract infection with Pseudomonas aeruginosa. Microbiol. Immunol. 2001, 45, 9–15. [Google Scholar]
- Mayberry-Carson, K.J.; Tober-Meyer, B.; Lambe, D.W., Jr.; Costerton, J.W. An electron microscopic study of the effect of clindamycin therapy on bacterial adherence and glycocalyx formation in experimental Staphylococcus aureus osteomyelitis. Microbios 1986, 48, 189–206. [Google Scholar]
- Eerenberg, J.P.; Patka, P.; Haarman, H.J.; Dwars, B.J. A new model for posttraumatic osteomyelitis in rabbits. J. Invest. Surg. 1994, 7, 453–465. [Google Scholar] [CrossRef]
- Del Pozo, J.L.; Rouse, M.S.; Euba, G.; Kang, C.I.; Mandrekar, J.N.; Steckelberg, J.M.; Patel, R. The electricidal effect is active in an experimental model of Staphylococcus epidermidis chronic foreign body osteomyelitis. Antimicrob. Agents Chemother. 2009, 53, 4064–4068. [Google Scholar] [CrossRef]
- Poelstra, K.A.; Barekzi, N.A.; Grainger, D.W.; Gristina, A.G.; Schuler, T.C. A novel spinal implant infection model in rabbits. Spine (Phila Pa 1976) 2000, 25, 406–410. [Google Scholar] [CrossRef]
- Li, D.; Gromov, K.; Soballe, K.; Puzas, J.E.; O'Keefe, R.J.; Awad, H.; Drissi, H.; Schwarz, E.M. Quantitative mouse model of implant-associated osteomyelitis and the kinetics of microbial growth, osteolysis, and humoral immunity. J. Orthop. Res. 2008, 26, 96–105. [Google Scholar] [CrossRef]
- Philipov, J.P.; Pascalev, M.D.; Aminkov, B.Y.; Grosev, C.D. Changes in serum carboxyterminal telopeptide of type I collagen in an experimental model of canine osteomyelitis. Calcif Tissue Int. 1995, 57, 152–154. [Google Scholar]
- Williams, D.L.; Haymond, B.S.; Woodbury, K.L.; Beck, J.P.; Moore, D.E.; Epperson, R.T.; Bloebaum, R.D. Experimental model of biofilm implant-related osteomyelitis to test combination biomaterials using biofilms as initial inocula. J. Biomed. Mater. Res. A 2012, 100, 1888–1900. [Google Scholar]
- Southwood, R.T.; Rice, J.L.; McDonald, P.J.; Hakendorf, P.H.; Rozenbilds, M.A. Infection in experimental hip arthroplasties. J. Bone Joint Surg. Br. 1985, 67, 229–231. [Google Scholar]
- Bergamini, T.M.; Bandyk, D.F.; Govostis, D.; Kaebnick, H.W.; Towne, J.B. Infection of vascular prostheses caused by bacterial biofilms. J. Vasc. Surg. 1988, 7, 21–30. [Google Scholar]
- Farooq, M.; Freischlag, J.; Kelly, H.; Seabrook, G.; Cambria, R.; Towne, J. Gelatin-sealed polyester resists Staphylococcus epidermidis biofilm infection. J. Surg. Res. 1999, 87, 57–61. [Google Scholar]
- Arad, E.; Navon-Venezia, S.; Gur, E.; Kuzmenko, B.; Glick, R.; Frenkiel-Krispin, D.; Kramer, E.; Carmeli, Y.; Barnea, Y. Novel Rat Model of Methicillin-Resistant Staphyloccocus aureus Infected Silicone Breast-Implants: A Study of Biofilm Pathogenesis. Plast Reconstr. Surg. 2013, 131, 205–214. [Google Scholar]
- Chilukuri, D.M.; Shah, J.C. Local delivery of vancomycin for the prophylaxis of prosthetic device-related infections. Pharm Res. 2005, 22, 563–572. [Google Scholar]
- Hessburg, P.C.; Truant, J.P.; Penn, W.P. Treatment of Pseudomonas keratitis in rabbits. Am. J. Ophthalmol. 1966, 61, 49–54. [Google Scholar]
- Fulcher, T.P.; Dart, J.K.; McLaughlin-Borlace, L.; Howes, R.; Matheson, M.; Cree, I. Demonstration of biofilm in infectious crystalline keratopathy using ruthenium red and electron microscopy. Ophthalmology 2001, 108, 1088–1092. [Google Scholar]
- Engelbert, M.; Gilmore, M.S. Fas ligand but not complement is critical for control of experimental Staphylococcus aureus Endophthalmitis. Invest. Ophthalmol. Vis. Sci. 2005, 46, 2479–2486. [Google Scholar]
- Elder, M.J.; Stapleton, F.; Evans, E.; Dart, J.K. Biofilm-related infections in ophthalmology. Eye (Lond) 1995, 9, 102–109. [Google Scholar] [CrossRef]
- Shea, P.R.; Virtaneva, K.; Kupko, J.J., 3rd; Porcella, S.F.; Barry, W.T.; Wright, F.A.; Kobayashi, S.D.; Carmody, A.; Ireland, R.M.; Sturdevant, D.E.; et al. Interactome analysis of longitudinal pharyngeal infection of cynomolgus macaques by group A Streptococcus. Proc. Natl. Acad. Sci. USA 2010, 107, 4693–4698. [Google Scholar] [CrossRef]
- Chole, R.A.; Faddis, B.T. Anatomical evidence of microbial biofilms in tonsillar tissues: a possible mechanism to explain chronicity. Arch. Otolaryngol. Head Neck Surg. 2003, 129, 634–636. [Google Scholar]
- Post, J.C.; Hiller, N.L.; Nistico, L.; Stoodley, P.; Ehrlich, G.D. The role of biofilms in otolaryngologic infections: update 2007. Curr. Opin. Otolaryngol. Head Neck Surg. 2007, 15, 347–351. [Google Scholar]
- McDuffie, R.S., Jr.; Kunze, M.; Barr, J.; Wolf, D.; Sze, C.I.; Shikes, R.; Sherman, M.; Gibbs, R.S. Chronic intrauterine and fetal infection with Gardnerella vaginalis. Am. J. Obstet Gynecol. 2002, 187, 1263–1266. [Google Scholar]
- Verstraelen, H.; Swidsinski, A. The biofilm in bacterial vaginosis: implications for epidemiology, diagnosis and treatment. Curr. Opin. Infect. Dis. 2013, 26, 86–89. [Google Scholar]
- Klein, M.; Obermaier, B.; Angele, B.; Pfister, H.W.; Wagner, H.; Koedel, U.; Kirschning, C.J. Innate immunity to pneumococcal infection of the central nervous system depends on toll-like receptor (TLR) 2 and TLR4. J. Infect. Dis. 2008, 198, 1028–1036. [Google Scholar]
- Neil, R.B.; Apicella, M.A. Clinical and laboratory evidence for Neisseria meningitidis biofilms. Future Microbiol. 2009, 4, 555–563. [Google Scholar]
- Ruellan, K.; Frijns, J.H.; Bloemberg, G.V.; Hautefort, C.; Van den Abbeele, T.; Lamers, G.E.; Herman, P.; Huy, P.T.; Kania, R.E. Detection of bacterial biofilm on cochlear implants removed because of device failure, without evidence of infection. Otol. Neurotol. 2010, 31, 1320–1324. [Google Scholar]
- Everaert, E.P.; van de Belt-Gritter, B.; van der Mei, H.C.; Busscher, H.J.; Verkerke, G.J.; Dijk, F.; Mahieu, H.F.; Reitsma, A. In vitro and in vivo microbial adhesion and growth on argon plasma-treated silicone rubber voice prostheses. J. Mater Sci. Mater Med. 1998, 9, 147–157. [Google Scholar] [CrossRef]
- Braxton, E.E., Jr.; Ehrlich, G.D.; Hall-Stoodley, L.; Stoodley, P.; Veeh, R.; Fux, C.; Hu, F.Z.; Quigley, M.; Post, J.C. Role of biofilms in neurosurgical device-related infections. Neurosurg. Rev. 2005, 28, 249–255. [Google Scholar]
- Wilson, S.K.; Costerton, J.W. Biofilm and penile prosthesis infections in the era of coated implants: a review. J. Sex. Med. 2012, 9, 44–53. [Google Scholar]
- Ginsberg, G.; Cope, C.; Shah, J.; Martin, T.; Carty, A.; Habecker, P.; Kaufmann, C.; Clerc, C.; Nuutinen, J.P.; Tormala, P. In vivo evaluation of a new bioabsorbable self-expanding biliary stent. Gastrointest Endosc. 2003, 58, 777–784. [Google Scholar]
- Guaglianone, E.; Cardines, R.; Vuotto, C.; Di Rosa, R.; Babini, V.; Mastrantonio, P.; Donelli, G. Microbial biofilms associated with biliary stent clogging. FEMS Immunol. Med. Microbiol. 2010, 59, 410–420. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lebeaux, D.; Chauhan, A.; Rendueles, O.; Beloin, C. From in vitro to in vivo Models of Bacterial Biofilm-Related Infections. Pathogens 2013, 2, 288-356. https://doi.org/10.3390/pathogens2020288
Lebeaux D, Chauhan A, Rendueles O, Beloin C. From in vitro to in vivo Models of Bacterial Biofilm-Related Infections. Pathogens. 2013; 2(2):288-356. https://doi.org/10.3390/pathogens2020288
Chicago/Turabian StyleLebeaux, David, Ashwini Chauhan, Olaya Rendueles, and Christophe Beloin. 2013. "From in vitro to in vivo Models of Bacterial Biofilm-Related Infections" Pathogens 2, no. 2: 288-356. https://doi.org/10.3390/pathogens2020288