Molybdenum Effects on Pitting Corrosion Resistance of FeCrMnMoNC Austenitic Stainless Steels
Abstract
:1. Introduction
2. Experimental Section
2.1. Materials and Mechanical Tests
2.2. Electrochemical Tests
3. Results and Discussion
3.1. Microstructure and Tensile Properties
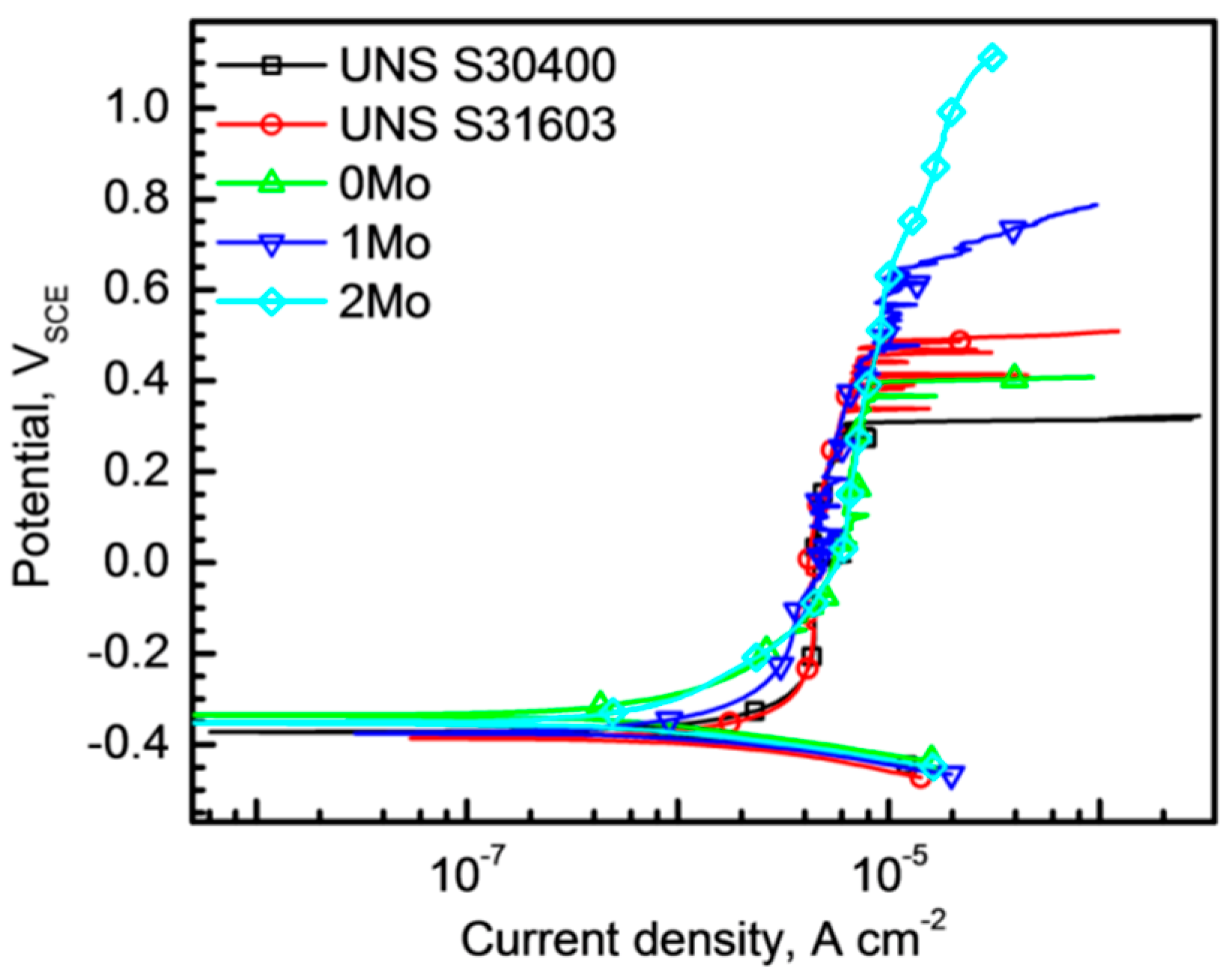
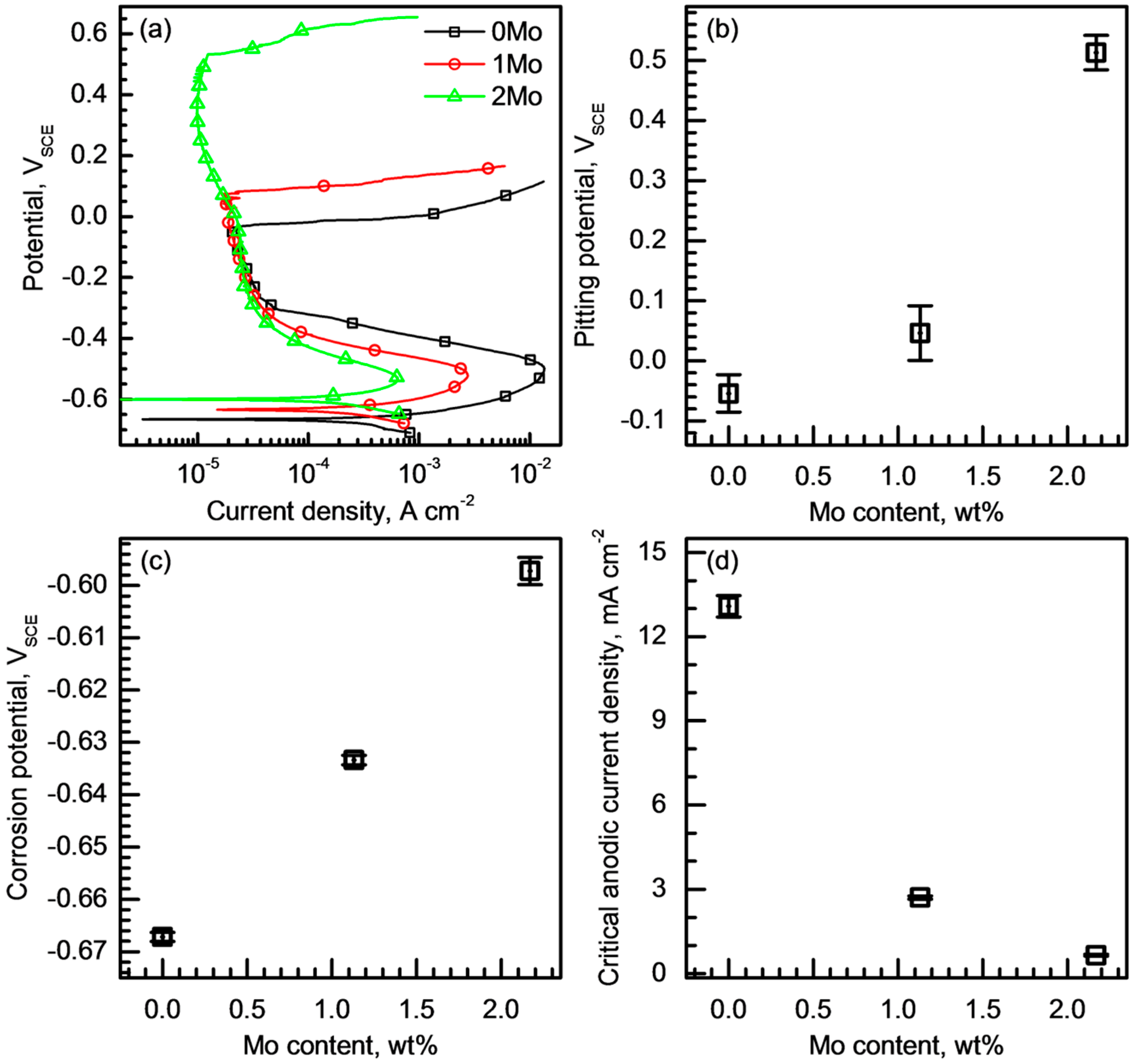
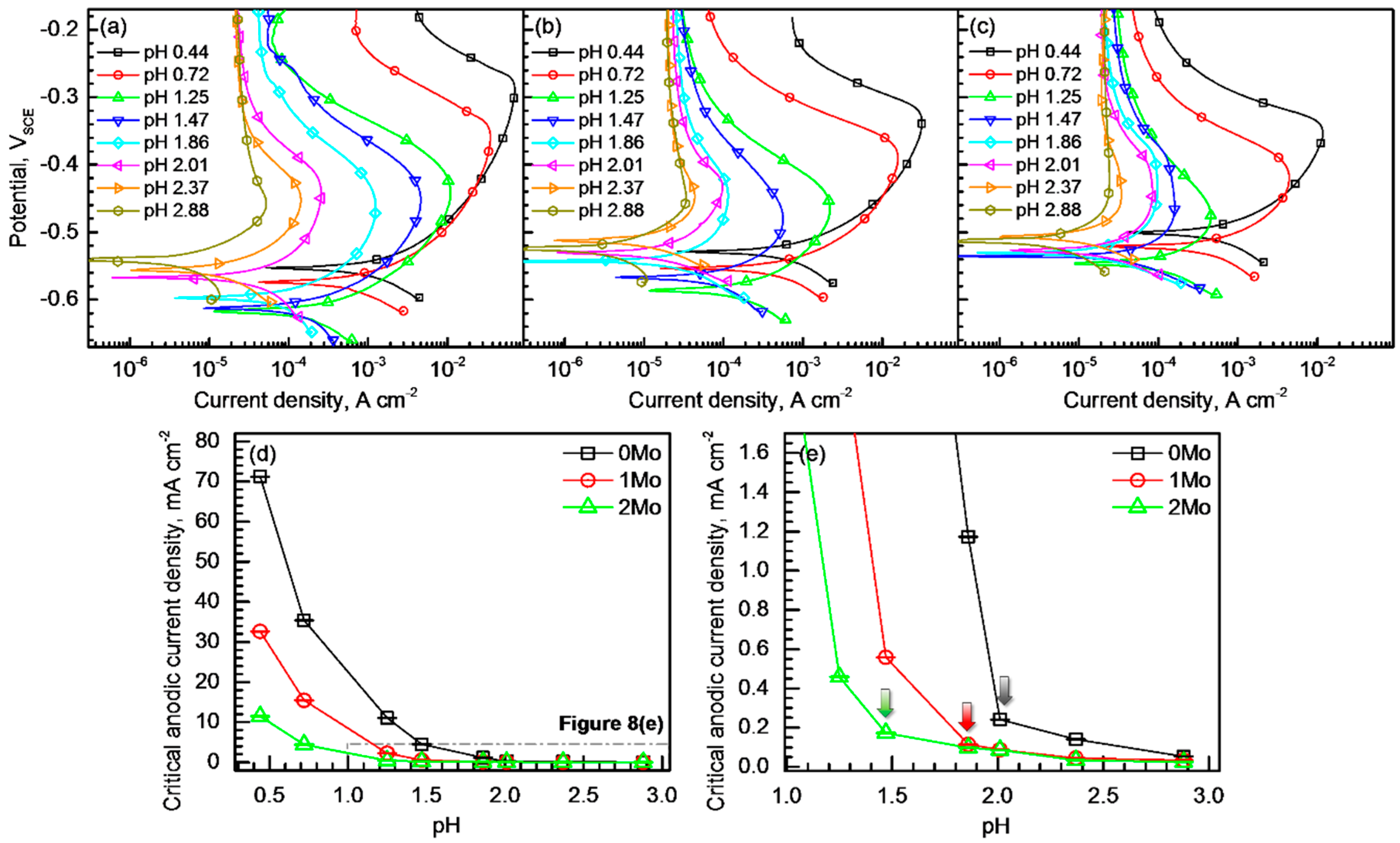
3.2. Pitting Corrosion Resistance
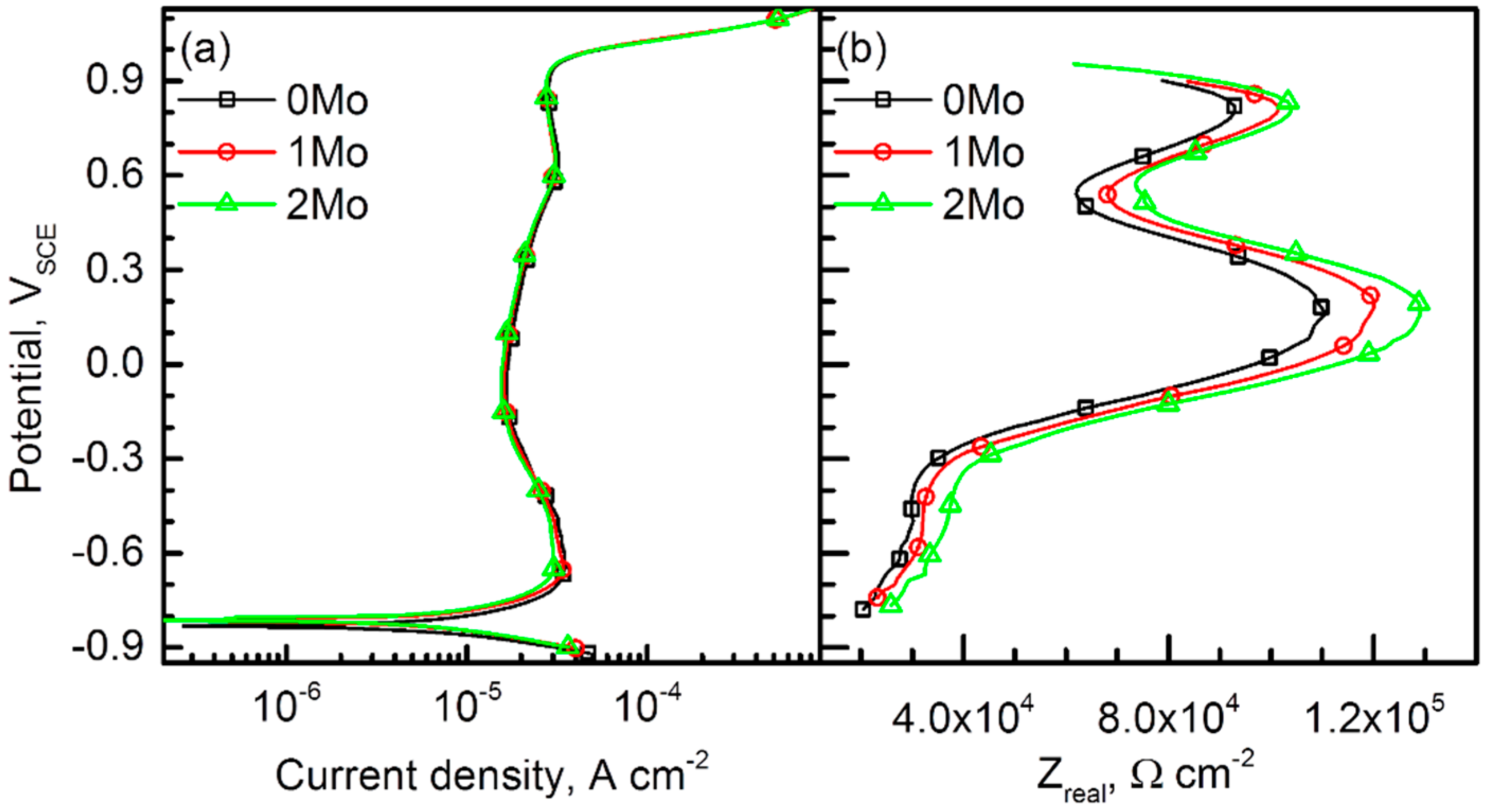
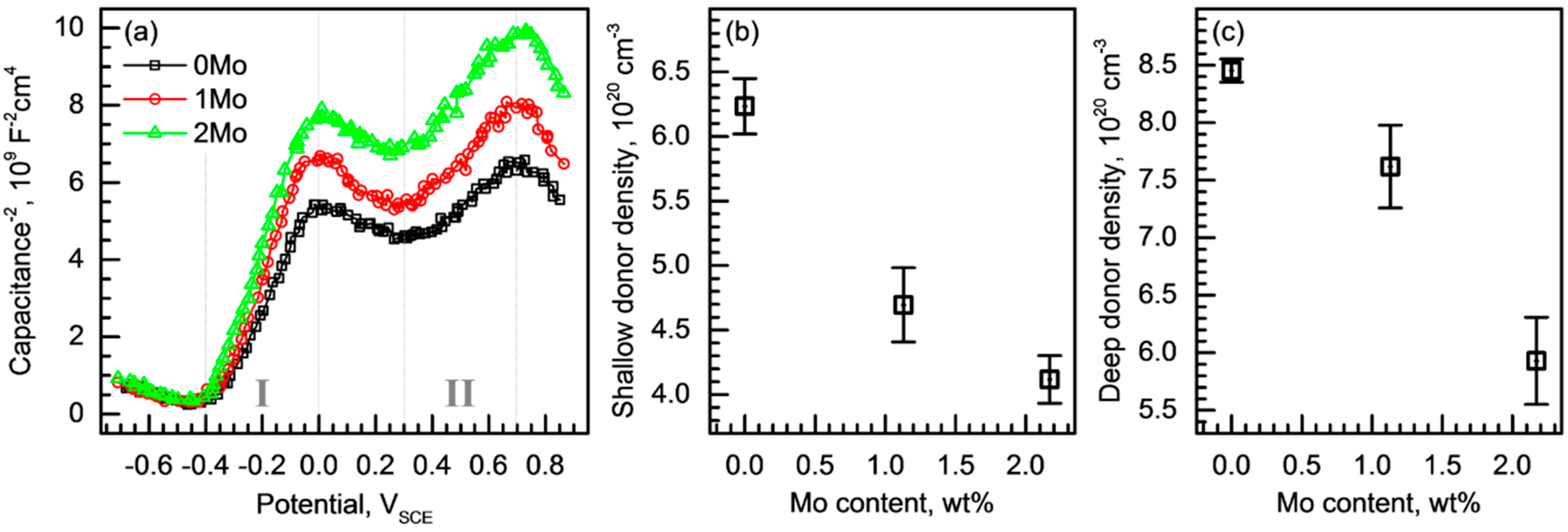
3.3. Passive Behavior
3.4. General Corrosion Behavior
4. Conclusions
- (1)
- Potentiodynamic polarization tests in chloride solutions revealed that the alloying Mo suppressed metastable pitting corrosion and raised both Epit and Erp of the alloys. In addition, it was found that the difference between the Epit and Erp decreased as the Mo content increased.
- (2)
- Passive film analysis through a resistance measurement and Mott-Schottky analysis indicated that the alloyed Mo increased the film resistance by decreasing the number of point defects in the passive film.
- (3)
- The alloyed Mo reduced the critical dissolution rate of the alloys in acidified chloride solutions, and the alloy with higher Mo content was able to resist active dissolution in stronger acid.
- (4)
- It is concluded that the alloying Mo enhanced pitting corrosion resistance of the alloy through increasing protectiveness of passive film and lowering pit propagation rate.
Author Contributions
Funding
Conflicts of Interest
References
- Thomann, U.I.; Uggowitzer, P.J. Wear-corrosion behavior of biocompatible austenitic stainless steels. Wear 2000, 239, 48–58. [Google Scholar] [CrossRef]
- Gavrilyuk, V.G.; Berns, H. High-strength Austenitic Stainless Steels. Met. Sci. Heat Treat. 2007, 49, 566–568. [Google Scholar] [CrossRef]
- Rawers, J.C. Alloying effects on the microstructure and phase stability of Fe–Cr–Mn steels. J. Mater. Sci. 2008, 43, 3618–3624. [Google Scholar] [CrossRef]
- Gavriljuk, V.G.; Shanina, B.D.; Berns, H. A physical concept for alloying steels with carbon + nitrogen. Mater. Sci. Eng. 2008, 481–482, 707–712. [Google Scholar] [CrossRef]
- Ha, H.-Y.; Lee, T.-H.; Oh, C.-S.; Kim, S.-J. Effects of combined addition of carbon and nitrogen on pitting corrosion behavior of Fe-18Cr-10Mn alloys. Scr. Mater. 2009, 61, 121–124. [Google Scholar] [CrossRef]
- Ha, H.-Y.; Lee, T.-H.; Oh, C.-S.; Kim, S.-J. Effects of carbon on the corrosion behaviour in Fe-18Cr-10Mn-N-C stainless steels. Steel Res. Int. 2009, 80, 488–492. [Google Scholar]
- Lee, T.-H.; Shin, E.; Oh, C.-S.; Ha, H.-Y.; Kim, S.-J. Correlation between stacking fault energy and deformation microstructure in high-interstitial-alloyed austenitic steels. Acta Mater. 2010, 58, 3173–3186. [Google Scholar] [CrossRef]
- Schymura, M.; Stegemann, R.; Fischer, A. Crack propagation behavior of solution annealed austenitic high interstitial steels. Int. J. Fatigue 2015, 79, 25–35. [Google Scholar] [CrossRef]
- Seifert, M.; Siebert, S.; Huth, S.; Theisen, W.; Berns, H. New Developments in Martensitic Stainless Steels Containing C + N. Steel Res. Int. 2015, 86, 1508–1516. [Google Scholar] [CrossRef]
- Niederhofer, P.; Richrath, L.; Huth, S.; Theisen, W. Influence of conventional and powder–metallurgical manufacturing on the cavitation erosion and corrosion of high interstitial CrMnCN austenitic stainless steels. Wear 2016, 360–361, 67–76. [Google Scholar] [CrossRef]
- Gavriljuk, V.G.; Berns, H. High Nitrogen Steels, 1st ed.; Springer: Berlin, Germany, 1999; Chapter 1–2. [Google Scholar]
- Levey, P.R.; van Bennekom, A. A mechanistic study of the effects of nitrogen on the corrosion properties of stainless steels. Corrosion 1995, 51, 911–921. [Google Scholar] [CrossRef]
- Jargelius-Pettersson, R.F.A. Electrochemical investigation of the influence of nitrogen alloying on pitting corrosion of austenitic stainless steels. Corros. Sci. 1999, 41, 1639–1664. [Google Scholar] [CrossRef]
- Ha, H.-Y.; Lee, T.-H.; Kim, S.-J. Effect of C fraction on corrosion properties of high interstitial alloyed stainless steels. Metall. Mater. Trans. A 2012, 43, 2999–3005. [Google Scholar] [CrossRef]
- Speidel, M.O. Nitrogen containing austenitic stainless steels. Mater. Sci. Eng. Technol. 2006, 37, 875–880. [Google Scholar] [CrossRef]
- Lee, T.-H.; Kim, S.-J.; Oh, C.-S.; Ha, H.-Y. High Strength and High Corrosion Coal Nitrogen Combined Addition Austenitic Stainless Steel and a Manufacturing Method Thereof (In Japanese). Patent JP 5272078 B2, 17 May 2013. [Google Scholar]
- Lee, T.-H.; Kim, S.-J.; Oh, C.-S.; Ha, H.-Y. High Strength/Corrosion Resistant Austenitic Stainless Steel with Carbon-Nitrogen Complex Additive, and Method for Manufacturing Same. Patent EP 2455508 B1, 23 November 2016. [Google Scholar]
- Lo, K.H.; Shek, C.H.; Lai, J.K.L. Recent developments in stainless steels. Mater. Sci. Eng. 2009, 65, 39–104. [Google Scholar] [CrossRef]
- Frankel, G.S. Pitting Corrosion of Metals—A Review of the Critical Factors. J. Electrochem. Soc. 1998, 145, 2186–2198. [Google Scholar] [CrossRef]
- Newman, R.C. The dissolution and passivation kinetics of stainless alloys containing molybdenum—1. Coulometric studies of Fe-Cr and Fe-Cr-Mo alloys. Corros. Sci. 1985, 25, 331–339. [Google Scholar] [CrossRef]
- Newman, R.C. The dissolution and passivation kinetics of stainless alloys containing molybdenum—II. Dissolution kinetics in artificial pits. Corros. Sci. 1985, 25, 341–350. [Google Scholar] [CrossRef]
- Kaneko, M.; Isaacs, H.S. Effects of molybdenum on the pitting of ferritic- and austenitic-stainless steels in bromide and chloride solutions. Corros. Sci. 2002, 44, 1825–1834. [Google Scholar] [CrossRef]
- Villanueva, D.M.E.; Junior, F.C.P.; Plaut, R.L.; Padilha, A.F. Comparative study on sigma phase precipitation of three types of stainless steels: austenitic, superferritic and duplex. Mater. Sci. Technol. 2006, 22, 1098–1104. [Google Scholar] [CrossRef]
- Sourmail, T. Precipitation in creep resistant austenitic stainless steels. Mater. Sci. Technol. 2001, 17, 1–14. [Google Scholar] [CrossRef]
- Weiss, B.; Stickler, R. Phase Instabilities During High Temperature Exposure of 316 Austenitic Stainless Steel. Metall. Mater. Trans. B 1972, 3, 851–866. [Google Scholar] [CrossRef]
- Sugimoto, K.; Sawada, Y. The role of molybdenum additions to austenitic stainless steels in the inhibition of pitting in acid chloride solutions. Corros. Sci. 1977, 17, 425–445. [Google Scholar] [CrossRef]
- Hashimoto, K.; Asami, K.; Teramoto, K. An X-ray photo-electron spectroscopic study on the role of molybdenum in increasing the corrosion resistance of ferritic stainless steels in HCl. Corros. Sci. 1979, 19, 3–14. [Google Scholar] [CrossRef]
- Clayton, C.R.; Lu, Y.C. A Bipolar Model of the Passivity of Stainless Steel: The Role of Mo Addition. J. Electrochem. Soc. 1986, 133, 2465–2473. [Google Scholar] [CrossRef]
- Landolt, D.; Mischler, S.; Vogel, A.; Mathieu, H.J. Chloride Ion Effects on Passive Films on FeCr and FeCrMo Studied by AES, XPS and SIMS. Corros. Sci. 1990, 31, 431–440. [Google Scholar] [CrossRef]
- Montemor, M.F.; Simoes, A.M.P.; Ferreira, M.G.S.; Da Cunha Belo, M. The role of Mo in the chemical composition and semiconductive behaviour of oxide flms formed on stainless steels. Corros. Sci. 1999, 41, 17–34. [Google Scholar] [CrossRef]
- Ilevbare, G.O.; Burstein, G.T. The role of alloyed molybdenum in the inhibition of pitting corrosion in stainless steels. Corros. Sci. 2001, 43, 485–513. [Google Scholar] [CrossRef]
- Bastidas, J.M.; Torres, C.L.; Cano, E.; Polo, J.L. Influence of molybdenum on passivation of polarised stainless steels in a chloride environment. Corros. Sci. 2002, 44, 625–633. [Google Scholar] [CrossRef] [Green Version]
- Tobler, W.J.; Vertanen, S. Effect of Mo species on metastable pitting of Fe18Cr alloys—A current transient analysis. Corros. Sci. 2006, 48, 1585–1607. [Google Scholar] [CrossRef]
- Li, D.G.; Wang, J.D.; Chen, D.R.; Liang, P. Molybdenum addition enhancing the corrosion behaviors of 316 L stainless steel in the simulated cathodic environment of proton exchange membrane fuel cell. Int. J. Hydrogen Energy 2015, 40, 5947–5957. [Google Scholar] [CrossRef]
- Loable, C.; Vicosa, I.N.; Mesquita, T.J.; Mantel, M.; Nogueira, R.P.; Berthome, G.; Chauveau, E.; Roche, V. Synergy between molybdenum and nitrogen on the pitting corrosion and passive film resistance of austenitic stainless steels as a pH-dependent effect. Mater. Chem. Phys. 2017, 186, 237–245. [Google Scholar] [CrossRef]
- Mesquita, T.J.; Chauveau, E.; Mantel, M.; Nogueira, R.P. A XPS study of the Mo effect on passivation behaviors for highly controlled stainless steels in neutral and alkaline conditions. Appl. Surf. Sci. 2013, 270, 90–97. [Google Scholar] [CrossRef]
- Vignal, V.; Olive, J.M.; Desjardins, D. Effect of molybdenum on passivity of stainless steels in chloride media using ex situ near field microscopy observations. Corros. Sci. 1999, 41, 869–884. [Google Scholar] [CrossRef]
- Mesquita, T.J.; Chauveau, E.; Mantel, M.; Kinsman, N.; Roche, V.; Nogueira, R.P. Lean duplex stainless steels—The role of molybdenum in pitting corrosion of concrete reinforcement studied with industrial and laboratory castings. Mater. Chem. Phys. 2012, 132, 967–972. [Google Scholar] [CrossRef]
- Olsson, C.-O.A. The influence of nitrogen and molybdenum on passive films formed on the austenoferritic stainless steel 2205 studied by AES and XPS. Corros. Sci. 1995, 37, 467–479. [Google Scholar] [CrossRef]
- Ha, H.-Y.; Lee, T.-H.; Kim, S.-J. Role of nitrogen in the active–passive transition behavior of binary Fe-Cr alloy system. Electrochim. Acta 2012, 80, 432–439. [Google Scholar] [CrossRef]
- Ha, H.-Y.; Jang, M.-H.; Lee, T.-H. Influences of Mn in solid solution on the pitting corrosion behaviour of Fe-23 wt. %Cr-based alloys. Electrochim. Acta 2016, 191, 864–875. [Google Scholar] [CrossRef]
- Krakowiak, S.; Darowicki, K.; Slepski, P. Impedance investigation of passive 304 stainless steel in the pit pre-initiation state. Electrochim. Acta 2005, 50, 2699–2704. [Google Scholar] [CrossRef]
- Nagarajan, S.; Rajendran, N. Crevice corrosion behaviour of superaustenitic stainless steels: Dynamic electrochemical impedance spectroscopy and atomic force microscopy studies. Corros. Sci. 2009, 51, 217–224. [Google Scholar] [CrossRef]
- American Society for Testing and Materials (ASTM). ASTM A276-06, Standard Specification for Stainless Steel Bars and Shapes; ASTM: West Conshohocken, PA, USA, 2016. [Google Scholar]
- Ha, H.-Y.; Kwon, H.-S. Effects of pH levels on the surface charge and pitting corrosion resistance of Fe. J. Electrochem. Soc. 2012, 159, C416–C421. [Google Scholar] [CrossRef]
- Piao, T.; Park, S.-M. Spectroelectrochemical studies of passivation and transpassive breakdown reactions of stainless steel. J. Electrochem. Soc. 1997, 144, 3371–3377. [Google Scholar] [CrossRef]
- Cho, E.A.; Kwon, H.S.; Macdonald, D.D. Photoelectrochemical analysis on the passive film formed on Fe–20Cr in pH 8.5 buffer solution. Electrochim. Acta 2002, 47, 1661–1668. [Google Scholar] [CrossRef]
- Macdonald, D.D. The Point Defect Model for the Passive State. J. Electrochem. Soc. 1992, 139, 3434–3449. [Google Scholar] [CrossRef]
- Macdonald, D.D. The history of the Point Defect Model for the passive state: A brief review of film growth aspects. Electrochim. Acta 2011, 56, 1761–1772. [Google Scholar] [CrossRef]
- Dean, M.H.; Stimming, U. The electronic properties of disordered passive films. Corros. Sci. 1989, 29, 199–211. [Google Scholar] [CrossRef]
- Fattah-alhosseini, A.; Vafaeian, S. Comparison of electrochemical behavior between coarse-grained and fine-grained AISI 430 ferritic stainless steel by Mott–Schottky analysis and EIS measurements. J. Alloy. Compd. 2015, 639, 301–307. [Google Scholar] [CrossRef]
- Ahn, S.J.; Kwon, H.S. Effects of solution temperature on electronic properties of passive film formed on Fe in pH 8.5 borate buffer solution. Electrochim. Acta 2004, 49, 3347–3353. [Google Scholar] [CrossRef]
- Sedek, P.; Brozda, J.; Gazdowicz, J. Pitting corrosion of the stainless steel ventilation duct in a roofed swimming pool. Eng. Fail. Anal. 2008, 15, 281–286. [Google Scholar] [CrossRef]
- Szala, M.; Beer-Lech, K.; Walczak, M. A study on the corrosion of stainless steel floor drains in an indoor swimming pool. Eng. Fail. Anal. 2017, 77, 31–38. [Google Scholar] [CrossRef]
- Alfonsson, E.; Mameng, S.H. The possibilities & limitations of austenitic and duplex stainless steels in chlorinated water systems. Nucl. Exch. 2012, 30–34. [Google Scholar]
- Olsson, J.; Snis, M. Duplex—A new generation of stainless steels for desalination plants. Desalination 2007, 205, 104–113. [Google Scholar] [CrossRef]



| Alloys | Fe | Cr | Mn | Mo | N | C | Yield Strength (MPa) | Tensile Strength (MPa) | Elongation (%) |
|---|---|---|---|---|---|---|---|---|---|
| 0Mo | Balance | 18.19 | 9.72 | - | 0.36 | 0.50 | 502.1 | 957.2 | 61.7 |
| 1Mo | 17.89 | 9.81 | 1.13 | 0.40 | 0.47 | 499.2 | 897.5 | 56.4 | |
| 2Mo | 18.10 | 9.47 | 2.17 | 0.38 | 0.48 | 529.0 | 979.9 | 62.1 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ha, H.-Y.; Lee, T.-H.; Bae, J.-H.; Chun, D.W. Molybdenum Effects on Pitting Corrosion Resistance of FeCrMnMoNC Austenitic Stainless Steels. Metals 2018, 8, 653. https://doi.org/10.3390/met8080653
Ha H-Y, Lee T-H, Bae J-H, Chun DW. Molybdenum Effects on Pitting Corrosion Resistance of FeCrMnMoNC Austenitic Stainless Steels. Metals. 2018; 8(8):653. https://doi.org/10.3390/met8080653
Chicago/Turabian StyleHa, Heon-Young, Tae-Ho Lee, Jee-Hwan Bae, and Dong Won Chun. 2018. "Molybdenum Effects on Pitting Corrosion Resistance of FeCrMnMoNC Austenitic Stainless Steels" Metals 8, no. 8: 653. https://doi.org/10.3390/met8080653






