Thermodynamic Reassessment of the Na-Cu and Na-K Binary Systems
Abstract
:1. Introduction
2. Thermodynamic Modeling
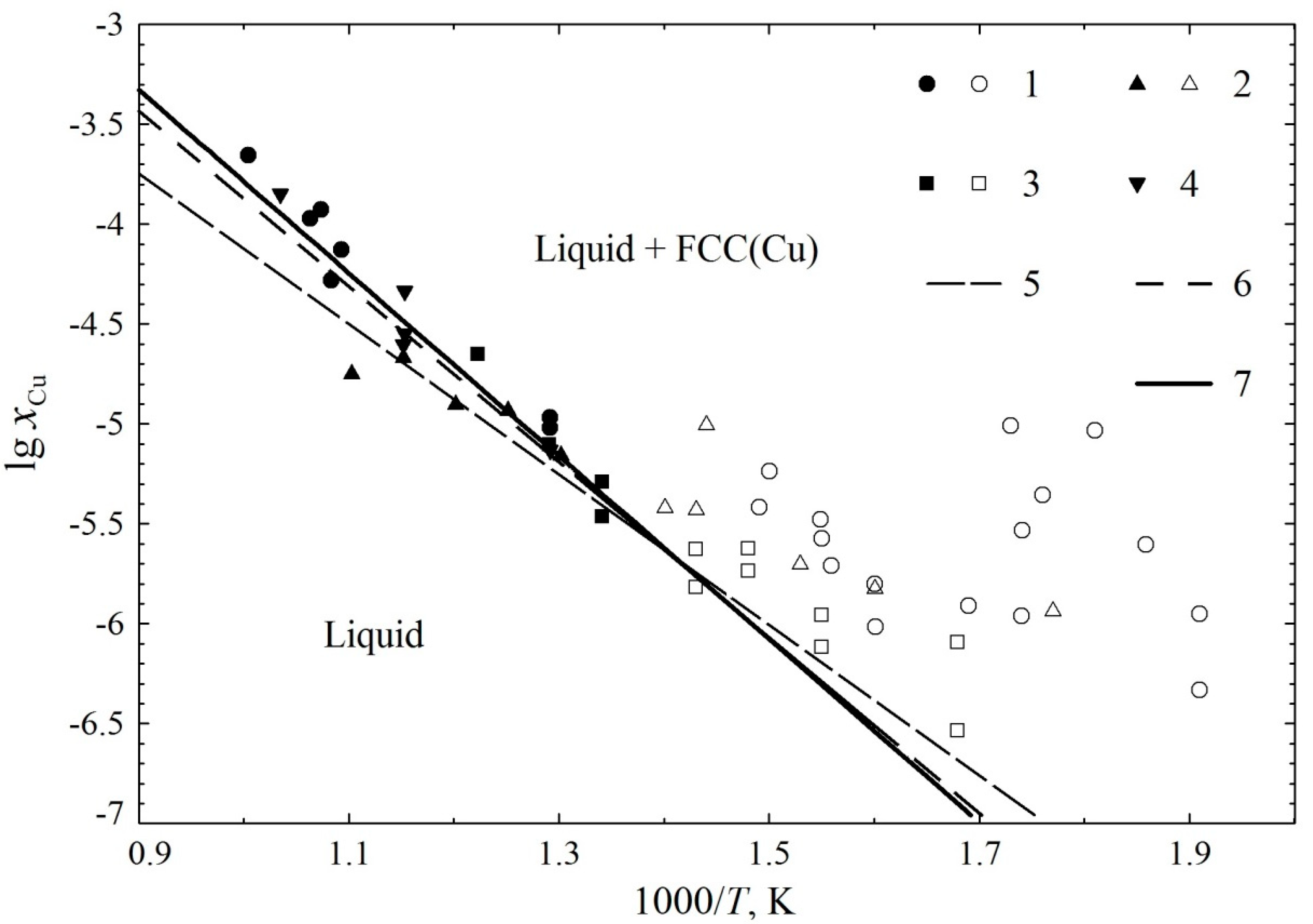
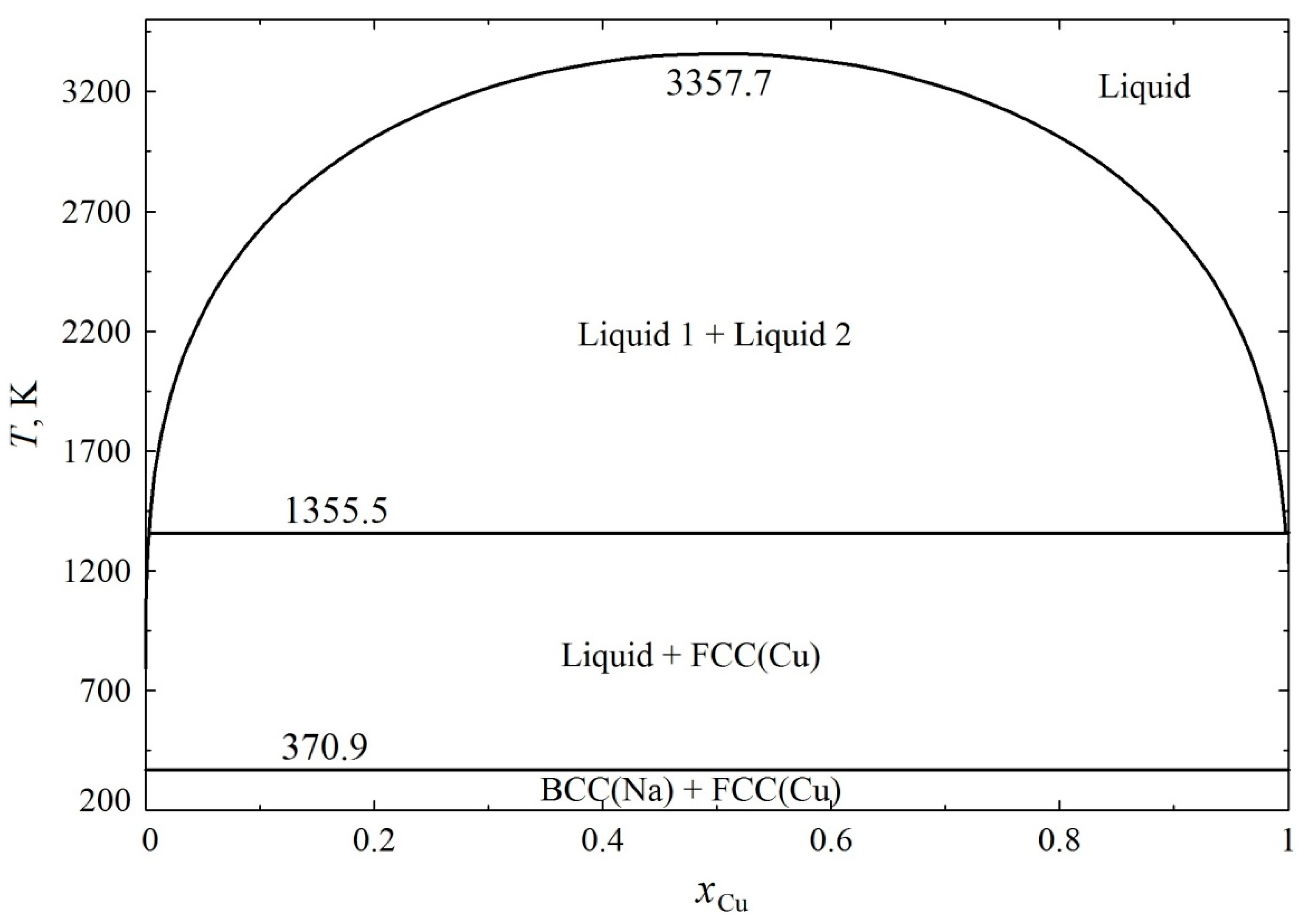
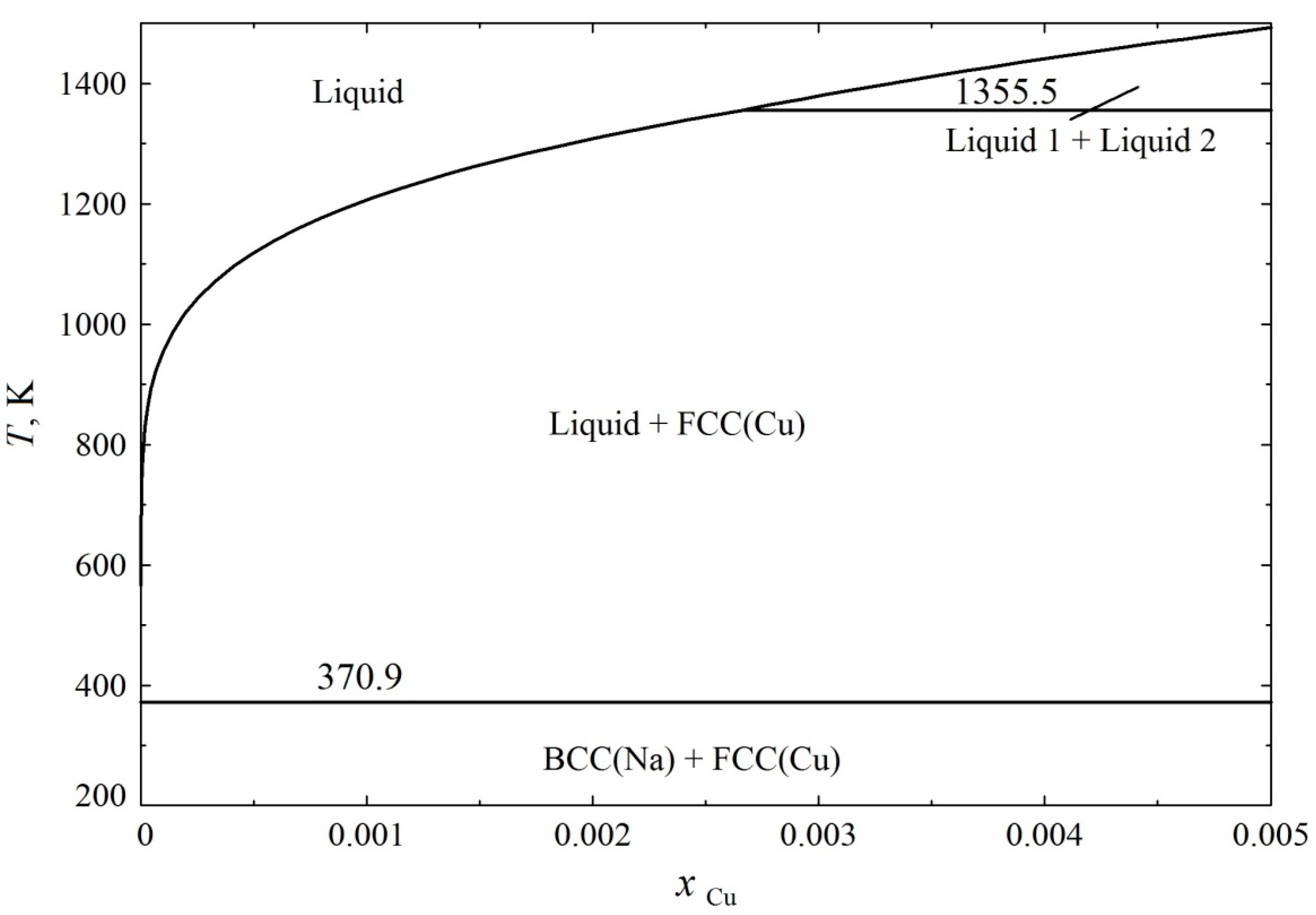
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Baldev, R.; Chellapandi, P.; Vasudeva Rao, P.R. Sodium Fast Reactors with Closed Fuel Cycle; CRC Press: Boca Raton, FL, USA, 2015; pp. 1–901. ISBN 9781466587694. [Google Scholar]
- Pelton, A.D. The Cu-K (copper-potassium) system. Bull. Alloy Phase Diagrams 1986, 7, 231. [Google Scholar] [CrossRef]
- Bale, C.W.; Chartrand, P.; Degterov, S.A.; Eriksson, G.; Hack, K.; Ben Mahfoud, R.; Melançon, J.; Pelton, A.D.; Petersen, S. FactSage Thermochemical Software and Databases. Calphad 2002, 26, 189–228. [Google Scholar] [CrossRef]
- Bale, C.W.; Bélisle, E.; Chartrand, P.; Decterov, S.A.; Eriksson, G.; Hack, K.; Jung, I.-H.; Kang, Y.-B.; Melançon, J.; Pelton, A.D.; et al. FactSage thermochemical software and databases—recent developments. Calphad 2009, 33, 295–311. [Google Scholar] [CrossRef]
- Bale, C.W.; Bélisle, E.; Chartrand, P.; Decterov, S.A.; Eriksson, G.; Gheribi, A.E.; Hack, K.; Jung, I.-H.; Kang, Y.-B.; Melançon, J.; Pelton, A.D.; et al. FactSage thermochemical software and databases, 2010–2016. Calphad 2016, 54, 35–53. [Google Scholar] [CrossRef]
- Dinsdale, A.T. SGTE data for pure elements. Calphad 1991, 15, 317–425. [Google Scholar] [CrossRef]
- Hillert, M. Partial Gibbs energies from Redlich-Kister polynomials. Thermochim. Acta 1988, 129, 71–75. [Google Scholar] [CrossRef]
- Hao, D.; Bu, M.; Wang, Y.; Tang, Y.; Gao, Q.; Wang, M.; Hu, B.; Du, Y. Thermodynamic modeling of the Na–X (X = Si, Ag, Cu, Cr) systems. J. Min. Metall. Sect. B 2012, 48, 273–282. [Google Scholar] [CrossRef]
- Pelton, A.D. The Cu-Na (copper-sodium) system. Bull. Alloy Phase Diagrams 1986, 7, 25–27. [Google Scholar] [CrossRef]
- Eichelberger, R.L.; McKisson, R.L. Solubility of Cu in Na; North American Rockwell Corporation: Springfield, VA, USA, 31 May 1968; AI-AEC-12671. [Google Scholar]
- Walker, R.A.; Pratt, J.N. The solubility of copper in liquid sodium. J. Nucl. Mater. 1969, 32, 340–345. [Google Scholar] [CrossRef]
- Singer, R.M.; Becker, W.; Weeks, J.R. Quarterly Progress Report of Work Sponsored by the Fuel and Materials Branch, April to June 1967; Brookhaven National Lab: Upton, NY, USA, 1967; BNL-50065. [Google Scholar]
- Singer, R.M.; Weeks, J.R. On the Solubilities of Cu, Ni, and Fe in Liquid Sodium. In Proceedings of the International Conference on Sodium Technology and Large Fast Reactor Design, 7–9 November 1968, Part I, Sessions of Sodium Technology, Argonne National Laboratories, ANL-7520; Clearinghouse for Federal Scientific and Technical Information: Springfield, VA, USA, 1969. [Google Scholar]
- Humphreys, J.R., Jr. Interdivision Document K-3-774; Los Alamos Scientific Laboratory: Los Alamos, NM, USA, 1958. [Google Scholar]
- Okamoto, H. Supplemental literature review of binary phase diagrams: B-La, B-Zn, Bi-La, Bi-Ti, Cd-Pr, Ce-Ga, Cu-Na, Ge-Ta, Ge-Y, H-Zr, Na-Si, and Pb-S. J. Phase Equilib. Diffus. 2015, 36, 644–655. [Google Scholar] [CrossRef]
- Kean, C.H. The pressure-temperature phase diagram of Na-K alloys and the effect of pressure on the resistance of the liquid phase. Phys. Rev. 1939, 55, 750–754. [Google Scholar] [CrossRef]
- Rinck, M.E. Diagrammes de solidification des alliages formés par deux métaux alcalins. Alliages sodium-potassium. C. R. Hebd. Séances Acad. sci. 1933, 197, 49–51. [Google Scholar]
- MacDonald, D.K.C.; Pearson, W.B.; Towle, L.T. An investigation of the sodium-potassium equilibrium diagram. Can. J. Phys. 1956, 34, 389–394. [Google Scholar] [CrossRef]
- Ott, J.B.; Goates, J.R.; Anderson, D.R.; Hall, H.T. Solid-liquid phase equilibria in the sodium + potassium system. Trans. Faraday Soc. 1969, 65, 2870–2878. [Google Scholar] [CrossRef]
- Van Rossen, G.L.C.M.; van Bleiswijk, H.Z. Über das zustandsdiagramm der kalium-natriumlegierungen. Anorg. Chem. 1912, 74, 152–156. [Google Scholar] [CrossRef]
- Lindberg, D.; Backman, R.; Hupa, M.; Chartrand, P. Thermodynamic Evaluation and Optimization of the (Na+K+S) System. J. Chem. Thermodyn. 2006, 38, 900–915. [Google Scholar] [CrossRef]
- Pelton, A.D.; Chartrand, P.; Eriksson, G. The modified quasi-chemical model: Part IV. Two-sublattice quadruplet approximation. Metall. Mater. Trans. A 2001, 32, 1409–1416. [Google Scholar] [CrossRef]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Trofimov, E.; Samoilova, O.; Zaitseva, O.; Vakhitova, E. Thermodynamic Reassessment of the Na-Cu and Na-K Binary Systems. Metals 2018, 8, 629. https://doi.org/10.3390/met8080629
Trofimov E, Samoilova O, Zaitseva O, Vakhitova E. Thermodynamic Reassessment of the Na-Cu and Na-K Binary Systems. Metals. 2018; 8(8):629. https://doi.org/10.3390/met8080629
Chicago/Turabian StyleTrofimov, Evgeny, Olga Samoilova, Olga Zaitseva, and Elena Vakhitova. 2018. "Thermodynamic Reassessment of the Na-Cu and Na-K Binary Systems" Metals 8, no. 8: 629. https://doi.org/10.3390/met8080629





