Metallization of Extruded Briquettes (BREX) in Midrex Process
Abstract
:1. Introduction
2. Materials and Methods
2.1. Raw Materials
2.2. Experimental Brex Production
2.3. Testing of the Brex Physical Properties
3. Results and Discussion
3.1. Mineralogical Study of the Reduced Brex
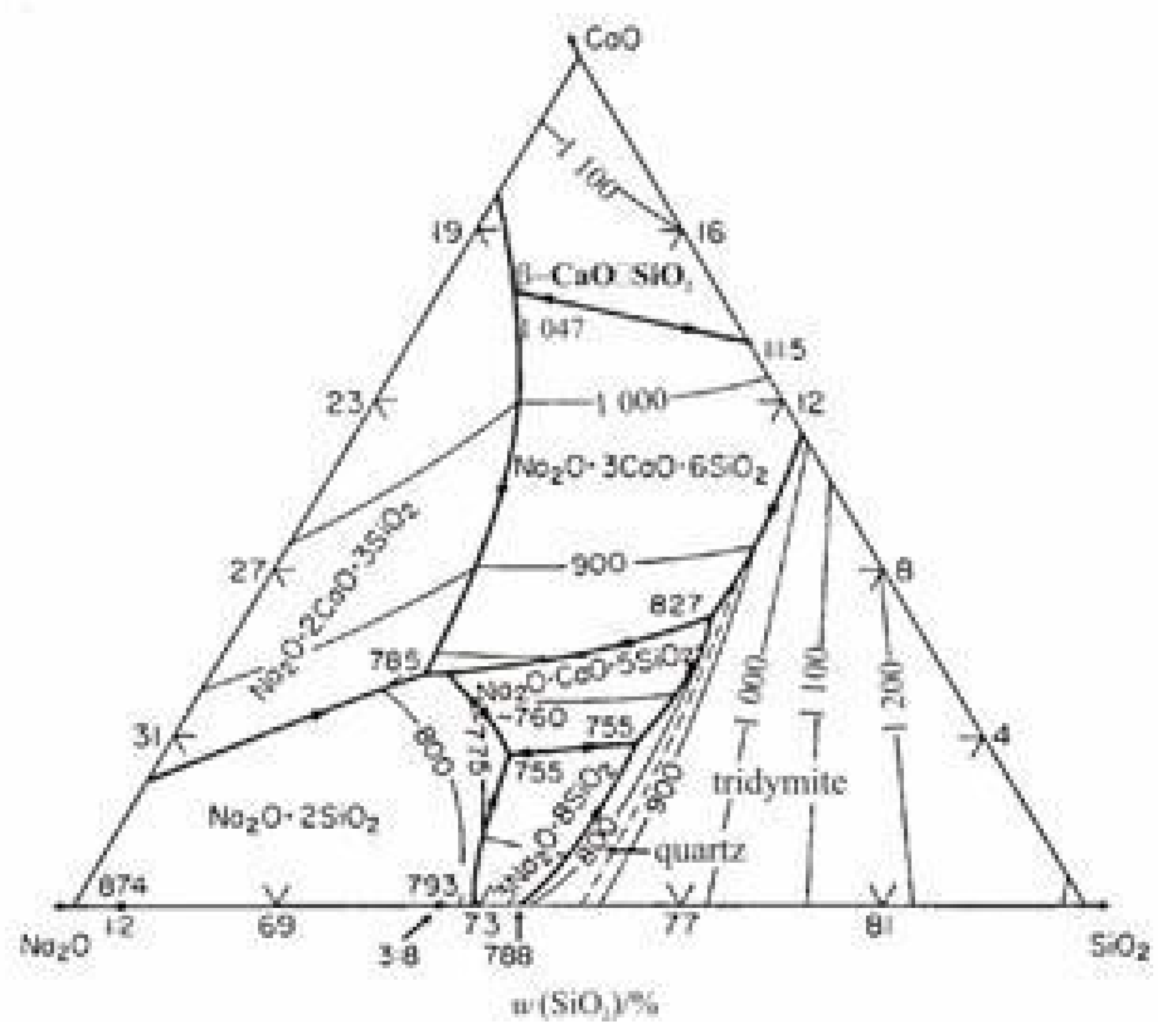
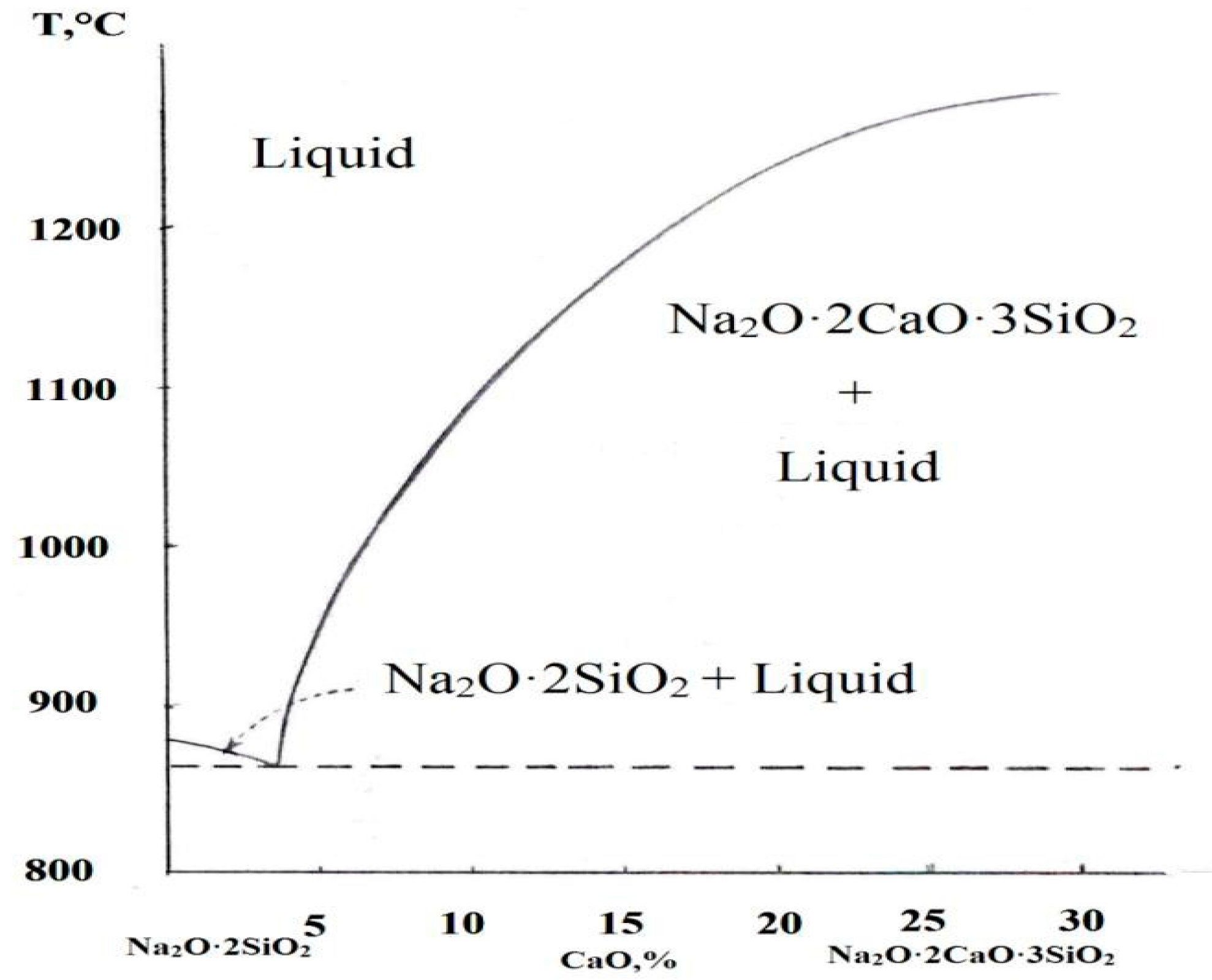
3.1.1. Mineralogical Analysis of the Reduced Cement-Bonded Brex
3.1.2. Mineralogical Analysis of the Reduced Lime-Bonded Brex
3.1.3. Mineralogical Analysis of the Reduced Brex with Magnesium Sulfate-Based Binder
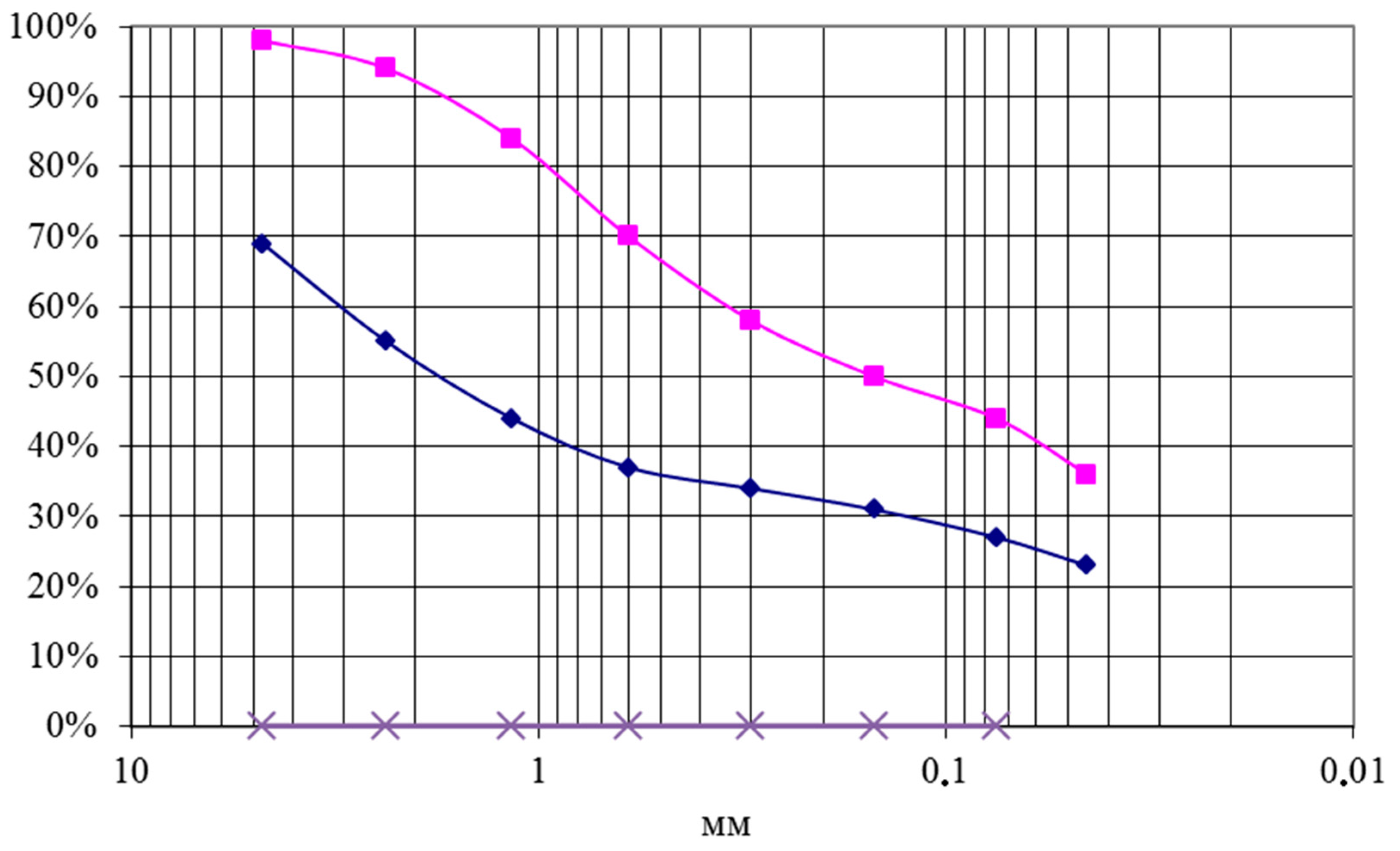
3.2. Morphological Study of the Porosity Change during Reduction
5. Conclusions
- Reduction of the brex with cement as binder was accompanied by the formation of a significant amount of fines which retarded their metallization due to clogging of the holes in the walls of the steel packages, which resulted in a violation of the conditions of the experiment.
- Low hot strength of brex with cement as a binder results from their tendency to swell as well as due to the low level of generation of silicate binders. It might be required to have cement content larger than 10% to avoid brex abnormal swelling and to promote for their sufficient hot strength.
- A sufficiently high value of the degree of metallization of the lime-bonded brex justifies further testing of such brex.
- Degree of metallization of the magnesium sulfate-bonded brex (96.71%) is comparable with the degree of metallization of the main product of DRI reactors-metallized pellets.
- During the reduction, brex microstructure changes, becoming close to the metal-silicate phase.
- There is the difference in the porosity change during the reduction depending on the type of the binder applied. In brex with magnesium binder, process of metallization was accompanied by the growth of the large pores shares.
- High Sulphur content of the brex with magnesium sulfate-based binder makes them unsuitable for Midrex reactors.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bizhanov, A.M.; Pavlov, A.V.; Chadaeva, O.; Dalmia, Y.; Mishra, B. High Temperature Reduction of the Stiff Vacuum Extrusion Briquettes under the ITmk3 Conditions. ISIJ Int. 2014, 54, 1450–1452. [Google Scholar] [CrossRef]
- Bizhanov, A.M.; Kurunov, I.F.; Podgorodetskyi, G.; Dashevskyi, V.; Pavlov, A.V.; Chadaeva, O. Extruded Briquettes—New Charge Component for the Manganese Ferroalloys Production. ISIJ Int. 2014, 54, 2206–2214. [Google Scholar] [CrossRef]
- Bizhanov, A.M.; Kurunov, I.F.; Dalmia, Y.; Mishra, B.; Mishra, S. Blast Furnace Operation with 100% Extruded Briquettes Charge. ISIJ Int. 2015, 55, 175–182. [Google Scholar] [CrossRef]
- Zhuravlev, V.F.; Zhitomirskaya, V.I. Binding properties of crystal hydrates of the sulfate type. J. Appl. Chem. USSR 1950, 23, 115–119. [Google Scholar]
- Bizhanov, A.M.; Kurunov, I.F.; Wakeel, A.K. Behavior of Extrusion Briquettes (Brex) in Midrex Reactors. Part 1. Metallurgist 2015, 59, 3–4. [Google Scholar] [CrossRef]
- Singh, M.; Bjorkman, B. Cold bond agglomerates of iron and steel plant by-products as burden material for blast furnaces. In Proceedings of the REWAS 99, San Sebastian, Spain, 5–9 September 1999; Volume 2, pp. 1539–1548. [Google Scholar]
- Berezhnoy, A.S. Multi Alkaline Oxide Systems; Naukova Dumka: Kiev, Ukraine, 1988; p. 200. [Google Scholar]
- Schwelberger, J.; Brunner, C.; Fleischanderl, A. Recycling of Ferrous By-Products in the Iron and Steel. In Proceedings of the 34th Biennale Conference of the Institute for Briquetting and Agglomeration, Scottsdale, AZ, USA, 8–11 November 2015. [Google Scholar]
- Sokolov, V.N.; Yurkovets, D.I.; Melnik, V.N.; Boyde, A.; Howell, P.G.T. 3D reconstruction of surface and subsurface structures of solids by SEM stereo images. In Proceedings of the Institute of Physics Electron Microscopy and Analysis Group Conference, Dundee, Scotland, UK, 5–7 September 2001. [Google Scholar]








| Chemical Compounds | Pellets Fine | Mill Scale | DRI Sludge | EAF Dust |
|---|---|---|---|---|
| Fetot | 65.00 | 70.0 | 66.2 | 29.68 |
| SiO2 | 2.50 | 1.00 | 2.14 | 4.25 |
| CaO | 1.30 | 0.15 | 4.38 | 19.74 |
| MgO | 0.75 | 0.10 | 0.69 | 24.27 |
| Al2O3 | 0.95 | 0.25 | 0.83 | 1.32 |
| MnO | 0.10 | 1.20 | 0.16 | 0.96 |
| S | 0.015 | 0.015 | 0.01 | 0.13 |
| Na2O + K2 | 0.034 | – | 0.33 | 1.42 |
| Charge Component | Brex #1 | Brex #2 | Brex #3 |
|---|---|---|---|
| Pellets fines | 50.0 | 50.0 | 50.0 |
| Sludge | 25.0 | 25.0 | 25.0 |
| Mill scale | 15.0 | 15.0 | 15.0 |
| EAF dust | 5.0 | 4.75 | 5.0 |
| Slaked lime | 5.0 | - | - |
| Portland cement | - | 5.0 | - |
| Magnesium binder | - | - | 5.0 |
| Bentonite | - | 0.25 | - |
| Elements and Oxides | Brex #1 | Brex #2 | Brex #3 |
|---|---|---|---|
| Fet | 62.47 | 61.30 | 62.61 |
| C | 1.49 | 1.40 | 1.05 |
| CaO | 9.24 | 8.41 | 4.30 |
| MgO | 3.61 | 3.07 | 6.24 |
| SiO2 | 2.45 | 4.91 | 2.76 |
| Al2O3 | 1.29 | 1.81 | 0.99 |
| TiO2 | 0.10 | 0.12 | 0.12 |
| V2O5 | 0.076 | 0.07 | 0.08 |
| MnO | 0.34 | 0.36 | 0.35 |
| P2O5 | 0.07 | 0.09 | 0.06 |
| S | 0.08 | 0.08 | 0.50 |
| Na2O + K2O | 0.19 | 0.93 | 0.83 |
| Cl | 0.04 | 0.03 | 0.02 |
| ZnO | 0.36 | 0.40 | 0.35 |
| Property | Brex #1 | Brex #2 | Brex #3 |
|---|---|---|---|
| Density, g/cm3 | 3.5 | 3.48 | 3.66 |
| Compressive strength, N/mm2 | 4.8 | 11.1 | 4.4 |
| Tensile splitting strength, N/mm2 | 1.5 | 1.4 | 1.2 |
| Porosity | 29.7 | 24.7 | 25.1 |
| Moisture content, % | 8.4 | 8.4 | 8.6 |
| Elements and Oxides | Brex #1 | Brex #2 | Brex #3 |
|---|---|---|---|
| Fet | 74.86 | 69.02 | 86.86 |
| Femet | 49.11 | 18.66 | 84.00 |
| Metallization, % | 65.60 | 26.96 | 96.71 |
| C | 1.75 | 0.89 | 1.02 |
| CaO | 9.11 | 6.71 | 5.07 |
| MgO | 2.93 | 2.24 | 7.81 |
| SiO2 | 3.41 | 4.35 | 4.04 |
| Al2O3 | 1.57 | 1.45 | 2.13 |
| TiO2 | 0.11 | 0.12 | 0.12 |
| V2O5 | 0.07 | 0.08 | 0.07 |
| MnO | 0.34 | 0.43 | 0.39 |
| P2O5 | 0.08 | 0.07 | 0.01 |
| S | 0.03 | 0.09 | 0.26 |
| Na2O + K2O | 0.48 | 0.51 | 0.74 |
| Cl | 0.09 | 0.01 | - |
| ZnO | 0.18 | 0.30 | 0.05 |
| BREX | Porosity SEM/SEM + CT | Contribution of Pores of Different Dimension Categories to Total Porosity (ptot), %. Diameter in Microns. | Maximum Diameter, μm | ||||
|---|---|---|---|---|---|---|---|
| ptot, % | D1 | D2 | D3 | D4 | D5 | Dmax | |
| <0.1 | 0.1–1.0 | 1.0–10 | 10–100 | >100 | |||
| Brex #1 | 31.6 | 0.6 | 8.6 | 25.6 | 65.2 | 0.0 | 57.6 |
| 37.3 | 0.5 | 7.2 | 21.4 | 66.8 | 4.1 | 407.2 | |
| Brex #1 (reduced) | 32.9 | 0.2 | 9.0 | 38.4 | 52.4 | 0.0 | 70.94 |
| 38.3 | 0.1 | 7.7 | 32.4 | 49.8 | 10.0 | 504.49 | |
| Brex #2 | 31.2 | 0.6 | 8.0 | 30.1 | 61.3 | 0.0 | 45.78 |
| 35.7 | 0.5 | 7.0 | 26.2 | 61.3 | 5.0 | 299.96 | |
| Brex #2 (reduced) | 37.9 | 0.2 | 9.3 | 31.1 | 59.1 | 0.3 | 100.51 |
| 40.4 | 0.2 | 8.9 | 29.5 | 55.2 | 6.2 | 583.77 | |
| Brex #3 | 31.8 | 0.6 | 5.8 | 28.4 | 65.2 | 0.0 | 62.92 |
| 32.9 | 0.5 | 5.5 | 27.2 | 65.3 | 1.5 | 191.89 | |
| Brex #3 (reduced) | 32.8 | 0.2 | 3.2 | 35.7 | 60.9 | 0.0 | 71.75 |
| 39.3 | 0.2 | 2.6 | 28.9 | 58.0 | 10.3 | 736.41 | |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bizhanov, A.; Malysheva, T. Metallization of Extruded Briquettes (BREX) in Midrex Process. Metals 2017, 7, 259. https://doi.org/10.3390/met7070259
Bizhanov A, Malysheva T. Metallization of Extruded Briquettes (BREX) in Midrex Process. Metals. 2017; 7(7):259. https://doi.org/10.3390/met7070259
Chicago/Turabian StyleBizhanov, Aitber, and Tatyana Malysheva. 2017. "Metallization of Extruded Briquettes (BREX) in Midrex Process" Metals 7, no. 7: 259. https://doi.org/10.3390/met7070259





