Reaction Behavior of Phosphorus in Coal-Based Reduction of an Oolitic Hematite Ore and Pre-Dephosphorization of Reduced Iron
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Reduction and Dephosphorization Experiments
2.3. Analysis and Characterization
3. Results and Discussion
3.1. Reaction Behavior of Phosphorus
3.1.1. Effect of Reduction Temperature
3.1.2. Effect of Reduction Time
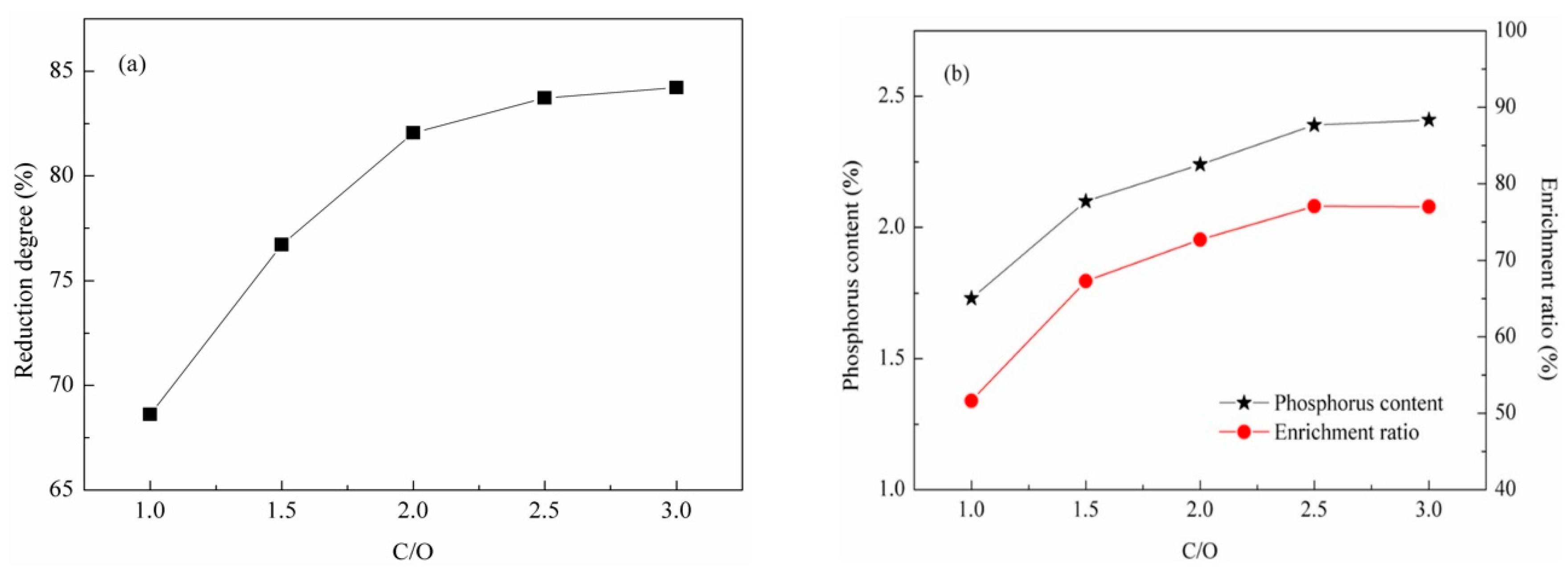
3.1.3. Effect of C/O Molar Ratio
3.2. Phase Transformation of Phosphorus
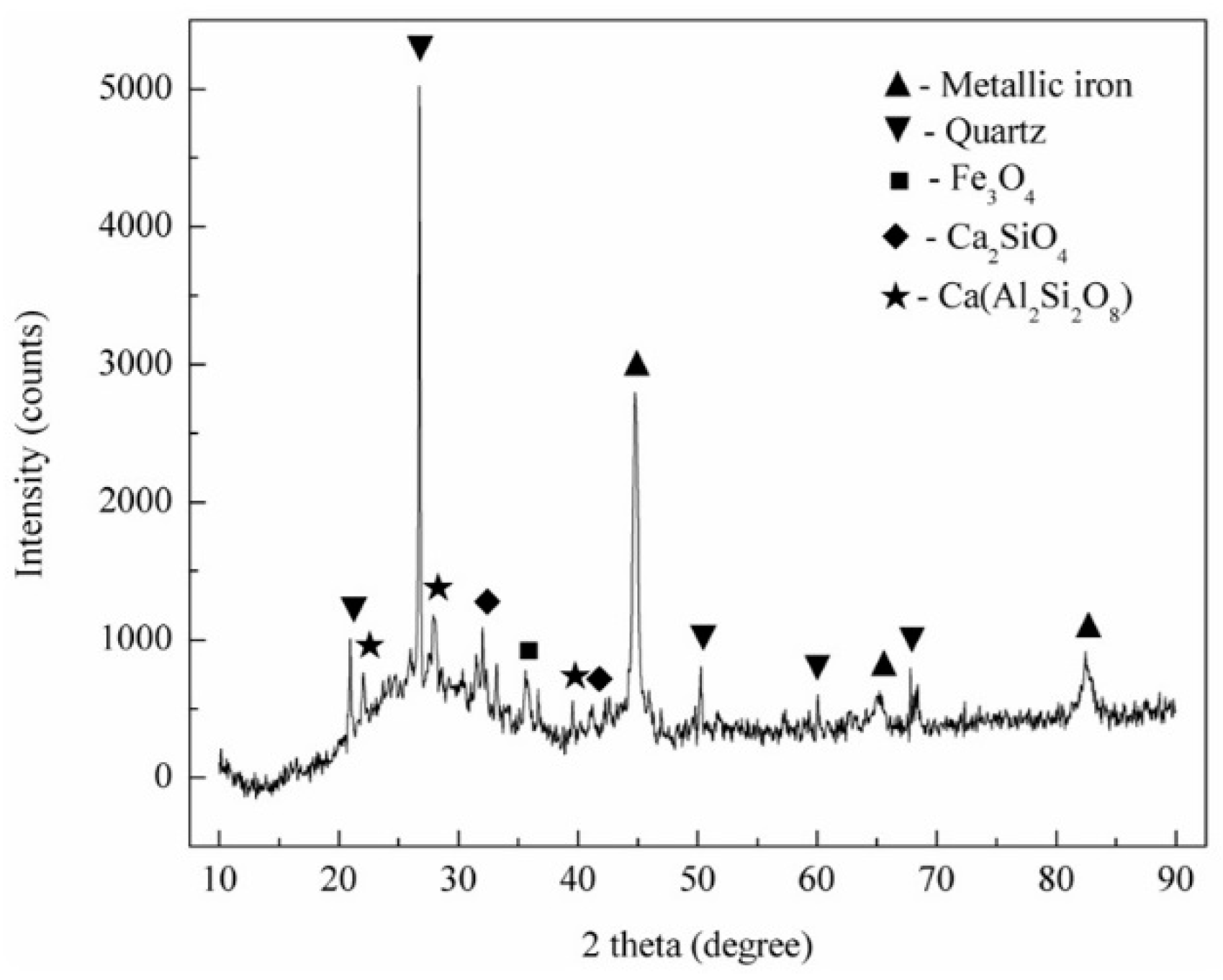
3.2.1. Phase Compositions of Reduction Products
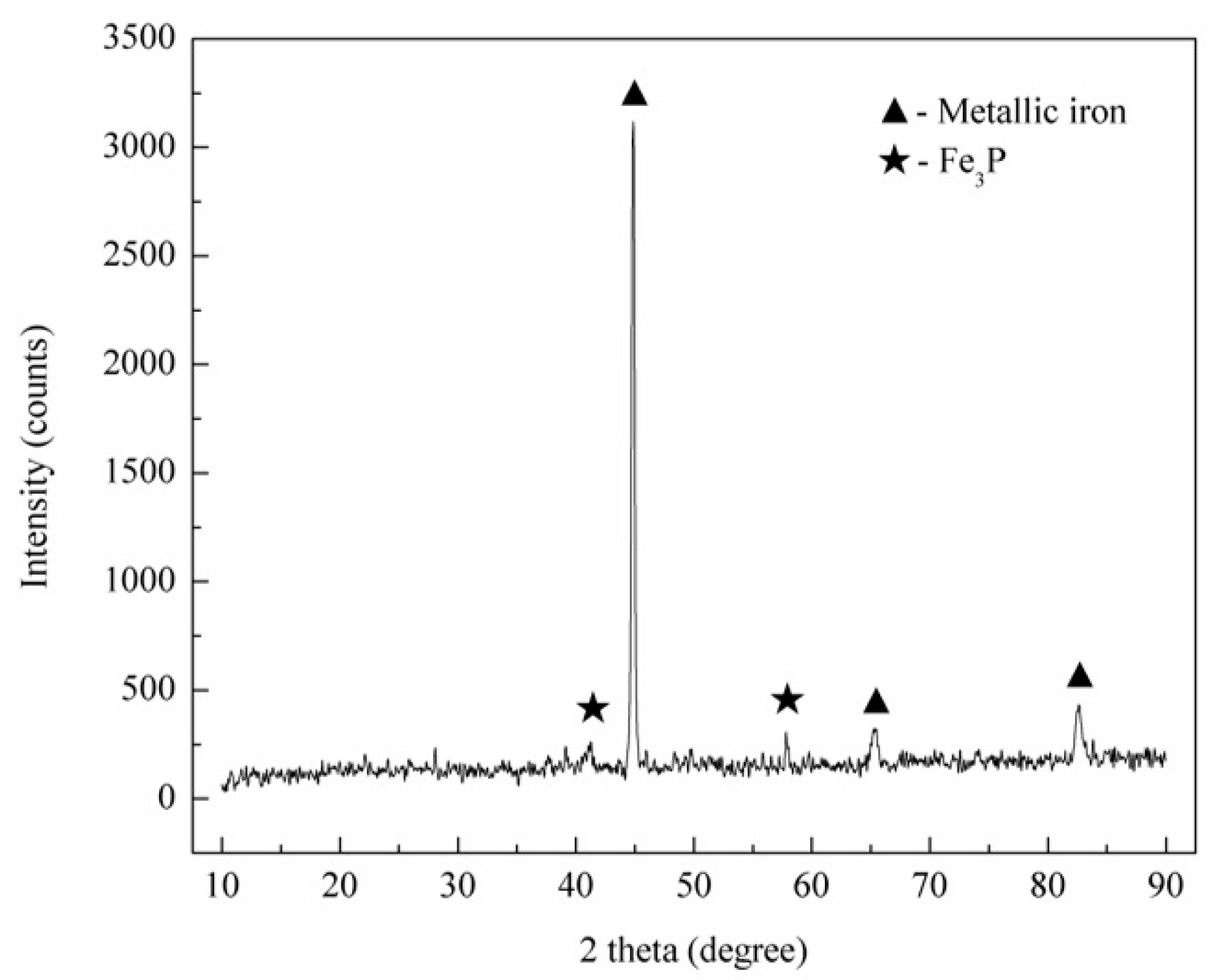
3.2.2. Phase Compositions of Reduced Iron
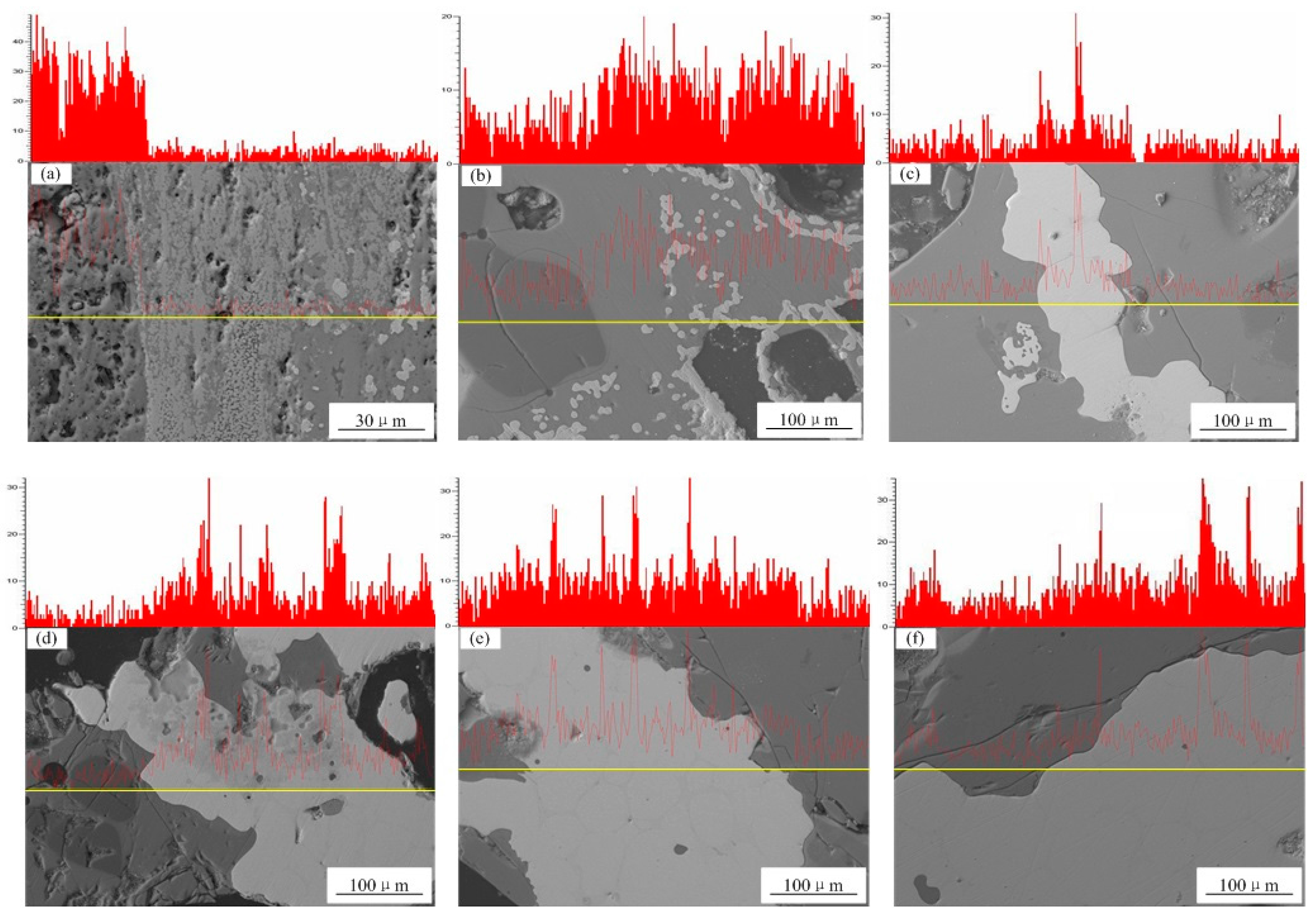
3.3. Migration Behavior of Phosphorus
3.4. Pre-Dephosphorization of Reduced Iron
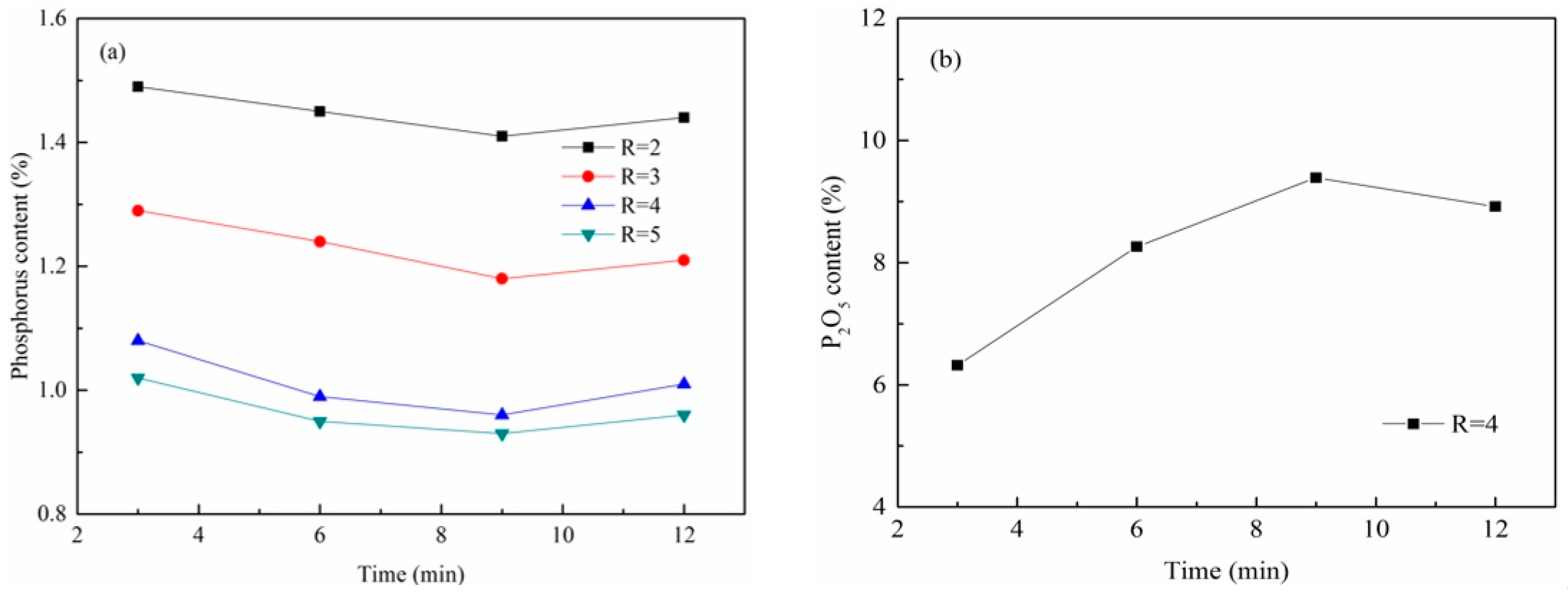
3.4.1. Effect of Basicity
3.4.2. Effect of FetO Content
4. Conclusions
- (1)
- The reduction of apatite to phosphorus and the phosphorus enrichment in reduced iron were enhanced by a higher reduction temperature, longer reduction time, and higher C/O molar ratio. The reduction degree of apatite, phosphorus content, and enrichment ratio of phosphorus in reduced iron were about 84%, 2.4% and 77%, respectively, at 1548 K with a C/O molar ratio 2.5 and reduction time of 60 min.
- (2)
- During the reduction process, the apatite in high phosphorus-containing oolitic hematite ore was reduced to phosphorus and Ca2SiO4 (or Ca(Al2Si2O8) in the presence of SiO2 and Al2O3. Most of the reduced phosphorus was enriched in reduced iron and existed in the form of Fe3P.
- (3)
- Line scanning results showed that phosphorus primarily existed in slag phase at a reduction time less than 10 min. When the reduction time increased to 40 min from 10 min, increased amounts of phosphorus migrated into iron phase from slag phase. The enrichment of phosphorus in reduced iron was negligible at reduction times 50 min and greater.
- (4)
- The phosphorus content of the molten iron decreased with increasing basicity or FetO content of the CaO-based slag. The phosphorus content was decreased below 0.3% at 1873 K with a basicity of 4 and 55% FetO content after 9–12 min dephosphorization. Meanwhile, the P2O5 content of dephosphorization slag increased above 15%, suggesting its use as a phosphate fertilizer. The results of our research indicate that the comprehensive utilization of high phosphorus-containing oolitic hematite ore is possible. It should be noted that the molten iron with less than 0.3% phosphorus is still higher than market specifications. The further dephosphorization of the molten iron will be studied by designing a suitable slag system.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Gao, P.; Sun, Y.S.; Ren, D.Z.; Han, Y.X. Growth of metallic iron particles during coal-based reduction of a rare earths-bearing iron ore. Miner. Metall. Proc. 2013, 30, 74–78. [Google Scholar]
- Li, Y.J.; Sun, Y.S.; Han, Y.X.; Gao, P. Coal-based reduction mechanism of low-grade laterite ore. Trans. Nonferrous Met. Soc. China 2013, 23, 3428–3433. [Google Scholar] [CrossRef]
- Tang, H.Q.; Guo, Z.C.; Zhao, Z.L. Phosphorus removal of high phosphorus iron ore by gas-based reduction and melt separation. J. Iron Steel Res. Int. 2010, 17, 1–6. [Google Scholar] [CrossRef]
- Weissberger, S.; Zimmels, Y.; Lin, I.J. Mechanism of growth of metallic phase in direct reduction of iron bearing oolites. Metall. Mater. Trans. B 1986, 17, 433–442. [Google Scholar] [CrossRef]
- Özdemir, Ö.; Deutsch, E.R. Magnetic properties of oolitic iron ore on Bell Island, Newfoundland. Earth Planet. Sci. Lett. 1984, 69, 427–441. [Google Scholar] [CrossRef]
- Abro, M.I.; Pathan, A.G.; Mallah, A.H. Liberation of oolitic hematite grains from iron ore, Dilband Mines Pakistan. Mehran Univ. Res. J. Eng. Technol. 2011, 30, 329–338. [Google Scholar]
- Sun, Y.S.; Han, Y.X.; Gao, P.; Ren, D.Z. Distribution behavior of phosphorus in the coal-based reduction of high-phosphorus-content oolitic iron ore. Int. J. Min. Met. Mater. 2014, 21, 331–338. [Google Scholar] [CrossRef]
- Wu, J.; Wen, Z.J.; Cen, M.J. Development of technologies for high phosphorus oolitic hematite utilization. Steel Res. Int. 2011, 82, 494–500. [Google Scholar]
- Szekely, J.; Poveromo, J.J. A mathematical and physical representation of the raceway region in the iron blast furnace. Metall. Mater. Trans. B 1975, 6, 119–130. [Google Scholar] [CrossRef]
- Li, G.H.; Zhang, S.H.; Rao, M.J.; Zhang, Y.B.; Jiang, T. Effects of sodium salts on reduction roasting and Fe-P separation of high-phosphorus oolitic hematite ore. Int. J. Miner. Process. 2013, 124, 26–34. [Google Scholar] [CrossRef]
- Nunes, A.P.L.; Pinto, C.L.L.; Valadāo, G.E.S.; Viana, P.R.M. Floatability studies of wavellite and preliminary results on phosphorus removal from a Brazilian iron ore by froth flotation. Miner. Eng. 2012, 39, 206–212. [Google Scholar] [CrossRef]
- Yu, K.P.; Yu, Y.F.; Xu, X.Y. Separation behavior and mechanism of hematite and collophane in the presence of collector RFP-138. Trans. Nonferrous Met. Soc. China 2013, 23, 501–507. [Google Scholar] [CrossRef]
- Xia, W.T.; Ren, Z.D.; Gao, Y.F. Removal of phosphorus from high phosphorus iron ores by selective HCl leaching method. J. Iron Steel Res. Int. 2011, 18, 1–4. [Google Scholar] [CrossRef]
- Muhammed, M.; Zhang, Y. A hydrometallurgical process for the dephosphorization of iron ore. Hydrometallurgy 1989, 21, 277–292. [Google Scholar] [CrossRef]
- Zhang, Y.; Muhammed, M. The removal of phosphorus from iron ore by leaching with nitric acid. Hydrometallurgy 1989, 21, 255–275. [Google Scholar] [CrossRef]
- Ionkov, K.; Gaydardzhiev, S.; de Araujo, A.C.; Bastin, D.; Lacoste, M. Amenability for processing of oolitic iron ore concentrate for phosphorus removal. Miner. Eng. 2013, 46–47, 119–127. [Google Scholar] [CrossRef]
- Wang, J.C.; Shen, S.B.; Kang, J.H.; Li, H.X.; Guo, Z.C. Effect of ore solid concentration on the bioleaching of phosphorus from high-phosphorus iron ores using indigenous sulfur-oxidizing bacteria from municipal wastewater. Process Biochem. 2010, 45, 1624–1631. [Google Scholar]
- Delvasto, P.; Ballester, A.; Muñoz, J.A.; González, F.; Blázquez, M.L.; Igual, J.M.; Valverde, A.; García-Balboa, C. Mobilization of phosphorus from iron ore by the bacterium Burkholderia caribensis FeGL03. Miner. Eng. 2009, 22, 1–9. [Google Scholar] [CrossRef]
- Delvasto, P.; Valverde, A.; Ballester, A.; Muñoz, J.A.; González, F.; Blázquez, M.L.; Igual, J.M.; García-Balboa, C. Diversity and activity of phosphate bioleaching bacteria from a high-phosphorus iron ore. Hydrometallurgy 2008, 92, 124–129. [Google Scholar] [CrossRef]
- Yu, Y.F.; Qi, C.Y. Magnetizing roasting mechanism and effective ore dressing process for oolitic hematite ore. J. Wuhan Univ. Technol-Mat. Sci. 2011, 26, 177–182. [Google Scholar] [CrossRef]
- Yu, W.; Sun, T.C.; Liu, Z.Z.; Kou, J.; Xu, C.Y. The effects of particle sizes of iron ore and coal on the strength and reduction of high phosphorus oolitic hematite coal composite briquettes. ISIJ Int. 2014, 54, 56–62. [Google Scholar] [CrossRef]
- Sun, Y.S.; Gao, P.; Han, Y.X.; Ren, D.Z. Reaction behavior of iron minerals and metallic iron particles growth in coal-based reduction of an oolitic iron ore. Ind. Eng. Chem. Res. 2013, 52, 2323–2329. [Google Scholar] [CrossRef]
- Li, K.Q.; Ni, W.; Zhu, M.; Zheng, M.J.; Li, Y. Iron extraction from oolitic iron ore by a deep reduction process. J. Iron Steel Res. Int. 2011, 18, 9–13. [Google Scholar] [CrossRef]
- Sun, Y.S.; Han, Y.X.; Gao, P.; Wang, Z.H.; Ren, D.Z. Recovery of iron from high phosphorus oolitic iron ore using coal-based reduction followed by magnetic separation. Int. J. Miner. Metall. Mater. 2013, 20, 411–419. [Google Scholar] [CrossRef]
- Li, Y.L.; Sun, T.C.; Zou, A.H.; Xu, C.Y. Effect of coal levels during direct reduction roasting of high phosphorus oolitic hematite ore in a tunnel kiln. Int. J. Min. Sci. Technol. 2012, 22, 323–328. [Google Scholar] [CrossRef]
- Manning, C.P.; Fruehan, R.J. The behavior of phosphorus in direct-reduced iron and hot briquetted iron. Iron Steelmak. 2003, 30, 62–72. [Google Scholar]
- Xu, C.Y.; Sun, T.C.; Kou, J.; Li, Y.L.; Mo, X.L.; Tang, L.G. Mechanism of phosphorus removal in beneficiation of high phosphorous oolitic hematite by direct reduction roasting with dephosphorization agent. Trans. Nonferrous Met. Soc. China 2012, 22, 2806–2812. [Google Scholar] [CrossRef]
- Yu, W.; Sun, T.C.; Kou, J.; Wei, Y.X.; Xu, C.Y.; Liu, Z.Z. The Function of Ca(OH)2 and Na2CO3 as additive on the reduction of high-phosphorus oolitic hematite-coal mixed pellets. ISIJ Int. 2013, 53, 427–433. [Google Scholar] [CrossRef]
- Tian, Z.H.; Li, B.H.; Zhang, X.M.; Jiang, Z.H. Double slag operation dephosphorization in BOF for producing low phosphorus steel. J. Iron Steel Res. Int. 2009, 16, 6–14. [Google Scholar] [CrossRef]
- Diao, J.; Xie, B.; Wang, Y.H.; Guo, X. Recovery of phosphorus from dephosphorization slag produced by duplex high phosphorus hot metal refining. ISIJ Int. 2012, 52, 955–959. [Google Scholar] [CrossRef]
- Han, Y.X.; Ren, D.Z.; Sun, Y.S.; Gao, P. Phosphorus migration behaviors in reduction of high phosphorus oolitic hematite ore. Iron Steel 2013, 48, 7–11. [Google Scholar]
- Elias, M.; Yasushi, S.; Mitsutaka, H. Phosphorus gasification from sewage sludge during carbothermic reduction. ISIJ Int. 2008, 48, 912–917. [Google Scholar]
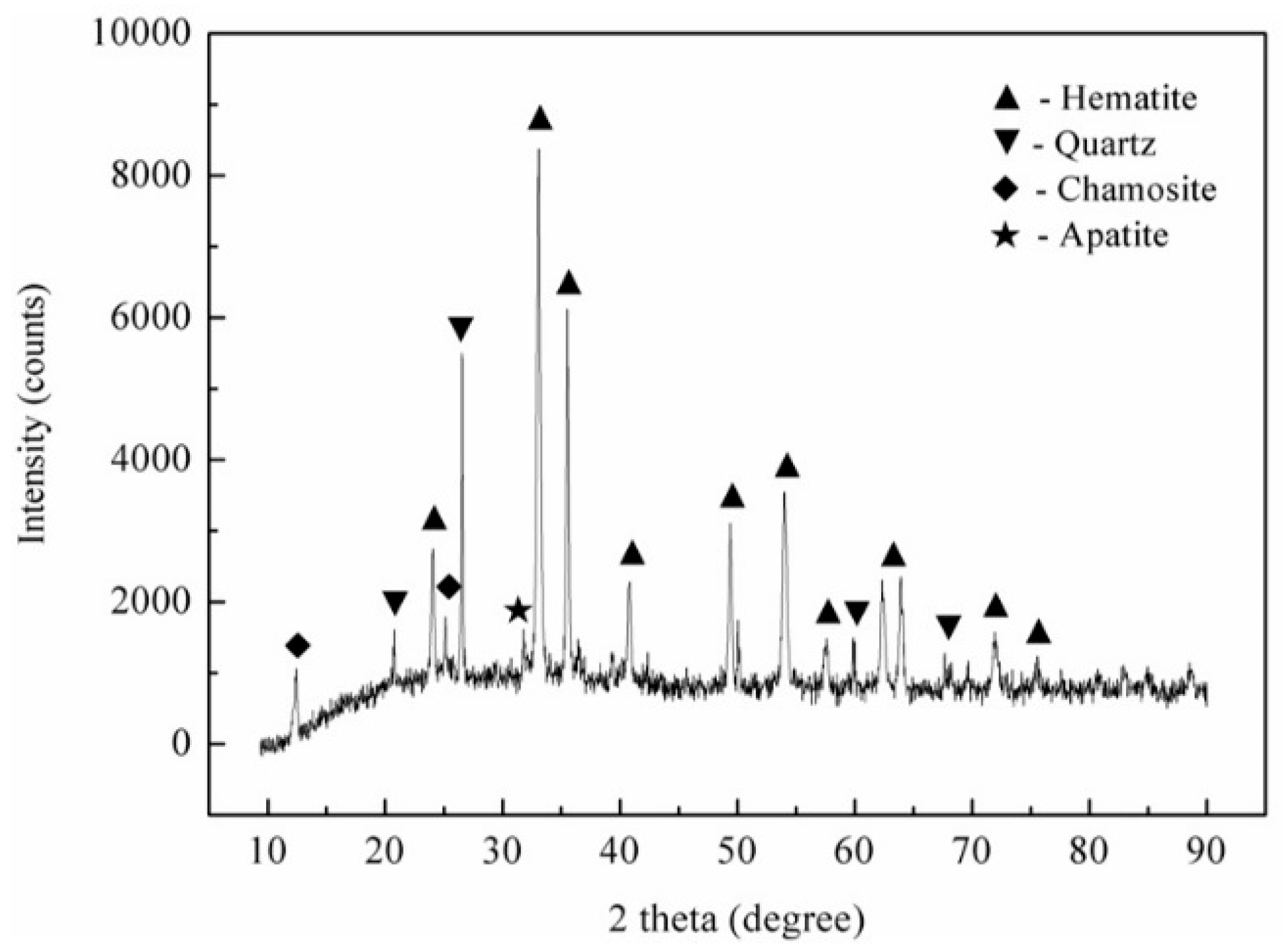





| Constituents | TFe | FeO | SiO2 | Al2O3 | CaO | MgO | P | S | TiO2 | K | Mn |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Content | 42.21 | 4.31 | 21.80 | 5.47 | 4.33 | 0.59 | 1.31 | 0.13 | 0.19 | 0.41 | 0.20 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gao, P.; Li, G.-F.; Han, Y.-X.; Sun, Y.-S. Reaction Behavior of Phosphorus in Coal-Based Reduction of an Oolitic Hematite Ore and Pre-Dephosphorization of Reduced Iron. Metals 2016, 6, 82. https://doi.org/10.3390/met6040082
Gao P, Li G-F, Han Y-X, Sun Y-S. Reaction Behavior of Phosphorus in Coal-Based Reduction of an Oolitic Hematite Ore and Pre-Dephosphorization of Reduced Iron. Metals. 2016; 6(4):82. https://doi.org/10.3390/met6040082
Chicago/Turabian StyleGao, Peng, Guo-Feng Li, Yue-Xin Han, and Yong-Sheng Sun. 2016. "Reaction Behavior of Phosphorus in Coal-Based Reduction of an Oolitic Hematite Ore and Pre-Dephosphorization of Reduced Iron" Metals 6, no. 4: 82. https://doi.org/10.3390/met6040082





