Expression of Resistance in Amaranthus spp. (Caryophyllales: Amaranthaceae): Effects of Selected Accessions on the Behaviour and Biology of the Amaranth Leaf-Webber, Spoladea recurvalis (Lepidoptera: Crambidae)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Amaranth Accessions
2.2. Amaranth Leaf-Webber (Spoladea recurvalis F.) Colony
2.3. Choice Bioassay
2.4. No-Choice Bioassay
2.5. Effect of Accession on Weight Gain of Amaranth Leaf-Webber
2.6. Effects of Selected Amaranth Accessions on the Development of Amaranth Leaf-Webber and Adult Longevity
2.7. Data Analysis
3. Results
3.1. Morphological Characteristics of Amaranth Accessions
3.2. Oviposition by S. recurvalis in Choice Situation
3.3. Oviposition by S. recurvalis in No-Choice Condition
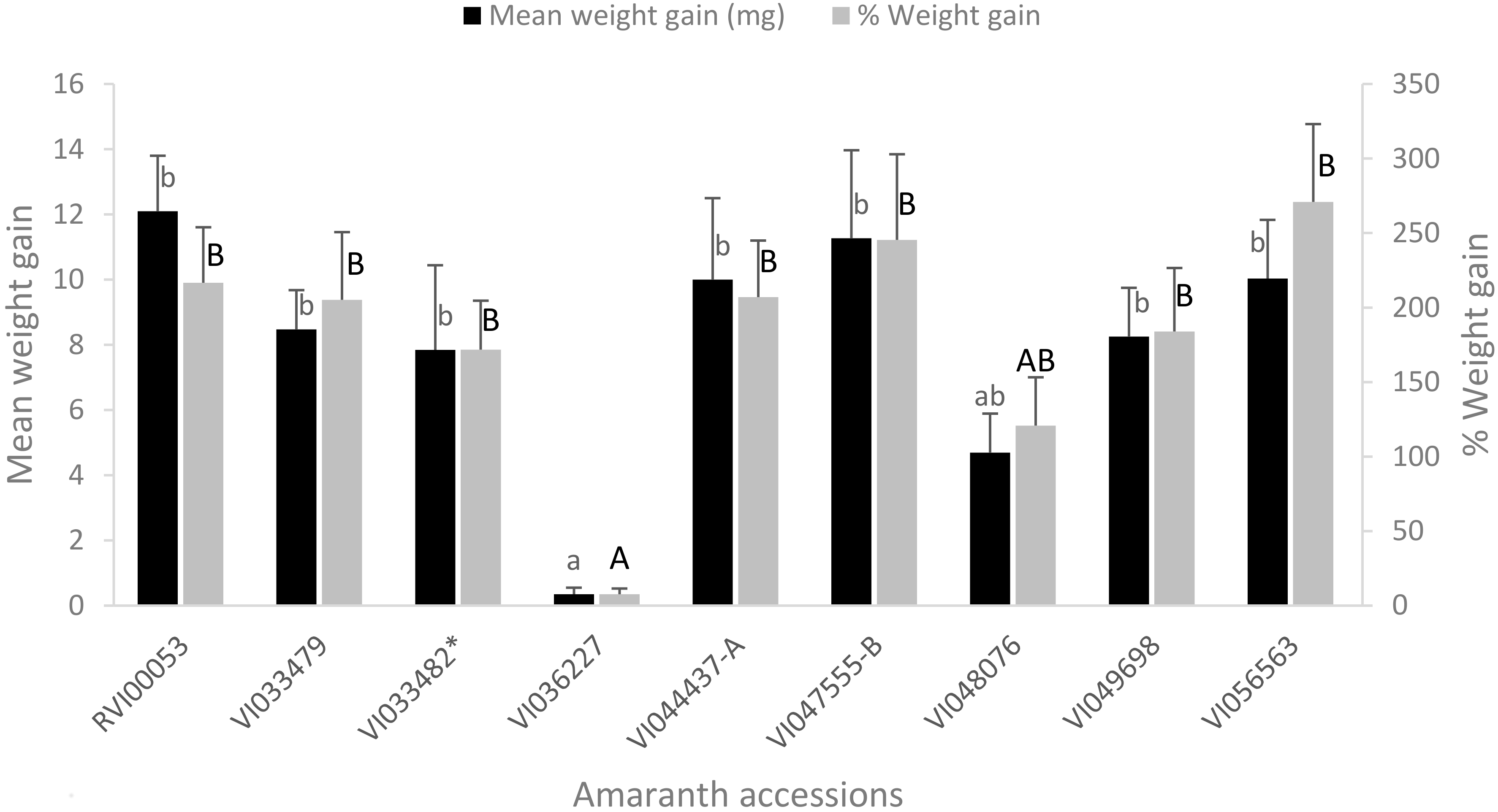
3.4. Weight Gain by Larvae of S. recurvalis after 48 h of Feeding on the Selected Amaranth Accessions
3.5. Development Time of S. recurvalis on the Selected Amaranth Accessions
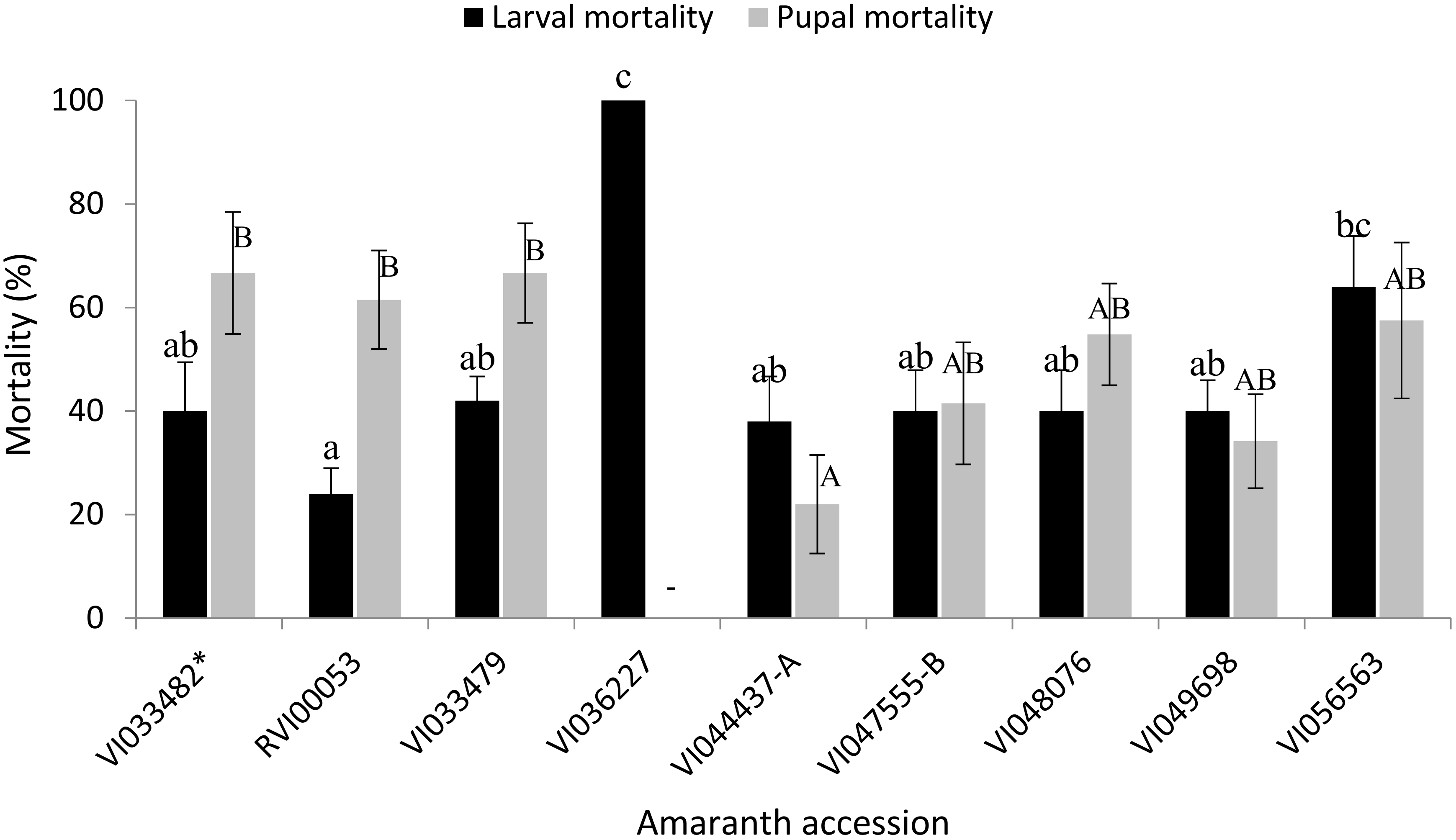
3.6. Mortality Rates and Instantaneous Rate of Increase of S. recurvalis on Selected Amaranth Accessions
3.7. Adult Longevity, Fecundity, Egg Viability and Sex Ratios of S. recurvalis
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Amicarelli, V.; Camaggio, G. Amaranthus: A crop to rediscover. Forum Ware Int. 2012, 2, 4–11. [Google Scholar]
- Ezeh, A.; Ogedegbe, A.; Ogedegbe, S. Insect pest occurrence on cultivated Amaranthus spp. in Benin city, Edo state, Nigeria. J. Appl. Sci. Environ. Manag. 2015, 19, 335–339. [Google Scholar] [CrossRef]
- Dinssa, F.; Hanson, P.; Dubois, T.; Tenkouano, A.; Stoilova, T.; Hughes, J.; Keating, J. AVRDC—The world vegetable center’s women-oriented improvement and development strategy for traditional African vegetables in sub-saharan Africa. Eur. J. Hortic. Sci. 2016, 81, 91–105. [Google Scholar] [CrossRef]
- Jeyasankar, A.; Gokilamani, D. Biology and eco-friendly control of amaranths pests, Hymenia recurvalis Fabricius and Psara basalis Walker (Lepidoptera: Crambidae). Int. J. Acad. Stud. 2016, 2, 218–230. [Google Scholar]
- Adebooye, O.; Opabode, J. Status of conservation of the indigenous leaf vegetables and fruits of Africa. Afr. J. Biotechnol. 2004, 3, 700–705. [Google Scholar]
- Costea, M.; Weaver, S.E.; Tardif, F.J. The biology of Canadian weeds. 130. Amaranthus retroflexus L., A. powellii S. Watson and A. hybridus L. Can. J. Plant Sci. 2004, 84, 631–668. [Google Scholar] [CrossRef]
- Niveyro, S.L.; Mortensen, A.G.; Fomsgaard, I.S.; Salvo, A. Differences among five amaranth varieties (Amaranthus spp.) regarding secondary metabolites and foliar herbivory by chewing insects in the field. Arthropod-Plant Interact. 2013, 7, 235–245. [Google Scholar] [CrossRef]
- Ouma, M. Indigenous Vegetable Production and Utilization in Suba District, Kenya: Improving Health, Unlocking the Wealth in Suba District. Bio Vision-TTU, ICIPE 2004. Available online: http://www.infonet-biovision.org/sites/default/files/indigenous_veg_production_suba_district_ouma.pdf (accessed on 5 January 2018).
- Gikonyo, N.; Mburu, M.; Kenji, G.; Mwasaru, A. Properties of a complementary food based on amaranth grain (Amaranthus cruentus) grown in Kenya. J. Agric. Food Tech. 2011, 1, 153–178. [Google Scholar]
- Liu, F.; Stützel, H. Leaf expansion, stomatal conductance, and transpiration of vegetable amaranth (Amaranthus sp.) in response to soil drying. J. Am. Soc. Hortic. Sci. 2002, 127, 878–883. [Google Scholar]
- Aderolu, I.; Omooloye, A.; Okelana, F. Occurrence, abundance and control of the major insect pests associated with amaranths in Ibadan, Nigeria. Entomol. Ornithol. Herpetol. 2013, 2, 112. [Google Scholar] [CrossRef]
- James, B.; Atcha-Ahowé, C.; Godonou, I.; Baimey, H.; Goergen, H.; Sikirou, R.; Toko, M. Integrated Pest Management in Vegetable Production: A Guide for Extension Workers in West Africa; IITA: Ibadan, Nigeria, 2010. [Google Scholar]
- Kagali, R.N.; Kioko, E.N.; Osiemo, Z.; Muya, S.; Wachera, C. Insect abundance and diversity on cultivated Amaranthus spp. (amaranthacea) in Meru county, Kenya. Am. Int. J. Contemp. Res. 2013, 3, 110–116. [Google Scholar]
- Mureithi, D.; Mworia, J.; Meyhöfer, R.; Murungi, L.; Losenge, T.; Akutse, K.; Ekesi, S.; Fiaboe, K. Survey for pest and natural enemies of amaranth and African nightshades in Kenya and Tanzania. In Proceedings of the TROPENTAG 2015, Humboldt-Universität, Berlin, Germany, 16–18 September 2015. [Google Scholar]
- Sharma, G.; Ramamurthy, V. A Checklist of Lepidopterous Pests of Vegetables in INDIA. 2009. Available online: www.zsi.gov.in/zoological-survey-of-india/zsi-data/checklist/index.htm (accessed on 1 February 2018).
- Clarke-Harris, D.; Fleischer, S.; Fuller, C.; Bolton, J. Evaluation of the efficacy of new chemistries for controlling major Lepidoptera pests on vegetable amaranth in Jamaica. CARDI Rev. 2004, 4, 12–19. [Google Scholar]
- García, A.A.; Huato, M.Á.D.; Lara, M.H.; Sáenz-de-Cabezón, F.J.; Pérez-Moreno, I.; Marco-Mancebón, V.; López-Olguín, J.F. Insect occurrence and losses due to phytophagous species in the amaranth Amaranthus hypocondriacus L. crop in Puebla, Mexico. Afr. J. Agric. Res. 2011, 6, 5924–5929. [Google Scholar]
- Niveyro, S.; Salvo, A. Taxonomic and functional structure of phytophagous insect communities associated with grain amaranth. Neotrop. Entomol. 2014, 43, 532–540. [Google Scholar] [CrossRef] [PubMed]
- Batra, H.; Bhattacherjee, N. Occurrence of Hymenia recurvalis (Fabricius) (Lepidoptera: Pyalidae) as a bad pest of some leaf vegetables. Indian J. Entomol. 1960, 22, 128–130. [Google Scholar]
- Mureithi, D.M.; Komi, F.K.; Ekesi, S.; Meyhöfer, R. Important arthropod pests on leafy amaranth (Amaranthus viridis, A. tricolor and A. blitum) and broad-leafed African nightshade (Solanum scabrum) with a special focus on host-plant ranges. Afr. J. Hortic. Sci. 2017, 11, 1–17. [Google Scholar]
- Othim, S.; Agbodzavu, K.; Kahuthia-Gathu, R.; Akutse, K.; Muchemi, S.; Ekesi, S.; Fiaboe, K. Performance of Apanteles hemara (Hymenoptera: Braconidae) on two amaranth leaf-webbers: Spoladea recurvalis and Udea ferrugalis (Lepidoptera: Crambidae). Environ. Entomol. 2017, 46, 1284–1291. [Google Scholar] [CrossRef] [PubMed]
- Othim, S.T.; Kahuthia-Gathu, R.; Akutse, K.; Foba, C.; Fiaboe, K. Seasonal occurrence of amaranth lepidopteran defoliators and effect of attractants and amaranth lines in their management. J. Appl. Entomol. 2018. [Google Scholar] [CrossRef]
- Clarke-Harris, D.; Fleischer, S.; Fender, A. Major pests of callaloo. In Identification Guide; The Pennsylvania State University: State College, PA, USA, 1998. [Google Scholar]
- Grovida, F. Vegetable Pests. 2015. Available online: http://www.grovida.us/vegetable-pests.html (accessed on 26 May 2015).
- Arivudainambi, S.; Selvamuthukumaran, T.; Baskaran, P. Efficacy of herbal extracts in management of amaranth leaf caterpillar. Int. J. Veg. Sci. 2010, 16, 167–173. [Google Scholar] [CrossRef]
- Chahal, K.; Singh, B.; Kang, B.; Battu, R.; Joia, B. Insecticide residues in farmgate vegetable samples in Punjab. Pestic. Res. J. 1997, 9, 256–260. [Google Scholar]
- Gitonga, Z.M.; Chabi-Olaye, A.; Mithöfer, D.; Okello, J.J.; Ritho, C.N. Control of invasive Liriomyza leafminer species and compliance with food safety standards by small scale snow pea farmers in Kenya. Crop Prot. 2010, 29, 1472–1477. [Google Scholar] [CrossRef]
- Srinivasan, R. Integrating biopesticides in pest management strategies for tropical vegetable production. J. Biopestic. 2012, 5, 36–45. [Google Scholar]
- Landolt, P.; Jang, E.; Carvalho, L.; Pogue, M. Attraction of pest moths (Lepidoptera: Noctuidae, Crambidae) to floral lures on the island of hawaii. Proc. Hawaii. Entomol. Soc. 2011, 43, 49–58. [Google Scholar]
- Othim, S.T.; Srinivasan, R.; Kahuthia-Gathu, R.; Dubois, T.; Dinssa, F.; Ekesi, S.; Fiaboe, K. Screening for pest resistance against important lepidopteran and stem weevil pests attacking amaranth in Tanzania. Euphytica 2018. under review. [Google Scholar]
- Stark, J.D.; Banks, J.E. Population-level effects of pesticides and other toxicants on arthropods. Ann. Rev. Entomol. 2003, 48, 505–519. [Google Scholar] [CrossRef] [PubMed]
- Gatehouse, J.A. Plant resistance towards insect herbivores: A dynamic interaction. New Phytol. 2002, 156, 145–169. [Google Scholar] [CrossRef]
- Kumar, S.; Atri, C.; Sangha, M.K.; Banga, S. Screening of wild crucifers for resistance to mustard aphid, lipaphis erysimi (kaltenbach) and attempt at introgression of resistance gene (s) from Brassica fruticulosa to Brassica juncea. Euphytica 2011, 179, 461–470. [Google Scholar] [CrossRef]
- Akaneme, F.; Ani, G. Morphological assessment of genetic variability among accessions of amaranthus hybridus. World Appl. Sci. J. 2013, 28, 568–577. [Google Scholar]
- Jared, J.J.; Murungi, L.K.; Wesonga, J.; Torto, B. Steroidal glycoalkaloids: Chemical defence of edible african nightshades against the tomato red spider mite, tetranychus evansi (acari: Tetranychidae). Pest Manag. Sci. 2016, 72, 828–836. [Google Scholar] [CrossRef] [PubMed]
- Jiang, C.-X.; Wright, R.; Woo, S.; DelMonte, T.; Paterson, A. Qtl analysis of leaf morphology in tetraploid gossypium (cotton). Theor. Appl. Genet. 2000, 100, 409–418. [Google Scholar] [CrossRef]
- War, A.R.; Paulraj, M.G.; Ahmad, T.; Buhroo, A.A.; Hussain, B.; Ignacimuthu, S.; Sharma, H.C. Mechanisms of plant defense against insect herbivores. Plant Signal. Behav. 2012, 7, 1306–1320. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hillier, N.; Dixon, P.; Larson, D. Trap captures of male grapholita libertina (lepidoptera: Tortricidae) moths: Relationship to larval numbers and damage in wild lingonberry1. Environ. Entomol. 2004, 33, 405–417. [Google Scholar] [CrossRef]
- Martínez, G.; Soler, R.; Dicke, M. Behavioral ecology of oviposition-site selection in herbivorous true bugs. In Advances in the Study of Behavior, 1st ed.; Jane-Brockmann, H., Roper, T.J., Naguib, M., Barrett, L., Eds.; Elsevier: New York, NY, USA, 2013; Volume 45, pp. 175–207. ISBN 9780124071865. [Google Scholar]
- Jaenike, J. Host specialization in phytophagous insects. Annu. Rev. Ecol. Syst. 1990, 21, 243–273. [Google Scholar] [CrossRef]
- Mardani-Talaei, M.; Nouri-Ganbalani, G.; Naseri, B.; Hassanpour, M. Life history studies of the beet armyworm, Spodoptera exigua (Hübner) (Lepidoptera: Noctuidae) on 10 corn hybrids. J. Entomol. Res. Soc. 2012, 14, 9–18. [Google Scholar]
- Seham, S.; Mostafa, F.; Aly, F.; Hussein, S. Certain biological aspects, threshold of development and thermal units for Hymenia recurvalis (Fab.), (Lepidoptera: Pyrallidae). Arab Univ. J. Agric. Sci. 2006, 14, 447–456. [Google Scholar]
- Bhattacherjee, N.; Ramdas, M. Bionomics, biology and control of Hymenia recurvalis (Fabricius)(Pyralidae: Lepidoptera). Indian J. Entomol. 1964, 26, 176–183. [Google Scholar]
- Simmonds, M.S. Flavonoid-insect interactions: Recent advances in our knowledge. Phytochemistry 2003, 64, 21–30. [Google Scholar] [CrossRef]
- Summers, C.B.; Felton, G.W. Prooxidant effects of phenolic acids on the generalist herbivore Helicoverpa zea (Lepidoptera: Noctuidae): Potential mode of action for phenolic compounds in plant anti-herbivore chemistry. Insect Biochem. Mol. Biol. 1994, 24, 943–953. [Google Scholar] [CrossRef]
- Liu, Z.; Li, D.; Gong, P.; Wu, K. Life table studies of the cotton bollworm, Helicoverpa armigera (Hübner)(Lepidoptera: Noctuidae), on different host plants. Environ. Entomol. 2004, 33, 1570–1576. [Google Scholar] [CrossRef]
- Hsu, Y.-C.; Srinivasan, R. Desert horse purslane weed as an alternative host for amaranth leaf webber, Hymenia recurvalis in Taiwan. Formos. Entomol. 2012, 32, 297–302. [Google Scholar]
- Pande, Y. Some observations on the bionomics of Hymenia recurvalis F. (Lepid., Pyralidae) feeding on Trianthema monogyna and Amaranthus viridis in India. J. Appl. Entomol. 1972, 72, 362–366. [Google Scholar] [CrossRef]
- Shirai, Y. Flight activity, reproduction, and adult nutrition of the beet webworm, Spoladea recurvalis (Lepidoptera: Pyralidae). Appl. Entomol. Zool. 2006, 41, 405–414. [Google Scholar] [CrossRef]
- Cortesero, A.; Stapel, J.; Lewis, W. Understanding and manipulating plant attributes to enhance biological control. Biol. Control 2000, 17, 35–49. [Google Scholar] [CrossRef]
- Pappas, M.L.; Broekgaarden, C.; Broufas, G.D.; Kant, M.R.; Messelink, G.J.; Steppuhn, A.; Wäckers, F.; van Dam, N.M. Induced plant defences in biological control of arthropod pests: A double-edged sword. Pest Manag. Sci. 2017, 73, 1780–1788. [Google Scholar] [CrossRef] [PubMed]

| Gene Bank Code | Species | Type | Leaf Colour | Leaf Shape | Country of Origin | Number of Branches per Plant (mean) | Plant Height (mean) cm | Leaf Width (mean) cm | Leaf Length (mean) cm | Petiole Length (mean) cm |
|---|---|---|---|---|---|---|---|---|---|---|
| VI033482 * | A. tricolor L. | Accession | Green | Reniform | Malaysia | 9.0 | 100.9 | 10.6 | 19.3 | 5.0 |
| RVI00002 | A. cruentus L. | Line | Green | Ovate | Zambia | 12.5 | 122.6 | 6.6 | 16.5 | 10.2 |
| RVI00005 | A. dubius Mart. | Line | Light reddish | Ovate | Tanzania | 12.2 | 140.3 | 6.0 | 12.5 | 7.6 |
| RVI00027 | Amaranthus sp. 1 | Line | Green | Ovate | Malawi | 7.3 | 96.3 | 6.2 | 11.1 | 7.8 |
| RVI00053 | A. dubius Mart. | Line | Green | Ovate | Uganda | 11.0 | 167.0 | 8.7 | 15.6 | 8.0 |
| VI033477 | Amaranthus sp. 2 | Accession | Reddish | Ovate | Malaysia | 9.2 | 99.7 | 7.9 | 12.3 | 5.2 |
| VI033479 | Amaranthus sp. 3 | Accession | Green | Ovate | Malaysia | 11.3 | 100.8 | 4.9 | 7.6 | 4.2 |
| VI033487 | A. cruentus L. | Accession | Green | Reniform | Malaysia | 13.5 | 128.7 | 5.6 | 7.7 | 6.6 |
| VI036225 | A. graecizans L. | Accession | Green | Ovate | Hungary | 15.4 | 77.2 | 1.6 | 3.2 | 2.3 |
| VI036227 | A. blitoides Watson | Accession | Green | Oblanceolate | Hungary | 15.8 | 67.4 | 1.2 | 3.2 | 1.6 |
| VI044367 | A. cruentus L. | Accession | Green | Lanceolate | Tanzania | 9.1 | 123.5 | 5.8 | 13.7 | 10.2 |
| VI044369 | A. hypochondriacus L. | Accession | Green | Lanceolate | Ghana | 13.7 | 129.0 | 6.1 | 17.0 | 9.2 |
| VI044388 | A. graecizans L. | Accession | Green | Oblanceolate | India | 14.6 | 89.8 | 2.3 | 4.3 | 2.5 |
| VI044432 | A. viridis L. | Accession | Green | Cordate | Indonesia | 11.0 | 102.9 | 4.5 | 6.9 | 4.0 |
| VI044437-A | A. cruentus L. | Accession | Green | Lanceolate | Malaysia | 11.5 | 89.5 | 5.5 | 13.0 | 7.2 |
| VI044473 | A. palmeri Watson | Accession | Green | Obovate | Senegal | 9.0 | 80.1 | 2.2 | 4.4 | 2.4 |
| VI046233-A | Amaranthus sp. 4 | Accession | Reddish | Lanceolate | Vietnam | 8.0 | 142.1 | 6.8 | 17.0 | 10.6 |
| VI047517-B | A. tricolor L. | Accession | Green | Ovate | Bangladesh | 12.9 | 119.8 | 8.1 | 15.9 | 7.7 |
| VI047555-B | A. tricolor L. | Accession | Green | Lanceolate | Vietnam | 10.9 | 135.6 | 4.5 | 13.4 | 4.8 |
| VI048076 | A. tricolor L. | Accession | Green | Cordate | Bangladesh | 13.1 | 130.1 | 8.0 | 13.5 | 7.1 |
| VI048864-A | A. viridis L. | Accession | Green | Cordate | Thailand | 10.2 | 95.1 | 4.1 | 5.8 | 3.6 |
| VI048919 | Amaranthus sp. 5 | Accession | Green | Ovate | Thailand | 11.6 | 126.1 | 3.7 | 7.0 | 4.3 |
| VI049242 | Amaranthus sp. 6 | Accession | Green | Ovate | Thailand | 11.6 | 87.8 | 4.4 | 5.8 | 3.7 |
| VI049502 | Amaranthus sp. 7 | Accession | Green | Cordate | Thailand | 10.0 | 103.1 | 4.7 | 6.8 | 4.1 |
| VI049504 | Amaranthus sp. 8 | Accession | Green | Lanceolate | Thailand | 12.1 | 134.2 | 3.0 | 6.6 | 3.2 |
| VI049530 | Amaranthus sp. 9 | Accession | Green | Ovate | Thailand | 10.4 | 89.2 | 4.3 | 6.4 | 3.9 |
| VI049639 | A. viridis L. | Accession | Green | Ovate | Thailand | 11.3 | 91.7 | 4.2 | 6.1 | 3.5 |
| VI049698 | A. viridis L. | Accession | Green | Ovate | Thailand | 12.4 | 100.5 | 3.9 | 5.5 | 3.4 |
| VI050609-A | A. tricolor L. | Accession | Variegated | Cordate | Vietnam | 9.8 | 129.1 | 9.2 | 11.7 | 6.8 |
| VI050609-B | A. tricolor L | Accession | Variegated | Ovate | Vietnam | 8.5 | 140.9 | 9.6 | 15.0 | 6.1 |
| VI054569 | A. gracilis Desf. | Accession | Green | Ovate | Philippines | 11.0 | 95.1 | 4.5 | 7.2 | 3.9 |
| VI054798 | Amaranthus sp. 10 | Accession | Green | Ovate | Lao PDR | 12.4 | 89.3 | 4.1 | 6.2 | 3.3 |
| VI055127 | A. viridis L. | Accession | Green | Ovate | Malaysia | 11.4 | 108.1 | 5.6 | 10.3 | 6.6 |
| VI055128 | A. viridis L. | Accession | Green | Cordate | Malaysia | 10.7 | 123.4 | 5.0 | 7.0 | 3.9 |
| VI055135 | A. viridis L | Accession | Green | Cordate | Malaysia | 10.8 | 92.0 | 5.0 | 7.2 | 4.1 |
| VI056563 | Amaranthus sp. 11 | Accession | Reddish | Ovate | Bangladesh | 9.7 | 136.9 | 9.0 | 17.1 | 8.3 |
| Mean | 11.2 | 110.7 | 5.5 | 10.0 | 5.5 | |||||
| p-value | 0.053 | <0.001 | <0.001 | <0.001 | <0.001 | |||||
| LSD (5%) | 4.38 | 26.74 | 1.4 | 3.69 | 2.33 |
| Set-up A | Set-up B | ||||
|---|---|---|---|---|---|
| Gene Bank Code | Number of Eggs | Relative Risk | Gene Bank Code | Number of Eggs | Relative Risk |
| VI033482 * | 25.67 ± 7.62a | VI033482* | 29.00 ± 7.95b | ||
| VI033487 | 11.33 ± 4.57b | 0.44 | VI048919 | 40.20 ± 16.41a | 1.39 |
| VI044388 | 10.00 ± 2.92bc | 0.39 | VI050609-B | 34.67 ± 9.47ab | 1.2 |
| VI036227 | 8.83 ± 3.93bcd | 0.34 | VI033477 | 15.00 ± 4.87c | 0.52 |
| RVI00027 | 7.33 ± 2.85be | 0.29 | VI049504 | 11.40 ± 6.21cd | 0.39 |
| RVI00005 | 6.17 ± 2.30cf | 0.24 | VI047517-B | 9.83 ± 4.48cde | 0.34 |
| VI048076 | 6.17 ± 1.64cf | 0.24 | VI056563 | 9.83 ± 3.51cde | 0.34 |
| VI049639 | 4.33 ± 1.74def | 0.17 | VI055127 | 6.00 ± 3.04def | 0.21 |
| RVI00002 | 4.00 ± 2.08ef | 0.16 | VI046233-A | 6.17 ± 3.82def | 0.21 |
| RVI00053 | 3.83 ± 1.70ef | 0.15 | VI049530 | 6.67 ± 3.48def | 0.23 |
| VI036225 | 3.67 ± 2.01ef | 0.14 | VI050609-A | 6.67 ± 2.89def | 0.23 |
| VI044473 | 3.33 ± 1.54ef | 0.13 | VI047555-B | 3.67 ± 1.87ef | 0.13 |
| VI044369 | 3.20 ± 2.03ef | 0.12 | VI055128 | 3.50 ± 2.05ef | 0.12 |
| VI044367 | 2.83 ± 0.83ef | 0.11 | VI054798 | 2.83 ± 2.46f | 0.1 |
| VI044437-A | 2.40 ± 1.03ef | 0.09 | VI055135 | 2.50 ± 0.92f | 0.09 |
| VI049698 | 2.33 ± 0.56f | 0.09 | VI048864-A | 2.33 ± 1.76f | 0.08 |
| VI044432 | 1.50 ± 0.56f | 0.06 | VI033479 | 2.17 ± 1.78f | 0.07 |
| VI054569 | 1.50 ± 0.85f | 0.06 | VI049242 | 2.17 ± 1.17f | 0.07 |
| VI049502 | 1.50 ± 0.81f | 0.05 | |||
| Gene Bank Code | No Choice | Relative Risk | Choice | Relative Risk | χ2 | df | p Value |
|---|---|---|---|---|---|---|---|
| VI033482 * | 59.63 ± 10.49eA | - | 21.58 ± 5.30dB | - | 127.96 | 1 | <0.001 |
| RVI00053 | 30.00 ± 6.52bcA | 0.50 | 3.83 ± 1.70abB | 0.18 | 183.54 | 1 | <0.001 |
| VI036227 | 40.20 ± 10.24cdA | 0.67 | 8.83 ± 3.93cB | 0.41 | 170.15 | 1 | <0.001 |
| VI044437-A | 29.33 ± 10.24bA | 0.49 | 2.40 ± 1.03abB | 0.11 | 282.65 | 1 | <0.001 |
| VI047555-B | 29.40 ± 10.27bcA | 0.49 | 3.67 ± 1.87abB | 0.17 | 181.96 | 1 | <0.001 |
| VI048076 | 18.50 ± 6.63aA | 0.31 | 6.17 ± 1.64bcB | 0.29 | 72.01 | 1 | <0.001 |
| VI049698 | 42.80 ± 18.14dA | 0.72 | 2.33 ± 0.56aB | 0.11 | 347.38 | 1 | <0.001 |
| VI056563 | 37.20 ± 5.54bcdA | 0.62 | 9.83 ± 3.51cB | 0.46 | 129.6 | 1 | <0.001 |
| χ2 =192.75 | χ2 =281.29 | ||||||
| df = 7, 37 | df = 7, 45 | ||||||
| p < 0.001 | p < 0.001 |
| Gene Bank Code | Larval Development Time | Pupal Development Time | Total Development Time | Early Stage Larval Mortality (%) |
|---|---|---|---|---|
| VI033482 * | 13.43 ± 0.30a | 7.45 ± 0.69a | 20.00 ± 0.8a | 8.0 ± 3.27bc |
| RVI00053 | 13.45 ± 0.37a | 6.60 ± 0.34a | 18.60 ± 0.45a | 4.0 ± 0.51c |
| VI033479 | 13.24 ± 0.23a | 6.11 ± 0.35a | 18.67 ± 0.44a | 8.0 ± 3.27bc |
| VI036227 | NA | NA | NA | 100.0 ± 0.0a |
| VI044437-A | 13.29 ± 0.29a | 5.86 ± 0.17a | 18.82 ± 0.28a | 14.0 ± 5.21bc |
| VI047555-B | 13.77 ± 0.36a | 6.16 ± 0.21a | 19.11 ± 0.24a | 24.0 ± 6.53bc |
| VI048076 | 13.77 ± 0.43a | 6.20 ± 0.43a | 19.00 ± 0.45a | 22.0 ± 7.57bc |
| VI049698 | 13.23 ± 0.30a | 6.43 ± 0.39a | 19.24 ± 0.36a | 12.0 ± 6.11bc |
| VI056563 | 14.00 ± 0.56a | 6.63 ± 0.42a | 19.75 ± 0.53a | 34.0 ± 11.57b |
| χ2 = 1.066 | χ2 = 3.348 | χ2 = 1.042 | F = 12.22 | |
| df = 7, 228 | df = 7, 112 | df = 7, 112 | df = 8, 81 | |
| p = 0.994 | p = 0.851 | p = 0.994 | p < 0.001 |
| Gene Bank Code | Adult Longevity (Days) | Odds Ratio | Egg Viability (%) | Fecundity/Female at 4-d-old | Proportion of Females (%) |
|---|---|---|---|---|---|
| VI047555-B | 8.70 ± 0.61a | 0.9 | 91.25 ± 1.93a | 13.06 ± 1.20ab | 51.79 ± 7.03a |
| VI033482 * | 9.69 ± 0.85ab | 95.49 ± 1.50a | 22.67 ± 3.67a | 59.17 ± 8.86a | |
| VI044437-A | 10.85 ± 0.52b | 1.12 | 97.79 ± 3.13a | 11.09 ± 0.80b | 44.97 ± 8.01a |
| VI033479 | 10.85 ± 0.85b | 1.12 | 97.62 ± 1.61a | 10.97 ± 1.71b | 45.49 ± 4.51a |
| RVI00053 | 11.00 ± 0.79bc | 1.14 | 97.53 ± 1.51a | 13.40 ± 1.74ab | 66.91 ± 8.09a |
| VI049698 | 11.22 ± 0.49bc | 1.16 | 96.25 ± 2.33a | 23.75 ± 3.50a | 46.03 ± 6.31a |
| VI056563 | 12.63 ± 0.82cd | 1.3 | 95.90 ± 4.10a | 14.67 ± 5.67ab | 49.77 ± 5.83a |
| VI048076 | 14.25 ± 0.82d | 1.47 | 94.66 ± 2.59a | 8.50 ± 0.74b | 56.76 ± 13.44a |
| p < 0.001 | p = 0.527 | p = 0.002 | p = 0.638 | ||
| df = 7, 380 | df = 7, 32 | df = 7,14 | df = 7,25 | ||
| χ2 = 92.51 | F = 0.89 | F = 6.07 | F = 0.74 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Othim, S.T.O.; Ramasamy, S.; Kahuthia-Gathu, R.; Dubois, T.; Ekesi, S.; Fiaboe, K.K.M. Expression of Resistance in Amaranthus spp. (Caryophyllales: Amaranthaceae): Effects of Selected Accessions on the Behaviour and Biology of the Amaranth Leaf-Webber, Spoladea recurvalis (Lepidoptera: Crambidae). Insects 2018, 9, 62. https://doi.org/10.3390/insects9020062
Othim STO, Ramasamy S, Kahuthia-Gathu R, Dubois T, Ekesi S, Fiaboe KKM. Expression of Resistance in Amaranthus spp. (Caryophyllales: Amaranthaceae): Effects of Selected Accessions on the Behaviour and Biology of the Amaranth Leaf-Webber, Spoladea recurvalis (Lepidoptera: Crambidae). Insects. 2018; 9(2):62. https://doi.org/10.3390/insects9020062
Chicago/Turabian StyleOthim, Stephen T. O., Srinivasan Ramasamy, Ruth Kahuthia-Gathu, Thomas Dubois, Sunday Ekesi, and Komi K. M. Fiaboe. 2018. "Expression of Resistance in Amaranthus spp. (Caryophyllales: Amaranthaceae): Effects of Selected Accessions on the Behaviour and Biology of the Amaranth Leaf-Webber, Spoladea recurvalis (Lepidoptera: Crambidae)" Insects 9, no. 2: 62. https://doi.org/10.3390/insects9020062






