Environment vs. Plant Ontogeny: Arthropod Herbivory Patterns on European Beech Leaves along the Vertical Gradient of Temperate Forests in Central Germany
Abstract
:1. Introduction
2. Materials and Methods
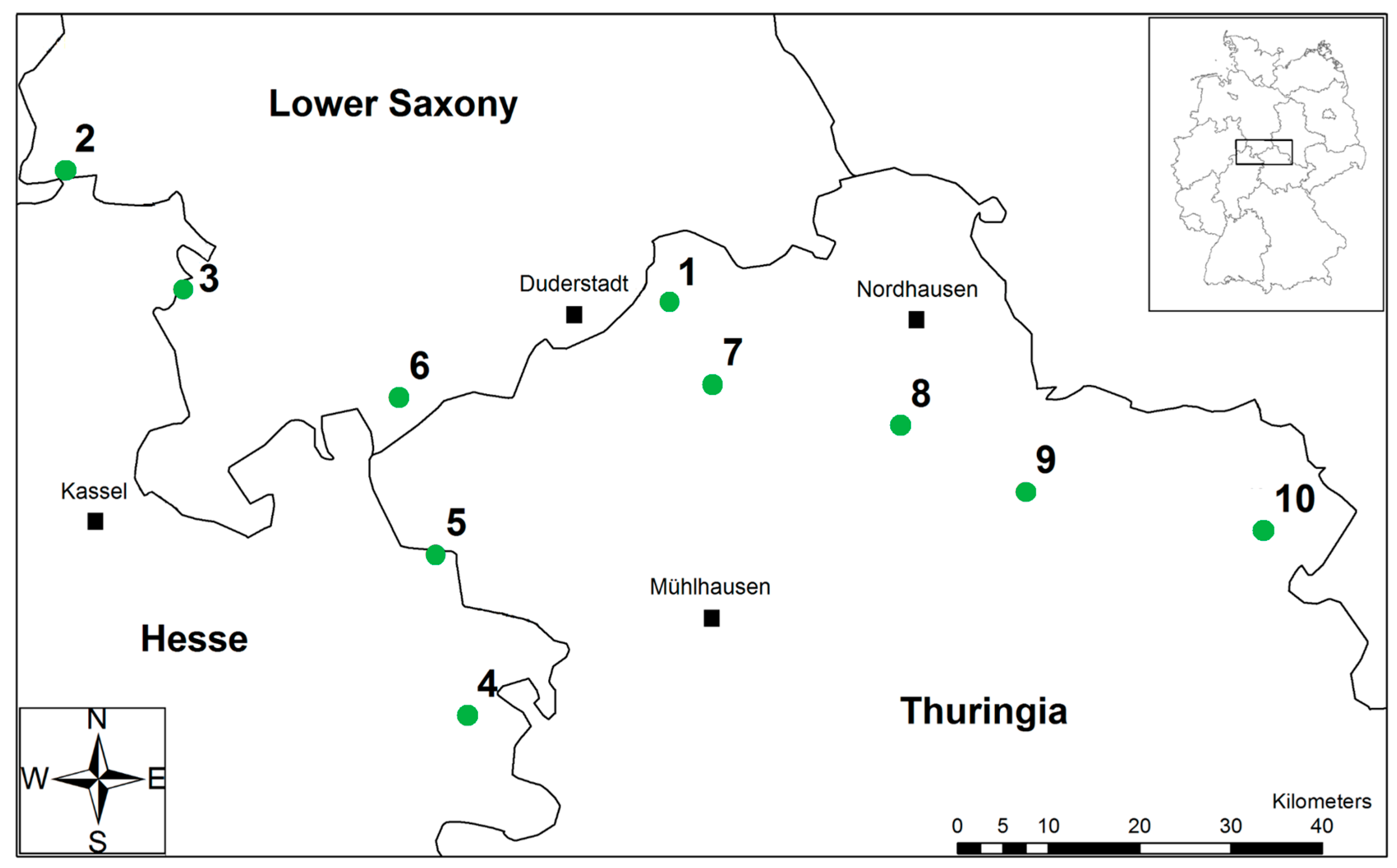
2.1. Study Site
2.2. Microclimate and Leaf Trait Data
2.3. Herbivory Data
2.4. Statistical Analyses
3. Results
3.1. Herbivore Feeding Traces
3.2. Feeding Trace Composition
3.3. Effects of Microclimate, Leaf Traits, and Plant Ontogenetic Stage
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Appendix A
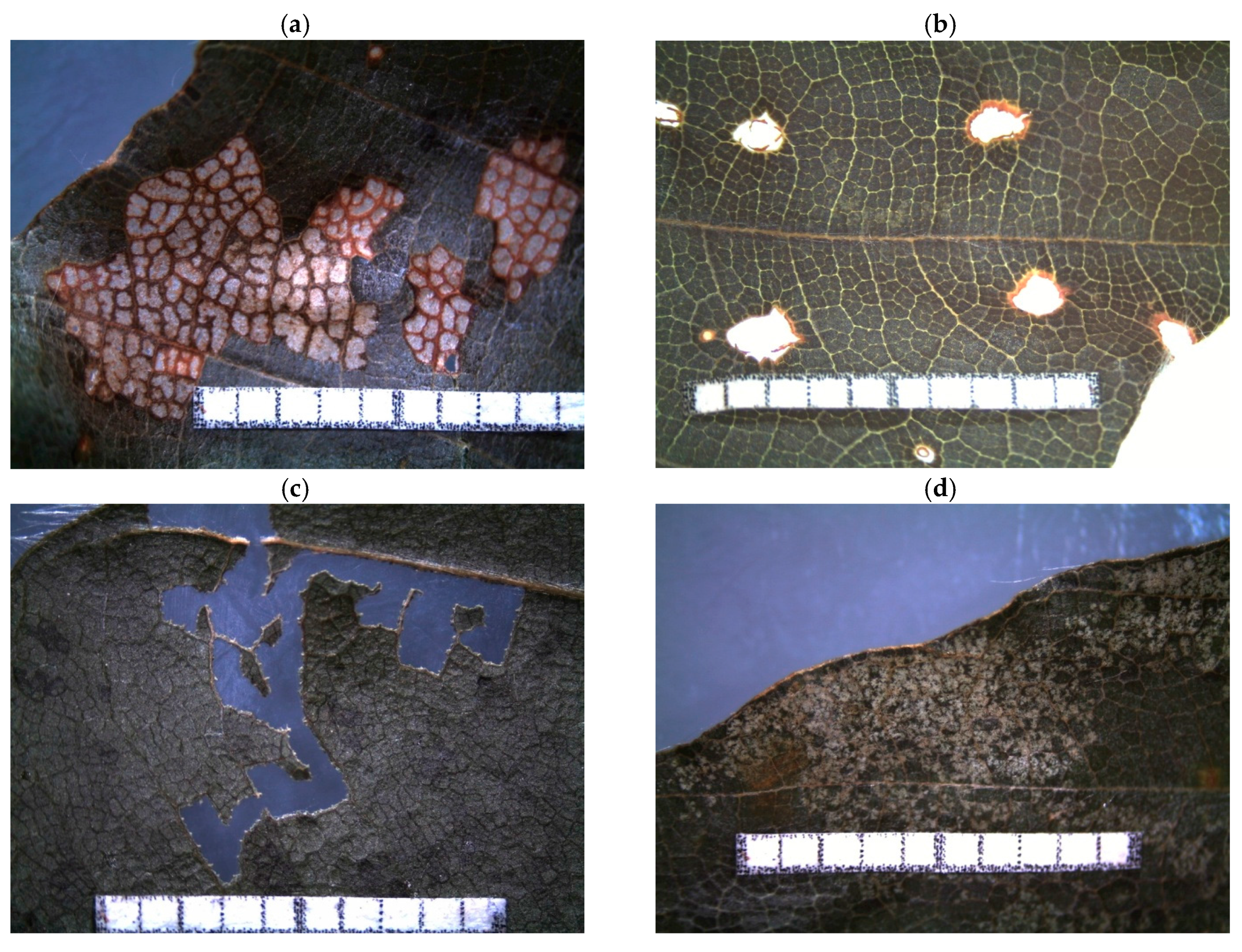

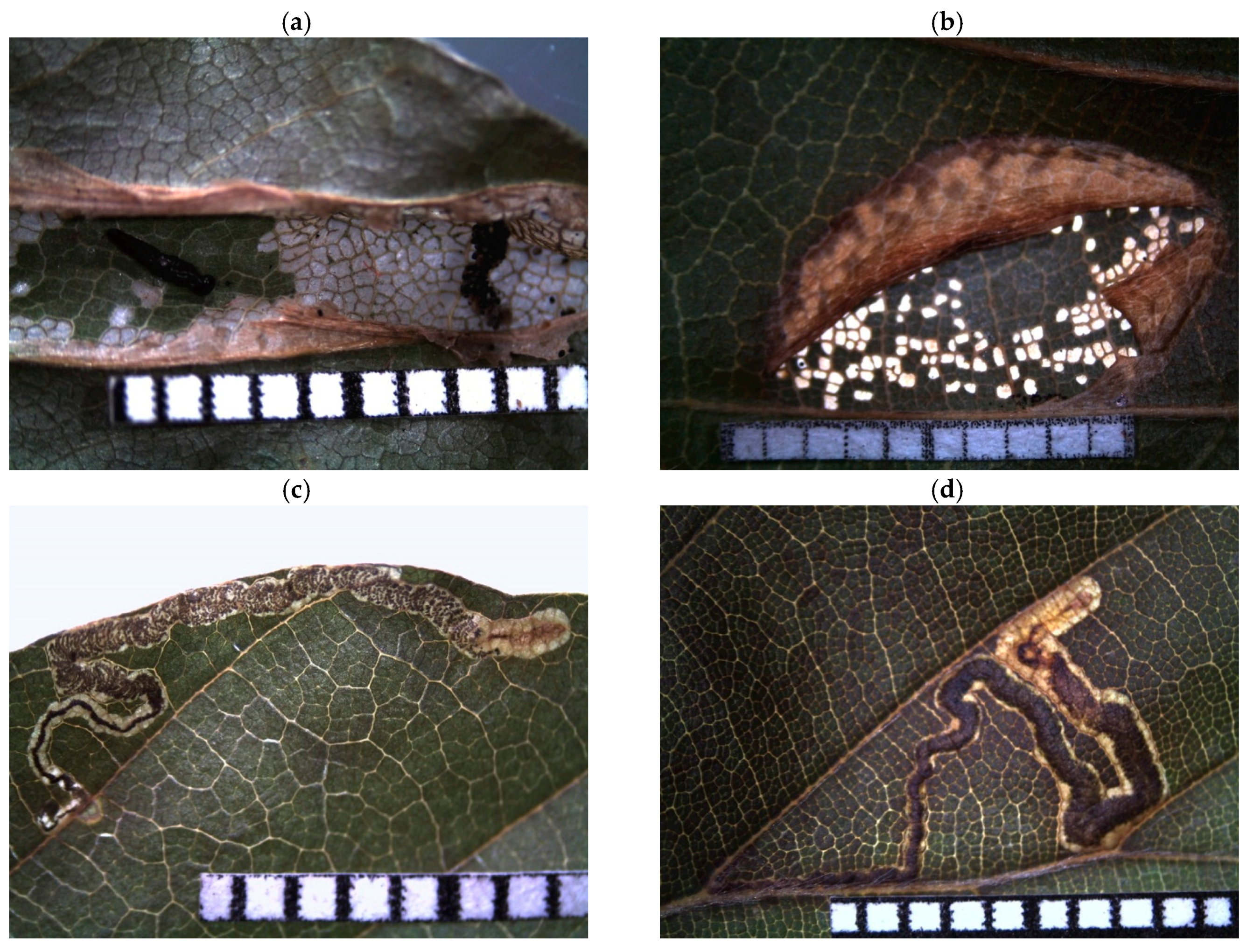

Appendix B
| Site | Beech | Leaf-Chewing | Sap-Sucking | Leaf-Mining | Gall-Inducing | ||||
|---|---|---|---|---|---|---|---|---|---|
| Individuals | Small Circles | Labyrinth | Whitish Spots | Tubular Mine | Leaf Edge Gall | Haired Vein Gall | Pannose Spot | Ovate Gall | |
| WB-N | juvenile (US) | + | + | + | + | + | + | ||
| adult (LC) | + | + | + | + | + | + | |||
| adult (UC) | + | + | + | + | + | + | |||
| WB-S | juvenile (US) | + | + | + | + | + | + | + | |
| adult (LC) | + | + | + | + | + | + | + | ||
| adult (UC) | + | + | + | + | + | + | |||
| TE-N | juvenile (US) | + | + | + | + | + | + | + | + |
| adult (LC) | + | + | + | + | + | + | + | ||
| adult (UC) | + | + | + | + | + | + | + | + | |
| TE-S | juvenile (US) | + | + | + | + | + | + | + | + |
| adult (LC) | + | + | + | + | + | + | + | + | |
| adult (UC) | + | + | + | + | + | + | |||
| KBVB-N | juvenile (us) | + | + | + | + | + | + | + | |
| adult (lc) | + | + | + | + | + | + | + | ||
| adult (uc) | + | + | + | + | + | + | |||
| KBVB-S | juvenile (US) | + | + | + | + | + | + | + | + |
| adult (LC) | + | + | + | + | + | + | |||
| adult (UC) | + | + | + | + | + | + | |||
| SS-N | juvenile (US) | + | + | + | + | + | + | + | + |
| adult (LC) | + | + | + | + | + | ||||
| adult (UC) | + | + | + | + | + | + | + | ||
| SS-S | juvenile (US) | + | + | + | + | + | + | + | |
| adult (LC) | + | + | + | + | + | + | |||
| adult (UC) | + | + | + | + | + | + | |||
| HGB-N | juvenile (US) | + | + | + | + | + | + | ||
| adult (LC) | + | + | + | + | + | ||||
| adult (UC) | + | + | + | + | + | ||||
| HGB-S | juvenile (US) | + | + | + | + | + | + | + | + |
| adult (LC) | + | + | + | + | + | + | + | ||
| adult (UC) | + | + | + | + | |||||
| BB-N | juvenile (US) | + | + | + | + | + | + | ||
| adult (LC) | + | + | + | + | + | + | |||
| adult (UC) | + | + | + | + | + | + | |||
| BB-S | juvenile (US) | + | + | + | + | + | + | + | + |
| adult (LC) | + | + | + | + | + | + | + | ||
| adult (UC) | + | + | + | + | + | + | + | ||
| HB-N | juvenile (US) | + | + | + | + | + | + | + | |
| adult (LC) | + | + | + | + | + | + | + | ||
| adult (UC) | + | + | + | + | + | + | + | ||
| HB-S | juvenile (US) | + | + | + | + | + | + | ||
| adult (LC) | + | + | + | + | + | + | |||
| adult (UC) | + | + | + | + | + | + | |||
| FK-N | juvenile (US) | + | + | + | + | + | + | ||
| adult (LC) | + | + | + | + | + | + | |||
| adult (UC) | + | + | + | + | + | + | |||
| FK-S | juvenile (US) | + | + | + | + | + | + | ||
| adult (LC) | + | + | + | + | + | + | |||
| adult (UC) | + | + | + | + | + | + | + | ||
| HDB-N | juvenile (US) | + | + | + | + | + | + | + | |
| adult (LC) | + | + | + | + | + | ||||
| adult (UC) | + | + | + | + | + | + | + | ||
| HDB-S | juvenile (US) | + | + | + | + | + | + | ||
| adult (LC) | + | + | + | + | + | + | |||
| adult (UC) | + | + | + | + | |||||
| EL-N | juvenile (US) | + | + | + | + | + | + | ||
| adult (LC) | + | + | + | + | + | + | |||
| adult (UC) | + | + | + | + | + | + | + | ||
| EL-S | juvenile (US) | + | + | + | + | + | + | + | |
| adult (LC) | + | + | + | + | + | + | + | + | |
| adult (UC) | + | + | + | + | + | + | + | ||
Appendix C
| Parameter | Beech Individual | ||
|---|---|---|---|
| Juvenile (US) | Adult (LC) | Adult (UC) | |
| Microclimate: | |||
| temperature (°C) 1 | 17.9 (17.6,18.2) a | 18.8 (18.6,19.1) b | 19.9 (19.6,20.6) c |
| relative air humidity (%) 2 | 83 (80,86) a | 74 (72,76) b | 69 (68,70) c |
| Leaf traits: | |||
| SLA (m2 kg−1) 2 | 38 (35,42) a | 28 (24,33) b | 16 (15,18) c |
| leaf area (cm2) 2 | 23 (21,24) a | 30 (23,34) b | 20 (19,23) a |
| N (mg g−1) 1 | 22 (21,24) a | 23 (22,24) a | 21 (20,22) b |
| C (mg g−1) 1 | 475 (472,748) a | 478 (476,480) b | 483 (481,486) c |
| C/N ratio (g g−1) 2 | 21.3 (19.8,22.1) a | 21.0 (19.7,22.2) a | 23.4 (22.5,24.4) b |
| chlorophyll (CCI) 1 | 13.8 (12.6,14.3) a | 13.5 (13.1,15.6) a | 13.2 (12.0,14.2) a |
Appendix D
- Call: metaMDS(comm = species, k = 2)
- global Multidimensional Scaling using monoMDS
- Data: wisconsin(sqrt(species))
- Distance: bray
- Dimensions: 2
- Stress: 0.1870401
- Stress type 1, weak ties
- Two convergent solutions found after 20 tries
- Scaling: centring, PC rotation, halfchange scaling
- Species: expanded scores based on ‘wisconsin(sqrt(species))
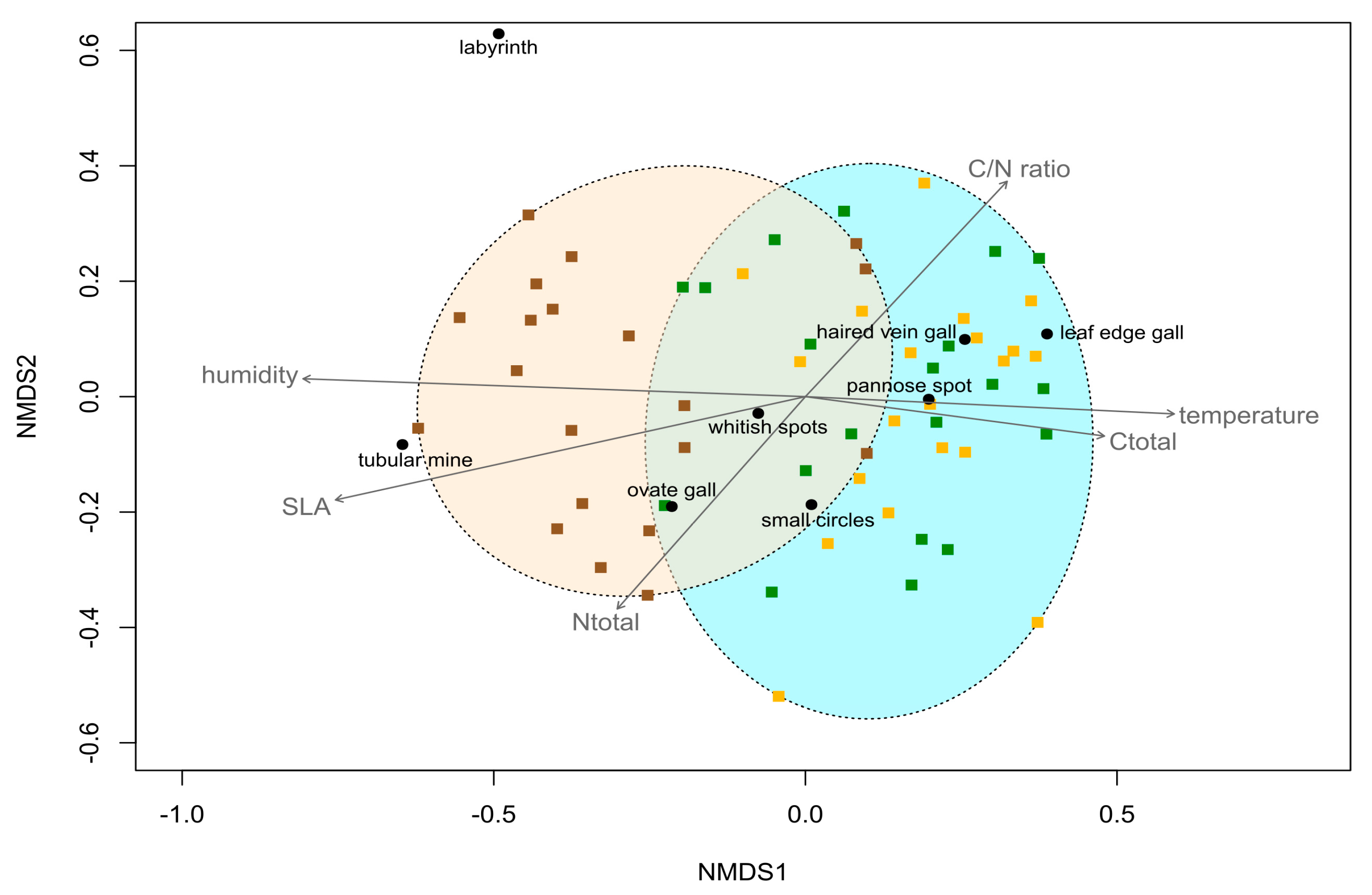
- ***VECTORS
- NMDS1 NMDS2 r2 Pr(>r)
- temp_dayav 0.99872 -0.05053 0.2489 0.001 ***
- humid_dayav -0.99926 0.03837 0.4612 0.001 ***
- SLA -0.97297 -0.23095 0.4257 0.001 ***
- Ctotal 0.99002 -0.14094 0.1665 0.006 **
- Ntotal -0.63438 -0.77302 0.1605 0.008 **
- C.N 0.65518 0.75547 0.1729 0.007 **
- chloro -0.87043 -0.49229 0.0544 0.234
- Permutation: free
- Number of permutations: 999
- 3 observations deleted due to missingness
- ***VECTORS
- NMDS1 NMDS2 r2 Pr(>r)
- temperature 0.99872 -0.05051 0.2489 0.001 ***
- humidity -0.99926 0.03836 0.4612 0.001 ***
- SLA -0.97296 -0.23095 0.4257 0.001 ***
- C 0.99002 -0.14094 0.1665 0.007 **
- N -0.63438 -0.77302 0.1605 0.009 **
- C.N 0.65518 0.75548 0.1729 0.005 **
- Permutation: free
- Number of permutations: 999
- 3 observations deleted due to missingness

Appendix E








References
- Crawley, M.J. Herbivory—The Dynamics of Animal-Plant Interactions; Blackwell Scientific Publications: Oxford, UK, 1983. [Google Scholar]
- Mulder, C.P.H.; Koricheva, J.; Huss-Danell, K.; Högberg, P.; Joshi, J. Insects affect relationships between plant species richness and ecosystem processes. Ecol. Lett. 1999, 2, 237–246. [Google Scholar] [CrossRef]
- Kozlov, M.V.; Zvereva, E.L. Background Insect Herbivory: Impacts, Patterns and Methodology. In Progress in Botany Volume 79; Progress in Botany; Springer: Cham, Switzerland, 2017; pp. 313–355. [Google Scholar]
- Zvereva, E.L.; Lanta, V.; Kozlov, M.V. Effects of sap-feeding insect herbivores on growth and reproduction of woody plants: A meta-analysis of experimental studies. Oecologia 2010, 163, 949–960. [Google Scholar] [CrossRef] [PubMed]
- Tal, O.; Freiberg, M.; Morawetz, W. Micro-climatic variability in the canopy of a temperate forest. In Canopy Arthropod Research in Europe; Floren, A., Schmidl, J., Eds.; Bioform Entomology: Nuremberg, Germany, 2008; pp. 49–59. [Google Scholar]
- Ulyshen, M.D. Arthropod vertical stratification in temperate deciduous forests: Implications for conservation-oriented management. For. Ecol. Manag. 2011, 261, 1479–1489. [Google Scholar] [CrossRef]
- Wilmers, F.; Ellenberg, H. Das Mikroklima in den untersuchten Beständen. In Ökosystemforschung. Ergebnisse des Sollingprojekts: 1966–1986; Ellenberg, H., Mayer, R., Schauermann, J., Eds.; Ulmer: Stuttgart, Germany, 1986; pp. 67–76. (In Germany) [Google Scholar]
- Tylianakis, J.M.; Didham, R.K.; Bascompte, J.; Wardle, D.A. Global change and species interactions in terrestrial ecosystems. Ecol. Lett. 2008, 11, 1351–1363. [Google Scholar] [CrossRef] [PubMed]
- Fowler, S.V. Differences in insect species richness and faunal composition of birch seedlings, saplings and trees: The importance of plant architecture. Ecol. Entomol. 1985, 10, 159–169. [Google Scholar] [CrossRef]
- Kearsley, M.J.C.; Whitham, T.G. Developmental changes in resistance to herbivory: Implications for individuals and populations. Ecology 1989, 70, 422–434. [Google Scholar] [CrossRef]
- Lowman, M.D. Herbivory in Australian Rain Forests, with Particular Reference to the Canopies of Doryphora sassafras (Monimiaceae). Biotropica 1992, 24, 263–272. [Google Scholar] [CrossRef]
- Basset, Y. Communities of insect herbivores foraging on saplings versus mature trees of Pourouma bicolor (Cecropiaceae) in Panama. Oecologia 2001, 129, 253–260. [Google Scholar] [CrossRef] [PubMed]
- Coley, P.D.; Barone, J.A. Herbivory and plant defenses in tropical forests. Annu. Rev. Ecol. Syst. 1996, 27, 305–335. [Google Scholar] [CrossRef]
- Lawson, E.J.R.; Poethig, R.S. Shoot development in plants: Time for a change. Trends Genet. 1995, 11, 263–268. [Google Scholar] [CrossRef]
- Poethig, R.S. Phase change and the regulation of shoot morphogenesis in plants. Science 1990, 250, 923–930. [Google Scholar] [CrossRef] [PubMed]
- Jones, C.S. An essay on juvenility, phase change, and heteroblasty in seed plants. Int. J. Plant Sci. 1999, 160, S105–S111. [Google Scholar] [CrossRef] [PubMed]
- Loney, P.E.; McARTHUR, C.; Potts, B.M.; Jordan, G.J. How does ontogeny in a Eucalyptus species affect patterns of herbivory by Brushtail Possums? Funct. Ecol. 2006, 20, 982–988. [Google Scholar] [CrossRef]
- Donaldson, J.R.; Stevens, M.T.; Barnhill, H.R.; Lindroth, R.L. Age-related shifts in leaf chemistry of clonal aspen (Populus tremuloides). J. Chem. Ecol. 2006, 32, 1415–1429. [Google Scholar] [CrossRef] [PubMed]
- Neilson, E.H.; Goodger, J.Q.D.; Woodrow, I.E. Novel aspects of cyanogenesis in Eucalyptus camphora subsp. humeana. Funct. Plant Biol. 2006, 33, 487–496. [Google Scholar] [CrossRef]
- Elger, A.; Lemoine, D.G.; Fenner, M.; Hanley, M.E. Plant ontogeny and chemical defence: Older seedlings are better defended. Oikos 2009, 118, 767–773. [Google Scholar] [CrossRef]
- Boege, K.; Marquis, R.J. Facing herbivory as you grow up: The ontogeny of resistance in plants. Trends Ecol. Evol. 2005, 20, 441–448. [Google Scholar] [CrossRef] [PubMed]
- Barton, K.E.; Koricheva, J. The ontogeny of plant defense and herbivory: Characterizing general patterns using meta-analysis. Am. Nat. 2010, 175, 481–493. [Google Scholar] [CrossRef] [PubMed]
- Stamp, N. Out of the quagmire of plant defense hypotheses. Q. Rev. Biol. 2003, 78, 23–55. [Google Scholar] [CrossRef] [PubMed]
- Basset, Y. Influence of leaf traits on the spatial distribution of arboreal arthropods within an overstorey rainforest tree. Ecol. Entomol. 1992, 17, 8–16. [Google Scholar] [CrossRef]
- Stiegel, S.; Entling, M.H.; Mantilla-Contreras, J. Reading the Leaves’ Palm: Leaf traits and herbivory along the microclimatic gradient of forest layers. PLOS ONE 2017, 12, e0169741. [Google Scholar] [CrossRef] [PubMed]
- Ali, J.G.; Agrawal, A.A. Specialist versus generalist insect herbivores and plant defense. Trends Plant Sci. 2012, 17, 293–302. [Google Scholar] [CrossRef] [PubMed]
- Andrew, N.R.; Hughes, L. Herbivore damage along a latitudinal gradient: Relative impacts of different feeding guilds. Oikos 2005, 108, 176–182. [Google Scholar] [CrossRef]
- Peeters, P.J. Correlations between leaf structural traits and the densities of herbivorous insect guilds. Biol. J. Linn. Soc. 2002, 77, 43–65. [Google Scholar] [CrossRef]
- Fortin, M.; Mauffette, Y. The suitability of leaves from different canopy layers for a generalist herbivore (Lepidoptera: Lasiocampidae) foraging on sugar maple. Can. J. For. Res. 2002, 32, 379–389. [Google Scholar] [CrossRef]
- Murakami, M.; Wada, N. Difference in leaf quality between canopy trees and seedlings affects migration and survival of spring-feeding moth larvae. Can. J. For. Res. 1997, 27, 1351–1356. [Google Scholar] [CrossRef]
- Horchler, P.J.; Morawetz, W. Canopy structure and its effect on canopy organisms: A generel introduction and some first findings of the Leipzig Canopy Crane Project with special reference to vertical stratification. In Canopy Arthropod Research in Europe; Floren, A., Schmidl, J., Eds.; Bioform Entomology: Nuremberg, Germany, 2008; pp. 31–48. [Google Scholar]
- Price, P.W.; Fernandes, G.W.; Lara, A.C.F.; Brawn, J.; Barrios, H.; Wright, M.G.; Ribeiro, S.P.; Rothcliff, N. Global patterns in local number of insect galling species. J. Biogeogr. 1998, 25, 581–591. [Google Scholar] [CrossRef]
- Parker, G.G. Structure and microclimate of forest canopies. In Forest canopies: A Review of Research on a Biological Frontier; Lowman, M.D., Nadkarni, N.M., Eds.; Academic Press: San Diego, CA, USA, 1995; pp. 73–106. [Google Scholar]
- Basset, Y.; Aberlenc, H.-P.; Barrios, H.; Curletti, G. Arthropod diel activity and stratification. In Arthropods of Tropical Forests—Spatio-temporal Dynamics and Resource Use in the Canopy; Basset, Y., Novotny, V., Miller, S.E., Kitching, R., Eds.; University Press: Cambridge, UK, 2003; pp. 304–314. [Google Scholar]
- Cornelissen, J.H.C.; Lavorel, S.; Garnier, E.; Diaz, S.; Buchmann, N.; Gurvich, D.E.; Reich, P.B.; Ter Steege, H.; Morgan, H.D.; van der Heijden, M.G.A.; et al. A handbook of protocols for standardised and easy measurement of plant functional traits worldwide. Aust. J. Bot. 2003, 51, 335–380. [Google Scholar] [CrossRef]
- Kleyer, M.; Bekker, R.M.; Knevel, I.C.; Bakker, J.P.; Thompson, K.; Sonnenschein, M.; Poschlod, P.; van Groenendael, J.M.; Klimeš, L.; Klimešová, J.; et al. The LEDA Traitbase: A database of life-history traits of the Northwest European flora. J. Ecol. 2008, 96, 1266–1274. [Google Scholar] [CrossRef]
- Coley, P.D.; Bryant, J.P.; Chapin, F.S. Resource availability and plant antiherbivore defense. Science 1985, 230, 895–899. [Google Scholar] [CrossRef] [PubMed]
- Van den Berg, A.K.; Perkins, T.D. Evaluation of a portable chlorophyll meter to estimate chlorophyll and nitrogen contents in sugar maple (Acer saccharum Marsh.) leaves. For. Ecol. Manag. 2004, 200, 113–117. [Google Scholar] [CrossRef]
- Peoples, M.B.; Dalling, M.J. The interplay between proteolysis and amino acid metabolism during senescence and nitrogen reallocation. In Senescence and Aging in Plants; Noodén, L.D., Leopold, A.C., Eds.; Academic Press: San Diego, CA, USA, 1988; pp. 181–217. [Google Scholar]
- Smith, C.M.S.; Bowie, M.H.; Hahner, J.L.; Boyer, S.; Kim, Y.-N.; Zhong, H.-T.; Abbott, M.; Rhodes, S.; Sharp, D.; Dickinson, N. Punakaiki Coastal Restoration Project: A case study for a consultative and multidisciplinary approach in selecting indicators of restoration success for a sand mining closure site, West Coast, New Zealand. CATENA 2016, 136, 91–103. [Google Scholar] [CrossRef]
- Vanbergen, A.J.; Woodcock, B.A.; Heard, M.S.; Chapman, D.S. Network size, structure and mutualism dependence affect the propensity for plant–pollinator extinction cascades. Funct. Ecol. 2017, 31, 1285–1293. [Google Scholar] [CrossRef]
- Kozlov, M.V.; Zvereva, E.L.; Zverev, V. Impacts of Point Polluters on Terrestrial Biota, 1st ed.; Springer: Amsterdam, The Netherlands, 2009. [Google Scholar]
- Kozlov, M.V.; Zvereva, E.L. Variations in the effects of local foliar damage on life span of individual leaves of downy birch (Betula pubescens). Botany 2014, 92, 477–484. [Google Scholar] [CrossRef]
- Ellis, W.N. Leafminers and Plant Galls of Europe. Available online: http://www.bladmineerders.nl/index.htm (accessed on 18 April 2017).
- Hochschule Weihenstephan-Triesdorf ARBOFUX—Diagnose-Datenbank für Gehölze. Available online: http://www.arbofux.de/ (accessed on 18 April 2017).
- Kimber, I. UKMoths—Guide to the Moths of Great Britain and Ireland. Available online: http://www.ukmoths.org.uk/ (accessed on 18 April 2017).
- Pitkin, B.; Ellis, W.N.; Plant, C.; Edmunds, R. The Leaf and Stem Mines of British Flies and Other Insects. Available online: http://www.ukflymines.co.uk/ (accessed on 18 April 2017).
- Rennwald, E.; Rodeland, J. Lepiforum: Bestimmung von Schmetterlingen (Lepidoptera) und Ihren Präimaginalstadien. Available online: http://www.lepiforum.de/ (accessed on 18 April 2017).
- Alford, D.V. Pests of Ornamental Trees, Shrubs and Flowers: A Colour Handbook, 2nd ed.; Manson Publishing: London, UK, 2012. [Google Scholar]
- Gossner, M.M.; Pašalić, E.; Lange, M.; Lange, P.; Boch, S.; Hessenmöller, D.; Müller, J.; Socher, S.A.; Fischer, M.; Schulze, E.-D.; et al. Differential responses of herbivores and herbivory to management in temperate european beech. PLoS ONE 2014, 9, e104876. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- R Development Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2017. [Google Scholar]
- Legendre, P.; Legendre, L. Numerical Ecology; Elsevier: Amsterdam, The Netherlands, 1998. [Google Scholar]
- Oksanen, J.; Blanchet, F.G.; Friendly, M.; Kindt, R.; Legendre, P.; McGlinn, D.; Minchin, P.R.; O’Hara, R.B.; Simons, G.L.; Solymos, P.; et al. Vegan: An Introduction to Ordination, R Package Version 2.4-2. 2017. Available online: https://CRAN.R-project.org/package=goeveg (accessed on 18 April 2017).
- Friedmann, G.; Schellenberg, J. Functions for Community Data and Ordinations, R Package Version 0.3.3. 2017. Available online: https://CRAN.R-project.org/package=goeveg (accessed on 18 April 2017).
- Bates, D.; Maechler, M.; Bolker, B.; Walker, S.; Christensen, R.H.B.; Singmann, H.; Dai, B.; Grothendieck, G.; Green, P. lme4: Linear Mixed-Effects Models using “Eigen” and S4. 2017. Available online: https://CRAN.R-project.org/package=lme4 (accessed on 18 April 2017).
- Bartoń, K. MuMIn: Multi-Model Inference. 2016. Available online: https://CRAN.R-project.org/package=MuMIn (accessed on 18 April 2017).
- Clarke, K.R. Non-parametric multivariate analyses of changes in community structure. Aust. J. Ecol. 1993, 18, 117–143. [Google Scholar] [CrossRef]
- Pritchard, I.M.; James, R. Leaf mines: Their effect on leaf longevity. Oecologia 1984, 64, 132–139. [Google Scholar] [CrossRef] [PubMed]
- Bussotti, F.; Grossoni, P.; Bottacci, A. Sclerophylly in beech (Fagus sylvatica L.) trees: Its relationship with crown transparency, nutritional status and summer drought. Forestry 1997, 267–272. [Google Scholar] [CrossRef]
- Feeny, P. Seasonal Changes in Oak Leaf Tannins and Nutrients as a Cause of Spring Feeding by Winter Moth Caterpillars. Ecology 1970, 51, 565–581. [Google Scholar] [CrossRef]
- Southwood, T.R.E.; Brown, V.K.; Reader, P.M. Leaf palatability, life expectancy and herbivore damage. Oecologia 1986, 70, 544–548. [Google Scholar] [CrossRef] [PubMed]
- Van Bael, S.A.; Brawn, J.D.; Robinson, S.K. Birds defend trees from herbivores in a Neotropical forest canopy. Proc. Natl. Acad. Sci. USA 2003, 100, 8304–8307. [Google Scholar] [CrossRef] [PubMed]
- Domínguez, C.A.; Dirzo, R.; Bullock, S.H. On the Function of Floral Nectar in Croton suberosus (Euphorbiaceae). Oikos 1989, 56, 109–114. [Google Scholar] [CrossRef]
- Boege, K. Herbivore Attack in Casearia nitida Influenced by Plant Ontogenetic Variation in Foliage Quality and Plant Architecture. Oecologia 2005, 143, 117–125. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, G.W.; Price, P.W. The adaptive significance of insect gall distribution: Survivorship of species in xeric and mesic habitats. Oecologia 1992, 90, 14–20. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, S.P.; Basset, Y. Gall-Forming and Free-Feeding Herbivory along Vertical Gradients in a Lowland Tropical Rainforest: The Importance of Leaf Sclerophylly. Ecography 2007, 30, 663–672. [Google Scholar] [CrossRef]
- Stork, N.E.; Hammond, P.M.; Russell, B.L.; Hadwen, W.L. The spatial distribution of beetles within the canopies of oak trees in Richmond Park, U.K. Ecol. Entomol. 2001, 26, 302–311. [Google Scholar] [CrossRef]
- Peeters, P.J.; Read, J.; Sanson, G.D. Variation in the guild composition of herbivorous insect assemblages among co-occurring plant species. Austral Ecol. 2001, 26, 385–399. [Google Scholar] [CrossRef]
- Kozlov, M.V.; Zverev, V.; Zvereva, E.L. Combined effects of environmental disturbance and climate warming on insect herbivory in mountain birch in subarctic forests: Results of 26-year monitoring. Sci. Total Environ. 2017, 601–602, 802–811. [Google Scholar] [CrossRef] [PubMed]
- Kass, R.E.; Raftery, A.E. Bayes factors. J. Am. Stat. Assoc. 1995, 90, 773–795. [Google Scholar] [CrossRef]

| Feeding Trace 1 | Description | Guild 2 | Leaf Side 3 | Probable Arthropod Species 1 |
|---|---|---|---|---|
| (a) Exophagous: | ||||
| windows | scraping damage on leaf surface | ch | Diurnea fagella (Denis and Schiffermüller, 1775) | |
| small circles | missing leaf area as small circles | ch | Orchestes fagi (Linnaeus, 1758) | |
| labyrinth | missing leaf area in form of labyrinths | ch | Phyllobius argentatus (Linnaeus, 1758) | |
| whitish spots | leaf flecked with whitish spots | s | ADS | Fagocyba cruenta (Herrich-Schäffer, 1838) |
| wax wool | waxed threads on leaf surface | s | ABS | Phyllaphis fagi (Linnaeus, 1767) |
| (b) Endophagous: | ||||
| tubular mine | tubular shaped mine between lateral veins | m | ABS | Phyllonorycter maestingella (Müller, 1764) |
| oval mine | oval shaped mine between lateral veins | m | ABS | Phyllonorycter messaniella (Zeller, 1846) |
| line crossing veins | wide corridor mine crossing lateral veins | m | Stigmella hemargyrella (Kollar, 1832) | |
| line between veins | zigzag mine between lateral veins | m | Stigmella tityrella (Stainton, 1854) | |
| leaf edge gall | gall causing rolled-up leaf edges | g | Acalitus stenaspis (Nalepa, 1891) | |
| haired vein gall | haired gall along lateral leaf veins | g | ADS | Aceria nervisequa (Canestrini, 1891) |
| pannose spot | pannose spot between lateral veins | g | ABS | Aceria nervisequa faginea (Nalepa, n.d.) |
| haired brownish gall | cylindrical, haired brownish gall | g | ADS | Hartigiola annulipes (Hartig, 1839) |
| ovate gall | ovate, acuminated gall (green to red) | g | ADS | Mikiola fagi (Hartig, 1839) |
| pleated gall | swollen, pleated leaf tissue forming a pouch | g | ADS | Phegomyia fagicola (Kieffer, 1901) |
| Feeding Guild | Feeding Trace | Attack Leaves (%) | ||
|---|---|---|---|---|
| Overall | Juvenile Beech | Adult Beech | ||
| leaf-chewing | small circles | 36 (29,65) a | 39 (30,54) a | 35 (28,72) a |
| labyrinth | 0 (0,7) b,c | 6 (0,13) b | 0 (0,2) b,d | |
| sap-sucking | whitish spots | 41 (27,60) a | 72 (59,78) a | 32 (23,49) a |
| leaf-mining | tubular mine | 0 (0,2) c | 3 (2,4) b | 0 (0,0) b |
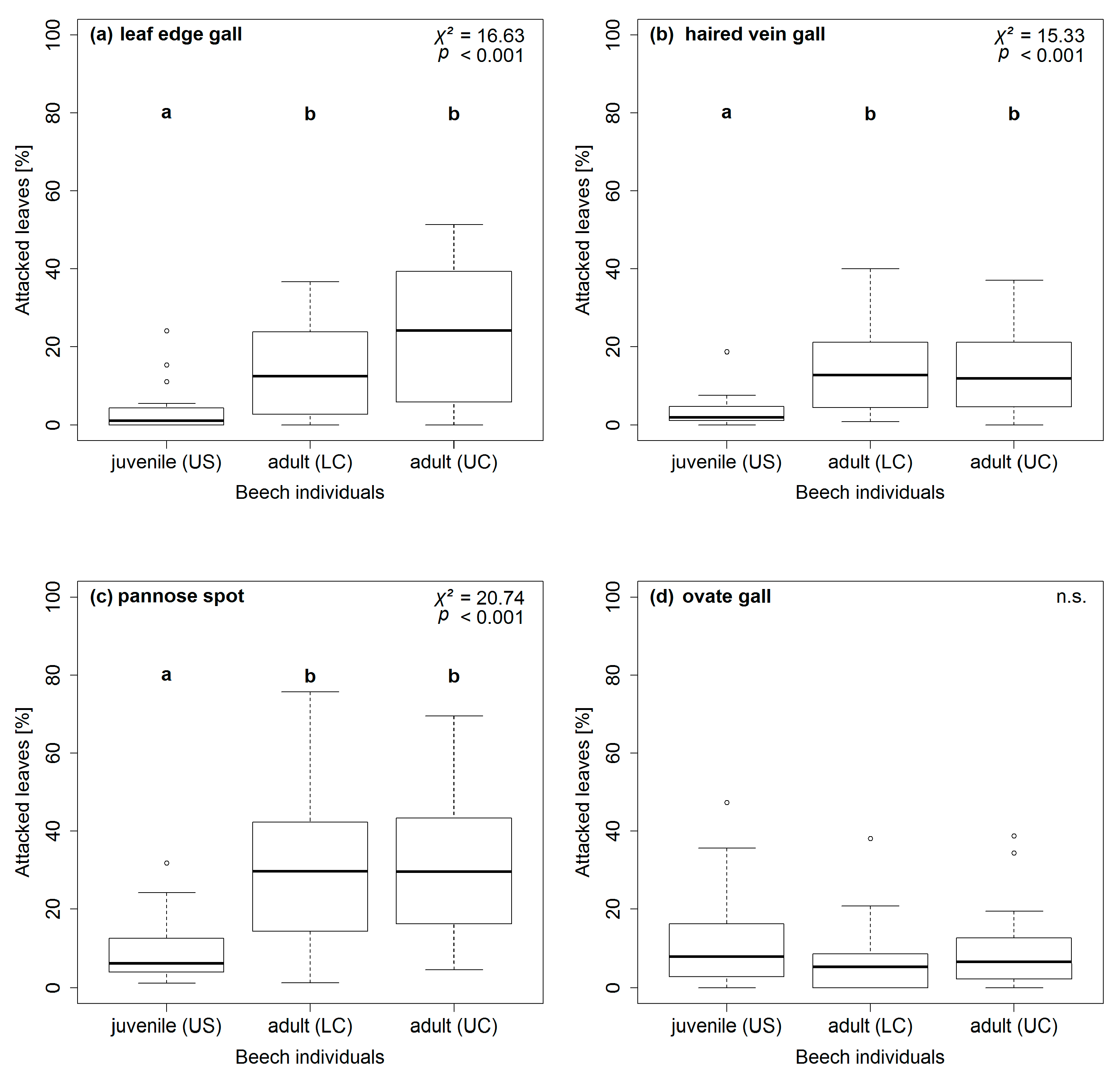
| gall-inducing | leaf edge gall | 9 (1,24) d,e | 1 (0,4) b | 19 (3,32) c,d,e |
| haired vein gall | 7 (2,16) d | 2 (1,4) b | 12 (5,21) e | |
| pannose spot | 18 (7,35) e | 6 (4,12) b | 30 (15,42) a,c | |
| ovate gall | 6 (2,13) b,d | 8 (3,15) b | 13 (4,21) d,e | |
| Model | d.f. | F-Values 1,2 | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Parameters | Exophagous | Endophagous | ||||||||
| n. | d. | Small Circles | Labyrinth | Whitish Spots | Tubular Mine | Leaf Edge Gall | Haired Vein Gall | Pannose Spot | Ovate Gall | |
| humidity | 1 | 55 | 1.29 | |||||||
| N content | 1 | 55 | 0.98 | |||||||
| N content + stage | 2 | 54 | 20.50 *** | 16.99 *** | 14.62 *** | 18.15 *** | ||||
| CN + stage | 2 | 54 | 17.72 *** | 21.08 *** | 16.31 *** | 17.25 *** | ||||
| stage | 1 | 55 | 0.19 | 9.79 ** | 30.85 *** | 37.25 *** | 17.96 *** | 18.64 *** | 27.56 *** | 1.33 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stiegel, S.; Mantilla-Contreras, J. Environment vs. Plant Ontogeny: Arthropod Herbivory Patterns on European Beech Leaves along the Vertical Gradient of Temperate Forests in Central Germany. Insects 2018, 9, 9. https://doi.org/10.3390/insects9010009
Stiegel S, Mantilla-Contreras J. Environment vs. Plant Ontogeny: Arthropod Herbivory Patterns on European Beech Leaves along the Vertical Gradient of Temperate Forests in Central Germany. Insects. 2018; 9(1):9. https://doi.org/10.3390/insects9010009
Chicago/Turabian StyleStiegel, Stephanie, and Jasmin Mantilla-Contreras. 2018. "Environment vs. Plant Ontogeny: Arthropod Herbivory Patterns on European Beech Leaves along the Vertical Gradient of Temperate Forests in Central Germany" Insects 9, no. 1: 9. https://doi.org/10.3390/insects9010009






