Fighting Arbovirus Transmission: Natural and Engineered Control of Vector Competence in Aedes Mosquitoes
Abstract
:1. Introduction
2. Natural Control of Arbovirus Transmission
2.1. Mosquitoes Naturally Resistant to Arbovirus Transmission
2.2. Insect-Specific Endosymbionts to Decrease Mosquito Competence for Arboviruses
2.2.1. Bacteria and Their Influence on Arbovirus Transmission: The Case of Wolbachia
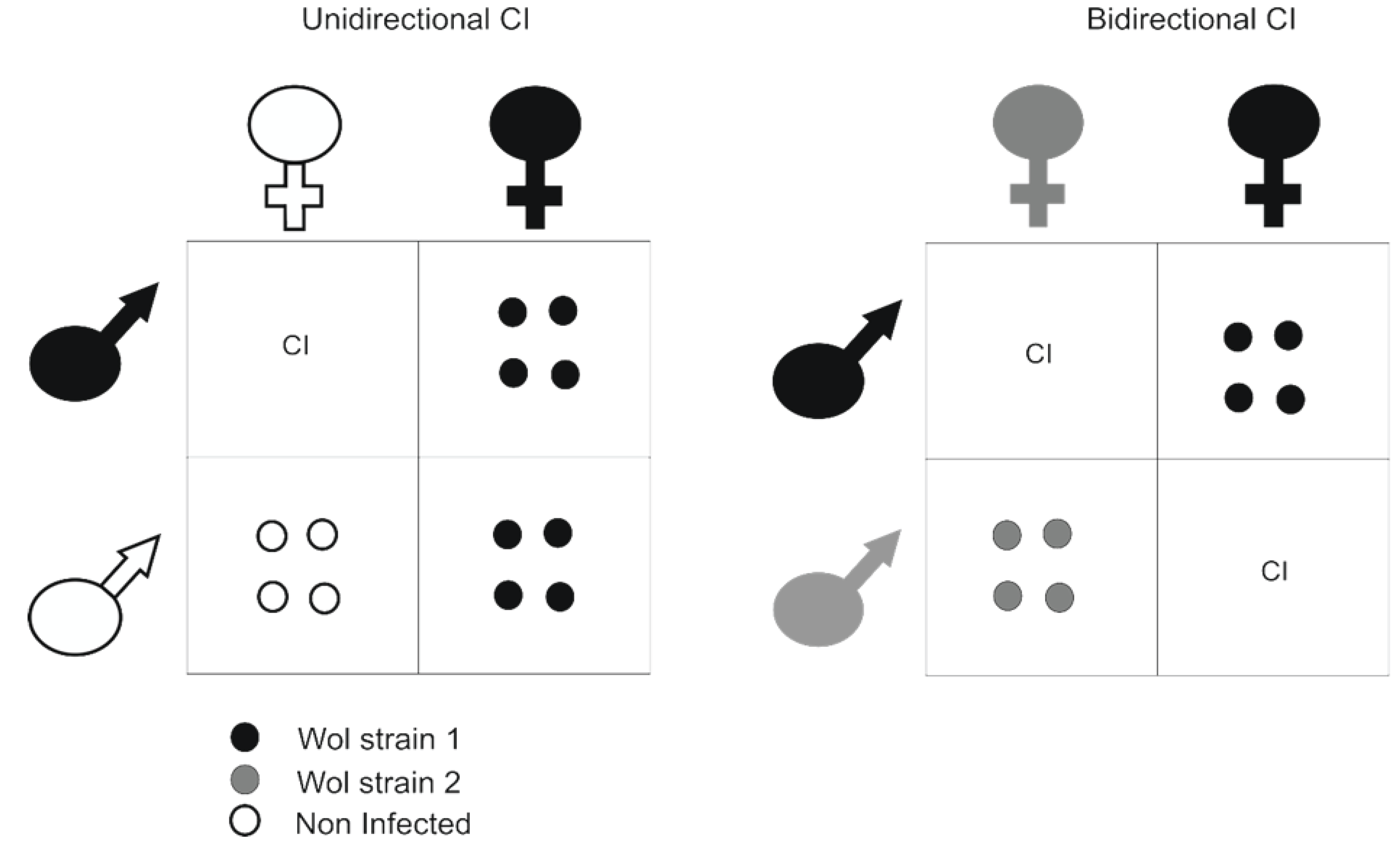
2.2.2. Insect-Specific Viruses (ISVs) and Their Influence on Arbovirus Transmission
| Arbovirus | ISV | Experimental Host | Experimental Outline | Effect on ISV | Effect on Arbovirus | Reference |
|---|---|---|---|---|---|---|
| DENV2 (flavivirus) | DNV (ambi-densovirus) | Ae. albopictus (adults & larvae) | DNV persistent infected mosquitoes, followed by DENV2 infection | Titer increase 2–3 log | 100× lower titer | [103] |
| WNV (flavivirus) MVEV (flavivirus), RRV (alphavirus) | PCV (flavivirus) | Ae. albopictus C6/36 | Arbovirus 6–7 dp PCV infection | Lower titer for WNV and MVEV No effect on RRV | [97] | |
| WNV (flavivirus) | CxFv (flavivirus) | Ae. albopictus C6/36 | Arbovirus 48 hp CxFV infection | 1 log lower titer at 108 hp infection. Other time points no effect | [91] | |
| WNV (flavivirus) | CxFv (flavivirus) | Cx. quinquefasciatus | CxFv persistent infected mosquitoes, followed by WNV infection | Delay in dissemination | [91] | |
| DENV2 (flavivirus) | AalDNV (ambi-densovirus) | Ae. albopictus C6/36 | Acute AalDNV infection, followed by DENV2 infection Persistent AalDNV infection, followed by DENV2 infection | Increased CPE Decreased CPE | [96] | |
| WNV (flavivirus) | CxFv-Izabal (flavivirus) | Ae. albopictus C6/36 | WNV 48 hp CxFV infection | Lower WNV titer from 4 dp infection (not significant) | [98] | |
| WNV (flavivirus) | CxFv-Izabal (flavivirus) | Cx. quinquefasciatus Honduras/Sebring | Co-infected by injection | CxFv in salivary glands (Honduras Cx.quinquefasciatus) if co-infected with WNV only | Increased WNV transmission | [98] |
| WNV (flavivirus) JEV (flavivirus) SLEV (flavivirus) | NHUV (flavivirus) | Ae. albopictus C6/36 | Arbovirus post or co-infected with NHUV | Lower titers | [102] |
2.3. Entomopathogenic Fungi and Their Effect on Competence for Arboviruses
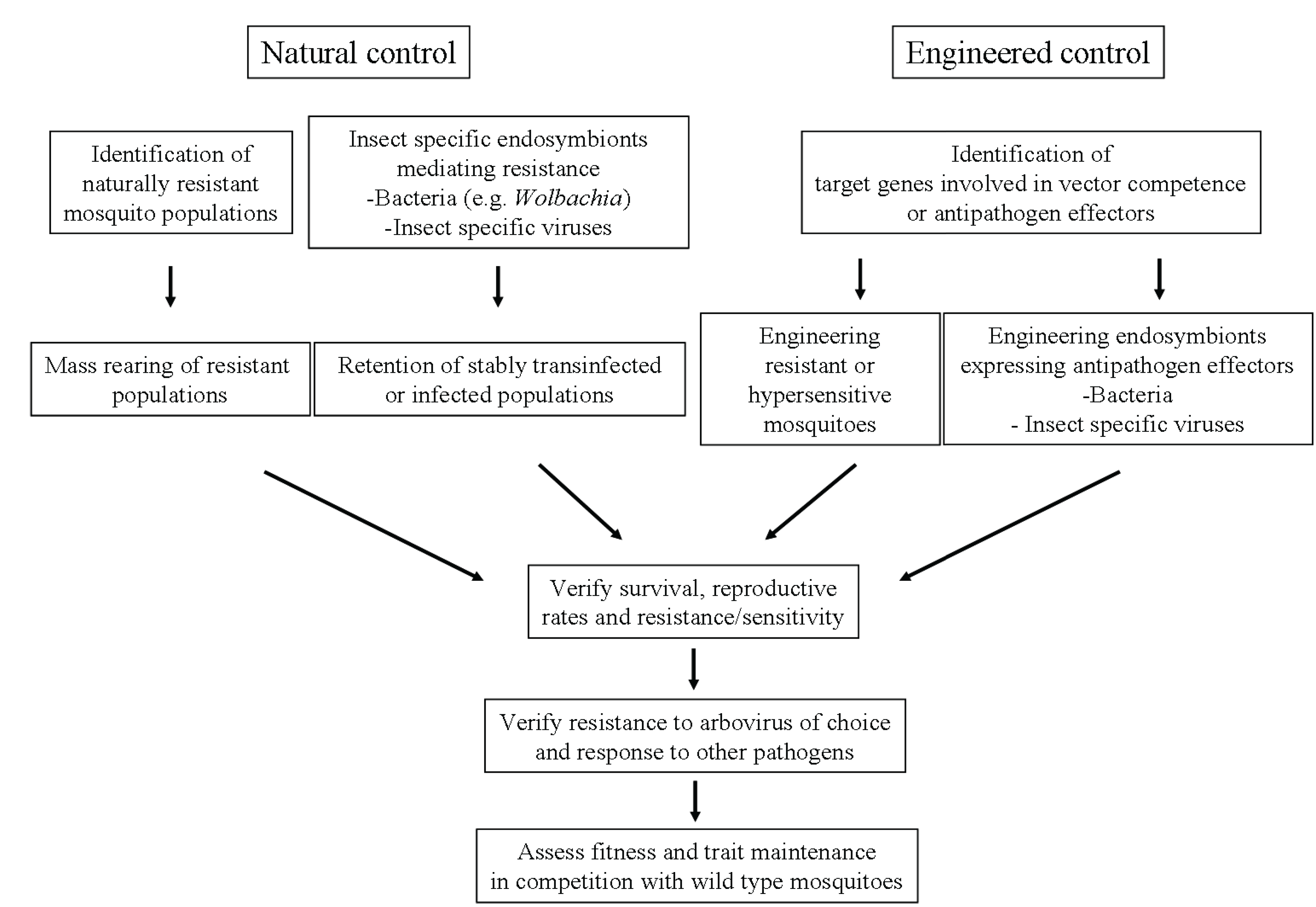
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bhatt, S.; Gething, P.W.; Brady, O.J.; Messina, J.P.; Farlow, A.W.; Moyes, C.L.; Drake, J.M.; Brownstein, J.S.; Hoen, A.G.; Sankoh, O.; et al. The global distribution and burden of dengue. Nature 2013, 496, 504–507. [Google Scholar] [CrossRef] [PubMed]
- Gould, E.A.; Higgs, S. Impact of climate change and other factors on emerging arbovirus diseases. Trans. R. Soc. Trop. Med. Hyg. 2009, 103, 109–121. [Google Scholar] [CrossRef] [PubMed]
- Weaver, S.C.; Reisen, W.K. Present and future arboviral threats. Antivir. Res. 2010, 85, 328–345. [Google Scholar] [CrossRef] [PubMed]
- Rezza, G. Dengue and chikungunya: Long-distance spread and outbreaks in naive areas. Pathog. Glob. Health 2014, 108, 349–355. [Google Scholar] [CrossRef] [PubMed]
- Tomasello, D.; Schlagenhauf, P. Chikungunya and dengue autochthonous cases in Europe, 2007–2012. Travel Med. Infect. Dis. 2013, 11, 274–284. [Google Scholar] [CrossRef] [PubMed]
- Jelinek, T. Trends in the epidemiology of dengue fever and their relevance for importation to Europe. Euro Surveill. 2009, 14. [Google Scholar] [PubMed]
- Reiter, P. Yellow fever and dengue: A threat to Europe? Euro Surveill. 2010, 15, 19509. [Google Scholar] [PubMed]
- Lindgren, E. Return of malaria, outbreaks of dengue fever. Possible health effects of climate changes in Europe, according to the WHO. Lakartidningen 2000, 97. [Google Scholar] [PubMed]
- Tsetsarkin, K.A.; Vanlandingham, D.L.; McGee, C.E.; Higgs, S. A single mutation in chikungunya virus affects vector specificity and epidemic potential. PLoS Pathog. 2007, 3, e201. [Google Scholar] [CrossRef] [PubMed]
- Vazeille, M.; Moutailler, S.; Coudrier, D.; Rousseaux, C.; Khun, H.; Huerre, M.; Thiria, J.; Dehecq, J.S.; Fontenille, D.; Schuffenecker, I.; et al. Two Chikungunya isolates from the outbreak of La Reunion (Indian Ocean) exhibit different patterns of infection in the mosquito, Aedes albopictus. PLoS ONE 2007, 2, e1168. [Google Scholar] [CrossRef] [PubMed]
- Schuffenecker, I.; Iteman, I.; Michault, A.; Murri, S.; Frangeul, L.; Vaney, M.C.; Lavenir, R.; Pardigon, N.; Reynes, J.M.; Pettinelli, F.; et al. Genome microevolution of chikungunya viruses causing the Indian Ocean outbreak. PLoS Med. 2006, 3, e263. [Google Scholar] [CrossRef] [PubMed]
- Coffey, L.L.; Failloux, A.B.; Weaver, S.C. Chikungunya virus-vector interactions. Viruses 2014, 6, 4628–4663. [Google Scholar] [CrossRef] [PubMed]
- Powers, A.M. Risks to the Americas associated with the continued expansion of chikungunya virus. J. Gen. Virol. 2015, 96, 1–5. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Progress and Prospects for the Use of Genetically Modified Mosquitoes to Inhibit Disease Transmission; World Health Organization: Geneva, Switzerland, 2009. [Google Scholar]
- Vontas, J.; Kioulos, E.; Pavlidi, N.; Morou, E.; Della Torre, A.; Ranson, H. Insecticide resistance in the major dengue vectors Aedes albopictus and Aedes aegypti. Pestic. Biochem. Physiol. 2012, 104, 126–131. [Google Scholar] [CrossRef]
- Dusfour, I.; Thalmensy, V.; Gaborit, P.; Issaly, J.; Carinci, R.; Girod, R. Multiple insecticide resistance in Aedes aegypti (Diptera: Culicidae) populations compromises the effectiveness of dengue vector control in French Guiana. Mem. Inst. Oswaldo Cruz 2011, 106, 346–352. [Google Scholar] [CrossRef] [PubMed]
- Norris, L.C.; Norris, D.E. Insecticide resistance in Culex quinquefasciatus mosquitoes after the introduction of insecticide-treated bed nets in Macha, Zambia. J. Vector Ecol. 2011, 36, 411–420. [Google Scholar] [CrossRef] [PubMed]
- Kamgang, B.; Marcombe, S.; Chandre, F.; Nchoutpouen, E.; Nwane, P.; Etang, J.; Corbel, V.; Paupy, C. Insecticide susceptibility of Aedes aegypti and Aedes albopictus in Central Africa. Parasit. Vectors 2011, 4, 79. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.L.; Battle, K.E.; Hay, S.I.; Barker, C.M.; Scott, T.W.; McKenzie, F.E. Ross, Macdonald, and a theory for the dynamics and control of mosquito-transmitted pathogens. PLoS Pathog. 2012, 8, e1002588. [Google Scholar] [CrossRef] [PubMed]
- Hardy, J.L.; Houk, E.J.; Kramer, L.D.; Reeves, W.C. Intrinsic factors affecting vector competence of mosquitoes for arboviruses. Ann. Rev. Entomol. 1983, 28, 229–262. [Google Scholar] [CrossRef]
- Black, W.C.; Bennett, K.E.; Gorrochotegui-Escalante, N.; Barillas-Mury, C.V.; Fernandez-Salas, I.; de Lourdes Munoz, M.; Farfan-Ale, J.A.; Olson, K.E.; Beaty, B.J. Flavivirus susceptibility in Aedes aegypti. Arch. Med. Res. 2002, 33, 379–388. [Google Scholar] [CrossRef] [PubMed]
- Ciota, A.T.; Kramer, L.D. Vector-virus interactions and transmission dynamics of West Nile virus. Viruses 2013, 5, 3021–3047. [Google Scholar] [CrossRef] [PubMed]
- Lambrechts, L. Quantitative genetics of Aedes aegypti vector competence for dengue viruses: Towards a new paradigm? Trends Parasitol. 2011, 27, 111–114. [Google Scholar] [CrossRef] [PubMed]
- Tabachnick, W.J. Nature, nurture and evolution of intra-species variation in mosquito arbovirus transmission competence. Int. J. Environ. Res. Public Health 2013, 10, 249–277. [Google Scholar] [CrossRef] [PubMed]
- Ciota, A.T.; Kramer, L.D. Insights into arbovirus evolution and adaptation from experimental studies. Viruses 2010, 2, 2594–2617. [Google Scholar] [CrossRef] [PubMed]
- Jupatanakul, N.; Sim, S.; Dimopoulos, G. The Insect Microbiome Modulates Vector Competence for Arboviruses. Viruses 2014, 6, 4294–4313. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, A.A.; Iturbe-Ormaetxe, I.; Callahan, A.G.; Phillips, B.L.; Billington, K.; Axford, J.K.; Montgomery, B.; Turley, A.P.; O’Neill, S.L. Stability of the wMel Wolbachia Infection following invasion into Aedes aegypti populations. PLoS Negl. Trop. Dis. 2014, 8, e3115. [Google Scholar] [CrossRef] [PubMed]
- Lacroix, R.; McKemey, A.R.; Raduan, N.; Kwee Wee, L.; Hong Ming, W.; Guat Ney, T.; Rahidah, A.A.S.; Salman, S.; Subramaniam, S.; Nordin, O.; et al. Open field release of genetically engineered sterile male Aedes aegypti in Malaysia. PLoS One 2012, 7, e42771. [Google Scholar] [CrossRef] [PubMed]
- Alphey, L. Genetic control of mosquitoes. Annu. Rev. Entomol. 2014, 59, 205–224. [Google Scholar] [CrossRef] [PubMed]
- Sim, S.; Jupatanakul, N.; Dimopoulos, G. Mosquito Immunity against Arboviruses. Viruses 2014, 6, 4479–4504. [Google Scholar] [CrossRef] [PubMed]
- Pike, A.D.; Cirimotich, C.; Dimopoulos, G. Impact of transgenic immune deployment on mosquito fitness. In Ecology of Parasite-Vector Interactions, Ecology and Control of Vector-Borne Diseases; Takken, W., Koenraadt, C.J.M., Eds.; Wageningen Academic Publishers: Wageningen, Gelderland, The Netherlands, 2013; pp. 19–33. [Google Scholar]
- Rainey, S.M.; Shah, P.; Kohl, A.; Dietrich, I. Understanding the Wolbachia-mediated inhibition of arboviruses in mosquitoes: Progress and challenges. J. Gen. Virol. 2014, 95, 517–530. [Google Scholar] [CrossRef] [PubMed]
- Vaidyanathan, R.; Scott, T.W. Apoptosis in mosquito midgut epithelia associated with West Nile virus infection. Apoptosis 2006, 11, 1643–1651. [Google Scholar] [CrossRef] [PubMed]
- Bennett, K.E.; Flick, D.; Fleming, K.H.; Jochim, R.; Beaty, B.J.; Black, W.C. Quantitative trait loci that control dengue-2 virus dissemination in the mosquito Aedes aegypti. Genetics 2005, 170, 185–194. [Google Scholar] [CrossRef] [PubMed]
- Bennett, K.E.; Beaty, B.J.; Black, W.C. Selection of D2S3, an Aedes aegypti (Diptera: Culicidae) strain with high oral susceptibility to Dengue 2 virus and D2MEB, a strain with a midgut barrier to Dengue 2 escape. J. Med. Entomol. 2005, 42, 110–119. [Google Scholar] [CrossRef] [PubMed]
- Bosio, C.F.; Fulton, R.E.; Salasek, M.L.; Beaty, B.J.; Black, W.C. Quantitative trait loci that control vector competence for dengue-2 virus in the mosquito Aedes aegypti. Genetics 2000, 156, 687–698. [Google Scholar] [PubMed]
- Gomez-Machorro, C.; Bennett, K.E.; del Lourdes Munoz, M.; Black, W.C. Quantitative trait loci affecting dengue midgut infection barriers in an advanced intercross line of Aedes aegypti. Insect. Mol. Biol. 2004, 13, 637–648. [Google Scholar] [CrossRef] [PubMed]
- Mercado-Curiel, R.F.; Black, W.C.; Munoz Mde, L. A dengue receptor as possible genetic marker of vector competence in Aedes aegypti. BMC Microbiol. 2008, 8, 118. [Google Scholar] [CrossRef] [PubMed]
- Lambrechts, L.; Quillery, E.; Noel, V.; Richardson, J.H.; Jarman, R.G.; Scott, T.W.; Chevillon, C. Specificity of resistance to dengue virus isolates is associated with genotypes of the mosquito antiviral gene Dicer-2. Proc. Biol. Sci. 2013, 280, 20122437. [Google Scholar] [CrossRef] [PubMed]
- Behura, S.K.; Gomez-Machorro, C.; deBruyn, B.; Lovin, D.D.; Harker, B.W.; Romero-Severson, J.; Mori, A.; Severson, D.W. Influence of mosquito genotype on transcriptional response to dengue virus infection. Funct. Integr. Genomics 2014, 14, 581–589. [Google Scholar] [CrossRef] [PubMed]
- Souza-Neto, J.A.; Sim, S.; Dimopoulos, G. An evolutionary conserved function of the JAK-STAT pathway in anti-dengue defense. Proc. Natl. Acad. Sci. USA 2009, 106, 17841–17846. [Google Scholar] [CrossRef] [PubMed]
- Behura, S.K.; Gomez-Machorro, C.; Harker, B.W.; deBruyn, B.; Lovin, D.D.; Hemme, R.R.; Mori, A.; Romero-Severson, J.; Severson, D.W. Global cross-talk of genes of the mosquito Aedes aegypti in response to dengue virus infection. PLoS Negl. Trop. Dis. 2011, 5, e1385. [Google Scholar] [CrossRef] [PubMed]
- Shin, D.; Civana, A.; Acevedo, C.; Smartt, C.T. Transcriptomics of differential vector competence: West Nile virus infection in two populations of Culex pipiens quinquefasciatus linked to ovary development. BMC Genomics 2014, 15, 513. [Google Scholar] [CrossRef] [PubMed]
- Sim, S.; Jupatanakul, N.; Ramirez, J.L.; Kang, S.; Romero-Vivas, C.M.; Mohammed, H.; Dimopoulos, G. Transcriptomic profiling of diverse Aedes aegypti strains reveals increased basal-level immune activation in dengue virus-refractory populations and identifies novel virus-vector molecular interactions. PLoS Negl. Trop. Dis. 2013, 7, e2295. [Google Scholar] [CrossRef] [PubMed]
- Armbruster, P.; Hutchinson, R.A.; Linvell, T. Equivalent inbreeding depression under laboratory and field conditions in a tree-hole-breeding mosquito. Proc. Biol. Sci. 2000, 267, 1939–1945. [Google Scholar] [CrossRef] [PubMed]
- Hilgenboecker, K.; Hammerstein, P.; Schlattmann, P.; Telschow, A.; Werren, J.H. How many species are infected with Wolbachia?—A statistical analysis of current data. Fems Microbiol. Lett. 2008, 281, 215–220. [Google Scholar] [CrossRef] [PubMed]
- Zug, R.; Hammerstein, P. Still a host of hosts for Wolbachia: Analysis of recent data suggests that 40% of terrestrial arthropod species are infected. PLoS ONE 2012, 7, e38544. [Google Scholar] [CrossRef] [PubMed]
- Werren, J.H.; Baldo, L.; Clark, M.E. Wolbachia: Master manipulators of invertebrate biology. Nat. Rev. Microbiol. 2008, 6, 741–751. [Google Scholar] [CrossRef] [PubMed]
- Sinkins, S.P. Wolbachia and cytoplasmic incompatibility in mosquitoes. Insect Biochem. Mol. Biol. 2004, 34, 723–729. [Google Scholar] [CrossRef] [PubMed]
- Laven, H. Eradication of Culex pipiens fatigans through cytoplasmic incompatibility. Nature 1967, 216, 383–384. [Google Scholar] [CrossRef] [PubMed]
- O’Connor, L.; Plichart, C.; Sang, A.C.; Brelsfoard, C.L.; Bossin, H.C.; Dobson, S. L. Open release of male mosquitoes infected with a wolbachia biopesticide: Field performance and infection containment. PLoS Negl. Trop. Dis. 2012, 6, e1797. [Google Scholar] [CrossRef] [PubMed]
- Ricci, I.; Cancrini, G.; Gabrielli, S.; D’Amelio, S.; Favi, G. Searching for Wolbachia (Rickettsiales: Rickettsiaceae) in mosquitoes (Diptera: Culicidae): Large polymerase chain reaction survey and new identifications. J. Med. Entomol. 2002, 39, 562–567. [Google Scholar] [CrossRef] [PubMed]
- Kittayapong, P.; Baisley, K.J.; Baimai, V.; O’Neill, S.L. Distribution and diversity of Wolbachia infections in Southeast Asian mosquitoes (Diptera: Culicidae). J. Med. Entomol. 2000, 37, 340–345. [Google Scholar] [CrossRef] [PubMed]
- Sinkins, S.P.; Braig, H.R.; O’Neill, S.L. Wolbachia pipientis: Bacterial density and unidirectional cytoplasmic incompatibility between infected populations of Aedes albopictus. Exp. Parasitol. 1995, 81, 284–291. [Google Scholar] [CrossRef] [PubMed]
- Glaser, R.L.; Meola, M.A. The native Wolbachia endosymbionts of Drosophila melanogaster and Culex quinquefasciatus increase host resistance to West Nile virus infection. PLoS ONE 2010, 5, e11977. [Google Scholar] [CrossRef] [PubMed]
- Baldini, F.; Segata, N.; Pompon, J.; Marcenac, P.; Robert Shaw, W.; Dabire, R.K.; Diabate, A.; Levashina, E.A.; Catteruccia, F. Evidence of natural Wolbachia infections in field populations of Anopheles gambiae. Nat. Commun. 2014, 5, 3985. [Google Scholar] [CrossRef] [PubMed]
- McMeniman, C.J.; Lane, R.V.; Cass, B.N.; Fong, A.W.; Sidhu, M.; Wang, Y.F.; O’Neill, S.L. Stable introduction of a life-shortening Wolbachia infection into the mosquito Aedes aegypti. Science 2009, 323, 141–144. [Google Scholar] [CrossRef] [PubMed]
- Walker, T.; Johnson, P.H.; Moreira, L.A.; Iturbe-Ormaetxe, I.; Frentiu, F.D.; McMeniman, C.J.; Leong, Y.S.; Dong, Y.; Axford, J.; Kriesner, P.; et al. The wMel Wolbachia strain blocks dengue and invades caged Aedes aegypti populations. Nature 2011, 476, 450–453. [Google Scholar] [CrossRef] [PubMed]
- Suh, E.; Mercer, D.R.; Fu, Y.; Dobson, S.L. Pathogenicity of life-shortening Wolbachia in Aedes albopictus after transfer from Drosophila melanogaster. Appl. Environ. Microbiol. 2009, 75, 7783–7788. [Google Scholar] [CrossRef] [PubMed]
- Yeap, H.L.; Axford, J.K.; Popovici, J.; Endersby, N.M.; Iturbe-Ormaetxe, I.; Ritchie, S.A.; Hoffmann, A.A. Assessing quality of life-shortening Wolbachia-infected Aedes aegypti mosquitoes in the field based on capture rates and morphometric assessments. Parasit. Vectors 2014, 7, 58. [Google Scholar] [CrossRef] [PubMed]
- Hedges, L.M.; Brownlie, J.C.; O’Neill, S.L.; Johnson, K.N. Wolbachia and virus protection in insects. Science 2008, 322, 702. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, L.; Ferreira, A.; Ashburner, M. The bacterial symbiont Wolbachia induces resistance to RNA viral infections in Drosophila melanogaster. PLoS Biol. 2008, 6, e2. [Google Scholar] [CrossRef] [PubMed]
- Bian, G.; Xu, Y.; Lu, P.; Xie, Y.; Xi, Z. The endosymbiotic bacterium Wolbachia induces resistance to dengue virus in Aedes aegypti. PLoS Pathog. 2010, 6, e1000833. [Google Scholar] [CrossRef] [PubMed]
- Bian, G.; Zhou, G.; Lu, P.; Xi, Z. Replacing a native Wolbachia with a novel strain results in an increase in endosymbiont load and resistance to dengue virus in a mosquito vector. PLoS Negl. Trop. Dis. 2013, 7, e2250. [Google Scholar] [CrossRef] [PubMed]
- Blagrove, M.S.; Arias-Goeta, C.; Di Genua, C.; Failloux, A.B.; Sinkins, S.P. A Wolbachia wMel transinfection in Aedes albopictus is not detrimental to host fitness and inhibits Chikungunya virus. PLoS Negl. Trop. Dis. 2013, 7, e2152. [Google Scholar] [CrossRef] [PubMed]
- Hussain, M.; Lu, G.; Torres, S.; Edmonds, J.H.; Kay, B.H.; Khromykh, A.A.; Asgari, S. Effect of Wolbachia on replication of West Nile virus in a mosquito cell line and adult mosquitoes. J. Virol. 2013, 87, 851–858. [Google Scholar] [CrossRef] [PubMed]
- Van den Hurk, A.F.; Hall-Mendelin, S.; Pyke, A.T.; Frentiu, F.D.; McElroy, K.; Day, A.; Higgs, S.; O’Neill, S. L. Impact of Wolbachia on infection with chikungunya and yellow fever viruses in the mosquito vector Aedes aegypti. PLoS Negl. Trop. Dis. 2012, 6, e1892. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moreira, L.A.; Iturbe-Ormaetxe, I.; Jeffery, J.A.; Lu, G.; Pyke, A.T.; Hedges, L.M.; Rocha, B.C.; Hall-Mendelin, S.; Day, A.; Riegler, M.; et al. A Wolbachia symbiont in Aedes aegypti limits infection with dengue, Chikungunya, and Plasmodium. Cell 2009, 139, 1268–1278. [Google Scholar] [CrossRef] [PubMed]
- Mousson, L.; Zouache, K.; Arias-Goeta, C.; Raquin, V.; Mavingui, P.; Failloux, A.B. The native Wolbachia symbionts limit transmission of dengue virus in Aedes albopictus. PLoS Negl. Trop. Dis. 2012, 6, e1989. [Google Scholar] [CrossRef] [PubMed]
- Mousson, L.; Martin, E.; Zouache, K.; Madec, Y.; Mavingui, P.; Failloux, A.B. Wolbachia modulates Chikungunya replication in Aedes albopictus. Mol. Ecol. 2010, 19, 1953–1964. [Google Scholar] [CrossRef] [PubMed]
- Osborne, S.E.; Iturbe-Ormaetxe, I.; Brownlie, J.C.; O’Neill, S.L.; Johnson, K.N. Antiviral protection and the importance of Wolbachia density and tissue tropism in Drosophila simulans. Appl. Environ. Microbiol. 2012, 78, 6922–6929. [Google Scholar] [CrossRef] [PubMed]
- Lu, P.; Bian, G.; Pan, X.; Xi, Z. Wolbachia induces density-dependent inhibition to dengue virus in mosquito cells. PLoS Negl. Trop. Dis. 2012, 6, e1754. [Google Scholar] [CrossRef] [PubMed]
- McMeniman, C.J.; Lane, A.M.; Fong, A.W.; Voronin, D.A.; Iturbe-Ormaetxe, I.; Yamada, R.; McGraw, E.A.; O’Neill, S.L. Host adaptation of a Wolbachia strain after long-term serial passage in mosquito cell lines. Appl. Environ. Microbiol. 2008, 74, 6963–6969. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sinkins, S.P. Wolbachia and arbovirus inhibition in mosquitoes. Future Microbiol. 2013, 8, 1249–1256. [Google Scholar] [CrossRef] [PubMed]
- Moreira, L.A. Wolbachia in Aedes mosquitoes: Towards biological control of vector-borne diseases. In Ecology of Parasite-Vector Interactions, Ecology and Control of Vector-Borne Diseases; Takken, W., Koenraadt, C.J.M., Eds.; Wageningen Academic Publishers: Wageningen, Gelderland, The Netherlands, 2013; pp. 155–166. [Google Scholar]
- Rances, E.; Ye, Y.H.; Woolfit, M.; McGraw, E.A.; O’Neill, S.L. The relative importance of innate immune priming in Wolbachia-mediated dengue interference. PLoS Pathog. 2012, 8, e1002548. [Google Scholar] [CrossRef] [PubMed]
- Pan, X.; Zhou, G.; Wu, J.; Bian, G.; Lu, P.; Raikhel, A.S.; Xi, Z. Wolbachia induces reactive oxygen species (ROS)-dependent activation of the Toll pathway to control dengue virus in the mosquito Aedes aegypti. Proc. Natl. Acad. Sci. USA 2012, 109, E23–E31. [Google Scholar] [CrossRef] [PubMed]
- Osborne, S.E.; Leong, Y.S.; O’Neill, S.L.; Johnson, K.N. Variation in antiviral protection mediated by different Wolbachia strains in Drosophila simulans. PLoS Pathog. 2009, 5, e1000656. [Google Scholar] [CrossRef] [PubMed]
- Rances, E.; Johnson, T.K.; Popovici, J.; Iturbe-Ormaetxe, I.; Zakir, T.; Warr, C.G.; O’Neill, S.L. The toll and Imd pathways are not required for wolbachia-mediated dengue virus interference. J. Virol. 2013, 87, 11945–11949. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, A.A.; Montgomery, B.L.; Popovici, J.; Iturbe-Ormaetxe, I.; Johnson, P.H.; Muzzi, F.; Greenfield, M.; Durkan, M.; Leong, Y.S.; Dong, Y.; et al. Successful establishment of Wolbachia in Aedes populations to suppress dengue transmission. Nature 2011, 476, 454–457. [Google Scholar] [CrossRef] [PubMed]
- Frentiu, F.D.; Zakir, T.; Walker, T.; Popovici, J.; Pyke, A.T.; van den Hurk, A.; McGraw, E.A.; O’Neill, S.L. Limited dengue virus replication in field-collected Aedes aegypti mosquitoes infected with Wolbachia. PLoS Negl. Trop. Dis. 2014, 8, e2688. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Engelstadter, J.; Telschow, A. Cytoplasmic incompatibility and host population structure. Heredity 2009, 103, 196–207. [Google Scholar] [CrossRef] [PubMed]
- Schofield, P.; Chaplain, M.; Hubbard, S. Mathematical modelling of host-parasitoid systems: Effects of chemically mediated parasitoid foraging strategies on within- and between-generation spatio-temporal dynamics. J. Theor. Biol. 2002, 214, 31–47. [Google Scholar] [CrossRef] [PubMed]
- Vavre, F.; Charlat, S. Making (good) use of Wolbachia: What the models say. Curr. Opin. Microbiol. 2012, 15, 263–268. [Google Scholar] [CrossRef] [PubMed]
- Hancock, P.A.; Godfray, H.C. Modelling the spread of Wolbachia in spatially heterogeneous environments. J. R. Soc. Interface R. Soc. 2012, 9, 3045–3054. [Google Scholar] [CrossRef]
- Hughes, H.; Britton, N.F. Modelling the use of Wolbachia to control dengue fever transmission. Bull. Math. Biol. 2013, 75, 796–818. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Turelli, M. Cytoplasmic incompatibility in populations with overlapping generations. Evol. Int. J. Org. Evol. 2010, 64, 232–241. [Google Scholar] [CrossRef]
- Hughes, G.L.; Dodson, B.L.; Johnson, R.M.; Murdock, C.C.; Tsujimoto, H.; Suzuki, Y.; Patt, A.A.; Cui, L.; Nossa, C.W.; Barry, R.M.; et al. Native microbiome impedes vertical transmission of Wolbachia in Anopheles mosquitoes. Proc. Natl. Acad. Sci. USA 2014, 111, 12498–12503. [Google Scholar] [CrossRef] [PubMed]
- Cook, S.; Moureau, G.; Kitchen, A.; Gould, E.A.; de Lamballerie, X.; Holmes, E.C.; Harbach, R.E. Molecular evolution of the insect-specific flaviviruses. J. Gen. Virol. 2012, 93, 223–234. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Junglen, S.; Drosten, C. Virus discovery and recent insights into virus diversity in arthropods. Curr. Opin. Microbiol. 2013, 16, 507–513. [Google Scholar] [CrossRef] [PubMed]
- Bolling, B.G.; Olea-Popelka, F.J.; Eisen, L.; Moore, C.G.; Blair, C.D. Transmission dynamics of an insect-specific flavivirus in a naturally infected Culex pipiens laboratory colony and effects of co-infection on vector competence for West Nile virus. Virology 2012, 427, 90–97. [Google Scholar] [CrossRef] [PubMed]
- Wei, W.; Shao, D.; Huang, X.; Li, J.; Chen, H.; Zhang, Q.; Zhang, J. The pathogenicity of mosquito densovirus (C6/36DNV) and its interaction with dengue virus type II in Aedes albopictus. Am. J. Trop. Med. Hyg. 2006, 75, 1118–1126. [Google Scholar] [PubMed]
- Saiyasombat, R.; Bolling, B.G.; Brault, A.C.; Bartholomay, L.C.; Blitvich, B.J. Evidence of efficient transovarial transmission of Culex flavivirus by Culex pipiens (Diptera: Culicidae). J. Med. Entomol. 2011, 48, 1031–1038. [Google Scholar] [CrossRef] [PubMed]
- Lutomiah, J.J.; Mwandawiro, C.; Magambo, J.; Sang, R.C. Infection and vertical transmission of Kamiti river virus in laboratory bred Aedes aegypti mosquitoes. J. Insect Sci. 2007, 7, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Scott, J.C.; Brackney, D.E.; Campbell, C.L.; Bondu-Hawkins, V.; Hjelle, B.; Ebel, G.D.; Olson, K.E.; Blair, C.D. Comparison of dengue virus type 2-specific small RNAs from RNA interference-competent and -incompetent mosquito cells. PLoS Negl. Trop. Dis. 2010, 4, e848. [Google Scholar] [CrossRef] [PubMed]
- Burivong, P.; Pattanakitsakul, S.N.; Thongrungkiat, S.; Malasit, P.; Flegel, T.W. Markedly reduced severity of Dengue virus infection in mosquito cell cultures persistently infected with Aedes albopictus densovirus (AalDNV). Virology 2004, 329, 261–269. [Google Scholar] [CrossRef] [PubMed]
- Hobson-Peters, J.; Yam, A.W.; Lu, J.W.; Setoh, Y.X.; May, F.J.; Kurucz, N.; Walsh, S.; Prow, N.A.; Davis, S.S.; Weir, R.; et al. A new insect-specific flavivirus from northern Australia suppresses replication of West Nile virus and Murray Valley encephalitis virus in co-infected mosquito cells. PLoS ONE 2013, 8, e56534. [Google Scholar] [CrossRef] [PubMed]
- Kent, R.J.; Crabtree, M.B.; Miller, B.R. Transmission of West Nile virus by Culex quinquefasciatus say infected with Culex Flavivirus Izabal. PLoS Negl. Trop. Dis. 2010, 4, e671. [Google Scholar] [CrossRef] [PubMed]
- Newman, C.M.; Cerutti, F.; Anderson, T.K.; Hamer, G.L.; Walker, E.D.; Kitron, U.D.; Ruiz, M.O.; Brawn, J.D.; Goldberg, T.L. Culex flavivirus and West Nile virus mosquito coinfection and positive ecological association in Chicago, United States. Vector Borne Zoonotic Dis. 2011, 11, 1099–1105. [Google Scholar] [CrossRef] [PubMed]
- Zou, G.; Zhang, B.; Lim, P.Y.; Yuan, Z.; Bernard, K.A.; Shi, P. Y. Exclusion of West Nile virus superinfection through RNA replication. J. Virol. 2009, 83, 11765–11776. [Google Scholar] [CrossRef] [PubMed]
- Karpf, A.R.; Lenches, E.; Strauss, E.G.; Strauss, J.H.; Brown, D.T. Superinfection exclusion of alphaviruses in three mosquito cell lines persistently infected with Sindbis virus. J. Virol. 1997, 71, 7119–7123. [Google Scholar] [PubMed]
- Kenney, J.L.; Solberg, O.D.; Langevin, S.A.; Brault, A.C. Characterization of a novel insect-specific flavivirus from Brazil: Potential for inhibition of infection of arthropod cells with medically important flaviviruses. J. Gen. Virol. 2014, 95, 2796–2808. [Google Scholar] [CrossRef] [PubMed]
- Mosimann, A.L.; Bordignon, J.; Mazzarotto, G.C.; Motta, M.C.; Hoffmann, F.; Santos, C.N. Genetic and biological characterization of a densovirus isolate that affects dengue virus infection. Mem. Inst. Oswaldo Cruz 2011, 106, 285–292. [Google Scholar] [CrossRef] [PubMed]
- Blanford, S.; Chan, B.H.; Jenkins, N.; Sim, D.; Turner, R.J.; Read, A.F.; Thomas, M.B. Fungal pathogen reduces potential for malaria transmission. Science 2005, 308, 1638–1641. [Google Scholar] [CrossRef] [PubMed]
- Scholte, E.J.; Knols, B.G.; Takken, W. Infection of the malaria mosquito Anopheles gambiae with the entomopathogenic fungus Metarhizium anisopliae reduces blood feeding and fecundity. J. Invertebr. Pathol. 2006, 91, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Scholte, E.J.; Ng’habi, K.; Kihonda, J.; Takken, W.; Paaijmans, K.; Abdulla, S.; Killeen, G.F.; Knols, B.G. An entomopathogenic fungus for control of adult African malaria mosquitoes. Science 2005, 308, 1641–1642. [Google Scholar] [CrossRef] [PubMed]
- Scholte, E.J.; Njiru, B.N.; Smallegange, R.C.; Takken, W.; Knols, B.G. Infection of malaria (Anopheles gambiae s.s.) and filariasis (Culex quinquefasciatus) vectors with the entomopathogenic fungus Metarhizium anisopliae. Malar. J. 2003, 2. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Knols, B.G.; Bukhari, T.; Farenhorst, M. Entomopathogenic fungi as the next-generation control agents against malaria mosquitoes. Future Microbiol. 2010, 5, 339–341. [Google Scholar] [CrossRef] [PubMed]
- Scholte, E.J.; Takken, W.; Knols, B.G. Infection of adult Aedes aegypti and Ae. albopictus mosquitoes with the entomopathogenic fungus Metarhizium anisopliae. Acta Trop. 2007, 102, 151–158. [Google Scholar] [CrossRef] [PubMed]
- Darbro, J.M.; Johnson, P.H.; Thomas, M.B.; Ritchie, S.A.; Kay, B.H.; Ryan, P.A. Effects of Beauveria bassiana on survival, blood-feeding success, and fecundity of Aedes aegypti in laboratory and semi-field conditions. Am. J. Trop. Med. Hyg. 2012, 86, 656–664. [Google Scholar] [CrossRef] [PubMed]
- De Paula, A.R.; Souza Brito, E.; Pereira, C.R.; Pinheiro Carrera, M.; Samuels, R.I. Susceptibility of adult Aedes aegypti (Diptera: Culicidae) to infection by Metarhizium anisopliae and Beauveria bassiana: Prospects for Dengue vector control. Biocontrol Sci. Technol. 2008, 18, 1017–1025. [Google Scholar] [CrossRef]
- Dong, Y.; Morton, J.C., Jr.; Ramirez, J.L.; Souza-Neto, J.A.; Dimopoulos, G. The entomopathogenic fungus Beauveria bassiana activate toll and JAK-STAT pathway-controlled effector genes and anti-dengue activity in Aedes aegypti. Insect Biochem. Mol. Biol. 2012, 42, 126–132. [Google Scholar] [CrossRef] [PubMed]
- Garza-Hernandez, J.A.; Rodriguez-Perez, M.A.; Salazar, M.I.; Russell, T.L.; Adeleke, M.A.; de Luna-Santillana Ede, J.; Reyes-Villanueva, F. Vectorial capacity of Aedes aegypti for dengue virus type 2 is reduced with co-infection of Metarhizium anisopliae. PLoS Negl. Trop. Dis. 2013, 7, e2013. [Google Scholar] [CrossRef] [PubMed]
- Shin, S.W.; Kokoza, V.; Bian, G.; Cheon, H.M.; Kim, Y.J.; Raikhel, A.S. REL1, a homologue of Drosophila dorsal, regulates toll antifungal immune pathway in the female mosquito Aedes aegypti. J. Biol. Chem. 2005, 280, 16499–16507. [Google Scholar] [CrossRef] [PubMed]
- Xi, Z.; Ramirez, J.L.; Dimopoulos, G. The Aedes aegypti toll pathway controls dengue virus infection. PLoS Pathog. 2008, 4, e1000098. [Google Scholar] [CrossRef] [PubMed]
- Mitri, C.; Vernick, K.D. Anopheles gambiae pathogen susceptibility: The intersection of genetics, immunity and ecology. Curr. Opin. Microbiol. 2012, 15, 285–291. [Google Scholar] [CrossRef] [PubMed]
- Mnyone, L.L.; Lyimo, I.N.; Lwetoijera, D.W.; Mpingwa, M.W.; Nchimbi, N.; Hancock, P.A.; Russell, T.L.; Kirby, M.J.; Takken, W.; Koenraadt, C.J. Exploiting the behaviour of wild malaria vectors to achieve high infection with fungal biocontrol agents. Malar. J. 2012, 11, 87. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Paula, A.R.; Carolino, A.T.; Silva, C.P.; Pereira, C.R.; Samuels, R.I. Testing fungus impregnated cloths for the control of adult Aedes aegypti under natural conditions. Parasit. Vectors 2013, 6, 256. [Google Scholar] [CrossRef] [PubMed]
- Carolino, A.T.; Paula, A.R.; Silva, C.P.; Butt, T.M.; Samuels, R.I. Monitoring persistence of the entomopathogenic fungus Metarhizium anisopliae under simulated field conditions with the aim of controlling adult Aedes aegypti (Diptera: Culicidae). Parasit. Vectors 2014, 7, 198. [Google Scholar] [CrossRef] [PubMed]
- Scholte, E.J.; Knols, B.G.; Samson, R.A.; Takken, W. Entomopathogenic fungi for mosquito control: A review. J. Insect Sci. 2004, 4, 19. [Google Scholar] [CrossRef] [PubMed]
- Nene, V.; Wortman, J.R.; Lawson, D.; Haas, B.; Kodira, C.; Tu, Z. J.; Loftus, B.; Xi, Z.; Megy, K.; Grabherr, M.; et al. Genome sequence of Aedes aegypti, a major arbovirus vector. Science 2007, 316, 1718–1723. [Google Scholar] [CrossRef] [PubMed]
- Holt, R.A.; Subramanian, G.M.; Halpern, A.; Sutton, G.G.; Charlab, R.; Nusskern, D.R.; Wincker, P.; Clark, A.G.; Ribeiro, J.M.; Wides, R.; et al. The genome sequence of the malaria mosquito Anopheles gambiae. Science 2002, 298, 129–149. [Google Scholar] [CrossRef] [PubMed]
- Neafsey, D.E. Genome sequencing sheds light on emerging drug resistance in malaria parasites. Nat. Genet. 2013, 45, 589–590. [Google Scholar] [CrossRef] [PubMed]
- Neafsey, D.E.; Waterhouse, R.M.; Abai, M.R.; Aganezov, S.S.; Alekseyev, M.A.; Allen, J.E.; Amon, J.; Arcà, B.; Arensburger, P.; Artemov, G.; et al. Highly evolvable malaria vectors: The genomes of 16 Anopheles mosquitoes. Science 2014, 347. [Google Scholar] [CrossRef]
- Arensburger, P.; Megy, K.; Waterhouse, R.M.; Abrudan, J.; Amedeo, P.; Antelo, B.; Bartholomay, L.; Bidwell, S.; Caler, E.; Camara, F.; et al. Sequencing of Culex quinquefasciatus establishes a platform for mosquito comparative genomics. Science 2010, 330, 86–88. [Google Scholar] [CrossRef] [PubMed]
- Alphey, L. Natural and engineered mosquito immunity. J. Biol. 2009, 8, 40. [Google Scholar] [CrossRef] [PubMed]
- Gabrieli, P.; Smidler, A.; Catteruccia, F. Engineering the control of mosquito-borne infectious diseases. Genome Biol. 2014, 15, 535. [Google Scholar] [CrossRef] [PubMed]
- Olson, K.E.; Alphey, L.; Carlson, J.O.; James, A.A. Genetic approaches in Aedes aegypti for control of dengue: An overview. In Bridging Laboratory and Field Research for Genetic Control of Disease Vectors; Knols, B., Louis, C., Eds.; Springer: Heidelberg, Germany, 2007; pp. 77–87. [Google Scholar]
- Fragkoudis, R.; Attarzadeh-Yazdi, G.; Nash, A.A.; Fazakerley, J.K.; Kohl, A. Advances in dissecting mosquito innate immune responses to arbovirus infection. J. Gen. Virol. 2009, 90, 2061–2072. [Google Scholar] [CrossRef] [PubMed]
- Blair, C.D.; Olson, K.E. Mosquito immune responses to arbovirus infections. Curr. Opin. Insect Sci. 2014, 3, 22–29. [Google Scholar] [CrossRef] [PubMed]
- Prasad, A.N.; Brackney, D.E.; Ebel, G.D. The role of innate immunity in conditioning mosquito susceptibility to West Nile virus. Viruses 2013, 5, 3142–3170. [Google Scholar] [CrossRef] [PubMed]
- Blair, C.D. Mosquito RNAi is the major innate immune pathway controlling arbovirus infection and transmission. Future Microbiol. 2011, 6, 265–277. [Google Scholar] [CrossRef] [PubMed]
- Donald, C.L.; Kohl, A.; Schnettler, E. New Insights into Control of Arbovirus Replication and Spread by Insect RNA Interference Pathways. Insects 2012, 3, 511–531. [Google Scholar] [CrossRef]
- Keene, K.M.; Foy, B.D.; Sanchez-Vargas, I.; Beaty, B.J.; Blair, C.D.; Olson, K.E. RNA interference acts as a natural antiviral response to O’nyong-nyong virus (Alphavirus; Togaviridae) infection of Anopheles gambiae. Proc. Natl. Acad. Sci. USA 2004, 101, 17240–17245. [Google Scholar] [CrossRef] [PubMed]
- Campbell, C.L.; Keene, K.M.; Brackney, D.E.; Olson, K.E.; Blair, C.D.; Wilusz, J.; Foy, B.D. Aedes aegypti uses RNA interference in defense against Sindbis virus infection. BMC Microbiol. 2008, 8, 47. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Vargas, I.; Scott, J.C.; Poole-Smith, B.K.; Franz, A.W.; Barbosa-Solomieu, V.; Wilusz, J.; Olson, K.E.; Blair, C.D. Dengue virus type 2 infections of Aedes aegypti are modulated by the mosquito’s RNA interference pathway. PLoS Pathog. 2009, 5, e1000299. [Google Scholar] [CrossRef] [PubMed]
- Waldock, J.; Olson, K.E.; Christophides, G.K. Anopheles gambiae antiviral immune response to systemic O’nyong-nyong infection. PLoS Negl. Trop. Dis. 2012, 6, e1565. [Google Scholar] [CrossRef] [PubMed]
- Schnettler, E.; Donald, C.L.; Human, S.; Watson, M.; Siu, R.W.; McFarlane, M.; Fazakerley, J.K.; Kohl, A.; Fragkoudis, R. Knockdown of piRNA pathway proteins results in enhanced Semliki Forest virus production in mosquito cells. J. Gen. Virol. 2013, 94, 1680–1689. [Google Scholar] [CrossRef] [PubMed]
- McFarlane, M.; Arias-Goeta, C.; Martin, E.; O’Hara, Z.; Lulla, A.; Mousson, L.; Rainey, S.M.; Misbah, S.; Schnettler, E.; Donald, C.L.; et al. Characterization of Aedes aegypti innate-immune pathways that limit Chikungunya virus replication. PLoS Negl. Trop. Dis. 2014, 8, e2994. [Google Scholar] [CrossRef] [PubMed]
- Khoo, C.C.; Piper, J.; Sanchez-Vargas, I.; Olson, K.E.; Franz, A.W. The RNA interference pathway affects midgut infection- and escape barriers for Sindbis virus in Aedes aegypti. BMC Microbiol. 2010, 10, 130. [Google Scholar] [CrossRef] [PubMed]
- Khoo, C.C.; Doty, J.B.; Heersink, M.S.; Olson, K.E.; Franz, A.W. Transgene-mediated suppression of the RNA interference pathway in Aedes aegypti interferes with gene silencing and enhances Sindbis virus and dengue virus type 2 replication. Insect Mol. Biol. 2013, 22, 104–114. [Google Scholar] [CrossRef] [PubMed]
- Khoo, C.C.; Doty, J.B.; Held, N.L.; Olson, K.E.; Franz, A.W. Isolation of midgut escape mutants of two American genotype dengue 2 viruses from Aedes aegypti. Virol. J. 2013, 10, 257. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, P.M.; Rico-Hesse, R. Efficiency of dengue serotype 2 virus strains to infect and disseminate in Aedes aegypti. Am. J. Trop. Med. Hyg. 2003, 68, 539–544. [Google Scholar] [PubMed]
- Salazar, M.I.; Loroño-Pino, M.A.; Farfán-Ale, J.A.; Olson, K.E.; Beaty, B.J. American and American/Asian genotypes of dengue virus differ in mosquito infection efficiency: Candidate molecular determinants of productive vector infection. Rev. Biomed. 2010, 21, 121–135. [Google Scholar]
- Carissimo, G.; Pondeville, E.; McFarlane, M.; Dietrich, I.; Mitri, C.; Bischoff, E.; Antoniewski, C.; Bourgouin, C.; Failloux, A.B.; Kohl, A.; et al. Antiviral immunity of Anopheles gambiae is highly compartmentalized, with distinct roles for RNA interference and gut microbiota. Proc. Natl. Acad. Sci. USA 2015, 112, E176–185. [Google Scholar] [CrossRef] [PubMed]
- Vodovar, N.; Bronkhorst, A.W.; van Cleef, K.W.; Miesen, P.; Blanc, H.; van Rij, R.P.; Saleh, M.C. Arbovirus-derived piRNAs exhibit a ping-pong signature in mosquito cells. PLoS ONE 2012, 7, e30861. [Google Scholar] [CrossRef] [PubMed]
- Brackney, D.E.; Beane, J.E.; Ebel, G.D. RNAi targeting of West Nile virus in mosquito midguts promotes virus diversification. PLoS Pathog. 2009, 5, e1000502. [Google Scholar] [CrossRef] [PubMed]
- Morazzani, E.M.; Wiley, M.R.; Murreddu, M.G.; Adelman, Z.N.; Myles, K.M. Production of virus-derived ping-pong-dependent piRNA-like small RNAs in the mosquito soma. PLoS Pathog. 2012, 8, e1002470. [Google Scholar] [CrossRef] [PubMed]
- Hess, A.M.; Prasad, A.N.; Ptitsyn, A.; Ebel, G.D.; Olson, K.E.; Barbacioru, C.; Monighetti, C.; Campbell, C.L. Small RNA profiling of Dengue virus-mosquito interactions implicates the PIWI RNA pathway in anti-viral defense. BMC Microbiol. 2011, 11, 45. [Google Scholar] [CrossRef] [PubMed]
- Leger, P.; Lara, E.; Jagla, B.; Sismeiro, O.; Mansuroglu, Z.; Coppée, J.Y.; Bonnefoy, E.; Bouloy, M. Dicer-2 and Piwi-mediated RNA interference in Rift Valley fever virus infected mosquito cells. J. Virol. 2013, 87, 1631–1648. [Google Scholar] [CrossRef] [PubMed]
- Siomi, M.C.; Sato, K.; Pezic, D.; Aravin, A.A. PIWI-interacting small RNAs: The vanguard of genome defence. Nat. Rev. Mol. Cell Biol. 2011, 12, 246–258. [Google Scholar] [CrossRef] [PubMed]
- Campbell, C.L.; Black, W.C.t.; Hess, A.M.; Foy, B.D. Comparative genomics of small RNA regulatory pathway components in vector mosquitoes. BMC Genomics 2008, 9, 425. [Google Scholar] [CrossRef] [PubMed]
- Ambros, V. MicroRNA pathways in flies and worms: Growth, death, fat, stress, and timing. Cell 2003, 113, 673–676. [Google Scholar] [CrossRef] [PubMed]
- Ghildiyal, M.; Xu, J.; Seitz, H.; Weng, Z.; Zamore, P.D. Sorting of Drosophila small silencing RNAs partitions microRNA * strands into the RNA interference pathway. RNA 2010, 16, 43–56. [Google Scholar] [CrossRef] [PubMed]
- Rubio, M.; Montanez, R.; Perez, L.; Milan, M.; Belles, X. Regulation of atrophin by both strands of the mir-8 precursor. Insect Biochem. Mol. Biol. 2013, 43, 1009–1014. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.S.; Smibert, P.; Westholm, J.O.; Jee, D.; Maurin, T.; Lai, E.C. Intertwined pathways for Argonaute-mediated microRNA biogenesis in Drosophila. Nucleic Acids Res. 2014, 42, 1987–2002. [Google Scholar] [CrossRef] [PubMed]
- Okamura, K.; Hagen, J.W.; Duan, H.; Tyler, D.M.; Lai, E.C. The mirtron pathway generates microRNA-class regulatory RNAs in Drosophila. Cell 2007, 130, 89–100. [Google Scholar] [CrossRef] [PubMed]
- Zhuo, Y.; Gao, G.; Shi, J.A.; Zhou, X.; Wang, X. miRNAs: Biogenesis, origin and evolution, functions on virus-host interaction. Cell Physiol. Biochem. 2013, 32, 499–510. [Google Scholar] [CrossRef] [PubMed]
- Skalsky, R.L.; Vanlandingham, D.L.; Scholle, F.; Higgs, S.; Cullen, B.R. Identification of microRNAs expressed in two mosquito vectors, Aedes albopictus and Culex quinquefasciatus. BMC Genomics 2010, 11, 119. [Google Scholar] [CrossRef] [PubMed]
- Slonchak, A.; Hussain, M.; Torres, S.; Asgari, S.; Khromykh, A.A. Expression of mosquito microRNA Aae-miR-2940–5p is downregulated in response to West Nile virus infection to restrict viral replication. J. Virol. 2014, 88, 8457–8467. [Google Scholar] [CrossRef] [PubMed]
- Campbell, C.L.; Harrison, T.; Hess, A.M.; Ebel, G.D. MicroRNA levels are modulated in Aedes aegypti after exposure to Dengue-2. Insect Mol. Biol. 2014, 23, 132–139. [Google Scholar] [CrossRef] [PubMed]
- Yan, H.; Zhou, Y.; Liu, Y.; Deng, Y.; Chen, X. miR-252 of the Asian tiger mosquito Aedes albopictus regulates dengue virus replication by suppressing the expression of the dengue virus envelope protein. J. Med. Virol. 2014, 86, 1428–1436. [Google Scholar] [CrossRef] [PubMed]
- Shrinet, J.; Jain, S.; Jain, J.; Bhatnagar, R.K.; Sunil, S. Next generation sequencing reveals regulation of distinct Aedes microRNAs during chikungunya virus development. PLoS Negl. Trop. Dis. 2014, 8, e2616. [Google Scholar] [CrossRef] [PubMed]
- Hussain, M.; Asgari, S. MicroRNA-like viral small RNA from Dengue virus 2 autoregulates its replication in mosquito cells. Proc. Natl. Acad. Sci. USA 2014, 111, 2746–2751. [Google Scholar] [CrossRef] [PubMed]
- Hussain, M.; Torres, S.; Schnettler, E.; Funk, A.; Grundhoff, A.; Pijlman, G.P.; Khromykh, A.A.; Asgari, S. West Nile virus encodes a microRNA-like small RNA in the 3' untranslated region which up-regulates GATA4 mRNA and facilitates virus replication in mosquito cells. Nucleic Acids Res. 2012, 40, 2210–2223. [Google Scholar] [CrossRef] [PubMed]
- Franz, A.W.; Sanchez-Vargas, I.; Adelman, Z.N.; Blair, C.D.; Beaty, B.J.; James, A.A.; Olson, K.E. Engineering RNA interference-based resistance to dengue virus type 2 in genetically modified Aedes aegypti. Proc. Natl. Acad. Sci. USA 2006, 103, 4198–4203. [Google Scholar] [CrossRef] [PubMed]
- Franz, A.W.; Sanchez-Vargas, I.; Raban, R.R.; Black, W.C.t.; James, A.A.; Olson, K.E. Fitness impact and stability of a transgene conferring resistance to dengue-2 virus following introgression into a genetically diverse Aedes aegypti strain. PLoS Negl. Trop. Dis. 2014, 8, e2833. [Google Scholar] [CrossRef] [PubMed]
- Mathur, G.; Sanchez-Vargas, I.; Alvarez, D.; Olson, K.E.; Marinotti, O.; James, A.A. Transgene-mediated suppression of dengue viruses in the salivary glands of the yellow fever mosquito, Aedes aegypti. Insect Mol. Biol. 2010, 19, 753–763. [Google Scholar] [CrossRef] [PubMed]
- Merkling, S.H.; van Rij, R.P. Beyond RNAi: Antiviral defense strategies in Drosophila and mosquito. J. Insect Physiol. 2013, 59, 159–170. [Google Scholar] [CrossRef] [PubMed]
- Christophides, G.K.; Zdobnov, E.; Barillas-Mury, C.; Birney, E.; Blandin, S.; Blass, C.; Brey, P.T.; Collins, F.H.; Danielli, A.; Dimopoulos, G.; et al. Immunity-related genes and gene families in Anopheles gambiae. Science 2002, 298, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Waterhouse, R.M.; Kriventseva, E.V.; Meister, S.; Xi, Z.; Alvarez, K.S.; Bartholomay, L.C.; Barillas-Mury, C.; Bian, G.; Blandin, S.; Christensen, B.M.; et al. Evolutionary dynamics of immune-related genes and pathways in disease-vector mosquitoes. Science 2007, 316, 1738–1743. [Google Scholar] [CrossRef] [PubMed]
- Hillyer, J.F. Mosquito immunity. Adv. Exp. Med. Biol. 2010, 708, 218–238. [Google Scholar] [PubMed]
- Clayton, A.M.; Dong, Y.; Dimopoulos, G. The Anopheles innate immune system in the defense against malaria infection. J. Innate Immun. 2014, 6, 169–181. [Google Scholar] [CrossRef] [PubMed]
- Luplertlop, N.; Surasombatpattana, P.; Patramool, S.; Dumas, E.; Wasinpiyamongkol, L.; Saune, L.; Hamel, R.; Bernard, E.; Sereno, D.; Thomas, F.; et al. Induction of a peptide with activity against a broad spectrum of pathogens in the Aedes aegypti salivary gland, following Infection with Dengue Virus. PLoS Pathog. 2011, 7, e1001252. [Google Scholar] [CrossRef] [PubMed]
- Sim, S.; Ramirez, J.L.; Dimopoulos, G. Dengue virus infection of the Aedes aegypti salivary gland and chemosensory apparatus induces genes that modulate infection and blood-feeding behavior. PLoS Pathog. 2012, 8, e1002631. [Google Scholar] [CrossRef] [PubMed]
- Sanders, H.R.; Foy, B.D.; Evans, A.M.; Ross, L.S.; Beaty, B.J.; Olson, K.E.; Gill, S.S. Sindbis virus induces transport processes and alters expression of innate immunity pathway genes in the midgut of the disease vector, Aedes aegypti. Insect Biochem. Mol. Biol. 2005, 35, 1293–1307. [Google Scholar] [CrossRef] [PubMed]
- Avadhanula, V.; Weasner, B.P.; Hardy, G.G.; Kumar, J.P.; Hardy, R.W. A novel system for the launch of alphavirus RNA synthesis reveals a role for the Imd pathway in arthropod antiviral response. PLoS Pathog. 2009, 5, e1000582. [Google Scholar] [CrossRef] [PubMed]
- Fragkoudis, R.; Chi, Y.; Siu, R.W.; Barry, G.; Attarzadeh-Yazdi, G.; Merits, A.; Nash, A.A.; Fazakerley, J.K.; Kohl, A. Semliki Forest virus strongly reduces mosquito host defence signaling. Insect Mol. Biol. 2008, 17, 647–656. [Google Scholar] [CrossRef] [PubMed]
- Bian, G.; Shin, S.W.; Cheon, H.M.; Kokoza, V.; Raikhel, A.S. Transgenic alteration of Toll immune pathway in the female mosquito Aedes aegypti. Proc. Natl. Acad. Sci. USA 2005, 102, 13568–13573. [Google Scholar] [CrossRef] [PubMed]
- Shin, S.W.; Kokoza, V.; Lobkov, I.; Raikhel, A.S. Relish-mediated immune deficiency in the transgenic mosquito Aedes aegypti. Proc. Natl. Acad. Sci. USA 2003, 100, 2616–2621. [Google Scholar] [CrossRef] [PubMed]
- Kokoza, V.; Ahmed, A.; Cho, W.L.; Jasinskiene, N.; James, A.A.; Raikhel, A. Engineering blood meal-activated systemic immunity in the yellow fever mosquito, Aedes aegypti. Proc. Natl. Acad. Sci. USA 2000, 97, 9144–9149. [Google Scholar] [CrossRef] [PubMed]
- Kokoza, V.; Ahmed, A.; Woon Shin, S.; Okafor, N.; Zou, Z.; Raikhel, A.S. Blocking of Plasmodium transmission by cooperative action of Cecropin A and Defensin A in transgenic Aedes aegypti mosquitoes. Proc. Natl. Acad. Sci. USA 2010, 107, 8111–8116. [Google Scholar] [CrossRef] [PubMed]
- Antonova, Y.; Alvarez, K.S.; Kim, Y.J.; Kokoza, V.; Raikhel, A.S. The role of NF-kappaB factor REL2 in the Aedes aegypti immune response. Insect Biochem. Mol. Biol. 2009, 39, 303–314. [Google Scholar] [CrossRef] [PubMed]
- Kokoza, V.; Ahmed, A.; Wimmer, E.A.; Raikhel, A.S. Efficient transformation of the yellow fever mosquito Aedes aegypti using the piggyBac transposable element vector pBac[3xP3-EGFP afm]. Insect Biochem. Mol. Biol. 2001, 31, 1137–1143. [Google Scholar] [CrossRef] [PubMed]
- Kokoza, V.A.; Raikhel, A.S. Targeted gene expression in the transgenic Aedes aegypti using the binary Gal4-UAS system. Insect Biochem. Mol. Biol. 2011, 41, 637–644. [Google Scholar] [CrossRef] [PubMed]
- Salazar, M.I.; Richardson, J.H.; Sanchez-Vargas, I.; Olson, K.E.; Beaty, B.J. Dengue virus type 2: Replication and tropisms in orally infected Aedes aegypti mosquitoes. BMC Microbiol. 2007, 7, 9. [Google Scholar] [CrossRef] [PubMed]
- Dubrulle, M.; Mousson, L.; Moutailler, S.; Vazeille, M.; Failloux, A.B. Chikungunya virus and Aedes mosquitoes: Saliva is infectious as soon as two days after oral infection. PLoS ONE 2009, 4, e5895. [Google Scholar] [CrossRef] [PubMed]
- Terenius, O.; Marinotti, O.; Sieglaff, D.; James, A.A. Molecular genetic manipulation of vector mosquitoes. Cell Host Microbe 2008, 4, 417–423. [Google Scholar] [CrossRef] [PubMed]
- Hammond, A.M.; Nolan, T. Sex-, tissue- and stage-specific transgene expression. In Transgenic Insects: Techniques and Applications; Benedict, M.Q., Ed.; CABI Publishing: Wallingford, UK, 2014; pp. 29–50. [Google Scholar]
- Romoser, W.S.; Faran, M.E.; Bailey, C.L.; Lerdthusnee, K. An immunocytochemical study of the distribution of Rift Valley fever virus in the mosquito Culex pipiens. Am. J. Trop. Med. Hyg. 1992, 46, 489–501. [Google Scholar] [PubMed]
- Bowers, D.F.; Coleman, C.G.; Brown, D.T. Sindbis virus-associated pathology in Aedes albopictus (Diptera: Culicidae). J. Med. Entomol. 2003, 40, 698–705. [Google Scholar] [CrossRef] [PubMed]
- Girard, Y.A.; Klingler, K.A.; Higgs, S. West Nile virus dissemination and tissue tropisms in orally infected Culex pipiens quinquefasciatus. Vector Borne Zoonotic Dis. 2004, 4, 109–122. [Google Scholar] [CrossRef] [PubMed]
- Lerdthusnee, K.; Romoser, W.S.; Faran, M.E.; Dohm, D.J. Rift Valley fever virus in the cardia of Culex pipiens: An immunocytochemical and ultrastructural study. Am. J. Trop. Med. Hyg. 1995, 53, 331–337. [Google Scholar] [PubMed]
- Brault, A.C.; Foy, B.D.; Myles, K.M.; Kelly, C.L.; Higgs, S.; Weaver, S.C.; Olson, K.E.; Miller, B.R.; Powers, A.M. Infection patterns of o’nyong nyong virus in the malaria-transmitting mosquito, Anopheles gambiae. Insect Mol. Biol. 2004, 13, 625–635. [Google Scholar] [CrossRef] [PubMed]
- Parikh, G.R.; Oliver, J.D.; Bartholomay, L.C. A haemocyte tropism for an arbovirus. J. Gen. Virol. 2009, 90, 292–296. [Google Scholar] [CrossRef] [PubMed]
- Edwards, M.J.; Moskalyk, L.A.; Donelly-Doman, M.; Vlaskova, M.; Noriega, F.G.; Walker, V.K.; Jacobs-Lorena, M. Characterization of a carboxypeptidase A gene from the mosquito, Aedes aegypti. Insect Mol. Biol. 2000, 9, 33–38. [Google Scholar] [CrossRef] [PubMed]
- Moreira, L.A.; Edwards, M.J.; Adhami, F.; Jasinskiene, N.; James, A.A.; Jacobs-Lorena, M. Robust gut-specific gene expression in transgenic Aedes aegypti mosquitoes. Proc. Natl. Acad. Sci. USA 2000, 97, 10895–10898. [Google Scholar] [CrossRef] [PubMed]
- Zhao, B.; Kokoza, V.A.; Saha, T.T.; Wang, S.; Roy, S.; Raikhel, A.S. Regulation of the gut-specific carboxypeptidase: A study using the binary Gal4/UAS system in the mosquito Aedes aegypti. Insect Biochem. Mol. Biol. 2014, 54C, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Coates, C.J.; Jasinskiene, N.; Pott, G.B.; James, A.A. Promoter-directed expression of recombinant fire-fly luciferase in the salivary glands of Hermes-transformed Aedes aegypti. Gene 1999, 226, 317–325. [Google Scholar] [CrossRef] [PubMed]
- Fu, G.; Lees, R.S.; Nimmo, D.; Aw, D.; Jin, L.; Gray, P.; Berendonk, T.U.; White-Cooper, H.; Scaife, S.; Kim Phuc, H.; et al. Female-specific flightless phenotype for mosquito control. Proc. Natl. Acad. Sci. USA 2010, 107, 4550–4554. [Google Scholar] [CrossRef] [PubMed]
- Anderson, M.A.; Gross, T.L.; Myles, K.M.; Adelman, Z.N. Validation of novel promoter sequences derived from two endogenous ubiquitin genes in transgenic Aedes aegypti. Insect Mol. Biol. 2010, 19, 441–449. [Google Scholar]
- Choi, Y.J.; Fuchs, J.F.; Mayhew, G.F.; Yu, H.E.; Christensen, B.M. Tissue-enriched expression profiles in Aedes aegypti identify hemocyte-specific transcriptome responses to infection. Insect Biochem. Mol. Biol. 2012, 42, 729–738. [Google Scholar] [CrossRef] [PubMed]
- Klambt, C.; Glazer, L.; Shilo, B.Z. breathless, a Drosophila FGF receptor homolog, is essential for migration of tracheal and specific midline glial cells. Genes Dev. 1992, 6, 1668–1678. [Google Scholar] [CrossRef] [PubMed]
- Brand, A.H.; Perrimon, N. Targeted gene expression as a means of altering cell fates and generating dominant phenotypes. Development 1993, 118, 401–415. [Google Scholar] [PubMed]
- Southall, T.D.; Elliott, D.A.; Brand, A.H. The GAL4 System: A Versatile Toolkit for Gene Expression in Drosophila. CSH Protoc. 2008, 2008, 49. [Google Scholar]
- Labbe, G.M.; Nimmo, D.D.; Alphey, L. piggybac- and PhiC31-mediated genetic transformation of the Asian tiger mosquito, Aedes albopictus (Skuse). PLoS Negl. Trop. Dis. 2010, 4, e788. [Google Scholar] [CrossRef] [PubMed]
- Nimmo, D.D.; Alphey, L.; Meredith, J.M.; Eggleston, P. High efficiency site-specific genetic engineering of the mosquito genome. Insect Mol. Biol. 2006, 15, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Franz, A.W.; Jasinskiene, N.; Sanchez-Vargas, I.; Isaacs, A.T.; Smith, M.R.; Khoo, C.C.; Heersink, M.S.; James, A.A.; Olson, K.E. Comparison of transgene expression in Aedes aegypti generated by mariner Mos1 transposition and PhiC31 site-directed recombination. Insect Mol. Biol. 2011, 20, 587–598. [Google Scholar] [CrossRef] [PubMed]
- Aryan, A.; Anderson, M.A.; Myles, K.M.; Adelman, Z.N. TALEN-based gene disruption in the dengue vector Aedes aegypti. PLoS ONE 2013, 8, e60082. [Google Scholar] [CrossRef] [PubMed]
- DeGennaro, M.; McBride, C.S.; Seeholzer, L.; Nakagawa, T.; Dennis, E.J.; Goldman, C.; Jasinskiene, N.; James, A.A.; Vosshall, L.B. Orco mutant mosquitoes lose strong preference for humans and are not repelled by volatile DEET. Nature 2013, 498, 487–491. [Google Scholar] [CrossRef] [PubMed]
- Gratz, S.J.; Cummings, A.M.; Nguyen, J.N.; Hamm, D.C.; Donohue, L.K.; Harrison, M.M.; Wildonger, J.; O’Connor-Giles, K.M. Genome engineering of Drosophila with the CRISPR RNA-guided Cas9 nuclease. Genetics 2013, 194, 1029–1035. [Google Scholar] [CrossRef] [PubMed]
- Mali, P.; Yang, L.; Esvelt, K.M.; Aach, J.; Guell, M.; DiCarlo, J.E.; Norville, J.E.; Church, G.M. RNA-guided human genome engineering via Cas9. Science 2013, 339, 823–826. [Google Scholar] [CrossRef] [PubMed]
- Kistler, K.E.; Vosshall, L.B.; Matthews, B.J. Genome-engineering with CRISPR-Cas9 in the mosquito Aedes aegypti. BioRxiv 2014. [Google Scholar] [CrossRef]
- Stapleford, K.A.; Coffey, L.L.; Lay, S.; Borderia, A.V.; Duong, V.; Isakov, O.; Rozen-Gagnon, K.; Arias-Goeta, C.; Blanc, H.; Beaucourt, S.; et al. Emergence and transmission of arbovirus evolutionary intermediates with epidemic potential. Cell Host Microbe 2014, 15, 706–716. [Google Scholar] [CrossRef] [PubMed]
- Arias-Goeta, C.; Mousson, L.; Rougeon, F.; Failloux, A.B. Dissemination and transmission of the E1–226V variant of chikungunya virus in Aedes albopictus are controlled at the midgut barrier level. PLoS ONE 2013, 8, e57548. [Google Scholar] [CrossRef] [PubMed]
- Arias-Goeta, C.; Moutailler, S.; Mousson, L.; Zouache, K.; Thiberge, J.M.; Caro, V.; Rougeon, F.; Failloux, A.B. Chikungunya virus adaptation to a mosquito vector correlates with only few point mutations in the viral envelope glycoprotein. Infect. Genet. Evol. 2014, 24, 116–126. [Google Scholar] [CrossRef] [PubMed]
- Tsetsarkin, K.A.; Chen, R.; Yun, R.; Rossi, S.L.; Plante, K.S.; Guerbois, M.; Forrester, N.; Perng, G.C.; Sreekumar, E.; Leal, G.; et al. Multi-peaked adaptive landscape for chikungunya virus evolution predicts continued fitness optimization in Aedes albopictus mosquitoes. Nat. Commun. 2014, 5, 4084. [Google Scholar] [CrossRef] [PubMed]
- Colpitts, T.M.; Cox, J.; Vanlandingham, D.L.; Feitosa, F.M.; Cheng, G.; Kurscheid, S.; Wang, P.; Krishnan, M.N.; Higgs, S.; Fikrig, E. Alterations in the Aedes aegypti transcriptome during infection with West Nile, dengue and yellow fever viruses. PLoS Pathog. 2011, 7, e1002189. [Google Scholar] [CrossRef] [PubMed]
- Yasunaga, A.; Hanna, S.L.; Li, J.; Cho, H.; Rose, P.P.; Spiridigliozzi, A.; Gold, B.; Diamond, M.S.; Cherry, S. Genome-wide RNAi screen identifies broadly-acting host factors that inhibit arbovirus infection. PLoS Pathog. 2014, 10, e1003914. [Google Scholar] [CrossRef] [PubMed]
- Paradkar, P.N.; Trinidad, L.; Voysey, R.; Duchemin, J.B.; Walker, P.J. Secreted Vago restricts West Nile virus infection in Culex mosquito cells by activating the Jak-STAT pathway. Proc. Natl. Acad. Sci. USA 2012, 109, 18915–18920. [Google Scholar] [CrossRef] [PubMed]
- Paradkar, P.N.; Duchemin, J.B.; Voysey, R.; Walker, P.J. Dicer-2-dependent activation of Culex Vago occurs via the TRAF-Rel2 signaling pathway. PLoS Negl. Trop. Dis. 2014, 8, e2823. [Google Scholar] [CrossRef] [PubMed]
- Panda, D.; Pascual-Garcia, P.; Dunagin, M.; Tudor, M.; Hopkins, K.C.; Xu, J.; Gold, B.; Raj, A.; Capelson, M.; Cherry, S. Nup98 promotes antiviral gene expression to restrict RNA viral infection in Drosophila. Proc. Natl. Acad. Sci. USA 2014, 111, E3890–E3899. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Grant, G.; Sabin, L.R.; Gordesky-Gold, B.; Yasunaga, A.; Tudor, M.; Cherry, S. Transcriptional pausing controls a rapid antiviral innate immune response in Drosophila. Cell Host Microbe 2012, 12, 531–543. [Google Scholar] [CrossRef] [PubMed]
- Carter, J.R.; Keith, J.H.; Fraser, T.S.; Dawson, J.L.; Kucharski, C.A.; Horne, K.M.; Higgs, S.; Fraser, M.J., Jr. Effective suppression of dengue virus using a novel group-I intron that induces apoptotic cell death upon infection through conditional expression of the Bax C-terminal domain. Virol. J. 2014, 11, 111. [Google Scholar] [CrossRef] [PubMed]
- Lambrechts, L.; Scott, T.W. Mode of transmission and the evolution of arbovirus virulence in mosquito vectors. Proc. Biol. Sci. 2009, 276, 1369–1378. [Google Scholar] [CrossRef] [PubMed]
- Tripet, F.; Aboagye-Antwi, F.; Hurd, H. Ecological immunology of mosquito-malaria interactions. Trends Parasitol. 2008, 24, 219–227. [Google Scholar] [CrossRef] [PubMed]
- Myles, K.M.; Wiley, M.R.; Morazzani, E.M.; Adelman, Z.N. Alphavirus-derived small RNAs modulate pathogenesis in disease vector mosquitoes. Proc. Natl. Acad. Sci. USA 2008, 105, 19938–19943. [Google Scholar] [CrossRef] [PubMed]
- Cirimotich, C.M.; Scott, J.C.; Phillips, A.T.; Geiss, B.J.; Olson, K.E. Suppression of RNA interference increases alphavirus replication and virus-associated mortality in Aedes aegypti mosquitoes. BMC Microbiol. 2009, 9, 49. [Google Scholar] [CrossRef] [PubMed]
- Levis, R.; Hazelrigg, T.; Rubin, G.M. Effects of genomic position on the expression of transduced copies of the white gene of Drosophila. Science 1985, 229, 558–561. [Google Scholar] [CrossRef] [PubMed]
- Lycett, G.J.; Amenya, D.; Lynd, A. The Anopheles gambiae alpha-tubulin-1b promoter directs neuronal, testes and developing imaginal tissue specific expression and is a sensitive enhancer detector. Insect Mol. Biol. 2012, 21, 79–88. [Google Scholar] [CrossRef] [PubMed]
- Carballar-Lejarazu, R.; Jasinskiene, N.; James, A.A. Exogenous gypsy insulator sequences modulate transgene expression in the malaria vector mosquito, Anopheles stephensi. Proc. Natl. Acad. Sci. USA 2013, 110, 7176–7181. [Google Scholar] [CrossRef] [PubMed]
- Gray, C.E.; Coates, C.J. Cloning and characterization of cDNAs encoding putative CTCFs in the mosquitoes, Aedes aegypti and Anopheles gambiae. BMC Mol. Biol. 2005, 6, 16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eggleston, P.; Meredith, J.M. Docking systems for site-directed transgene integration. In Transgenic Insects: Techniques and Applications; Benedict, M.Q., Ed.; CABI Publishing: Wallingford, UK, 2014; pp. 51–65. [Google Scholar]
- Bischof, J.; Maeda, R.K.; Hediger, M.; Karch, F.; Basler, K. An optimized transgenesis system for Drosophila using germ-line-specific phiC31 integrases. Proc. Natl. Acad. Sci. USA 2007, 104, 3312–3317. [Google Scholar] [CrossRef] [PubMed]
- Franz, A.W.; Sanchez-Vargas, I.; Piper, J.; Smith, M.R.; Khoo, C.C.; James, A.A.; Olson, K.E. Stability and loss of a virus resistance phenotype over time in transgenic mosquitoes harbouring an antiviral effector gene. Insect Mol. Biol. 2009, 18, 661–672. [Google Scholar] [CrossRef] [PubMed]
- Lambrechts, L.; Koella, J.C.; Boete, C. Can transgenic mosquitoes afford the fitness cost? Trends Parasitol. 2008, 24, 4–7. [Google Scholar] [CrossRef] [PubMed]
- Marrelli, M.T.; Moreira, C.K.; Kelly, D.; Alphey, L.; Jacobs-Lorena, M. Mosquito transgenesis: What is the fitness cost? Trends Parasitol. 2006, 22, 197–202. [Google Scholar] [CrossRef] [PubMed]
- Irvin, N.; Hoddle, M.S.; O’Brochta, D.A.; Carey, B.; Atkinson, P.W. Assessing fitness costs for transgenic Aedes aegypti expressing the GFP marker and transposase genes. Proc. Natl. Acad. Sci. USA 2004, 101, 891–896. [Google Scholar] [CrossRef] [PubMed]
- Marrelli, M.T.; Li, C.; Rasgon, J.L.; Jacobs-Lorena, M. Transgenic malaria-resistant mosquitoes have a fitness advantage when feeding on Plasmodium-infected blood. Proc. Natl. Acad. Sci. USA 2007, 104, 5580–5583. [Google Scholar] [CrossRef] [PubMed]
- Isaacs, A.T.; Jasinskiene, N.; Tretiakov, M.; Thiery, I.; Zettor, A.; Bourgouin, C.; James, A.A. Transgenic Anopheles stephensi coexpressing single-chain antibodies resist Plasmodium falciparum development. Proc. Natl. Acad. Sci. USA 2012, 109, E1922–E1930. [Google Scholar] [CrossRef] [PubMed]
- Amenya, D.A.; Bonizzoni, M.; Isaacs, A.T.; Jasinskiene, N.; Chen, H.; Marinotti, O.; Yan, G.; James, A.A. Comparative fitness assessment of Anopheles stephensi transgenic lines receptive to site-specific integration. Insect Mol. Biol. 2010, 19, 263–269. [Google Scholar] [CrossRef] [PubMed]
- James, A.A. Gene drive systems in mosquitoes: Rules of the road. Trends Parasitol. 2005, 21, 64–67. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Magori, K.; Lloyd, A.L.; Gould, F. Introducing transgenes into insect populations using combined gene-drive strategies: Modeling and analysis. Insect Biochem. Mol. Biol. 2007, 37, 1054–1063. [Google Scholar] [CrossRef] [PubMed]
- Robert, M.A.; Okamoto, K.W.; Gould, F.; Lloyd, A.L. Antipathogen genes and the replacement of disease-vectoring mosquito populations: A model-based evaluation. Evol. Appl. 2014, 7, 1238–1251. [Google Scholar] [CrossRef] [PubMed]
- Ramsey, J.M.; Bond, J.G.; Macotela, M.E.; Facchinelli, L.; Valerio, L.; Brown, D.M.; Scott, T.W.; James, A.A. A regulatory structure for working with genetically modified mosquitoes: Lessons from Mexico. PLoS Negl. Trop. Dis. 2014, 8, e2623. [Google Scholar] [CrossRef] [PubMed]
- Coutinho-Abreu, I.V.; Zhu, K.Y.; Ramalho-Ortigao, M. Transgenesis and paratransgenesis to control insect-borne diseases: Current status and future challenges. Parasitol. Int. 2010, 59, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Lebedeva, P.; Zelenko, A.; Kuznetsova, M.; Gudzgorban, A. Studies on the demonstration of a viral infection in larvae of Aedes aegypti mosquitoes. Microbiol. JSU 1972, 34, 70–73. [Google Scholar]
- Afanasiev, B.N.; Ward, T.W.; Beaty, B.J.; Carlson, J.O. Transduction of Aedes aegypti mosquitoes with vectors derived from Aedes densovirus. Virology 1999, 257, 62–72. [Google Scholar] [CrossRef] [PubMed]
- Gu, J.B.; Dong, Y.Q.; Peng, H.J.; Chen, X.G. A recombinant AeDNA containing the insect-specific toxin, BmK IT1, displayed an increasing pathogenicity on Aedes albopictus. Am. J. Trop. Med. Hyg. 2010, 83, 614–623. [Google Scholar] [CrossRef] [PubMed]
- Gu, J.; Liu, M.; Deng, Y.; Peng, H.; Chen, X. Development of an efficient recombinant mosquito densovirus-mediated RNA interference system and its preliminary application in mosquito control. PLoS One 2011, 6, e21329. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, Y.; Niu, G.; Hughes, G.L.; Rasgon, J.L. A viral over-expression system for the major malaria mosquito Anopheles gambiae. Sci. Rep. 2014, 4, 5127. [Google Scholar] [PubMed]
- Ren, X.; Hoiczyk, E.; Rasgon, J.L. Viral paratransgenesis in the malaria vector Anopheles gambiae. PLoS Pathog. 2008, 4, e1000135. [Google Scholar] [CrossRef] [PubMed]
- Ren, X.; Rasgon, J.L. Potential for the Anopheles gambiae densonucleosis virus to act as an “evolution-proof” biopesticide. J. Virol. 2010, 84, 7726–7729. [Google Scholar] [CrossRef] [PubMed]
- Favia, G. Asaia paratransgenesis in mosquitoes. In Transgenic Insects: Techniques and Applications; Benedict, M.Q., Ed.; CABI Publishing: Wallingford, UK, 2014; pp. 227–238. [Google Scholar]
- Lampe, D.J.; Bongio, N.J. Paratransgenesis in mosquitoes and other insects: Microbial ecology and bacterial genetic considerations. In Transgenic Insects: Techniques and Applications; Benedict, M.Q., Ed.; CABI Publishing: Wallingford, UK, 2014; pp. 208–226. [Google Scholar]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kean, J.; Rainey, S.M.; McFarlane, M.; Donald, C.L.; Schnettler, E.; Kohl, A.; Pondeville, E. Fighting Arbovirus Transmission: Natural and Engineered Control of Vector Competence in Aedes Mosquitoes. Insects 2015, 6, 236-278. https://doi.org/10.3390/insects6010236
Kean J, Rainey SM, McFarlane M, Donald CL, Schnettler E, Kohl A, Pondeville E. Fighting Arbovirus Transmission: Natural and Engineered Control of Vector Competence in Aedes Mosquitoes. Insects. 2015; 6(1):236-278. https://doi.org/10.3390/insects6010236
Chicago/Turabian StyleKean, Joy, Stephanie M. Rainey, Melanie McFarlane, Claire L. Donald, Esther Schnettler, Alain Kohl, and Emilie Pondeville. 2015. "Fighting Arbovirus Transmission: Natural and Engineered Control of Vector Competence in Aedes Mosquitoes" Insects 6, no. 1: 236-278. https://doi.org/10.3390/insects6010236







