Distribution of the Primary Endosymbiont (Candidatus Uzinura Diaspidicola) Within Host Insects from the Scale Insect Family Diaspididae
Abstract
:1. Introduction
2. Experimental Section
2.1. Collection and Taxonomy
2.2. Incubation and Slide Mounting
2.3. Mounting of Slides
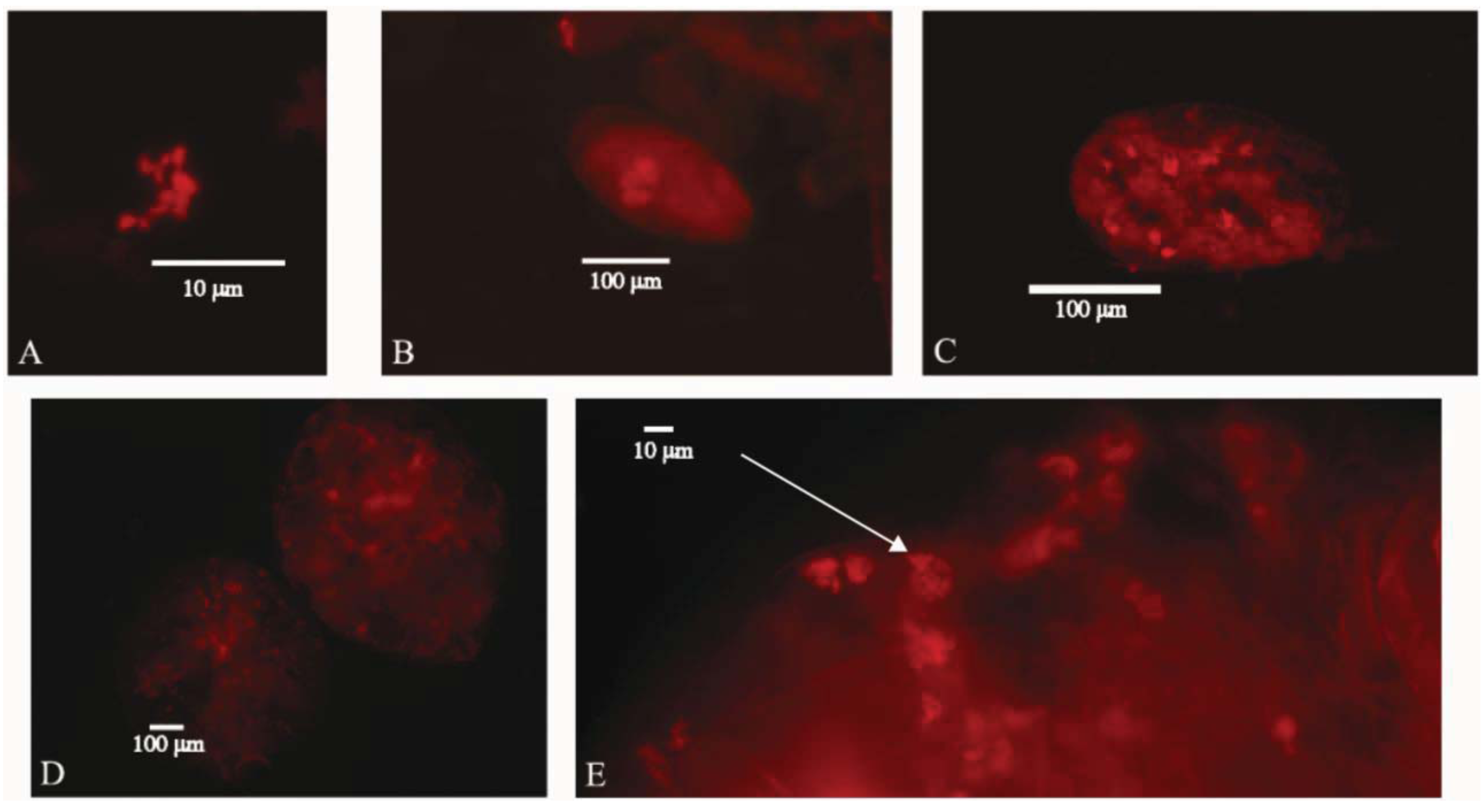
3. Results and Discussion
4. Conclusions
Acknowledgments
References
- Beardsley, J.W.; Gonzalez, R.H. The biology and ecology of armored scale insects. Annu. Rev. Entomol. 1975, 20, 47–73. [Google Scholar] [CrossRef]
- Ben-Dov, Y.; Miller, D.R.; Gibson, G.A.P. ScaleNet, Classification, 19 July 2009. Available online: http://www.sel.barc.usda.gov/SCALENET/classif.htm (accessed on 14 February 2012).
- Ben-Dov, Y. Diagnosis and Phylogenetic Relationships. In Armored Scale Insects, Their Biology, Natural Enemies and Control; Rosen, D., Ed.; Elsvier: New York, NY, USA, 1990; pp. 3–4. [Google Scholar]
- Brown, S.W.; Nur, U. Heterochromatic chromosomes in the coccids: The process of heterochromatization and the function of heterochromatin in the coccid insects are reviewed. Science 1964, 145, 130–136. [Google Scholar]
- Miller, D.R.; Davidson, J.A. Armored Scale Insect Pests of Trees and Shrubs; Cornell University Press: Ithaca, NY, USA, 2005; pp. 1–10. [Google Scholar]
- Anderson, J.C.; Gruwell, M.E.; Morse, G.E.; Normark, B.B. Cryptic diversity in the Aspidiotus nerii complex in Australia. Ann. Entomol. Soc. Am. 2010, 103, 844–854. [Google Scholar] [CrossRef]
- Thao, M.L.; Gullan, P.J.; Baumann., P. Secondary (γ-Proteobacteria) endosymbionts infect the primary (β-Proteobacteria) endosymbiont of mealybugs multiple times and coevolve with their hosts. Appl. Environ. Microbiol. 2002, 68, 3190–3197. [Google Scholar] [CrossRef]
- Gruwell, M.E.; Hardy, N.B.; Gullan, P.J.; Dittmar, K. Evolutionary relationships among primary endosymbionts of the mealybug subfamily Phenacoccinae (Hemiptera: Coccidae: Pseudococcidae). Appl. Environ. Microb. 2010, 76, 7521–7525. [Google Scholar] [CrossRef]
- Gruwell, M.E.; Morse, G.E.; Normark, B.B. Phylogenetic congruence of armored scale insects (Hemiptera: Diaspididae) and their primary endosymbionts from the phylum Bacteroidetes. Mol. Phylogenet. Evol. 2007, 44, 267–280. [Google Scholar] [CrossRef]
- Gruwell, M.E.; von Dohlen, C.D.; Patch, K.; Normark, B.B. Preliminary PCR Survey of Bacteria Associated with Scale Insects (Hemiptera: Coccoidea). In Proceedings of the Tenth International Symposium for Scale Insect Studies, Adana, Turkey, 19–23 April 2004; pp. 101–116.
- Metschnikoff, E. Embryologische studien an insekten. Z. Wissenschaftlich. Zool. 1866, 16, 389–500. [Google Scholar]
- Sulc, K. Symbiontsche Saccaromyceten der echten Cicaden. Sitzungsberichte der bohmischen Gesellschaft der Wissenschaften. 1910.
- Buchner, P. Endosymbiosis of Animals with Plant Microorganisms; John Wiley and Sons: New York, NY, USA, 1965; pp. 282–296. [Google Scholar]
- Tremblay, E. Beobachtung uber das Schicksal der Richtungskorper und die Endosymbioses bei einigen Cocciden. In Proceedings of the Eleventh International Congress of Entomology, Vienna, Austria, 17–25 August 1960; pp. 415–418.
- Tremblay, E. Coccoidea Endocytobiosis. In Insect Endocytobiosis: Morphology, Physiology, Genetics, Evolution; Schwemmler, W., Gassner, G., Eds.; CRC Press Inc.: Boca Raton, FL, USA, 1989; pp. 145–173. [Google Scholar]
- Tremblay, E. Endosymbionts. In Armored Scale Insects, Their Biology, Natural Enemies and Control; Rosen, D., Ed.; Elsvier: New York, NY, USA, 1990; pp. 275–283. [Google Scholar]
- von Dohlen, C.D.; Kohler, S.; Alsop, S.T.; McManus, W.R. Mealybug beta-proteobacterial endosymbionts contain gamma-proteobacterial symbionts. Nature 2001, 412, 782–791. [Google Scholar]
- Sabree, Z.L.; Kambhampati, S.; Moran, N. Nitrogen recycling and nutritional provisioning by Blattabacterium, the cockroach endosymbiont. Proc. Natl. Acad. Sci. USA 2009, 106, 19521–19526. [Google Scholar]
- Lopez-Sanchez, M.J.; Neef, A.; Pereto, J.; Patino-Navarrete, R.; Pignatelli, M.; Latorre, A.; Moya, A. Evolutionary convergence and nitrogen metabolism in Blattabacterium strain Bge, primary endosymbiont of the cockroach Blattella germanica. PLoS Genet. 2009, 5. [Google Scholar] [CrossRef]
- McCutcheon, J.P.; Moran, N.A. Parallel genomic evolution and metabolic interdependence in an ancient symbiosis. Proc. Natl. Acad. Sci. USA 2007, 104, 19392–19397. [Google Scholar] [CrossRef]
- Moran, N.A.; Tran, P.; Gerardo, N.M. Symbiosis and insect diversification: An ancient symbiont of sap-feeding insects from the bacterial phylum Bacteroidetes. Appl. Environ. Microbiol. 2007, 71, 8802–8810. [Google Scholar]
- Shigenobu, S.; Watanabe, H.; Hattori, M.; Sakaki, Y; Ishikawa, H. Genome sequence of the endocellular bacterial symbiont of aphids Buchnera sp. APS. Nature 2000, 407, 31–60. [Google Scholar] [CrossRef]
- Tamas, I.; Klasson, L.; Canback, B.; Näslund, A.K.; Eriksson, A.S.; Wernegreen, J.J.; Sandstrom, J.P.; Moran, N.A.; Andersson, S.G. Fifty million years of genomic stasis in endosymbiotic bacteria. Science 2002, 296, 2376–2379. [Google Scholar]
- Wu, D.; Daugherty, S.C.; van Aken, S.E.; Pai, G.H.; Watkins, K.L.; Khouri, H.; Tallon, L.J.; Zabroskyh, J.M.; Dunbar, H.E.; Tran, P.L.; et al. Metabolic complementarity and genomics of the dual bacterial symbiosis of sharpshooters. PLoS Biol. 2006, 4. [Google Scholar] [CrossRef]
- Nakabachi, A.; Yamashita, A.; Toh, H.; Ishikawa, H.; Dunbar, H.E.; Moran, N.A.; Hattori, M. The 160-kb genome of the bacterial endosymbiont Carsonella. Science 2008, 314. [Google Scholar] [CrossRef]
- Banks, H.J. Physiology and Biochemistry. In Armored Scale Insects, Their Biology, Natural Enemies and Control; Rosen, D., Ed.; Elsvier: New York, NY, USA, 1990; pp. 275–283. [Google Scholar]
- Foldi, I. Internal Anatomy. In Armored Scale Insects, Their Biology, Natural Enemies and Control; Rosen, D., Ed.; Elsvier: New York, NY, USA, 1990; pp. 65–80. [Google Scholar]
- Cochran, D.G. Nitrogen excretion in cockroaches. Annu. Rev. Entomol. 1985, 30, 29–49. [Google Scholar] [CrossRef]
- Mullins, D.E. An investigation into the Nitrogen Balance of an Insect Periplaneta americana (L.) with Specific Reference to Urate Storage and Mobilization, the Urate Storage Complex and Nitrogenous Excretory Products. Ph.D. Dissertation, Virginia Polytechnic Institute and State University, Blacksburg, VA, USA, 1971. [Google Scholar]
- Malke, H.; Schwartz, W. Production of aposymbiotic cockroaches by means of lysozyme. Nature 1964, 204, 1223–1224. [Google Scholar] [CrossRef]
- Cochran, D.G. Comparative analysis of excreta and fat body from various cockroach species. Comp. Biochem. Physiol. 1979, 64, 1–4. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Gruwell, M.E.; Flarhety, M.; Dittmar, K. Distribution of the Primary Endosymbiont (Candidatus Uzinura Diaspidicola) Within Host Insects from the Scale Insect Family Diaspididae. Insects 2012, 3, 262-269. https://doi.org/10.3390/insects3010262
Gruwell ME, Flarhety M, Dittmar K. Distribution of the Primary Endosymbiont (Candidatus Uzinura Diaspidicola) Within Host Insects from the Scale Insect Family Diaspididae. Insects. 2012; 3(1):262-269. https://doi.org/10.3390/insects3010262
Chicago/Turabian StyleGruwell, Matthew E., Meghan Flarhety, and Katharina Dittmar. 2012. "Distribution of the Primary Endosymbiont (Candidatus Uzinura Diaspidicola) Within Host Insects from the Scale Insect Family Diaspididae" Insects 3, no. 1: 262-269. https://doi.org/10.3390/insects3010262
APA StyleGruwell, M. E., Flarhety, M., & Dittmar, K. (2012). Distribution of the Primary Endosymbiont (Candidatus Uzinura Diaspidicola) Within Host Insects from the Scale Insect Family Diaspididae. Insects, 3(1), 262-269. https://doi.org/10.3390/insects3010262



